Bladder cancer is the sixth most common type of cancer in the United States.1 In 2021, the number of new cases of bladder cancer is estimated to be 83,730, which is 4.4% of all projected new cancer cases in the United States. The number of deaths from bladder cancer in 2021 is estimated at 17,200, which is 2.8% of all projected cancer-related deaths. The current number of new cases for men and women per 100,000 is 19.7, and the lifetime risk for bladder cancer is 2.4%.1
The incidence of bladder cancer is higher in men (34.2 per 100,000) than in women (8.5 per 100,000).1 The overall 5-year survival rate for bladder cancer is 77.1%, with a 6.4% survival rate for patients with distant disease. The median age of diagnosis of bladder cancer is 73 years.1 Some of the most frequently reported risks for bladder cancer include male sex, cigarette smoking, chronic infections and irritations of the bladder, occupational exposure to aromatic amines, and diabetes mellitus.2-4
Bladder cancer is staged based on the TNM (tumor, node, metastases) staging system.5 The treatment recommendations of the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology (NCCN Guidelines) for Bladder Cancer are grouped into non–muscle-invasive bladder cancer and muscle-invasive bladder cancer, including metastatic bladder cancer.6
Important factors included in determining the best treatment option for bladder cancer include biopsy findings of transurethral resection of bladder tumor specimens, histology, tumor grade, and the extent of invasion. More than 90% of urothelial tumors originate in the urinary bladder, 8% in the renal pelvis, and 2% in the ureter and urethra.6
In general, early-stage (ie, cTa and cT1) bladder cancer is managed with observation after transurethral resection for low-risk tumors, and with transurethral resection and adjuvant Bacillus Calmette-Guérin immunotherapy, or the administration of intravesical chemotherapy, including gemcitabine or mitomycin, for high-risk tumors.6
Cystectomy is also an option for high-risk non–muscle-invasive bladder cancer. Stages II and IIIA bladder cancer are generally managed by the administration of neoadjuvant cisplatin-based chemotherapy, followed by radical cystectomy and pelvic lymphadenectomy, or partial cystectomy and neoadjuvant chemotherapy for stage II bladder cancer.6
Concurrent chemoradiation and transurethral resection of bladder tumor can be used for bladder preservation. Cystectomy-ineligible patients should receive chemoradiation or radiation. Neoadjuvant chemotherapy is preferred to adjuvant chemotherapy for bladder cancer. Preferred neoadjuvant and adjuvant chemotherapy regimens include dose-dense methotrexate, vinblastine, doxorubicin, cisplatin (DDMVAC) or gemcitabine plus cisplatin. Cisplatin should not be substituted with carboplatin in the neoadjuvant setting. Stage IIIB bladder cancer is managed with chemotherapy for downstaging or with concurrent chemoradiation.6
Metastatic bladder cancer may be managed by additional lines of systemic chemotherapy after response to systemic therapy. Clinical trial enrollment is especially encouraged by the NCCN for patients in the second-line setting and subsequent lines of therapy.6
The first-line chemotherapy regimens used for the management of metastatic disease include gemcitabine plus cisplatin, or DDMVAC. These regimens are followed by maintenance therapy with avelumab in a patient with no disease progression after first-line platinum-containing chemotherapy. For cisplatin-ineligible patients, gemcitabine plus carboplatin followed by maintenance therapy with avelumab is a preferred option.6 For decades, the pharmacologic management of bladder cancer included immunotherapy (ie, Bacillus Calmette-Guérin) and chemotherapy agents alone. This situation has changed recently.
