Patients undergoing chemotherapy treatment in the outpatient setting often receive information about expected adverse effects related to chemotherapy from a variety of healthcare professionals, including nurses, pharmacists, and physicians. Although the repetition of providing this information may help the patient learn the expected adverse effects of their chemotherapy regimen, our experience has been that patients are apprehensive about self-managing chemotherapy-related adverse effects.
It is crucial that patients are educated about their chemotherapy and management of adverse effects prior to and during subsequent cycles of treatment to abate the anxiety and distress that may be associated with a lack of knowledge.1,2 In our experience, when speaking with patients in outpatient chemotherapy infusion centers, patients are not knowledgeable about how to self-monitor for chemotherapy-related adverse effects, including when to expect them and how to manage them effectively at home. In addition, we have observed that patients cannot state the names of the chemotherapy agents they are receiving, including the names of any supportive care premedications and the reason they are being used.
Although it is important that patients treated with chemotherapy be educated about what to expect from their chemotherapy regimen and appropriate use of supportive care medications, it is equally important that patients be assessed about their understanding of the counseling points provided to them.3,4 The lack of knowledge about their chemotherapy treatment and how to manage related adverse effects could lead to increased hospital admissions, increased morbidity, and decreased quality of life for the patient.3 A study by Weeks and colleagues showed that 69% of patients with advanced lung cancer and 81% of patients with advanced colorectal cancer did not report understanding that chemotherapy will not be a cure to their cancer.5
In related studies, clinical pharmacy services in the ambulatory oncology setting have been analyzed and led investigators to the conclusion that pharmacists are able to provide drug-related and consultative interventions. Drug-related interventions range from medication reconciliation to adverse effect management and prevention. Consultative interventions include patient education and answering drug information questions. In the study by Ruder and colleagues, during the patient education sessions, the pharmacist reviewed the treatment plan, adverse effects, and how to prevent them. Patient satisfaction was also assessed for the time spent with a pharmacist, and 28 patients (68%) strongly agreed that the medications recommended by the pharmacist for side effect management due to chemotherapy were beneficial.6
Iihara and colleagues reported that pharmacists can reduce physicians’ workload in outpatient oncology clinics and improve patient care. Pharmacists accomplished workload reduction by dedicating 75 hours per month to provide patient education. As a crucial team member, pharmacists were able to educate more patients and increase the prescriptions for supportive care medications each month.7 Tuffaha and colleagues evaluated ambulatory pediatric cancer patients and found that 247 (26%) of 939 interventions were related to pharmacist-provided patient counseling. This included education on the chemotherapy treatment plan, number of cycles, infusion time, common adverse effects, and recommendations to mitigate chemotherapy-related adverse effects.8 Similarly, Krzyzanowska and colleagues found that of 583 pharmacist interventions in an adult ambulatory clinic, 143 consisted of patient education.6 McKee and colleagues evaluated patient–pharmacist relationships and found that 86% of patients agreed that it is important to discuss treatment with a pharmacist and 84.3% of patients stated that they learned something new from an encounter with a pharmacist.9
To date, no study has evaluated ambulatory patient understanding of chemotherapy medications and management of supportive care medications. Based on the study by Weeks and colleagues, who found that a majority of oncology patients had poor insight into their treatment plan,5 we designed and implemented this prospective, survey-based study. The purpose of this study was to identify gaps in patients’ knowledge and their understanding of expected adverse effects related to chemotherapy treatment and the supportive care medications used to manage them. At the conclusion of this study, we hope to share further insight into the gaps that still exist in the education provided to patients regarding their chemotherapy.
The primary objective of this study was to evaluate patients’ knowledge of their chemotherapy regimen and management of expected adverse effects. Secondary objectives included assessing patients’ satisfaction with the education provided to them and the preferred method of receiving this education. The hypothesis of this study was that more than 50% of ambulatory oncology patients will not be confident in understanding their chemotherapy treatment and management of their chemotherapy- related adverse effects at home.
Methods
This was a multicenter, prospective, survey-based study involving 2 different outpatient chemotherapy infusion centers within the same university-based health system. One infusion center was part of a large teaching hospital, and consisted of 32 infusion chairs with 3 clinical staff pharmacists in the oncology satellite pharmacy dispensing chemotherapy to inpatients and outpatients. The other site was based in a smaller community hospital with a total of 22 infusion chairs, including 7 in a physician’s office owned by the community hospital. One clinical staff pharmacist dispensed all intravenous medications, including chemotherapy, to the main outpatient site at the community hospital, and the rest of the chemotherapy dispensed to the physician’s office was prepared by the nursing staff. In addition, an oncology pharmacy specialist was present at the community hospital outpatient infusion centers 2 days per week to provide patient education and other clinical services. All adult ambulatory cancer patients were identified for this study by viewing the outpatient infusion center schedule in order to identify patients currently receiving chemotherapy. All patients consented to participate in the study between December 2013 and May 2014. During the study period, patients were included if they were aged ≥18 years and currently receiving chemotherapy treatment. This study was approved by the Institutional Review Board.
