Filgrastim is a granulocyte colony-stimulating factor (G-CSF) that stimulates the bone marrow to produce and activate mature neutrophils and release them into the bloodstream to enhance cytotoxicity and prevent infections.1,2 In addition, filgrastim binds G-CSF receptors on myeloid cells to stimulate the proliferation and differentiation of neutrophils, and to shorten the neutrophil maturation period.3 Filgrastim (Neupogen) and its biosimilar filgrastim-sndz (Zarxio) are approved by the US Food and Drug Administration (FDA) for several indications in pediatric patients, including to reduce the duration of neutropenia in patients with nonmyeloid malignancies who had a bone marrow transplant (including those with slow engraftment), to decrease the incidence of infection in patients receiving myelosuppressive regimens associated with a high occurrence of severe febrile neutropenia, to increase survival in patients with hematopoietic syndrome of acute radiation syndrome (filgrastim only), to reduce the duration of chronic severe neutropenia, to help with peripheral blood progenitor-cell collection and therapy, and to reduce the time to neutrophil r
Filgrastim is a granulocyte colony-stimulating factor (G-CSF) that stimulates the bone marrow to produce and activate mature neutrophils and release them into the bloodstream to enhance cytotoxicity and prevent infections.1,2 In addition, filgrastim binds G-CSF receptors on myeloid cells to stimulate the proliferation and differentiation of neutrophils, and to shorten the neutrophil maturation period.3 Filgrastim (Neupogen) and its biosimilar filgrastim-sndz (Zarxio) are approved by the US Food and Drug Administration (FDA) for several indications in pediatric patients, including to reduce the duration of neutropenia in patients with nonmyeloid malignancies who had a bone marrow transplant (including those with slow engraftment), to decrease the incidence of infection in patients receiving myelosuppressive regimens associated with a high occurrence of severe febrile neutropenia, to increase survival in patients with hematopoietic syndrome of acute radiation syndrome (filgrastim only), to reduce the duration of chronic severe neutropenia, to help with peripheral blood progenitor-cell collection and therapy, and to reduce the time to neutrophil recovery and duration of fever in patients with acute myeloid leukemia.1,4
In 2015, filgrastim-sndz was the first biosimilar to receive FDA approval in the United States based on comparable efficacy, safety, and quality to its reference drug filgrastim, from which it differs by 1 inactive component.5 The Biologics Price Competition and Innovation Act of 2009 (BPCIA) created an abbreviated FDA approval process for biosimilars.6 Through this process, the manufacturer must demonstrate that the proposed drug has the same mechanism of action, route of administration, dosage forms, and dosage strengths as the reference drug, and that it is manufactured in facilities that meet the FDA’s standards.6 The BPCIA also requires that the biosimilar only be approved for the same indications and conditions as the reference drug.6 Biosimilars can decrease drug costs while providing safety and efficacy results that are consistent with their reference drug.6
When seeking FDA approval of a biosimilar, the manufacturer must prove structural and functional similarities between the 2 drugs, pharmacokinetic and pharmacodynamic similarities, and prove the biosimilar’s efficacy and safety.7 This process is done through laboratory validation and by studying the biosimilar in a population of patients receiving the reference drug that is sensitive to detect any difference between the biosimilar and the reference drug.7 Other labeled indications that were not explicitly studied may be extrapolated from the reference drug to the biosimilar. As a result, biosimilars can be approved for use in pediatric patients without ever being studied in a pediatric population.7
The PIONEER study included patients with breast cancer who were receiving myelosuppressive chemotherapy and filgrastim-sndz.2,8,9 The patients were separated into 4 arms: patients received only the reference drug, patients received only the biosimilar, or patients switched between the biosimilar and the reference drug, with one group starting treatment with a biosimilar, and the other group starting treatment with the reference drug.2,8,9
Data from the PIONEER study demonstrated noninferiority of the biosimilar to the reference drug based on the duration of severe neutropenia and the incidence of grade 4 neutropenia (absolute neutrophil count [ANC] <500 cells/mm3), and noninferiority of the reference drug to the patients in the groups who switched between the reference drug and the biosimilar based on the incidences of febrile neutropenia, infection, febrile neutropenia–related hospitalization, and bone pain.2,8,9
