Hematopoietic stem-cell transplant (HSCT) is a potentially lifesaving therapy for patients with hematologic malignancies. In patients undergoing allogeneic transplant, the optimal dosing of chemotherapy in conditioning regimens is important for reducing tumor burden and for providing immune modulation to prevent graft rejection.1-3 The ongoing obesity epidemic, however, has made the optimal dosing of chemotherapy a challenge in patients undergoing HSCT.
Efforts have been made to standardize chemotherapy dosing in obese patients to minimize adverse events and achieve therapeutic effects. In 2012, the American Society of Clinical Oncology recommended using full weight-based dosing of chemotherapy for obese adults, but did not address patients undergoing HSCT.1 In 2014, the American Society for Blood and Marrow Transplantation, now the American Society for Transplantation and Cellular Therapy (ASTCT), Practice Guideline Committee published a comprehensive review of the literature to provide guidance on conditioning chemotherapy dosing in obese patients undergoing HSCT.2
The ASTCT’s review, although comprehensive, primarily consists of evidence from retrospective reports and small case series, many of which lack adequate descriptions of patients’ height, weight, and body mass index (BMI).2 In addition, variable use of patient-specific pharmacokinetics for targeted chemotherapeutic dosing contributed to the heterogenous results. Because of these limitations in the supporting literature, the committee was unable to draw Level I or II evidence-based conclusions about optimal chemotherapy dosing of HSCT conditioning regimens in obese patients. Nevertheless, the panel’s consensus for busulfan- and fludarabine-containing regimens was to use total body weight when dosing based on body surface area (BSA).2
Chemotherapy underdosing is only one of many possible contributors to relapse. Disease type and remission status at the time of transplant are key prognostic factors when estimating overall survival.4,5 The Disease Risk Index (DRI) is a validated tool that stratifies patients undergoing HSCT into 4 risk groups—low, intermediate, high, and very high—based on the specific hematologic disease type and status.4,5
A large study that validated the DRI showed that patients who were categorized as high or very-high risk per the DRI classification had increased mortality rates (with 2-year OS of 34% and 24%, respectively) compared with patients who were stratified by the DRI as low or intermediate risk (with 2-year overall survival of 64% and 51%, respectively).5 The differences in overall survival among the 4 groups have previously been shown to result primarily from the incidence of disease relapse after transplant.4 The DRI is an important tool to use when assessing retrospectively outcomes in heterogeneous patient populations who have undergone HSCT.
In contrast to the ASTCT guidance,2 the standard practice at the University of Iowa Hospitals and Clinics has been to use a 25% adjusted body weight calculation to determine an adjusted BSA for dosing all adults receiving an ablative busulfan plus fludarabine regimen who are undergoing HSCT, regardless of obesity status.
The primary objective of our study was to determine if obese patients who were receiving ablative busulfan plus fludarabine conditioning regimen dosed by an adjusted BSA have earlier time-to-relapse than nonobese patients. The secondary outcomes included overall survival at 6 months and at 1 year.
Methods
This single-center, Institutional Review Board–approved retrospective study included all adults receiving a myeloablative conditioning regimen containing busulfan and fludarabine who underwent HSCT between September 2011 and June 2018 at the University of Iowa Hospitals and Clinics. An ablative busulfan and fludarabine regimen was defined as busulfan 130 mg/m2 daily for 4 days and fludarabine 40 mg/m2 daily for 4 days.
Dosing was based on a 25% adjusted body weight, calculated for obese and nonobese patients as 0.25 multiplied by actual body weight minus ideal body weight plus ideal body weight.
Patients were excluded if they received a reduced-intensity busulfan and fludarabine regimen, a dose reduction of either agent, busulfan dosed by mg/kg, or busulfan dosed based on patient-specific pharmacokinetics.
The patients were stratified into 2 groups—obese, defined as a BMI ≥30 kg/m2, and nonobese, BMI <30 kg/m2—to assess the differences in disease relapse and overall survival at 6 months and at 1 year after transplant.