Genomic Alterations and Molecular Testing
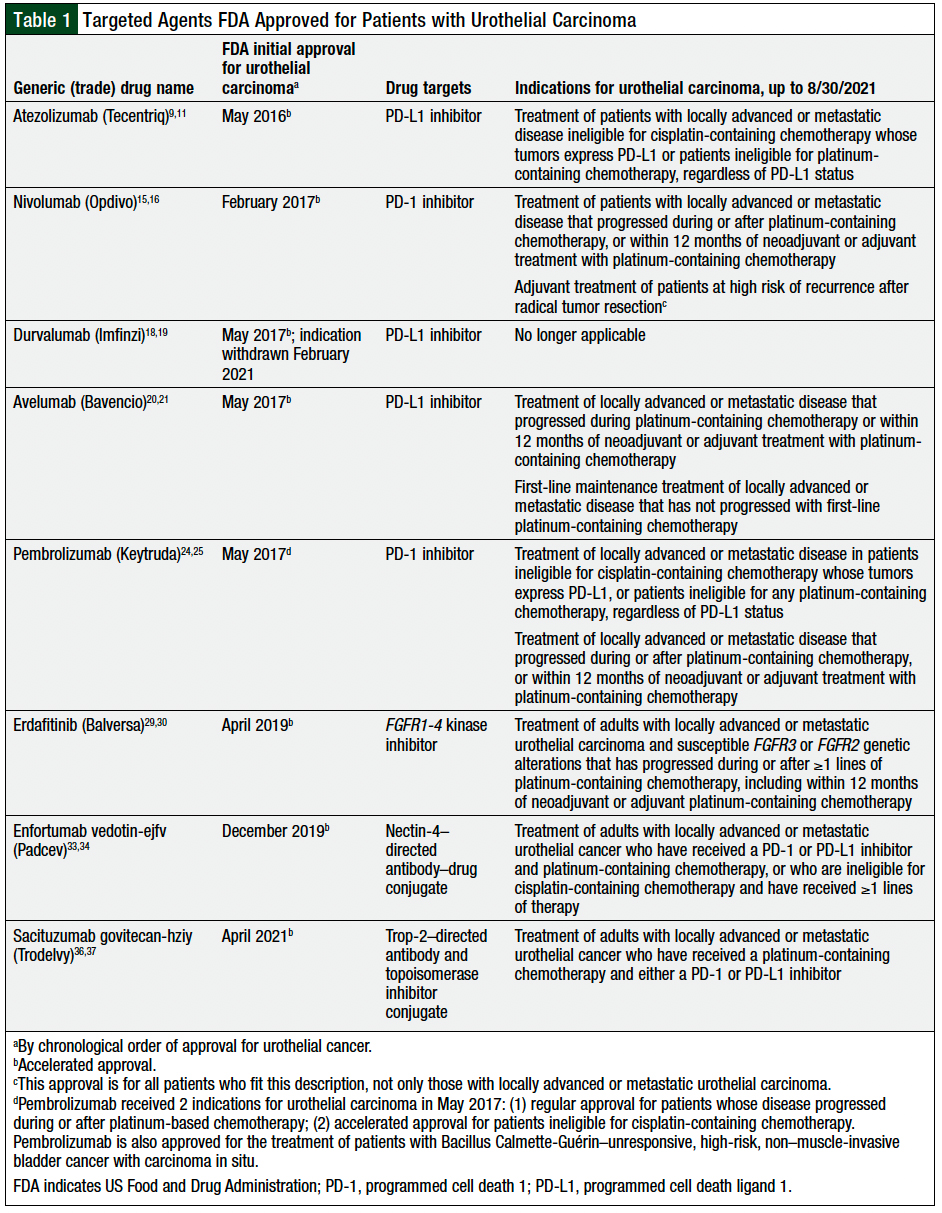
According to the Cancer Genome Atlas Research Network, bladder cancer is one of the cancer types with the highest number of genetic mutations and other genomic alterations.7,8 In the past several years, 8 different targeted therapies have been approved by the US Food and Drug Administration (FDA) for the treatment of advanced or metastatic urothelial carcinoma. These drugs target the following genomic alterations and biomarkers, including programmed cell death 1 (PD-1) or PD ligand 1 (PD-L1) expression, fibroblast growth factor receptors 1-4 (FGFR1-4) alterations, Nectin-4 expression, or the trophoblast cell-surface antigen 2 (Trop-2) biomarker.
