Immunotherapy, including programmed cell death protein-1 (PD-1) and PD ligand 1 (PD-L1) receptor antagonists, has revolutionized the treatment of many cancers. Currently, 3 PD-1 agents—nivolumab, pembrolizumab, and atezolizumab—have been approved by the US Food and Drug Administration (FDA) for the treatment of patients with lung cancer.1-3 In addition, 2 agents (avelumab and durvalumab) that inhibit PD-L1 receptors have been approved by the FDA for other types of cancer, along with 1 other agent (ipilimumab) that inhibits the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) receptor.4-6 These immune checkpoint inhibitors decrease tumor-cell signals, inhibiting T-cell activity and thereby increasing the antitumor effects of the patient’s immune system.1-6
The FDA approved nivolumab in September 2015 for weight-based dosing given every 14 days until unacceptable toxicities or disease progression.1 One year later, the FDA changed this indication to a fixed dose of intravenous (IV) 240 mg every 2 weeks.7 In May 2018, approximately 3 years after the original approval, the FDA approved the dose of IV nivolumab 480 mg given every 4 weeks.1,8 This approval of every 4-week dosing was based solely on pharmacokinetic data.9
Since the initial approval of ipilimumab for melanoma, PD-1 agents have received FDA approval for various solid and hematologic malignancies.1,2 Compared with chemotherapy, the immune checkpoint inhibitors have improved progression-free survival, overall survival, and duration of response, while decreasing acute side effects.1,10,11 However, concerns remain regarding their unique acute and chronic adverse reaction profiles. Immune-mediated adverse events have been documented in the skin, lung, gastrointestinal tract, cardiac tissues, neurologic system, and liver for patients who received treatment with immune checkpoint inhibitors.1-6 In addition, these agents may cause autoimmune endocrinopathies affecting the thyroid and the kidneys.1-6
Recently, the American Society of Clinical Oncology (ASCO) and the National Comprehensive Cancer Network (NCCN) published guidelines outlining the management of immune-related adverse drug events.12,13 The use of localized, low-dose steroids and interruption of therapy remain the hallmark of treatment for grade 1 or 2 immunotherapy-related adverse events. The presence of grade 3 or 4 immunotherapy-related adverse events warrants treatment with 1 mg/kg to 2 mg/kg daily of prednisone or its equivalent. If the adverse events are not improved within 3 days, both guidelines recommend the use of infliximab (for colitis, pneumonitis, or dermatitis) or mycophenolate mofetil (for hepatitis). It is also recommended to avoid infliximab in patients with acute hepatitis or transaminitis, because of the risk for hepatic failure with this agent.12,13
We present a case of severe transaminitis that resulted after the administration of the first dose of nivolumab 480 mg in a patient with non–small-cell lung cancer (NSCLC) adenocarcinoma. We then describe the management of transaminitis with systemic steroids and the antimetabolite mycophenolate mofetil.
Patient Case
The patient was an 81-year-old male who was initially diagnosed with muscular invasive bladder carcinoma in 2015. After resection of the tumor, he received radiation over the course of 1 year. He declined adjuvant chemotherapy and had since been observed without signs of progression. He was also diagnosed with NSCLC adenocarcinoma approximately 2 years later by fine-needle aspiration biopsy. A subsequent computed tomography (CT) scan showed metastatic disease, with invasion of bilateral lung nodules and lumbar spine. The patient also had a history of chronic obstructive pulmonary disease and atrial fibrillation. He did not have a history of tobacco use. Because of inadequate tissue biopsy, the patient opted to receive 3 cycles of carboplatin and pemetrexed for histologically confirmed adenocarcinoma before the results of tumor mutation or PD-L1 testing via liquid biopsy.
His disease progressed after 3 cycles of systemic chemotherapy with pemetrexed and carboplatin. At this point, tumor marker testing results showed no actionable tumor markers (testing was done for EGFR, ALK, and T790M mutations, as well as for PD-L1 levels). Because of the patient’s desire to maintain functional status, and NCCN Category 1 recommendation,14 it was decided to initiate nivolumab therapy. The IV dose of 480 mg every 28 days was chosen to limit the patient’s required visits to the infusion clinic. Baseline laboratory results before cycle 1 were all within normal limits. The patient received cycle 1 without any complaints of adverse reaction on the day of infusion.