The DRI category was identified for each patient, using our center’s electronic medical record (EMR). For patients who did not already have a DRI category assigned, the EMR was used to extract information regarding the patient’s disease type and status at the time of the transplant. A DRI category was then assigned from the 1 of 4 risk categories (ie, low, intermediate, high, very high) according to the validated risk scheme described earlier.4
Armand and colleagues suggest that a 3-group breakdown that combines very high risk and high risk into 1 group is preferable for a cohort size of ≤300 patients, whereas the 4-group breakdown is more appropriate for cohorts of >300 patients.5 Because we had a sample size of <300 patients, the 3-group risk breakdown (ie, low, intermediate, high or very high) was selected for our analysis.
Summary statistics of the demographic variables stratified by indicators of obesity and dose differences were calculated to test for any cohort imbalances. Continuous and categorical variables were compared using the Wilcoxon rank-sum test and the Pearson’s chi-square test, respectively. The time-to-event data were analyzed using Cox proportional hazards modeling, with censoring for patients who did not have relapsed disease or death by the end of 6 or 12 months or before another study-ending event.
The patient’s risk level (by DRI category), related transplant type, and their obesity status were considered when determining the optimal analytical models. For each outcome, the final analytical model was determined by selecting the covariate set that produced the smallest Akaike information criterion. The significance of the model covariates was assessed at alpha = 0.05.
Results
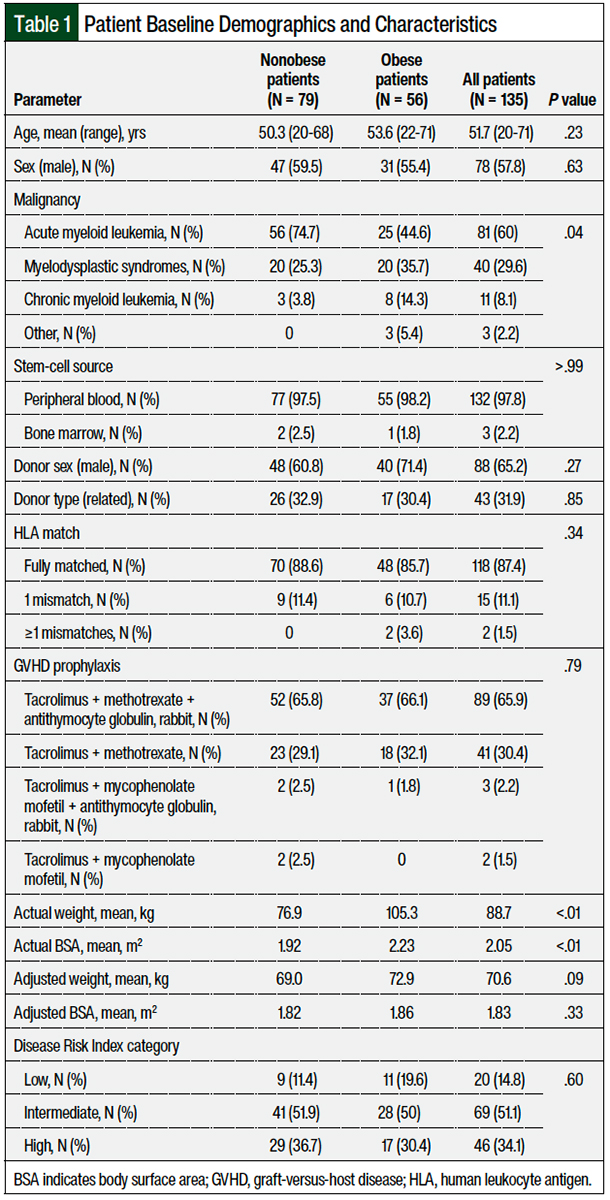
A total of 135 patients were included in the study, with 79 patients in the nonobese group and 56 patients in the obese group. Except for the anticipated difference between the average actual body weight and the actual BSA, patient demographics were similar between the 2 groups (Table 1). Despite the significant difference in actual body weight between the 2 groups, after adjusting the actual body weight and BSA using the 25% adjusted body weight calculation, the mean adjusted body weight and BSA were not significantly different. The distribution of patients in each DRI-based risk category was similar between the groups, with the majority of patients placed in the intermediate-risk category (51.1%), followed by the high-risk category (34.1%), and low-risk category (14.8%).