These 8 drugs approved by the FDA include the PD-L1 inhibitors atezolizumab, avelumab, and durvalumab (which was withdrawn in February 2021); the PD-1 inhibitors nivolumab and pembrolizumab; the Nectin-4 antibody–drug conjugate enfortumab vedotin-ejfv; the FGFR1-4 inhibitor erdafitinib; and the Trop-2 antibody and drug conjugate sacituzumab govitecan-hziy (Table 1).
Therefore, the NCCN guidelines currently recommend molecular testing for all patients diagnosed with stage IVA and stage IVB urothelial cancer. In addition, the NCCN recommends that molecular testing should be considered for patients with stage IIIB bladder cancer.6 In fact, molecular testing should be performed at the diagnosis of advanced-stage disease, which helps decision-making and prevents delays in starting therapy. Furthermore, according to the NCCN, all molecular testing should be performed in CLIA (Clinical Laboratory Improvement Amendments)-certified laboratories, because these molecular tests are complex.6
The goal of this review article is to describe the current FDA-approved targeted therapies for the treatment of urothelial carcinoma, the most common type of bladder cancer. Studies that led to the approval of these drugs are summarized, and the place in therapy of each agent in the current management of urothelial carcinoma is also provided.
Novel Agents for Advanced or Metastatic Urothelial Carcinoma of Bladder
In May 2016, atezolizumab was the first novel drug to be approved by the FDA for urothelial carcinoma.9 Atezolizumab is indicated for the treatment of advanced or metastatic urothelial carcinoma that progressed during or after platinum-containing chemotherapy or within 12 months of adjuvant or neoadjuvant chemotherapy in patients who are ineligible for cisplatin-based therapy and whose tumor expresses PD-L1 or who are ineligible for any platinum-based chemotherapy, regardless of PD-L1 expression.9,10
Atezolizumab is a monoclonal antibody that blocks interaction with PD-1 and B7.1 receptors, by binding to PD-L1. This binding causes PD-L1–mediated and PD-1–mediated inhibition to be released, which activates the antitumor response. This process does not induce antibody-dependent cellular cytotoxicity.11
Atezolizumab was approved by the FDA based on a single-arm, phase 2 clinical trial that included 310 patients with locally advanced or metastatic urothelial carcinoma.12 Atezolizumab was administered every 3 weeks at a dose of 1200 mg. Dose reductions were not allowed, but dose interruptions were allowed. Enrolled patients had locally advanced or metastatic urothelial carcinoma that was inoperable and that progressed after platinum-containing chemotherapy. The study’s primary end point was to assess the objective response rate (ORR). The patients were also assessed for PD-L1 expression.12 In all, 26% of patients with PD-L1 expression ≥5% had objective response, 18% of patients with PD-L1 expression ≥1% had a response, and 15% of all patients had an objective response. The median follow-up was 11.7 months, and ongoing responses were seen in 84% of patients.12
A total of 97% of the patients reported all-cause, any-grade adverse events, and 55% had a grade 3 or 4 event. Among the most common all-grade side effects were fatigue (30%), nausea (14%), decreased appetite (12%), pruritus (10%), pyrexia (9%), and diarrhea (8%). The most common (incidence, 2%) grade 3 or 4 side effect was fatigue, and other side effects, such as anemia, increased aspartate aminotransferase, pneumonitis, hypotension, hypertension, and colitis, had an incidence rate of 1%.12