Aside from the PIONEER clinical trial, few studies have compared the real-world efficacy of filgrastim and filgrastim-sndz, especially in pediatric patients. Within the United States, a single-center, retrospective study demonstrated similar efficacy and safety in adults receiving filgrastim with those receiving filgrastim-sndz for the prophylaxis of chemotherapy-induced neutropenia and neutrophil recovery after autologous hematopoietic stem-cell transplant (HSCT).10
In addition, 2 separate pharmaceutical benefit managers performed retrospective reviews of their healthcare claims data, which demonstrated similar efficacy between filgrastim and filgrastim-sndz based on the rates of febrile neutropenia.11,12 Data for real-world comparison of filgrastim biosimilars outside of the United States are scarce.13-15
A retrospective review of 29 pediatric patients in Italy demonstrated comparable efficacy for filgrastim-sndz and filgrastim in patients receiving G-CSF for peripheral blood stem-cell mobilization for autologous HSCT.14 A similar study of 118 healthy, primarily adult donors (N = 102 adults, N = 16 pediatric patients) in Mexico examined the use of 3 biosimilars to filgrastim (ie, Dextrifyl, Filatil, and Biofilgran) for stem-cell mobilization, and showed successful stem-cell collection rates without serious side effects.13
Finally, a study of 30 pediatric patients from Turkey compared filgrastim with its biosimilar Leucostim (recombinant human G-CSF) for prophylaxis of chemotherapy-induced neutropenia, showing similar efficacy outcomes between the 2 groups.15
The use of G-CSFs in pediatric patients will almost always be guided by clinical protocols. As in adults, G-CSF is recommended as the primary prophylaxis for pediatric patients at high risk for febrile neutropenia.16
In a meta-analysis, Wittman and colleagues evaluated the efficacy of prophylactic G-CSFs among children with a range of tumor types.17 Treatment with prophylactic G-CSF reduced the incidence of febrile neutropenia and the duration of severe neutropenia, hospitalization, and antibiotic use among children who received myelosuppressive chemotherapy. However, the use of prophylactic G-CSF did not decrease the incidence of documented infections.17 This 2006 study by Wittman and colleagues continues to be the benchmark for children receiving myelosuppressive chemotherapy.17 Although no differences were found in the rates of infection based on G-CSF use,17 in clinical practice, many pediatric regimens and pediatric clinical trials depend on rapid neutrophil count recovery to allow for intensive chemotherapy.
As Douglas S. Hawkins, MD, explained in a personal communication (on February 25, 2022), the Children’s Oncology Group allows for the use of biosimilars in its clinical trials, unless they are specifically prohibited in the study protocol; however, he noted that there is a lack of data showing comparable efficacy for such drugs in children.
On August 1, 2017, Children’s Hospital of Colorado converted its formulary G-CSF from filgrastim to filgrastim-sndz. With the increasing use of biosimilars in lieu of reference drugs in pediatric patients, it is important to evaluate the clinical outcomes to increase pharmacists’, patients’, and providers’ comfort and acceptance of biosimilars in this patient population.
The aim of our study was to compare the efficacy and safety of filgrastim and filgrastim-sndz in pediatric patients who receive short-acting G-CSF for the prophylaxis of chemotherapy-induced neutropenia or for post-HSCT engraftment.
Methods
This study was performed at Children’s Hospital of Colorado in Aurora, CO, a freestanding pediatric academic medical center. We retrospectively reviewed all inpatient dispenses of filgrastim between August 1, 2014, and July 31, 2017, and of filgrastim-sndz between August 1, 2017, and July 31, 2020. The data collection period was selected to align with the institution’s transition from filgrastim to filgrastim-sndz as its formulary agent in 2017, when all inpatients were automatically switched to the biosimilar agent.
All patients aged between <1 year and 22 years who received filgrastim or filgrastim-sndz for post-HSCT engraftment or for the prophylaxis of chemotherapy-induced neutropenia were included. Patients were excluded if (1) G-CSF was not started on day +0 or day +1 post-HSCT and at least 24 hours postchemotherapy, (2) they did not receive all doses of G-CSF from postchemotherapy to ANC recovery in the inpatient setting, or (3) they started G-CSF therapy before the cessation of chemotherapy (eg, fludarabine plus high-dose cytarabine and G-CSF).
Data on the patients’ baseline demographics, diagnosis and treatment, and ANC at the discontinuation of filgrastim G-CSF, as well as the total number of G-CSF doses received in each cycle of chemotherapy, and any reported adverse events were also collected.
The data for adverse events were assessed by searching the electronic medical records for the mention of common or serious adverse events associated with G-CSF (eg, progress note indicating potential side effect, cetirizine prescription with bone pain as the indication).