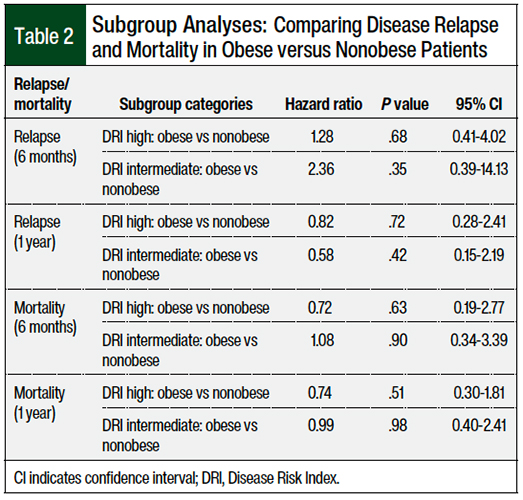
The patients were stratified based on their obesity status and DRI category to determine their risk for disease relapse or death at 6 months and at 1 year (Table 2). Overall, no significant difference was seen between the obese and nonobese groups.
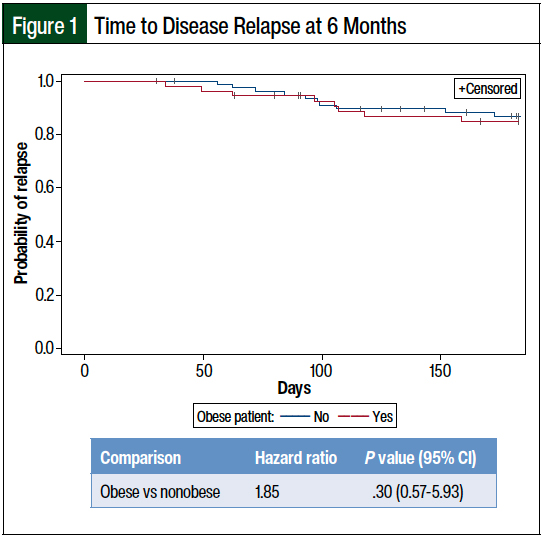
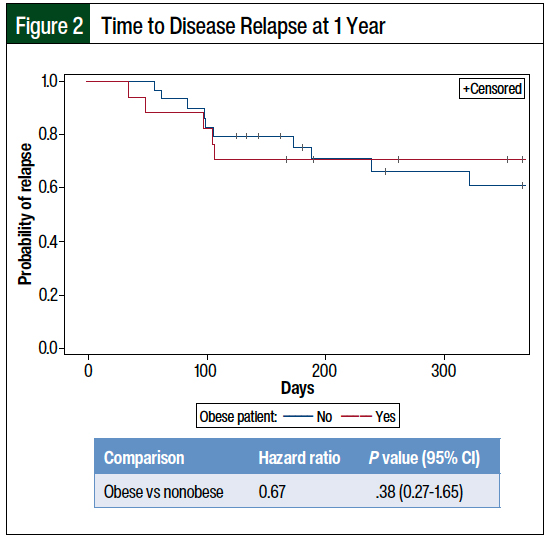
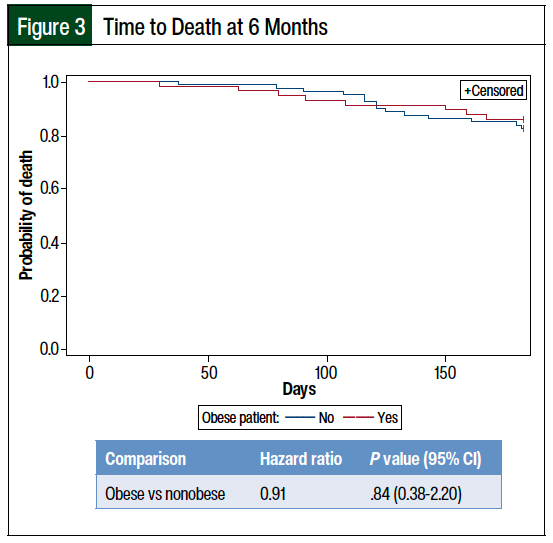
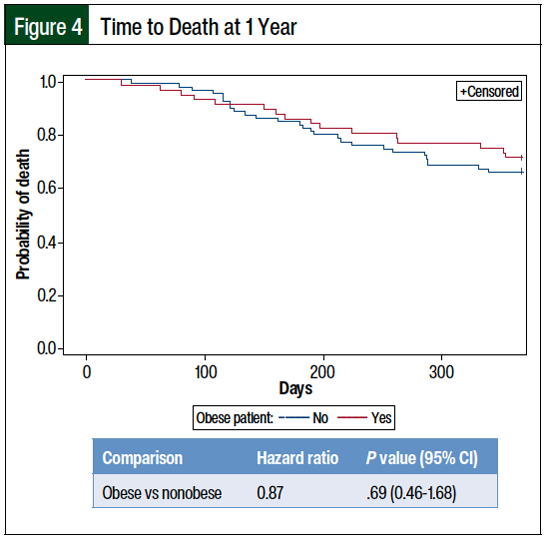
Specifically, obese patients were not found to have an increased risk for disease relapse at 6 months compared with nonobese patients (hazard ratio [HR], 1.85; P = .30). Similarly, obese patients were not more likely to have disease relapse at 1 year (HR, 0.67; P = .38), die at 6 months (HR, 0.91; P = .84), or die at 1 year (HR, 0.87; P = .69) than nonobese patients (Figures 1-4).
