Symptom Overview and Etiology
Cancer in women ages 15 to 19 years is relatively rare, but when it occurs, menstruation can result in clinically significant complications.1 During the care of hematologic malignancies, heavy menstrual bleeding in this patient population is commonly correlated with thrombocytopenia. The effects of chemotherapy, radiation, or bone marrow transplantation are also potential secondary causes of therapy-induced thrombocytopenia, leading to excessive menstrual bleeding.1,2 Finally, chemotherapy-related impacts on the hypothalamic-pituitary- gonadal axis can also lead to anovulatory uterine bleeding.1 Significant morbidity can occur, often leading to an increased need for regular transfusion support, with associated concerns for potential alloimmunization, and infectious risks.3 Therefore, the diagnosis and treatment of cancer in young women can present significant challenges related to the best management of their menstrual bleeding.
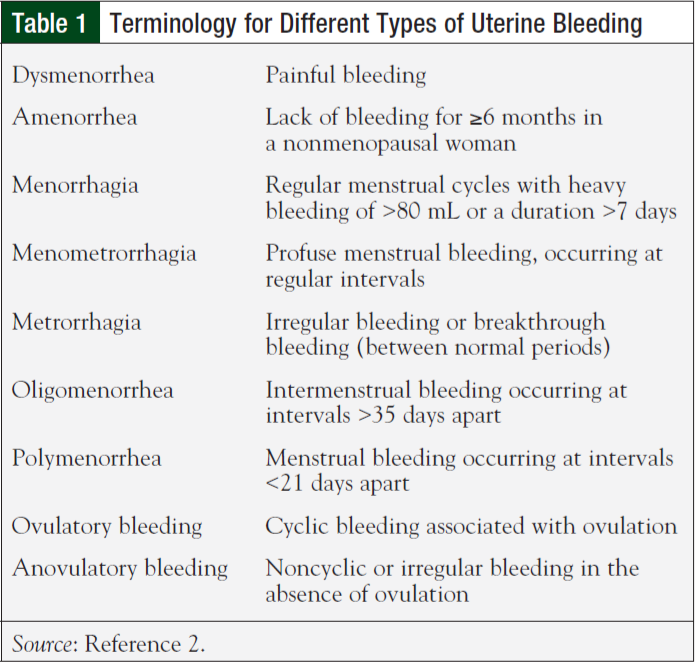
It is important to distinguish between the different types of uterine bleeding to better understand the potential extent of blood loss, and the best method for management (Table 1). Irregular menstrual cycles in the absence of ovulation (anovulatory bleeding) occur more commonly in adolescent and perimenopausal populations, and are often associated with heavy bleeding.2,4 This can be problematic, as patients may already present in an anemic state because of their hematologic malignancies. 1 Patients experiencing abnormal uterine bleeding (eg, menorrhagia, menometrorrhagia, or metrorrhagia) are also at a great risk for severe blood loss, and subsequent anemia.