All patients were notified that participation in the study was voluntary and that they could stop taking the survey at any time. To ensure the privacy and comfort of patients participating in the study, private rooms and isolated areas were available to complete the survey. Participation in the study required the patient to complete a survey containing 14 questions with an estimated completion time of 20 minutes.
The investigators reviewed the survey questionnaire with the patient in great detail and answered any questions about it. Patients were excluded from the study and not asked to complete the survey if they did not speak English, were sleeping, undergoing a nursing procedure, or wanted privacy; they did not have to complete the survey at their first meeting with the investigator. Patients were given the survey in the outpatient infusion center and were allowed to take the survey home to re-read and complete. Patients were also encouraged to discuss with family and friends if this would help with their decision to complete the survey.
Data collection consisted of patient responses to a nonvalidated survey created by the investigators (Figure). The survey questionnaire included the following 4 categories: patients’ knowledge of chemotherapy and premedications, patients’ understanding of the management of chemotherapy-related adverse effects, methods reported by patients for management of chemotherapy adverse effects at home, and patient satisfaction of chemotherapy education provided to them.
The sample size goal was 50 patients. Because the intent of this study was to use descriptive statistics to analyze the results of the survey questions, no sample size calculation was performed. In previously published studies involving surveys targeting oncology patients, the number of surveys analyzed was in the range of 55 to 150.5,6 Because of the time constraints with conducting this study, we aimed to achieve a sample size of 50 patients.
Results
A total of 92 patient surveys were distributed; 67 surveys received consent and were returned. All 67 patients were enrolled in the study. Primary end points of the study evaluated patients’ knowledge of their chemotherapy regimen and related adverse effects. Although most patients responded that they had a good understanding of the chemotherapy regimen they were receiving, which was defined as patients answering “strongly agree” or “agree” to that question, few patients were able to list all of the chemotherapy medications they were receiving— 91% and 52.2%, respectively. Patients responded similarly to the question regarding their understanding of premedications and the ability to list their premedications—97% and 29.8%, respectively. Similar inconsistencies were seen from patients completing the survey at both outpatient infusion centers (Table 1).
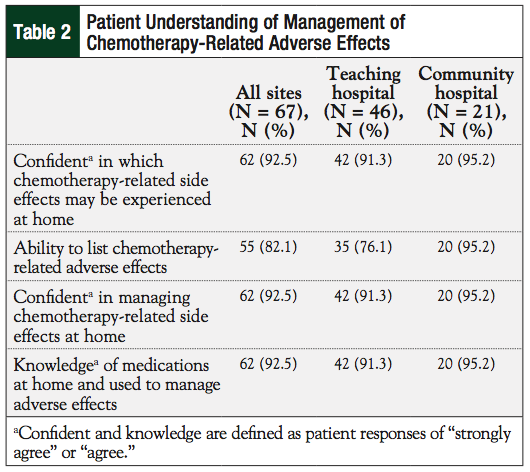
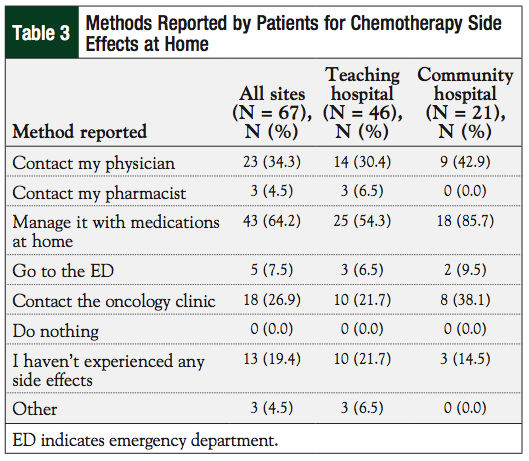
Results from questions relating to patients’ understanding of the management of chemotherapy-related adverse effects are available in Table 2. Patients treated at our smaller community hospital infusion center were more confident in knowing which chemotherapy-related side effects to expect at home and were able to list chemotherapy-related adverse effects in correlation. Patients at the larger teaching hospital infusion center were able to do the same; however, these patients were not able to list chemotherapy-related adverse effects to the same extent as those patients being treated at the community hospital. The majority of patients at both sites stated that if chemotherapy side effects did occur at home, most would manage it with the medications they had at home (Table 3).