To evaluate whether the patient’s obesity status affected outcomes more in certain subgroups, the rates of disease relapse and mortality were further assessed for obese versus nonobese patients within specific DRI categories (low, intermediate, and high). Nevertheless, still we found no significant difference between the obese and nonobese patients in terms of disease recurrence or death within any DRI category (Table 2).
Discussion
Determining the optimal chemotherapy dosing remains controversial in relation to obese patients, including those undergoing allogeneic HSCT, and clinical practice varies widely. The ASTCT’s 2014 position statement attempted to consolidate the available literature to provide a standardized approach for dosing chemotherapy preparative regimens in this patient population, yet it was unable to provide Level I evidence-based recommendations.2
Of the 18 clinical trials included in that position statement that assessed busulfan dosing, 8 studies did not report weight parameters, and 3 studies only evaluated pediatric populations.2 Two fludarabine-based studies were referenced, both of which used total body weight, but they, too, did not report patient weight parameters.2 The major limitations of this position statement include the retrospective nature of the studies considered, their small sample sizes, a lack of patient weight reporting, and inconsistencies in dosing strategies, pharmacokinetic monitoring, and median follow-up time.2
Surveys and discussion boards continue to illustrate the inconsistencies in dosing of conditioning chemotherapy regimen strategies across institutions and organizations. In 2015, the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation (EBMT) surveyed 56 centers to assess the current practice of conditioning chemotherapy dose adjustment in obese patients.3 Among the 56 centers, 45 (80%) stated that they adjust chemotherapy dosing for obese patients. Of these centers, 16 used actual body weight, 16 used a 25% adjusted body weight calculation, 10 used ideal body weight, 4 used a 40% adjusted body weight calculation, and other methods were used in 2 centers.3
In addition, among centers that used dose adjustments, 44% capped the BSA at 2 m2 for chemotherapy, based on BSA dosing.3 A total of 36 centers used the same approach for dose adjustments in myeloablative and reduced-intensity regimens or nonmyeloablative regimens, and 8 centers used smaller dose reductions for reduced-intensity chemotherapy or nonmyeloablative regimens.3 For dosing busulfan, 7 (13%) centers routinely monitored pharmacokinetics, 11 (20%) used ideal body weight, 17 (30%) used actual body weight, and 18 (32%) used adjusted body weight (15 used a 25% adjusted body weight calculation, and 3 used a 40% adjusted body weight calculation).3
Of note, many factors influence a patient’s risk for disease relapse after having allogeneic HSCT, aside from suboptimal chemotherapy dosing. The inclusion of DRI category status, which is a strong prognosticator of disease relapse rates and overall survival,5 was an attempt to control for these other variables in our study.