Treatment Options
Management of menstrual bleeding is dependent on multiple factors: severity of cancer and presentation; timing and type of cancer treatment; time between therapies; and severity and urgency of active bleeding.1,5 Patients presenting with severe disease may have significant thrombocytopenia, and onset of menses would likely result in an urgent need to manage associated bleeding.1 A patient not presenting with severe thrombocytopenia, but about to undergo treatment, would also benefit greatly from preventive therapies to suppress the regular occurrence of her cycle.1,2
Preventive Treatment
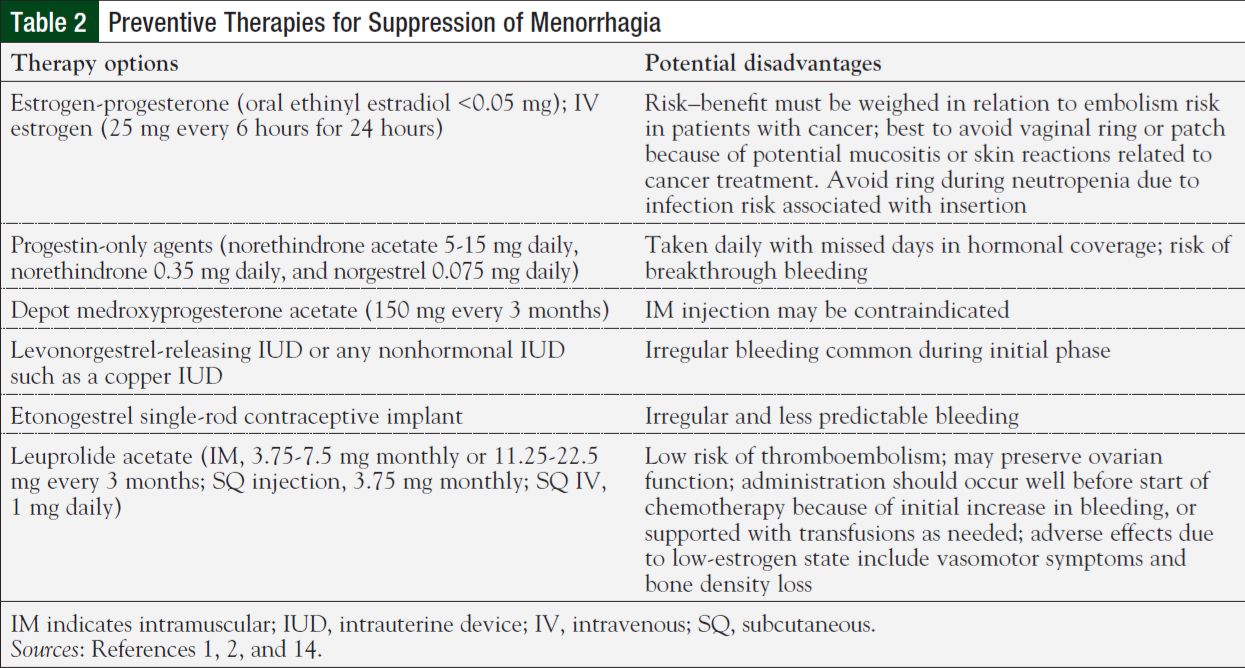
A variety of treatment options have been used to prevent the onset of menstrual bleeding (Table 2), versus attempting to halt active bleeding. Considerations that should be taken into account when deciding whether to provide preventive treatment include whether the patient can tolerate oral medication (as is the case with oral contraceptives), 2,3 contraindications to intramuscular (IM) injections, and safe timing of minor procedures (eg, intrauterine device [IUD] placement or rod implantation).1
Oral contraceptives include combination estrogen and progestin products, or progestin-only products.1 Importantly, these agents must be given continuously to prevent the onset of bleeding. There are no randomized studies assessing the efficacy and safety of these agents in women undergoing chemotherapy, and although combination hormone therapies are used regularly in this population, they do pose potential risks (eg, increased thromboembolic events, liver dysfunction, and venoocclusive disease in patients undergoing bone marrow transplantation).1,2,6 Progestin-only products pose less risk for thromboembolic events, but can be associated with more breakthrough bleeding.1
Concerns also rise for the patient’s ability to consistently receive oral therapy despite having chemotherapy- induced nausea/vomiting, and experience adequate absorption during times of diarrhea and/or mucositis.7,8 Alternative routes, such as vaginal rings or skin patches, should be avoided because of their potential to be impacted by chemotherapy-influenced absorption factors, specifically mucositis- and radiation-related skin reactions. 1,9 In addition, infection risk with insertion of a vaginal ring in neutropenic patients further limits the use of this contraceptive route.2
Long-acting agents (eg, medroxyprogesterone IM injections, levonorgestrel-releasing IUDs, and implantable etonogestrel rods) can be used, but timing of administration or implantation must be taken into account.1 High rates of expulsion during heavy bleeding would eliminate this method as a useful option for emergent cessation of excessive bleeding.10 Initial breakthrough bleeding often limits the use of IM medroxyprogesterone for emergent use, but it can be a useful option when initiated well before the onset of thrombocytopenia. IUDs and implantable rods as a viable option may be limited due to the irregular bleeding that can initially occur with these modalities, but it is a reasonable option to continue their use, if the patient already has them in place and is not experiencing bleeding issues.1