Based on our hospital’s current practice, engraftment for patients receiving G-CSF after HSCT is defined as ≥3 days with an ANC of >2500 cells/mm3. Per our institution’s definition, the number of days to achieve engraftment was the primary end point for patients undergoing HSCT.
In the patients receiving prophylaxis against chemotherapy-induced neutropenia, the number of days needed to achieve an ANC of 200 cells/mm3 and the number of febrile neutropenia episodes were the primary end points. The number of days to achieve an ANC of 200 cells/mm3 was chosen for this study because of our institution’s policies that allow high-risk patients, or those with treated infections, to discharge once they have nadired and have subsequently reached an ANC of 200 cells/mm3; these patients continue with G-CSF therapy in the outpatient setting until they reach full neutrophil count recovery.
Outpatients were not included in these data, because they do not consistently come to the clinic daily for G-CSF treatment or for laboratory testing; thus, the doses of G-CSF administered in the outpatient setting, and the exact date when the ANC goal was achieved, could not be confirmed by a retrospective review.
Febrile neutropenia was defined as a single oral temperature of >101°F (>38.3°C) or ≥100.4°F (≥38°C) for at least 1 hour and an ANC of <500 cells/mm3 or an ANC that is expected to continue to decrease to <500 cells/mm3.
Our calculation for statistical power indicated that a sample size of 136 patients who were receiving filgrastim or filgrastim-sndz was needed to detect equivalence between these 2 agents in terms of ANC recovery or engraftment within 2 days. Our calculation assumed a significance level of .05, a true difference of 0, and a standard deviation of 5.
Descriptive statistics were used to evaluate the baseline demographic data. The Mann–Whitney U test was used to compare the independent samples of nonparametric data (eg, number of filgrastim doses in patients undergoing autologous transplant), the 2-sample t-test was used to compare independent samples of parametric data (eg, patient age in each group), and the Fisher’s exact test was used for categorical data, such as febrile neutropenia episodes.
Results
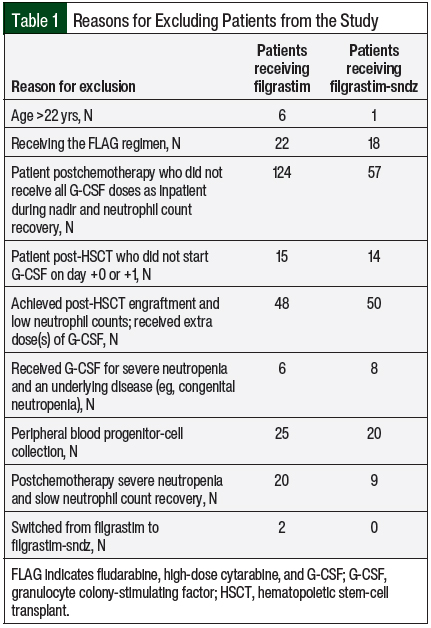
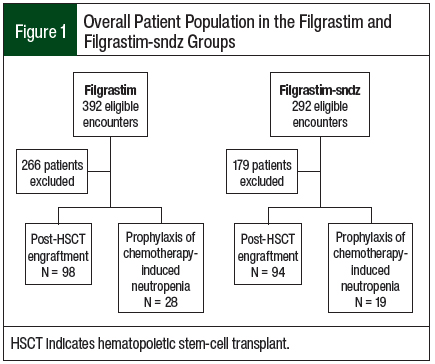
A total of 684 patient encounters were identified during the study time frame. Of those patients, 445 were excluded, leaving a total of 239 patient encounters for analysis. The reasons for study exclusion are listed in Table 1.