Although some objective responses were identified across all patients, a better response was observed in patients with PD-L1 expression (26%) than in patients without PD-L1 expression (9.5%). At the same time, the FDA also approved the Ventana PD-L1 (SP142) test for the detection of PD-L1 protein expression in tumors to determine which patients would most likely respond to treatment with atezolizumab.9
Atezolizumab was studied in the second-line setting for advanced or metastatic urothelial carcinoma. In this study, the primary end point was overall survival (OS) and atezolizumab was compared with chemotherapy. The median OS was not significantly different between the groups, at 11.1 months with atezolizumab versus 10.6 months with chemotherapy (P = .41).13
Atezolizumab was also studied in the first-line setting with and without platinum-based chemotherapy versus placebo plus platinum-based chemotherapy. The coprimary end points were OS and progression-free survival (PFS). The results of this study suggest that atezolizumab could be used in the first-line setting, in combination with chemotherapy, in patients who are eligible for platinum-based chemotherapy or as monotherapy in patients who are ineligible for chemotherapy.14
Nivolumab was granted accelerated approval by the FDA in February 2017 for patients with locally advanced or metastatic urothelial carcinoma whose disease progressed during or after platinum-based chemotherapy or within 12 months of receiving adjuvant or neoadjuvant treatment with platinum-based chemotherapy.15 The recommended dose of nivolumab for urothelial cancer is 240 mg intravenously every 2 weeks, or 480 mg intravenously every 4 weeks.16
Nivolumab is a human immunoglobulin (Ig)G4 monoclonal antibody that blocks interaction with PD-L1 and PD-L2 by binding to the PD-1 receptor. This causes PD-1 pathway–mediated inhibition to be released, which includes antitumor response.16
The approval of nivolumab was based on a multicenter, single-arm study of 270 patients (aged ≥18 years) with metastatic or nonresectable locally advanced urothelial carcinoma whose disease progressed or relapsed after at least 1 platinum regimen. Patients were placed in 3 different groups based on PD-1 expression of ≥5%, ≥1%, and <1%.17
Nivolumab was administered at a dose of 3 mg/kg every 2 weeks until disease progression, unacceptable toxicity, clinical worsening, or other protocol-particular reasons. The final analysis of the study included 265 patients, because there was not enough follow-up data on 5 of the patients. The study’s primary end point was ORR.17
The confirmed ORR was 19.6% in the total study population, including 28.4% in patients with PD-1 expression ≥5%, 23.8% in patients with PD-1 expression ≥1%, and 16.1% in patients with PD-1 expression <1%. Among the most common all-grade side effects were fatigue (17%), pruritus (9%), diarrhea (9%), decreased appetite (8%), hypothyroidism (8%), and nausea (<8%). Three (1%) patients died during the study from treatment-related adverse effects.17
In May 2017, the FDA granted durvalumab accelerated approval for the treatment of locally advanced or metastatic urothelial carcinoma in patients whose disease progressed during or after platinum-containing chemotherapy or within 12 months of receiving adjuvant or neoadjuvant platinum-based chemotherapy.18 Durvalumab is a human IgG1 kappa monoclonal antibody that blocks PD-1/PD-L1 and PD-L1/CD80 (B7.1) by binding to PD-L1.18
In February 2021, the FDA withdrew durvalumab’s indication for urothelial carcinoma, based on the results of the DANUBE study, which did not meet its primary end points, and the manufacturer voluntarily withdrew this indication in the United States.19
Avelumab was granted accelerated approval by the FDA in May 2017 for patients with locally advanced or metastatic urothelial carcinoma whose disease progressed during or after platinum-containing chemotherapy or within 12 months of adjuvant or neoadjuvant treatment with platinum-based chemotherapy.20,21