Leuprolide acetate, the gonadotropin-releasing hormone agonist, has received favorability because of its consistent ability to lead to amenorrhea.8 A systemic review reported amenorrhea rates as high as 96% when leuprolide was used. Additionally, a retrospective study of women of reproductive age undergoing chemotherapy found no incidence of moderate or severe menhorrhagia in patients receiving leuprolide versus depot medroxyprogesterone acetate (DMPA) (approximately 21.4%).11 Unfortunately, there is no consensus on dosing and timing of administration for this patient population. Daily intravenous (IV) dosing can be used if IM or subcutaneous injections of leuprolide are contraindicated becauseof a thrombocytopenic state.1 Although the risk for thromboembolism is low compared with hormonal options, it is important to be aware of the vasomotor symptoms and bone density losses that can result from the low-estrogen state induced by this agent.1,12,13 Progestin therapy can be added back to this treatment to help balance and reduce these side effects.12-14
Emergent Treatment
Life-threatening bleeding associated with thrombocytopenia secondary to hematologic malignancy or myelosuppressive therapy should be taken seriously, and appropriate treatment should be initiated to minimize morbidity.
Nonsteroidal anti-inflammatory drugs have been used in the general population to lessen the severity of menses bleeding through decreased prostaglandin production.2 Because of their antiplatelet effects, as well as the fact that they only reduce versus completely suppress bleeding, this class of agents is not recommended for use in oncology patients.
High-dose estrogen therapy has been successful in patient populations where thrombocytopenia is of concern. 1,15 Doses are characterized as >0.05 mg daily (high dose) or standard/low-dose estrogen (<0.05 mg). Lowdose therapy can be initiated and titrated up to high-dose if cessation does not occur or high-dose can be utilized if persistent bleeding occurs in the presence of low-dose therapy. Upon improvements in bleeding, estrogen therapy can be titrated down to standard or low-dose estrogen (<0.05 mg) to ensure continued menstrual control. 1,5,15 As for IV conjugated equine estrogen, a dose of 25 mg every 6 hours for 1 day can be used, and can then be transitioned to oral, high-dose therapy, and then lowdose therapy once bleeding is controlled.11 Similar to preventive therapy, when using estrogen agents, the risk of thromboembolism should be considered. DMPA, leuprolide, levonorgestrel-releasing IUDs, and implantable etonogestrel rods should be avoided in acute bleeding cases because of possible delay in onset of action, and/or potential to cause irregular bleeding.1,8,11
Oral medroxyprogesterone in doses ranging from 60 mg to 120 mg, given as 5 mg every 1 to 2 hours for the first day, followed by 20 mg daily for an additional 10 days, led to menstrual cessation in 24 adolescent patients.16 Additionally, alternative dosing entailing 20 mg 3 times daily for 7 days, followed by 20 mg daily for 3 weeks, also resulted in effective cessation in adult patients.17
The use of oral tranexamic acid is approved for use in the treatment of heavy menstrual bleeding,1 but has not been significantly studied in patients with hematologic malignancies.2 Although risk for thrombosis appears to be of little concern in the general population, it is difficult to extrapolate these risks to oncology patients. Many of these patients carry contraindications to tranexamic acid given their risk of thrombosis.1 Because of the limited data on tranexamic acid, and its potential risks in female oncology patients, risk–benefit analyses should be performed prior to its use.
Refractory Treatment
Most data available for refractory treatment is found primarily in nonmalignant hematologic disorders (eg, von Willebrand disease), making it challenging to extrapolate to this patient population.2 See Table 3 for a list of emergent and refractory therapies for suppression of menorrhagia. Experienced hematologists may find these agents helpful when faced with few alternatives.