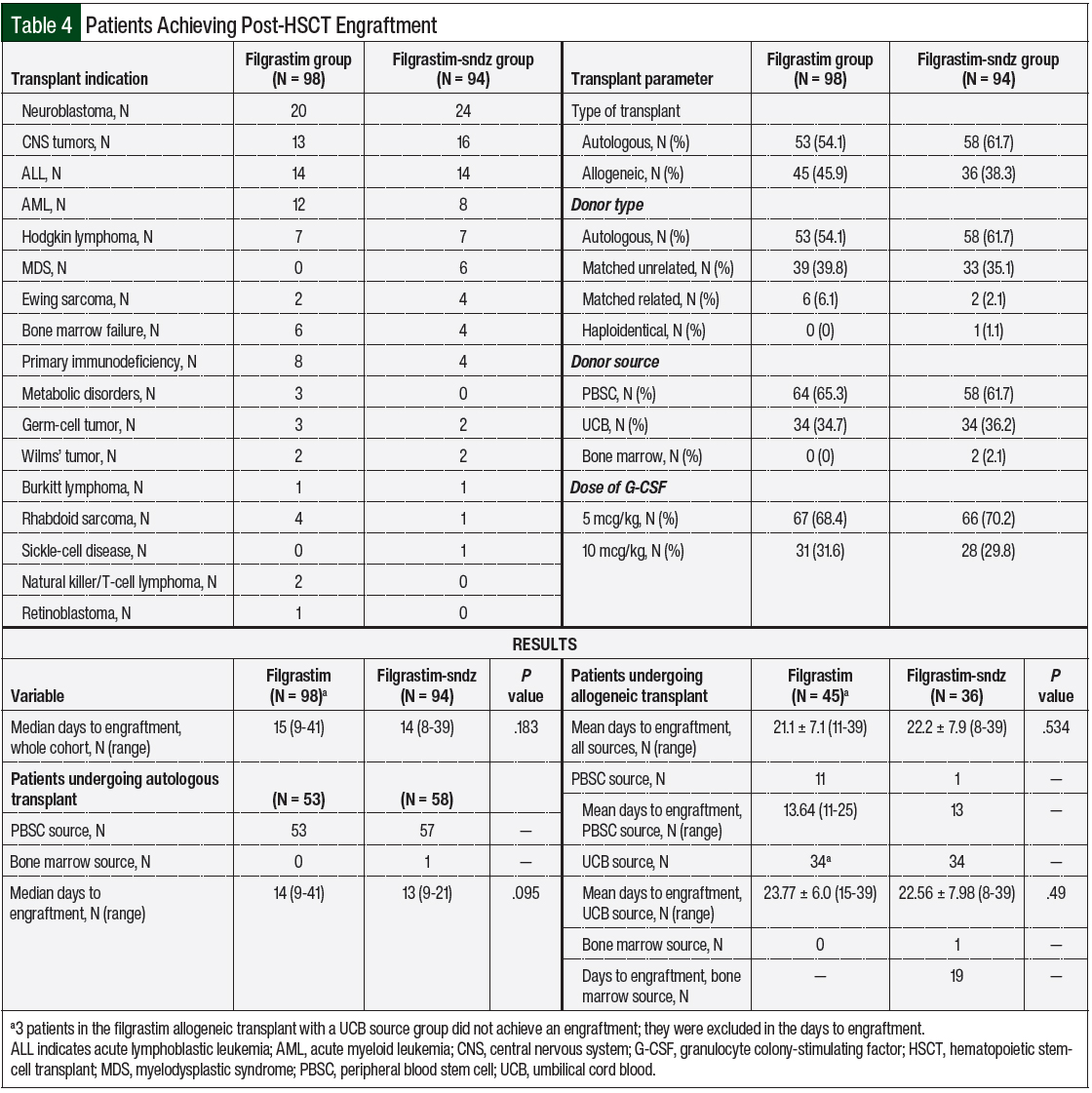
Of the patients who met the study inclusion criteria, 126 received filgrastim and 113 received filgrastim-sndz (Figure 1). In all, 28 patients in the filgrastim group and 19 patients in the filgrastim-sndz group received G-CSF for prophylaxis of chemotherapy-induced neutropenia. In addition, 98 patients who received filgrastim and 94 patients who received filgrastim-sndz achieved post-HSCT engraftment. Because of the low number of patients in each group, this study did not meet statistical power for significance.