Fourteen days after the first dose of nivolumab, the patient contacted the oncology pharmacist complaining of worsening shortness of breath, increased use of albuterol rescue inhaler, and weight gain of approximately 10 pounds. The patient had a CT angiography of the chest, which ruled out pulmonary embolism. Disease progression was again noted on imaging, with lung nodules and bone metastases increasing in size from approximately 1 month earlier.
Approximately 7 days later (21 days after cycle 1 of nivolumab) the patient began complaining of increased pain at the site of his spinal metastases, along with severe headaches, requiring approximately 6 acetaminophen 325-mg tablets daily. He noted significant worsening of activities of daily living and was admitted to the hospital through the emergency department for symptom management.
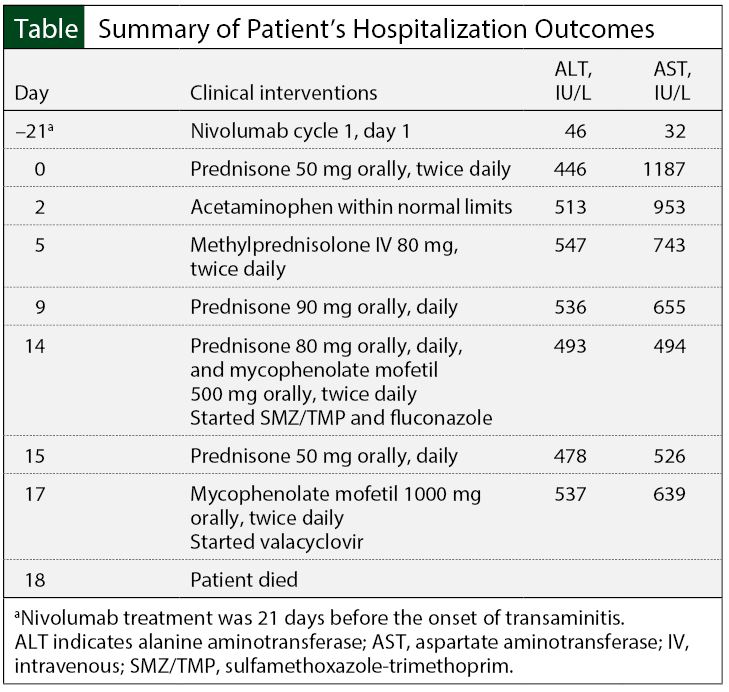
At admission, an abdominal ultrasound showed normal spleen, pancreas, bile duct, and gallbladder. Imaging showed the liver had increased echogenicity, which was consistent with steatosis or hepatocellular disease. In addition, liver function tests (LFTs) results included aspartate aminotransferase (AST) 1187 U/L (normal, 15 U/L-45 U/L), alanine aminotransferase (ALT) 446 U/L (normal, 5 U/L-40 U/L), total bilirubin 0.6 mg/dL (normal, 0.1 mg/dL-1.2 mg/dL), and alkaline phosphatase 139 U/L (normal, 40 U/L-125 U/L), consistent with grade 4 transaminitis.15
The patient started treatment with oral prednisone 50 mg twice daily (ie, 1.2 mg/kg daily). After 2 days of therapy, his AST levels decreased to 953 U/L, whereas the ALT levels increased slightly to 513 U/L. At that time, acetaminophen test results showed a level within normal limits. To increase the relative steroid potency and improve AST and ALT recovery, prednisone was discontinued, and he was given IV methylprednisolone 80 mg twice daily.
The patient’s AST and ALT levels remained elevated during 4 days of methylprednisolone therapy (AST range, 618 U/L-743 U/L; ALT range, 515 U/L-549 U/L). He continued to complain of severe myopathies, mood swings, and decreased appetite. On day 9 of his hospital stay, the patient was given oral prednisone 90 mg daily.
During the second week of his hospital stay, the patient’s AST and ALT remained in the range of 600 U/L-650 U/L and 530 U/L-576 U/L, respectively. On day 14 of hospitalization, he began treatment with oral mycophenolate mofetil 500 mg twice daily. Because of ongoing myopathies and psychiatric effects, oral prednisone was decreased to 80 mg daily. The patient also received oral sulfamethoxazole-trimethoprim 800-mg/160-mg tablets on Monday, Wednesday, and Friday, and oral fluconazole 200 mg daily for opportunistic infection prophylaxis, secondary to high-dose steroids.