In conclusion, menstrual suppression in female patients undergoing chemotherapy should be addressed to avoid significant morbidity. Although there are a number of treatment options available, more studies are needed to assess their efficacy and safety in this patient population. Agent and dose selection should be based on prevention versus emergent needs to stop bleeding, severity of existing or expected thrombocytopenia, and timing of chemotherapy. When possible, preventive therapy should be administered to ensure control is established prior to any bleeding risks.1
References
- American College of Obstetricians and Gynecologists. Committee opinion no. 606: options for prevention and management of heavy menstrual bleeding in adolescent patients undergoing cancer treatment. Obstet Gynecol. 2014;124:397-402.
- Bates JS, Buie LW, Woodis CB. Management of menorrhagia associated with chemotherapy-induced thrombocytopenia in women with hematologic malignancy. Pharmacotherapy. 2011;31:1092-1110.
- Adegite EA, Goyal RK, Murray PJ, et al. The management of menstrual suppression and uterine bleeding: a survey of current practices in the Pediatric Blood and Marrow Transplant Consortium. Pediatr Blood Cancer. 2012;59:553-557.
- Telner DE, Jakubovicz D. Approach to diagnosis and management of abnormal uterine bleeding. Can Fam Physician. 2007;53:58-64.
- Amsterdam A, Jakubowski A, Castro-Malaspina H, et al. Treatment of menorrhagia in women undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant. 2004;34:363-366.
- Hägglund H, Remberger M, Klaesson S, et al. Norethisterone treatment, a major risk-factor for veno-occlusive disease in the liver after allogenic bone marrow transplantation. Blood. 1998;92:4568-4572.
- Chiusolo P, Salutari P, Sica S, et al. Luteinizing hormone-releasing hormone analogue: leuprorelin acetate for the prevention of menstrual bleeding in premenopausal women undergoing stem cell transplantation. Bone Marrow Transplant. 1998;21:821-823.
- Quaas AM, Ginsburg ES. Prevention and treatment of uterine bleeding in hematologic malignancy. Eur J Obstet Gynecol Reprod Biol. 2007;134:3-8.
- Milroy C, Jones KP. Gynecologic care in hematopoietic stem cell transplant patients: a review. Obstet Gynecol Surv. 2010;65:668-679.
- Hidalgo M, Bahamondes L, Perrotti M, et al. Bleeding patterns and clinical performance of the levonorgestrel-releasing intrauterine system (Mirena) up to two years. Contraception. 2002;65:129-132.
- Meirow D, Rabinovici J, Katz D, et al. Prevention of severe menorrhagia in oncology patients with treatment-induced thrombocytopenia by luteinizing hormone-releasing hormone agonist and depo-medroxyprogesterone acetate. Cancer. 2006;107:1634-1641.
- Lhommé C, Brault P, Bourhis JH, et al. Prevention of menstruation with leuprorelin (GnRH agonist) in women undergoing myelosuppressive chemotherapy or radiochemotherapy for hematological malignancies: a pilot study. Leuk Lymphoma. 2001;42:1033-1041.
- Divasta AD, Laufer MR, Gordon CM. Bone density in adolescents treated with a GnRH agonist and add-back therapy for endometriosis. J Pediatr Adolesc Gynecol. 2007;20:293-297.
- Hornstein MD, Surrey ES, Weisberg GW, Casino LA. Leuprolide acetate depot and hormonal add-back in endometriosis: a 12-month study. Obstet Gynecol. 1998;91:16-24.
- Levens ED, Scheinberg P, DeCherney AH. Severe menorrhagia associated with thrombocytopenia. Obstet Gynecol. 2007;110:913-917.
- Aksu F, Madazli R, Budak E, et al. High-dose medroxyprogesterone acetate for the treatment of dysfunctional uterine bleeding in 24 adolescents. Aust N Z J Obstet Gynaecol. 1997;37:228-231.
- Munro MG, Mainor N, Basu R, et al. Oral medroxyprogesterone acetate and combination oral contraceptives for acute uterine bleeding: a randomized controlled trial. Obstet Gynecol. 2006;108:924-929.