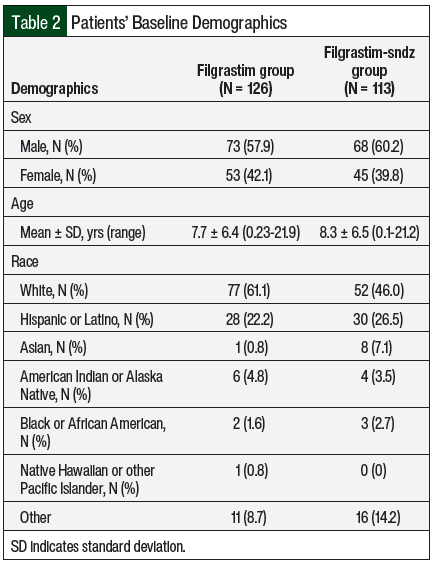
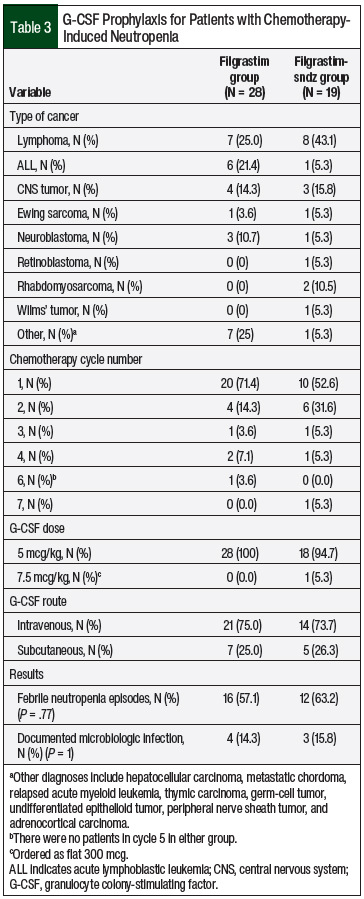
The baseline demographics were similar between the groups (Table 2). The patients receiving G-CSF for prophylaxis of chemotherapy-induced neutropenia had a similar distribution of cancer types across both treatment groups (Table 3).
The number of chemotherapy cycles during G-CSF administration was also collected to assess broadly the amount of chemotherapy previously received, which could affect bone marrow recovery. G-CSF administrations during cycles 1 through 7 were similar for the reference drug and the biosimilar drug (Table 3).
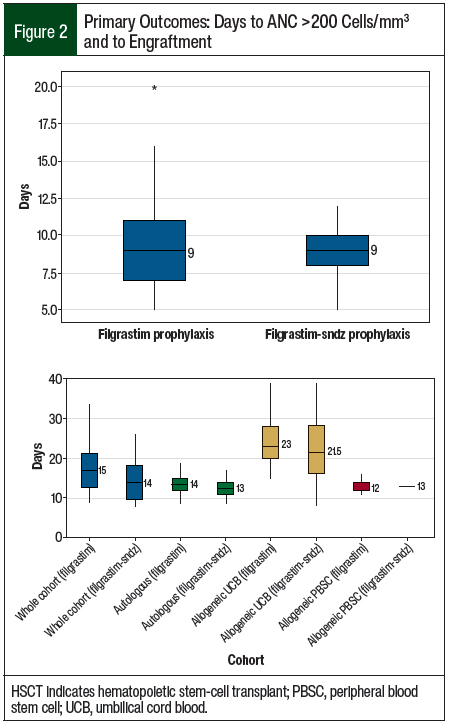
In the filgrastim group, 21 (75%) patients received G-CSF treatment intravenously compared with 14 (73.7%) patients in the filgrastim-sndz group. For the primary outcome, the median number of days to achieve an ANC of 200 cells/mm3 in each group was 9 (P = .978; Figure 2).
A total of 16 febrile neutropenia episodes occurred in the filgrastim group and 12 in the filgrastim-sndz group (P = .77; Table 3). A total of 4 patients in the filgrastim group and 3 patients in the filgrastim-sndz group had cultures positive for bacterial infection (P = 1). Similarly, the median number of doses a patient received before discharge or discontinuation of G-CSF treatment in the inpatient setting was 9 in each group (range: filgrastim, 4-26; filgrastim-sndz, 6-14; P = .907).
At the discontinuation of G-CSF treatment or at discharge from the hospital, whichever came first, the median ANC was 2125 cells/mm3 in the filgrastim group and 3360 cells/mm3 in the filgrastim-sndz group.
The patients receiving G-CSF for post-HSCT engraftment had a transplant for a variety of reasons (Table 4). Autologous transplant was more common than allogeneic transplant, representing 54.1% of patients in the filgrastim group and 61.7% in the filgrastim-sndz group. For patients receiving an allogeneic transplant, a matched unrelated donor was the most common donor type.
No significant difference was found in the days to achieve engraftment between the 2 agents (P = .183; Table 4). Patients in the filgrastim group achieved engraftment at a median of 15 days (range, 9-41 days) versus a median of 14 days in the filgrastim-sndz group (range, 8-39 days; Figure 2; P = .183). Of note, 3 patients in each group did not achieve engraftment and were excluded from the analysis. These groups were further broken down into allogeneic and autologous transplants (Table 4).