Of note, the patient’s serum creatinine was stable at 0.6 mg/dL (normal, 0.7 mg/dL-1.4 mg/dL) throughout his hospital stay. His blood glucose remained between 80 mg/dL and 100 mg/dL (normal, 60 mg/dL-105 mg/dL) and his blood pressure ranged from 118/61 mm Hg to 149/75 mm Hg. Furthermore, the patient’s white blood cells remained within normal limits (4.5 × 103 cells/mcL-11 × 103 cells/mcL). He also tested negative for hepatitis B and C.
The day after the initiation of mycophenolate mofetil 500 mg twice daily, the patient’s AST decreased to 494 U/L and the ALT decreased to 493 U/L. Because of increasing myopathies, the prednisone amount was decreased to 50 mg daily. Seventeen days into hospitalization, the AST and ALT increased again, which prompted an increase in oral mycophenolate mofetil dosing, to 1000 mg twice daily. Because of the increased dose of mycophenolate mofetil, the patient was given oral valacyclovir 1000 mg 3 times daily for cytomegalovirus prophylaxis. The Table provides a summary of the patient’s hospital course.
On day 18 of hospitalization, the patient went into cardiac arrest. After resuscitation with 3 doses of epinephrine and chest compressions, he was intubated. His family then made the decision to withdraw support and declined an autopsy. The patient died secondary to cardiac arrest, 18 days after hospitalization for transaminitis.
Discussion
In a recently pooled pharmacokinetic analysis of patients receiving IV nivolumab 480 mg every 4 weeks compared with IV 3 mg/kg every 2 weeks, the researchers concluded that any differences in efficacy or safety were not expected to be clinically meaningful.9 This study poster, which stratified pharmacokinetic findings across multiple tumor types, led to the FDA approval of 480-mg every 4-week dosing.8,9 Zhao and colleagues reported that the steady-state peak and time-averaged concentrations were increased compared with the every 2-week regimen. They also reported that the predicted trough levels were lower than the every 2-week regimen.9
The extended dosing interval of 480 mg every 4 weeks is ideal for patients who have difficulty coming to the oncology clinic bimonthly, or who want to have more time between treatments. The patient described here desired to continue working while receiving treatment. The decision was made to initiate every 4-week nivolumab therapy, even though this dosing regimen has not been well-tested in a large patient population. It is possible that the increased steady-state and time-averaged concentrations may be responsible for the rapid onset and severity of transaminitis in this patient.
Although our patient was older than the patients in the CheckMate 057 clinical trial (average age, 61 years in the nivolumab arm), his other characteristics were in line with the majority of patients in most other categories.10 Similarly, his first treatment was with a platinum-based doublet (100% in the study), he was a white (91%) male (52%) with only 1 previous treatment (88%) and had metastatic disease (93%).10 In addition, the use of nivolumab in the CheckMate 057 study led to improved survival for patients who did not have an EGFR mutation and who were receiving second-line therapy. By contrast, our patient’s age suggested that he was less likely to have a clear benefit from nivolumab therapy.10
These are crucial factors to consider, because the 480-mg every 4-week pharmacokinetic data were compared with pooled data that included the CheckMate 057 trial.9 This indicated to the authors the limited need for concern of grade 3 or 4 adverse reactions (such as transaminitis) with this increased dosing in our patient, because he was well-matched with the CheckMate 057 cohort.
In the CheckMate 057 study, only 1 incident of grade 3 or 4 transaminitis occurred.10 In comparison, in all patients in clinical trials who received nivolumab every 2 weeks, the incidence of transaminitis was only 1.8%.1 The average time to onset for nivolumab-induced transaminitis has been reported as 3.3 months (range, 6 days-9 months).1 For all PD-1 immunotherapies, the time to onset of hepatitis is reported as between 6 and 14 weeks.11
An analysis by Zarrabi and Wu showed that nivolumab therapy significantly increased the risk for all-grade and high-grade AST or ALT elevation compared with the control.16 When assessing all patients with autoimmune hepatitis, interruption of therapy occurred in 1% of patients, and permanent discontinuation in 0.7% of patients.1,16 These patients required prednisone for an average of 23 days, and 2 of the patients required mycophenolate mofetil before resolution of the hepatitis.1,16
Our patient was not the first who received treatment at our institution with the every 4-week nivolumab dosing. Several other patients were transitioned from the every 2-week dosing to the every 4-week dosing, but no other patients were (by now) initiated on the every 4-weeks dosing on cycle 1. Some of these patients had worsening rash when they were transitioned to the increased dosing, but the other patients did not have their treatment held or discontinued because of immunotherapy-related adverse events.