In June 2020, avelumab was approved as maintenance treatment of locally advanced or metastatic urothelial carcinoma that has not progressed during first-line platinum-containing chemotherapy.22
The dose of avelumab for urothelial carcinoma is 800 mg every 2 weeks. Avelumab is a human IgG1 lambda monoclonal antibody and a PD-L1–blocking antibody that blocks interaction between PD-L1 and its receptors, PD-1 and B7.1. This binding causes the release of the inhibitory effects of PD-L1 on the immune reaction, which reestablishes the immune responses that also include antitumor responses.21
Avelumab was approved based on the single-arm, multicenter study of 249 patients with locally advanced or metastatic urothelial carcinoma. Enrolled patients had disease progression after at least 1 previous platinum-based therapy. Patients were also allowed to participate if they were ineligible for cisplatin and might have received previous treatment in the perioperative setting, including platinum-naïve patients. Patients were not preselected based on PD-L1 expression. The study’s primary end point was confirmed best overall response.23
In the group of patients who received previous platinum-based therapy and who were followed for 6 months, 6% of patients had a complete response and 11% had a partial response.23 The most common grade 1 or 2 side effects were infusion-related reactions (29%), fatigue (14%), rash (14%), diarrhea (6%), asthenia (4%), decreased appetite (4%), hypothyroidism (4%), and pneumonitis (2%). The most common grade 3 or 4 side effects were fatigue (2%), asthenia (1%), and hypophosphatemia (1%).23 Patients should be premedicated with antihistamine and acetaminophen before the administration of the first 4 doses of avelumab.21
In May 2017, pembrolizumab received 2 FDA indications for the treatment of urothelial carcinoma.24 Pembrolizumab received regular approval for the treatment of locally advanced or metastatic urothelial carcinoma in patients whose disease progressed during or after platinum-based chemotherapy or within 12 months of adjuvant or neoadjuvant platinum-based chemotherapy.24,25 In addition, the FDA granted pembrolizumab accelerated approval for the treatment of locally advanced or metastatic urothelial carcinoma in patients who are ineligible for cisplatin-containing chemotherapy.24,25
Pembrolizumab is a humanized monoclonal IgG4 kappa antibody and a PD-1–blocking antibody that binds to PD-1 or PD-L1. This binding prevents interaction between PD-L1 and PD-L2, which results in the release of PD-1 pathway–mediated inhibition of immune response, as well as antitumor response.25 The recommended dose of pembrolizumab is 200 mg every 3 weeks or 400 mg every 6 weeks via intravenous infusion.25
The study that led to pembrolizumab’s regular approval was a multicenter, randomized clinical trial of patients with locally advanced or metastatic urothelial carcinoma whose disease progressed during or after platinum-containing chemotherapy. The patients were randomized in a 1:1 ratio to pembrolizumab or to investigator’s choice of chemotherapy, which included paclitaxel, docetaxel, or vinflunine.24,26
Patients who received pembrolizumab had significant improvements in OS and ORR. The median OS was 10.3 months for the pembrolizumab group versus 7.4 months for the chemotherapy group (P = .002). The pembrolizumab ORR was 21.1% versus 11.4% in the chemotherapy group (P = .001).24,26
The accelerated approval of pembrolizumab was based on a single-arm, open-label, phase 2 clinical trial of 370 patients with locally advanced or metastatic urothelial carcinoma; the patients were ineligible for cisplatin-based chemotherapy and did not receive previous systemic chemotherapy. The PD-L1 expression cutoff was set at 10%, which was unique for pembrolizumab.24,25,27