For patients undergoing an autologous transplant, the median number of days to achieve engraftment was 14 days (range, 9-41 days) in the filgrastim group and 13 days (range, 9-21 days) in the filgrastim-sndz group (P = .095). In the allogeneic transplant subset, the patients receiving filgrastim reached engraftment at a mean of 21.1 ± 7.1 days (range, 11-39 days) compared with 22.2 ± 7.9 days (range, 8-39 days) in the filgrastim-sndz group (P = .534).
Of the patients who had an autologous transplant, one had a bone marrow source and all the others had peripheral blood stem-cell source. This was expected, because our institution does not routinely use G-CSF in patients who receive stem cells from a bone marrow source. Of note, 3 patients in the filgrastim group who had an allogeneic umbilical cord blood transplant did not achieve engraftment and were excluded from the analysis.
We reviewed the electronic medical records to identify documented adverse events related to G-CSF. No serious adverse events were reported. Bone pain occurred in 23 (18.3%) patients who received filgrastim and in 28 (24.8%) patients who received filgrastim-sndz (P = .269).
Discussion
The use of G-CSFs decreases the duration and the severity of neutropenia, as well as the incidence of febrile neutropenia in patients (including children) receiving myelosuppressive chemotherapy regimens.1,4 G-CSF biosimilars have increased access to and decreased the costs of these medications; however, clinical data regarding the use of G-CSF biosimilars in children are limited.
Treatment with G-CSFs is used for a variety of indications at our institution, most often after HSCT, including for the prevention of chemotherapy-induced neutropenia in patients with low neutrophil counts after HSCT who have met the engraftment criteria (and thus their treatment with G-CSF was previously discontinued); for peripheral blood progenitor-cell collection; and for patients who have severe neutropenia because of another underlying condition (eg, congenital neutropenia, severe chronic neutropenia).
At our institution, G-CSF is typically only administered after HSCT if the patient has received an umbilical cord blood source (because of slower engraftment times) or is receiving an autologous transplant, although patients who do not meet this criterion may still receive G-CSF treatment. Of note, the use of G-CSF in patients with an allogeneic peripheral blood stem-cell source has not been typical practice over the past few years at our institution and is reflected in our data.
Finally, the use of G-CSF to prevent chemotherapy-induced neutropenia is primarily driven by the specific Children’s Oncology Group’s protocols in which patients are enrolled, and the majority of patients at our institution receive G-CSF in the outpatient setting.
The high rates of febrile neutropenia in this study were not surprising, given that our data only captured patients who remained in the hospital after receiving chemotherapy, suggesting that they might have had more medically complex disease or might have presented with more severe disease.
Since the initial PIONEER trial, few studies have compared the real-world efficacy of filgrastim with that of filgrastim-sndz, especially in pediatric patients. Zecchini and colleagues performed a single-center, retrospective review of hospitalized adults who received filgrastim or filgrastim-sndz for prophylaxis of chemotherapy-induced myelosuppression or neutrophil recovery after autologous HSCT.10 The results showed no difference in the duration of G-CSF therapy, white blood cell count, ANC at time of G-CSF discontinuation, or safety outcomes between the 2 treatment groups.10
Douglas and colleagues examined administrative claims of patients covered under Medicare Advantage Prescription Drug plan who were receiving G-CSF for prophylaxis of chemotherapy-induced febrile neutropenia.11 The results demonstrated noninferiority based on a diagnosis of febrile neutropenia for the 88 patients who were receiving filgrastim and the 101 patients who were receiving filgrastim-sndz, but the analysis of serious adverse events was not sufficient to establish noninferiority or to detect a significant difference between the 2 treatment groups.11
Similarly, Schwartzberg and colleagues examined the incidence of febrile neutropenia among patients with nonmyeloid cancer based on diagnosis codes and insurance claims.12 Their results showed that the incidence of febrile neutropenia was statistically equivalent between the patients who received filgrastim-sndz and those who received filgrastim during their first chemotherapy cycle.12
To our knowledge, there are currently no studies that examine the efficacy and safety of filgrastim versus filgrastim-sndz in children. Although a few pediatric studies have compared the reference drug filgrastim with its biosimilars in other countries,13-15 there is scant clinical evidence to support the use of filgrastim for post-HSCT engraftment or for prophylaxis of chemotherapy-induced neutropenia.
Because the majority of clinical evidence for the use of these drugs comes from the adult population, providers who treat pediatric patients may be hesitant to transition their patients to a biosimilar. Therefore, more robust data in this population are still needed.