Because of our patient’s unique travel and social situation, along with the FDA approval of the every 4-week dosing, the decision was made to initiate cycle 1 of nivolumab at the increased dose. Because of the unexpected time of onset (ie, 21 days after nivolumab administration), the severity of the transaminitis in our patient, and the lack of clinical evidence with the 480-mg dosing, his transaminitis was attributed to nivolumab therapy.
For the management of transaminitis, ASCO and NCCN recommend prednisone 1 mg/kg to 2 mg/kg daily for grade 3 or grade 4 toxicity. Without resolution in 3 days, they recommend mycophenolate mofetil 500 mg to 2000 mg daily.12,13 The ASCO guidelines recommend the use of antithymocyte globulin for hematologic and cardiac immunotherapy-related adverse events, but not for transaminitis.12
These recommendations come from multiple case reports of ipilimumab- and nivolumab-induced toxicities.17-22 Although the pathway for ipilimumab activity differs from that of nivolumab, it is reasonable to assume that the mechanism of autoimmune reactions involves T-cell activation in both instances. Because of the poor performance status of our patient, we believed that antithymocyte globulin was not appropriate for the management of his transaminitis.
In the case described by Ahmed and colleagues, the investigators chose to use antithymocyte globulin in addition to prednisone and mycophenolate mofetil to speed up the recovery of LFTs.17 This patient recovered within 2 weeks, and doses of prednisone and mycophenolate mofetil were gradually tapered off.17 In the case described by Tanaka and colleagues, the patient developed intolerance to methylprednisolone,18 similar to our patient. This led the providers to use mycophenolate mofetil and low-dose steroids for the management of ipilimumab-induced hepatitis. Ultimately, this patient’s LFTs recovered in approximately 2 weeks; however, she died as a result of progression of her metastatic melanoma.18
Mycophenolate mofetil is used for the treatment of autoimmune reactions, because it has pronounced effects on circulating T-lymphocytes.23 The active metabolite directly inhibits inosine monophosphate dehydrogenase, leading to decreased guanosine synthesis, an essential step in the proliferation of T-cells. With suppressed T-cells, one major concern is opportunistic infections.23 Infectious prophylaxis is not addressed in the Tanaka case, but it is mentioned in the various cases utilizing triple therapy for immunosuppression (ie, steroids, mycophenolate mofetil, and antithymocyte globulin).17-22
In addition, the NCCN and ASCO guidelines do not have specific recommendations for opportunistic infection prophylaxis.12,13 Our patient was receiving high doses of prednisone for an extended duration, along with mycophenolate mofetil, which we believed warranted opportunistic infection prophylaxis. Without absolute guidance, we chose sulfamethoxazole-trimethoprim and fluconazole for prophylaxis against Pneumocystis jirovecii fungal species while he was receiving steroids and lower doses of mycophenolate mofetil. It was determined that cytomegalovirus prophylaxis would not be initiated until the mycophenolate mofetil dose was increased to 2000 mg daily, following the protocol for solid organ transplant.24,25
Conclusion
In this patient case, the clinical team attributed the patient’s transaminitis to nivolumab. Although this causality could not be proved, we attributed the rapid onset to the every 4-week dosing. Further safety analyses are needed to evaluate the tolerability of this increased dose. Our institution requires tolerability of the every 2-week dose before the administration of every 4-week dosing, because of safety concerns.
This case highlights the importance of rapid and aggressive initiation of immunosuppressive therapy, particularly mycophenolate mofetil. Although liver dysfunction was not the cause of death in this patient, more rapid initiation of mycophenolate mofetil 1000-mg twice-daily dosing might have led to a quicker resolution of transaminitis. It is important to initiate prophylaxis for opportunistic infections in all patients whose immunotherapy-related adverse events are treated with steroids in combination with mycophenolate mofetil.
Author Disclosure Statement
Dr Stankowicz, Dr Banaszynski, and Mr Crawford have no conflicts of interest to report.
References
- Opdivo (nivolumab) injection, for intravenous use [prescribing information]. Princeton, NJ: Bristol-Myers Squibb Company; September 2019.
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Whitehouse Station, NJ: Merck & Co; January 2020.
- Tecentriq (atezolizumab) injection, for intravenous use [prescribing information]. South San Francisco, CA: Genentech; December 2019.