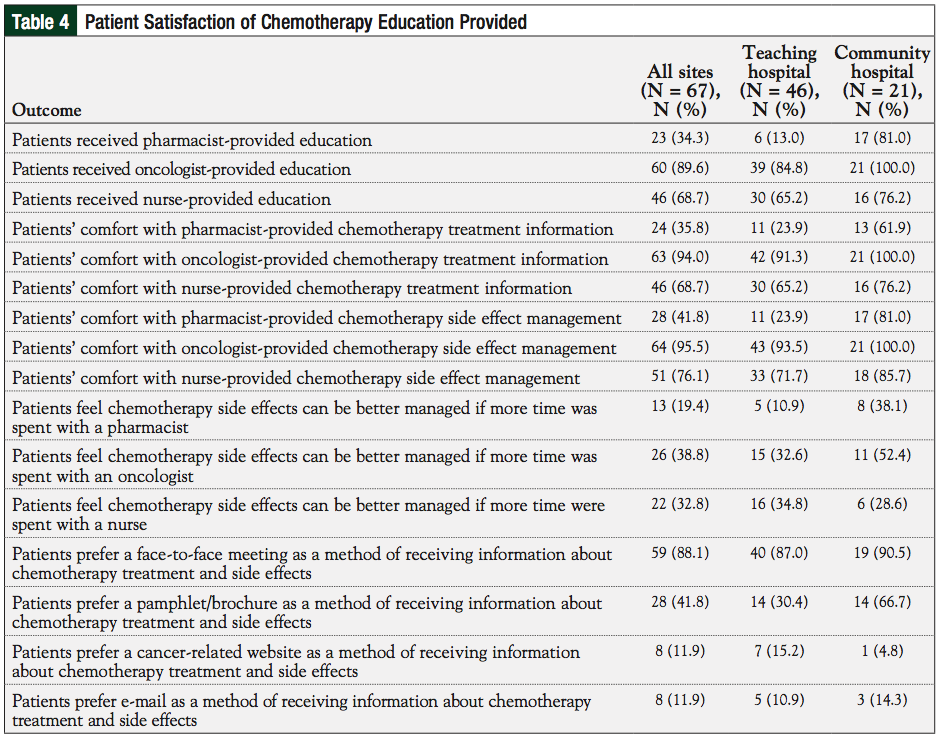
Survey questions evaluating patient satisfaction with the chemotherapy education provided showed pharmacists as having a minimal role in educating patients (34.3%); only 41.8% of patients felt most comfortable with pharmacists providing information on management of adverse effects. Patient preference for receiving education and side effect management by other healthcare professionals are also listed in Table 4. The majority (88.1%) of patients preferred a face-to-face meeting as a method of receiving chemotherapy education and adverse effect information. A small percentage (41.8%) of patients also preferred to receive a pamphlet or brochure.
Discussion
Despite that a majority of patients prefer a face-to-face meeting to discuss chemotherapy treatment and adverse effects, our large teaching hospital infusion center does not have a dedicated oncology pharmacist to provide education to patients, whereas our smaller community hospital has an oncology pharmacist on-site approximately 2 days per week.
Because of the volume of inpatient and outpatient chemotherapy at the teaching hospital, the role of the clinical staff pharmacist in the oncology satellite is mainly limited to distributive functions. At the moment, an outpatient oncology pharmacy specialist is not part of the pharmacy practice model. The analysis of these 2 centers showed that, in general, patients preferred oncologist-provided chemotherapy treatment and adverse effects information. However, more community hospital cancer patients, who were familiar with the role of the oncology pharmacist, preferred pharmacist-provided education and felt more comfortable consulting with a pharmacist for management of their chemotherapyrelated adverse effects.
In previous literature, patients have indicated a high importance but low satisfaction with pharmacist inquiry into the patient’s medication regimen.10 When pharmacists are available and able to follow-up with patients’ adverse effects management, there was a 74% positive response from patients.11 The majority of patients felt that pharmacist-run follow-up services allowed them to become more confident in managing medications on their own.11 The results of the secondary objective in this study as well as previous literature show a great potential for pharmacists to impact and improve patient care.