Uncertainty regarding biosimilar use in children was demonstrated in a study by Malter and colleagues who surveyed providers who treat inflammatory bowel disease to understand the knowledge gaps in patient treatment options, specifically regarding overall treatment availability and biosimilars.18 In that survey, providers who treat pediatric patients were significantly less comfortable recommending biosimilars compared with providers who treat adult patients (61% vs 36%, respectively; P ≤.05).18
In another study, Cohen and colleagues assessed the levels of awareness, knowledge, and perceptions of biosimilars among US specialty physicians who already prescribe biologics.19 The survey identified significant knowledge gaps regarding the use of biosimilars, including a lack of understanding that the safety and immunogenicity of biosimilars are comparable to the reference biologic drugs and a lack of understanding the rationale for the extrapolation of indications.19
These concepts are even more challenging in the context of pediatric care, given that children have unique pharmacokinetics and biologic development compared with adults, but the vast majority of pediatric indications are extrapolated from clinical studies of adults.
Studies conducted outside of the United States were also limited in pediatric data, and the drugs used in these studies are not routinely used in the United States13-15; in addition, 2 of those studies only examined the use of G-CSF for stem-cell mobilization.13,14
One potential reason for the hesitancy in using biosimilars in pediatric patients is providers’ concern about the allowed molecular differences between the biosimilar and the reference drug, and the impact that these differences may have on pharmacokinetics in a pediatric patient as opposed to an adult patient. Given the hesitancy of providers who treat pediatric patients about prescribing biosimilars, more robust data on available biosimilars are still needed to alleviate ongoing and future concerns for their use.
The primary benefit of biosimilars is that they create price competition with the reference drugs, which ultimately drives down the cost of these drugs. During our study period, a total of 4153 doses of filgrastim and 3067 doses of filgrastim-sndz were dispensed. Based on the average wholesale acquisition pricing (WAC), this represents more than $1 million in acquisition cost for filgrastim and nearly $500,000 for filgrastim-sndz. Because of the 50% reduction in WAC for filgrastim-sndz compared with filgrastim, the switch to a G-CSF biosimilar saved our hospital approximately $400,000 in drug acquisition cost over a 3-year period. This represents a significant savings, which can be reinvested in other patient care initiatives.
In addition, 2 other short-acting G-CSF biosimilars are FDA approved for use in the United States: the biosimilars filgrastim-aafi (Nivestym) and tbo-filgrastim (Granix), which further provide price competition and a potential for acquisition cost-savings for various institutions.
A long-acting formulation of G-CSF and its biosimilars are also available, but these were outside of the scope of our study. However, our data could further increase the comfort level of clinicians related to and acceptance of biosimilar drugs for long-acting G-CSF.
The results of this retrospective analysis further suggest that filgrastim-sndz and filgrastim are similarly safe and efficacious treatments in children.
Limitations
This study has several limitations, including its limited sample size from a single institution that prohibited us from reading the power for statistical significance.
In addition, the patient population was heterogenous in the biosimilar and in the reference drug groups, which made a direct comparison of the 2 groups challenging.
The retrospective nature of this study is another limitation, because it is hard to know if there were undocumented data that would have been pertinent to the results. For example, adverse events were only identified if they were specifically documented in a patient’s chart in a progress note, in the problem list, or via a prescription indication (eg, “as needed for bone pain”).
Conclusion
Our findings demonstrate similar efficacy and safety when comparing directly the use of filgrastim and filgrastim-sndz in pediatric patients. The patients receiving a G-CSF biosimilar or the reference drug for prophylaxis against chemotherapy-induced neutropenia had a similar median time to ANC of 200 cells/mm3, with both groups achieving this ANC at a median of 9 days. In the group receiving G-CSF for post-HSCT engraftment, the total number of doses to achieve engraftment was similar in both groups, regardless of the type of transplant they had. In addition, bone pain was the only reported adverse event, which occurred at similar rates in both groups.
The results of this retrospective review study support the use of filgrastim-sndz in pediatric patients and have the potential to improve patient, pharmacist, and provider confidence in the use of G-CSF biosimilars in pediatric patients. The incorporation of biosimilars into pediatric patient care provides an opportunity for increased drug access and economic sustainability for healthcare systems and for patients.
Author Disclosure Statement
Dr Rush, Dr Sabus, Dr Enabulele, and Dr Kim have no conflicts of interest to report.
References
- Neupogen (filgrastim) injection, for subcutaneous or intravenous use [prescribing information]. Amgen; February 2021. www.pi.amgen.com/-/media/Project/Amgen/Repository/pi-amgen-com/neupogen/neupogen_pi_hcp_english.pdf. Accessed December 14, 2022.