Pembrolizumab is indicated for the treatment of urothelial carcinoma in cisplatin-ineligible patients with a PD-L1 combined positive score of ≥10% or for patients who are ineligible for any platinum-based chemotherapy, regardless of PD-L1 status.24,25,27,28
In the group of patients with a PD-L1 combined positive score of ≥10%, 39% of patients had achieved objective response. In the group with a combined positive score of 1% to 10%, 20% of patients had achieved objective response, and in the group of PD-L1 combined positive score <1%, 11% of patients had achieved objective response. The most common all-grade adverse effects of pembrolizumab include fatigue (17%), pruritus (<15%), rash (<10%), diarrhea (8%), and nausea (<8%).27
Based on this trial, pembrolizumab received an indication for the treatment of locally advanced or metastatic urothelial carcinoma in patients ineligible for cisplatin-based therapy and with PD-L1 tumor expression or patients who are ineligible for any platinum-based therapy, regardless of PD-L1 expression.10,25,27
In April 2019, the FDA granted erdafitinib accelerated approval for the treatment of locally advanced or metastatic urothelial carcinoma in patients who have susceptible FGFR3 or FGFR2 mutations whose disease progressed during or within 12 months after platinum-based chemotherapy, including adjuvant or neoadjuvant platinum-based chemotherapy.29,30
Erdafitinib is a tyrosine kinase inhibitor of FGFR1-4, and it binds to RET, CSF1R, PDGFRA, PDGFRB, FLT4, KIT, and VEGFR2.30 Erdafitinib inhibits FGFR phosphorylation, which causes decreased cell viability in cells expressing FGFR genetic alterations.30 FGFR3 mutations have been linked to the carcinogenesis of bladder cancer.31
Erdafitinib was studied in a phase 2, open-label study that included patients with locally advanced and unresectable or metastatic urothelial carcinoma and at least 1 FGFR3 mutation or FGFR2 or FGFR3 fusion.32 All enrolled patients had disease that progressed during or after at least 1 course of chemotherapy or within 12 months after adjuvant or neoadjuvant chemotherapy. Patients who have not received chemotherapy were allowed into the study if they were ineligible to receive cisplatin. Patients who previously received immunotherapy were also allowed to enroll.32
The study had 2 different dosing regimens of erdafitinib, including 10 mg daily given 7 days on and 7 days off, and 6 mg given daily continuously. A planned interim analysis was performed, and the protocol was amended to have only 1 study group that received erdafitinib daily. For this single group, the starting dose was changed to 8 mg daily, with a dose increase to 9 mg daily in patients with serum phosphorus levels <5.5 mg/dL and no adverse effects attributable to the treatment. A total of 99 patients received this regimen, of whom 41 were able to tolerate dose escalation to 9 mg daily.32
The study’s primary end point was the confirmed response rate and the secondary end points were PFS, duration of response, OS, safety, response rate in biomarker-specific subgroups, and pharmacokinetics. A total of 3% of the patients had a confirmed complete response, and 37% of the patients had a confirmed partial response. In the group of 22 patients who previously received immunotherapy, 59% had a confirmed response.32