Limitations
Based on the results of the study, the hypothesis was refuted because more than 50% of patients were confident in their understanding of their chemotherapy treatment and management of their chemotherapy-related adverse effects at home. Although, the data did not support our hypothesis, data supporting the primary objective of assessing patients’ knowledge of their chemotherapy regimen and management of expected adverse effects provided a more accurate reflection of patients’ understanding and did not directly correlate with patients’ confidence level. Patients’ overconfidence could be confounded by the fact that this was a pharmacist-administered survey and was a limitation to this study.
Since this survey was nonvalidated, there is the possibility that the survey questions did not measure what they were designed to measure. Other limitations included fewer patients at the community infusion center compared with the large teaching hospital, which may skewed the results of the data, because a single patient response may have significantly altered the data. In addition, because of the voluntary nature of the consent and that patients can stop taking the survey at any time, not all questions were answered on all 67 surveys. The results of the lack of responses for a few questions may have altered the data outcomes as well.
Furthermore, not asking patients to list names of medications they use at home for chemotherapy-related adverse effect management or their indications in the assessment were other limitations in this study. We encountered a language barrier with some patients. Five patients did not consent to the survey at our teaching hospital infusion center because they did not speak English. In addition, 5 patients at our teaching hospital infusion center and 5 patients at our community hospital infusion center asked the study investigators to read the survey questions out loud and fill out the survey for the participants. This may have influenced patients’ responses in answering questions with having the investigator administrating the survey.
Another limitation to the study was that the investigators could not verify the accuracy of patient premedications and expected adverse effects of chemotherapy because the investigators did not use patient identifiers after administering the survey to verify the treatment regimen the patient received.
Conclusion
The results of this study will help to increase awareness of the gaps that exist in patients’ knowledge and management of chemotherapy-related adverse effects. This knowledge gap outlines opportunities that oncology pharmacists may be able to fill by providing much needed patient education and ongoing monitoring and management of chemotherapy-related adverse effects. Future studies analyzing the impact of pharmacist-provided education to patients with cancer in the ambulatory setting are warranted. To our knowledge, this was the first study to assess patient understanding of chemotherapy and related adverse effects. The results of our study provide further evidence of how oncology pharmacists can provide a valuable patient care service to cancer patients receiving chemotherapy in the outpatient setting.
Author Disclosure Statement
Dr Huynh and Dr Trovato reported no conflicts of interest.
Dr Huynh is a PGY-2 Oncology Pharmacy Practice Resident, and Dr Trovato is an Associate Professor, Department of Pharmacy Practice and Science, University of Maryland School of Pharmacy, Baltimore, MD.
References
1. National Comprehensive Cancer Network. NCCN Clinical Guidelines in Oncology. Distress Management Version 2. 2014. July 8, 2014.
2. Traeger L, Greer JA, Fernandez-Robles C, et al. Evidence-based treatment of anxiety in patients with cancer. J Clin Oncol. 2012;30:1197-1205.
3. Krzyzanowska MK, Treacy J, Maloney B, et al. Development of a patient registry to evaluate hospital admissions related to chemotherapy toxicity in a community cancer center. J Oncol Pract. 2005;1:15-19.
4. Neuss MN, Desch CE, McNiff KK, et al. A process for measuring the quality of cancer care: the Quality Oncology Practice Initiative. J Clin Oncol. 2005;23:6233-6239.
5. Weeks JC, Catalano PJ, Cronin A, et al. Patients’ expectations about effects of chemotherapy for advanced cancer. N Engl J Med. 2012;367:1616-1625.
6. Ruder AD, Smith DL, Madsen MT, Kass FH III. Is there a benefit to having a clinical oncology pharmacist on staff at a community oncology clinic? J Oncol Pharm Pract. 2011;17:425-432.
7. Iihara H, Ishihara M, Matsuura K, et al. Pharmacists contribute to the improved efficiency of medical practices in the outpatient cancer chemotherapy clinic. J Eval Clin Pract. 2012;18:753-760.
8. Tuffaha HW, Abdelhadi O, Omar SA. Clinical pharmacy services in the outpatient pediatric oncology clinics at a comprehensive cancer center. Int J Clin Pharm. 2012;34:27-31.
9. McKee M, Frei BL, Garcia A, et al. Impact of clinical pharmacy services on patients in an outpatient chemotherapy academic clinic. J Oncol Pharm Pract. 2011;17:387-394.
10. Gourdji I, McVey L, Loiselle C. Patients’ satisfaction and importance ratings of quality in an outpatient oncology center. J Nurs Care Qual. 2003;18:43-55.
11. Ryan N, Chambers C, Ralph C, et al. Evaluation of clinical pharmacists’ follow-up service in an oncology pain clinic. J Oncol Pharm Pract. 2013;19:151-158.