- Hegg R, Mattar A, Nunes de Matos-Neto J, et al. A phase III, randomized, non-inferiority study comparing the efficacy and safety of biosimilar filgrastim versus originator filgrastim for chemotherapy-induced neutropenia in breast cancer patients. Clinics (Sao Paulo). 2016;71:586-592.
- Mehta HM, Malandra M, Corey SJ. G-CSF and GM-CSF in neutropenia. J Immunol. 2015;195:1341-1349.
- Zarxio (filgrastim-sndz) injection, for subcutaneous or intravenous use [prescribing information]. Sandoz; August 2022. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=c0d1c22b-566b-4776-bdbf-00f96dad0cae&type=display. Accessed December 14, 2022.
- Sandoz. FDA approves first biosimilar Zarxio (filgrastimsndz). March 6, 2015. www.us.sandoz.com/news/media-releases/fda-approves-first-biosimilar-zarxiotm-filgrastimsndz. Accessed December 14, 2022.
- US Food and Drug Administration. Implementation of the Biologics Price Competition and Innovation Act of 2009. February 12, 2016. www.fda.gov/drugs/guidance-compliance-regulatory-information/implementation-biologics-price-competition-and-innovation-act-2009. Accessed December 14, 2022.
- US Food and Drug Administration. Scientific considerations in demonstrating biosimilarity to a reference product: guidance for industry. April 2015. www.fda.gov/media/82647/download. Accessed December 14, 2022.
- Blackwell K, Gascon P, Krendyukov A, et al. Safety and efficacy of alternating treatment with EP2006, a filgrastim biosimilar, and reference filgrastim: a phase III, randomised, double-blind clinical study in the prevention of severe neutropenia in patients with breast cancer receiving myelosuppressive chemotherapy. Ann Oncol. 2018;29:244-249.
- Blackwell K, Semiglazov V, Krasnozhon D, et al. Comparison of EP2006, a filgrastim biosimilar, to the reference: a phase III, randomized, double-blind clinical study in the prevention of severe neutropenia in patients with breast cancer receiving myelosuppressive chemotherapy. Ann Oncol. 2015;26:1948-1953.
- Zecchini J, Yum K, Steinberg A, et al. A single-center, retrospective analysis to compare the efficacy and safety of filgrastim-sndz to filgrastim for prophylaxis of chemotherapy-induced neutropenia and for neutrophil recovery following autologous stem cell transplantation. Support Care Cancer. 2018;26:1013-1016.
- Douglas AG, Schwab P, Lane D, et al. A comparison of brand and biosimilar granulocyte-colony stimulating factors for prophylaxis of chemotherapy-induced febrile neutropenia. J Manag Care Spec Pharm. 2017;23:1221-1226.
- Schwartzberg LS, Lal LS, Balu S, et al. Clinical outcomes of treatment with filgrastim versus a filgrastim biosimilar and febrile neutropenia-associated costs among patients with nonmyeloid cancer undergoing chemotherapy. J Manag Care Spec Pharm. 2018;24:976-984.
- Cesaro S, Tridello G, Prete A, et al. Biosimilar granulocyte–colony-stimulating factor for mobilization of autologous peripheral blood stem cells in pediatric hematology-oncology patients. Transfusion. 2015;55:246-252.
- Gómez-De León A, Bugarin-Estrada E, Colunga-Pedraza PR, et al. Efficacy of three filgrastim-intended copies for hematopoietic stem cell mobilization in healthy adult and pediatric donors in Mexico. J Clin Apher. 2019;34:537-544.
- Buyukavci M, Yildirim ZK. The comparison of the efficacy and safety of original and biosimilar filgrastim in prevention of chemotherapy-induced neutropenia in children with cancer. Eurasian J Med. 2019;51:112-115.
- Smith TJ, Bohlke K, Lyman GH, et al. Recommendations for the use of WBC growth factors: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2015;33:3199-3212.
- Wittman B, Horan J, Lyman GH, et al. Prophylactic colony-stimulating factors in children receiving myelosuppressive chemotherapy: a meta-analysis of randomized controlled trials. Cancer Treat Rev. 2006;32:289-303.
- Malter L, Jain A, Cohen BL, et al. Identifying IBD providers’ knowledge gaps using a prospective web-based survey. Inflamm Bowel Dis. 2020;26:1445-1450.
- Cohen H, Beydoun D, Chien D, et al. Awareness, knowledge, and perceptions of biosimilars among specialty physicians. Adv Ther. 2016;33:2160-2172.