In all, 13 patients discontinued treatment as a result of side effects, including detachment of the retinal pigment epithelium, hand–foot syndrome, dry mouth, and skin or nail side effects. The most common grade ≥3 adverse events were hyponatremia (11%), stomatitis (10%), asthenia (7%), nail dystrophy (6%), and urinary tract infections (5%). The rate of grade ≥3 adverse events was similar in patients who received the 9-mg dose and those who received the 8-mg dose. Dose reduction was needed in 55 patients, and the most common adverse reactions that required dose reductions were stomatitis (N = 16) and hyperphosphatemia (N = 9).32
Hyperphosphatemia, which was the most common side effect, was managed by either dose interruption (23%), dose reduction (9%), or the administration of phosphate binders, if needed.33 Dry skin was managed by administration of topical ointments, and nail side effects were managed with topical nail strengthener and with antibiotics or silver nitrate in more severe cases.34 Along with erdafitinib, the FDA approved a companion diagnostic test, the Therascreen FGFR RGQ RT-PCR Kit, to identify patients who are candidates for erdafitinib therapy.29
In December 2019, the FDA granted accelerated approval to enfortumab vedotin for the treatment of locally advanced or metastatic urothelial cancer in adults who previously received platinum-based chemotherapy and a PD-1 or PD-L1 inhibitor in the adjuvant or neoadjuvant setting.33,34 Enfortumab vedotin is a Nectin-4–directed antibody and microtubule inhibitor conjugate. Enfortumab vedotin binds to cells that express Nectin-4, which causes the release of a small-molecule microtubule-disrupting agent (monomethyl auristatin E) in the target cells and leads to the disruption of microtubules and to cell-cycle arrest and apoptosis.34
The approved dose of enfortumab vedotin is 1.25 mg/kg (maximum dose, 125 mg) intravenously on days 1, 8, and 15 of a 28-day cycle until disease progression or unacceptable toxicity.33,34
The multicenter, phase 2 study of enfortumab vedotin that led to its approval included 125 patients with locally advanced or metastatic urothelial carcinoma who previously received platinum-based chemotherapy and anti–PD-1 or anti–PD-L1 therapy.35 The study’s primary end point was ORR. The overall confirmed ORR was 44%, and 12% of those patients had complete response. Objective responses occurred in both patient groups. The patients who had a previous response to a PD-1 or PD-L1 inhibitor had an ORR of 56%, and the patients who did not have previous response to PD-1 or PD-L1 inhibition had an ORR of 41%.35
The most common any-grade side effects were fatigue (50%), alopecia (49%), decreased appetite (44%), dysgeusia (40%), peripheral sensory neuropathy (40%), nausea (39%), and diarrhea (32%).35 The most common grade ≥3 side effects were fatigue (6%), maculopapular rash (4%), peripheral sensory neuropathy (2%), nausea (2%), diarrhea (2%), decreased appetite (1%), and decreased weight (1%).35
In that study, peripheral neuropathy was mostly low grade and manageable, and the symptoms mostly self-resolved or were grade 1 at follow-up.35 Rash was also predominantly low-grade and was manageable with the administration of topical corticosteroids, oral antihistamines, and, in some cases, with the administration of systemic corticosteroids or dose reductions or delays of enfortumab vedotin. Hyperglycemia was mostly resolved or improved at follow-up.35
In April 2021, the FDA granted accelerated approval to sacituzumab govitecan for the treatment of locally advanced or metastatic urothelial cancer in patients who previously received platinum-based chemotherapy and a PD-1 or PD-L1 inhibitor.36 Sacituzumab govitecan is a Trop-2–directed antibody and topoisomerase inhibitor conjugate.37