The majority (51.1%) of patients in our study were categorized as DRI-based intermediate risk based on their disease type and stage, which is similar to the expected 55% reported in the literature.4 Similarly, all other risk groups were representative of the typical makeup of the transplant population reported in the literature.4
Our findings did not show a significant difference in disease relapse or mortality between the obese and nonobese patients, even when the DRI category was taken into consideration. However, it is possible that differences in disease relapse or mortality exist at the 2- or 4-year time point, as is demonstrated in the literature, but we chose shorter minimum follow-up time in our study to allow for inclusion of more patients in our data set.4,5
When our retrospective review was performed, the University of Iowa Hospitals and Clinics used a 25% adjusted body weight calculation to dose all patients who underwent allogeneic HSCT, including those with a BMI of <30 kg/m2. This was in contrast to the 2014 ASTCT consensus statement, which recommends a variety of modalities for dosing individual chemotherapy agents, including the use of 25% and 40% adjusted body weight calculations as well as total body weight.2
Using this mixed approach in a real-world setting can be difficult to implement, because of the complexity of incorporating multiple dosing rules into chemotherapy plans within our EMR and the potential for human error. Because of the use of adjusted body weight and the potential for underdosing of these myeloablative regimens, we theorized that obese patients would have a higher rate of disease relapse and/or mortality after undergoing HSCT.
The typical practice for pharmacists at the University of Iowa Hospitals and Clinics consists of notifying the provider if a patient’s most recent actual body weight differs from the dosing weight used to calculate a chemotherapy regimen by 10% compared with previous cycles. Although this 10% weight gain or loss may not always result in a large dose disparity, due diligence is warranted, given the narrow therapeutic index of chemotherapy.
Limitations
This study has several limitations. The retrospective nature of this study and the standard practice of using a 25% adjusted body weight calculation for all patients undergoing allogeneic HSCT at the University of Iowa Hospitals and Clinics meant that the nonobese patient population served as our comparator group rather than a population dosed based on actual body weight.
Because our institution was using a 25% adjusted body weight calculation on all patients undergoing allogeneic HSCT, we inadvertently identified 11 (13.9%) patients in the nonobese group who were administered chemotherapy doses of more than 10% less than the dose they would have received had actual BSA been used for the dose calculation. This large dose discrepancy in our comparator group (ie, the nonobese group) presents a notable limitation in our original study design, potentially masking a possible difference in disease relapse or mortality between the 2 groups had the nonobese patients been dosed according to actual BSA, as the ASTCT guidance suggests.2
As a consequence of this dose discrepancy finding, and to minimize the risk for underdosing nonobese patients undergoing allogeneic HSCT, the University of Iowa Hospitals and Clinics has since modified its practice to use actual body weight and BSA for conditioning chemotherapy dose calculations, using a 25% adjusted body weight calculation and adjusted BSA only when the body weight exceeds 125% of the ideal body weight.
In addition, the retrospective chart review nature of this study limited the sample size and the overall patient diversity. However, our sample provides an accurate representation of the current obesity epidemic in the United States. Of the 135 patients who were included in this study, 56 (41.5%) were obese, which is consistent with the reported obesity rate in Iowa (≥35%) and in the United States (42.4% in 2017-2018).6,7
Given the wide individual variability in pharmacokinetics, the narrow therapeutic window, and the lack of standardization across institutions, busulfan dosing remains a common topic of research.
Our institution does not routinely perform pharmacokinetic monitoring for busulfan dosing in adults, which is another limitation of this study. Whether because of facility limitations or an uncertainty of definitive benefit in the era of intravenous busulfan, forgoing busulfan therapeutic drug monitoring is not unusual in clinical practice, though.8 According to a large recent survey by the EBMT, only 17 (16%) of 106 responding transplant centers use therapeutic drug monitoring for busulfan dose adjustments in myeloablative conditioning regimens. Among those centers, there was marked variability in how therapeutic drug monitoring was performed, the timing of measurements, the units used, and the method of calculating the area under the curve.8
Furthermore, the use of busulfan therapeutic drug monitoring has been inconsistent with busulfan plus fludarabine regimens, because of variations in the total busulfan dose, fludarabine dose, use of antithymocyte globulin, and the type of postgrafting immunosuppression.9 The ASTCT’s 2016 evidence-based review on personalized busulfan dosing indicates certain scenarios when busulfan therapeutic drug monitoring should be used, but it notes that intravenous busulfan can be dosed without therapeutic drug monitoring when combined with a cumulative fludarabine dose of 120 mg/m2.9
Further complicating the busulfan therapeutic drug monitoring paradigm, multiple busulfan plasma exposure units are used clinically and are reported in publications, which can lead to misinterpretations and errors when capturing data and implementing protocols.10 The lack of standardization in dosing busulfan, the variability in reporting, and the lack of inclusion in multicenter databases have led to the production of an international busulfan plasma exposure units harmonization project.10 Moreover, the use of therapeutic drug monitoring for busulfan dosing would not obviate the need to define the most appropriate weight for determining the first dose of the conditioning regimen, as has been investigated recently by several centers.11-13
Another limitation to our study is that we did not evaluate nonrelapse mortality (ie, death without recurrent or progressive disease), and we did not measure outcomes beyond 1 year.