- Bavencio (avelumab) injection, for intravenous use [prescribing information]. Rockland, MA: EMD Serono/Pfizer; May 2019.
- Imfinzi (durvalumab) injection, for intravenous use [prescribing information]. Wilmington, DE: AstraZeneca Pharmaceuticals; August 2019.
- Yervoy (ipilimumab) injection, for intravenous use [prescribing information]. Princeton, NJ: Bristol-Myers Squibb Company; September 2019.
- US Food and Drug Administration. Modification of the dosage regimen for nivolumab. September 15, 2016. www.fda.gov/drugs/resources-information-approved-drugs/modification-dosage-regimen-nivolumab. Accessed February 28, 2020.
- Bristol-Myers Squibb’s Opdivo (nivolumab) now the first and only FDA-approved PD-1 inhibitor to offer every four-week dosing [news release]. Princeton, NJ: Bristol-Myers Squibb; March 6, 2018. https://news.bms.com/press-Release/corporatefinancial-news/bristol-m yers-squibbs-opdivo-niv olumab-now-first-and-only-fda-. Accessed March 2, 2020.
- Zhao X, Ivaturi V, Gopalakrishnan M, et al. A model-based exposure–response (E–R) assessment of a nivolumab (NIVO) 4-weekly (Q4W) dosing schedule across multiple tumor types. Poster presented at the American Association for Cancer Research annual meeting; April 1-5, 2017; Washingt on, DC.
- Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced non-squamous non–small-cell lung cancer. N Engl J Med. 2015;373:1627-1639.
- Puzanov I, Diab A, Abdallah K, et al; for the Society for Immunotherapy of Cancer Toxicity Management Working Group. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5:95. doi.org/10.1186/s40425-017-0300-z.
- Brahmer JR, Lacchetti C, Schneider BJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714-1768.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Management of Immunotherapy-Related Toxicities. Version 1.2020. December 16, 2019. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed February 28, 2020.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Non-Small Cell Lung Cancer. Version 2.2020. December 23, 2019. www.nccn.org/professionals/physician_gls/pdf/nscl.pdf. Accessed February 28, 2020.
- Department of Health & Human Services, National Institutes of Health, National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CT CAE_v5_Quick_Reference_ 8.5x11.pdf. Accessed October 23, 2018.
- Zarrabi K, Wu S. Increased risk of liver toxicity secondary to nivolumab therapy in the treatment of cancer. J Clin Oncol. 2017;35(Suppl 15). Abstract e14559.
- Ahmed T, Pandey R, Shah B, Black J. Resolution of ipilimumab induced severe hepatotoxicity with triple immunosuppressants therapy. BMJ Case Rep. 2015;2015. pii:bcr2014208102. doi.org/10.1136/bcr-2014-208102.
- Tanaka R, Fujisawa Y, Sae I, et al. Severe hepatitis arising from ipilimumab administration, following melanoma treatment with nivolumab. Jpn J Clin Oncol. 2017;47:175-178.
- Chmiel KD, Suan D, Liddle C, et al. Resolution of severe ipilimumab-induced hepatitis after antithymocyte globulin therapy. J Clin Oncol. 2011;29:e237-e240.
- Ange N, McDowall LM, Yun J, et al. Antithymocyte globulin for the treatment of checkpoint inhibitor mediated myocarditis. Intern Med J. 2017;47(Suppl 5):32. Abstract P93.
- Spänkuch I, Gassenmaier M, Tampouri I, et al. Severe hepatitis under combined immunotherapy: resolution under corticosteroids plus anti-thymocyte immunoglobulins. Eur J Cancer. 2017;81:203-205. Erratum in: Eur J Cancer. 2017;87:221.
- Tay RY, Blackley E, McLean C, et al. Successful use of equine anti-thymocyte globulin (ATGAM) for fulminant myocarditis secondary to nivolumab therapy. Br J Cancer. 2017;117:921-924.
- CellCept (mycophenolate mofetil) [prescribing information]. South San Francisco, CA: Genentech USA; December 2019.
- Hodson EM, Jones CA, Webster AC, et al. Antiviral medications to prevent cytomegalovirus disease and early death in recipients of solid-organ transplants: a systematic review of randomised controlled trials. Lancet. 2005;365:2105-2115.
- Kalil AC, Levitsky J, Lyden E, et al. Meta-analysis: the efficacy of strategies to prevent organ disease by cytomegalovirus in solid organ transplant recipients. Ann Intern Med. 2005;143:870-880.