Finally, we analyzed the dosing of an allogeneic HSCT conditioning regimen, but the degree of graft-versus-tumor effect in this setting also significantly influences patient outcomes. In the future, it may be more prudent to compare outcomes in obese and nonobese patients who are undergoing autologous HSCT, a scenario in which the optimal dosing of myeloablative chemotherapy is a more definitive determinant of disease relapse than with allogeneic HSCT.
Conclusion
Our findings did not show differences between nonobese and obese patients in the time to disease relapse or the rate of overall survival at 6 months and at 1 year, regardless of the DRI category status at the time of transplant. Of note, most recommendations regarding dosing chemotherapy in obese patients come from subgroup analyses of randomized clinical trials and observational studies comparing dosing strategies that use actual body weight with those that use adjusted body weight dosing. Because of the inconsistent chemotherapy dosing based on body weight (ie, actual, adjusted, or ideal weight) used in clinical practice, larger multicenter studies are needed to elucidate further any potential differences in outcomes between obese and nonobese patients who are undergoing stem-cell transplant.
Author Disclosure Statement
The authors have no conflicts of interest to report.
References
- Griggs JJ, Mangu PB, Anderson H, et al. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30:1553-1561.
- Bubalo J, Carpenter PA, Majhail N, et al. Conditioning chemotherapy dose adjustment in obese patients: a review and position statement by the American Society for Blood and Marrow Transplantation Practice Guideline Committee. Biol Blood Marrow Transplant. 2014;20:600-616.
- Shem-Tov N, Labopin M, Moukhtari L, et al. Chemotherapy dose adjustment for obese patients undergoing hematopoietic stem cell transplantation: a survey on behalf of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Oncologist. 2015;20:50-55.
- Armand P, Gibson CJ, Cutler C, et al. A disease risk index for patients undergoing allogeneic stem cell transplantation. Blood. 2012;120:905-913.
- Armand P, Kim HT, Logan BR, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123:3664-3671.
- Centers for Disease Control and Prevention. Adult Obesity Prevalence Maps. www.cdc.gov/obesity/data/prevalence-maps.html. Accessed September 22, 2018.
- Hales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of obesity and severe obesity among adults: United States, 2017–2018. National Center for Health Statistics Data Brief, No 360. Hyattsville, MD: National Center for Health Statistics; February 2020. www.cdc.gov/nchs/data/databriefs/db360-h.pdf. Accessed September 22, 2018.
- Ruutu T, van der Werf S, van Biezen A, et al. Use of busulfan in conditioning for allogeneic hematopoietic stem cell transplantation in adults: a survey by the Transplant Complications Working Party of the EBMT. Bone Marrow Transplant. 2019;54:2013-2019.
- Palmer J, McCune JS, Perales MA, et al. Personalizing busulfan-based conditioning: considerations from the American Society for Blood and Marrow Transplantation Practice Guidelines Committee. Biol Blood Marrow Transplant. 2016;22:1915-1925.
- McCune JS, Quinones CM, Ritchie J, et al. Harmonization of busulfan plasma exposure unit (BPEU): a community-initiated consensus statement. Biol Blood Marrow Transplant. 2019;25:1890-1897.
- Griffin SP, Wheeler SE, Wiggins LE, et al. Pharmacokinetic and clinical outcomes when ideal body weight is used to dose busulfan in obese hematopoietic stem cell transplant recipients. Bone Marrow Transplant. 2019;54:218-225.
- Clemmons AB, Evans S, DeRemer DL, Awan FT. Busulfan dosing (Q6 or Q24) with adjusted or actual body weight, does it matter? J Oncol Pharm Pract. 2015;21:425-432.
- Shalhoub SD, Taylor BJ, Piedra KM, et al. Busulfan pharmacokinetics using 25% adjusted body weight in obese stem cell transplant patients. Biol Blood Marrow Transplant. 2020;26(3 suppl):Abstract 612.