The approval of sacituzumab govitecan was based on results from the TROPHY clinical trial, a single-arm, multicenter study of patients with locally advanced or metastatic urothelial cancer who had received platinum-based chemotherapy and either a PD-1 or PD-L1 inhibitor. All the patients received intravenous sacituzumab govitecan 10 mg/kg on days 1 and 8 of a 21-day treatment cycle. The median duration of response in this study was 7.2 months, and the ORR was 27.7%, with complete response rate of 5.4% and partial response rate of 22.3%.36 Some of the most common adverse effects of sacituzumab govitecan include nausea, diarrhea, constipation, fatigue, and neutropenia.37
Selecting the Right Therapy
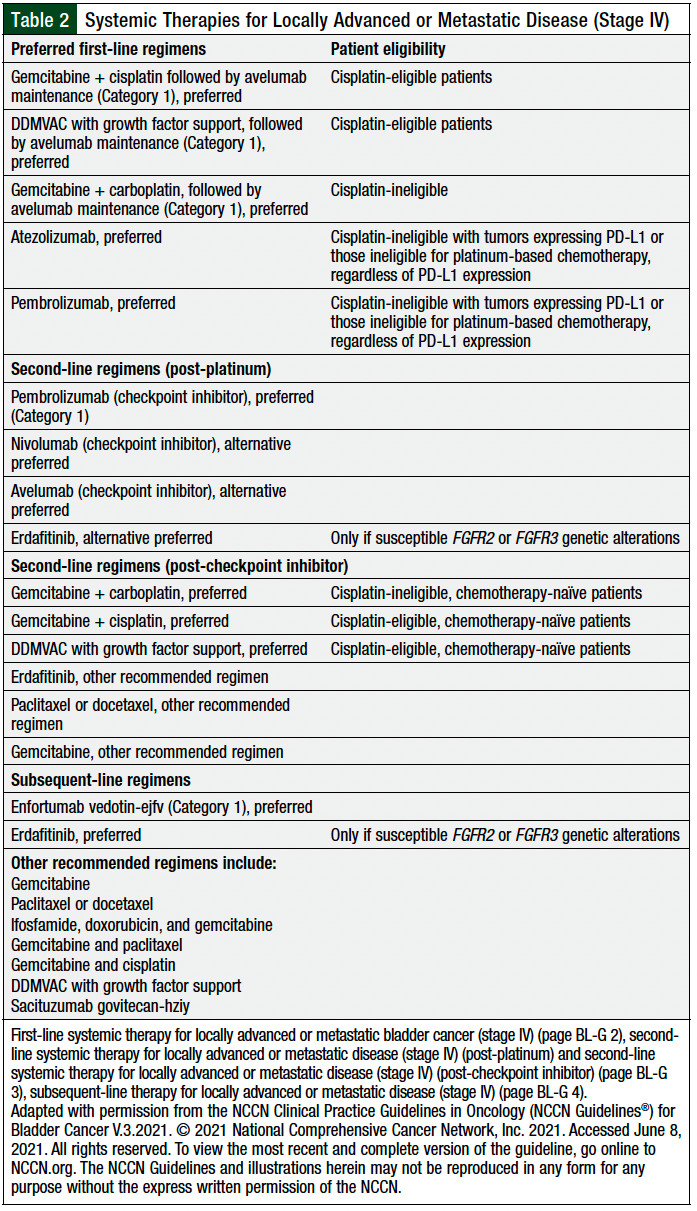
The NCCN guidelines for bladder cancer provide guidance on the current place in therapy of immune checkpoint inhibitors and other targeted agents for the treatment of patients with urothelial cancer, to help in the selection of the best therapy for each patient (Table 2).6
Avelumab is listed as preferred Category 1 recommendation for maintenance therapy after gemcitabine plus cisplatin preferred Category 1 regimen for cisplatin-eligible patients or after DDMVAC therapy that is supported with growth factors. Avelumab is recommended in this setting only for patients who have no disease progression with first-line platinum-containing chemotherapy (Table 2).6
Atezolizumab and pembrolizumab are the preferred choices as first-line systemic therapy for locally advanced or metastatic disease (stage IV) in patients whose tumors express PD-L1 or in patients who are ineligible for cisplatin therapy, regardless of PD-L1 expression.6
Pembrolizumab is the preferred (Category 1) agent, and nivolumab, avelumab, and erdafitinib are listed as alternative preferred regimens as second-line systemic therapy for locally advanced or metastatic disease (stage IV) after receiving platinum therapy (Table 2).6
Erdafitinib is recommended only for patients with susceptible FGFR2 or FGFR3 genetic alterations. Erdafitinib is also listed as a recommended second-line systemic therapy for locally advanced or metastatic bladder cancer after a checkpoint inhibitor, in addition to other recommended regimens, including paclitaxel or docetaxel or gemcitabine alone or in combination with paclitaxel in certain patients (Table 2).6
Enfortumab vedotin and erdafitinib are preferred regimens (Category 1), and sacituzumab govitecan is listed as one of the other recommended agents for subsequent-line systemic therapy for locally advanced or metastatic bladder cancer (Table 2).6
Novel Agents for Early-Stage Urothelial Cancer
In January 2020, the FDA approved pembrolizumab for the treatment of patients unresponsive to Bacillus Calmette-Guérin and who have high-risk, non–muscle-invasive bladder cancer with carcinoma in situ, with or without papillary tumors, and who are ineligible for or who have elected not to have cystectomy.38
Pembrolizumab was granted priority review for this indication, based on the KEYNOTE-057 trial’s interim analysis (the study’s estimated completion date is July 2023).38,39
The KEYNOTE-057 trial is a multicenter, single-arm study that included 148 patients, 96 of whom had carcinoma in situ, with or without papillary tumors, and were nonresponsive to Bacillus Calmette-Guérin immunotherapy.38 The study’s efficacy outcomes included complete response and duration of response. Of the 96 patients, 41% had a complete response, and 46% of them had a response that lasted ≥12 months.38
Conclusion
Bladder cancer is associated with a significant number of genetic mutations and other genomic alterations. These biomarkers are potential targets for novel therapies for urothelial carcinoma. Based on the discovery of genomic targets and mutations in bladder cancer, several novel agents targeting PD-1/PD-L1, FGFR1-4, Nectin-4, and Trop-2 have been approved by the FDA for advanced or metastatic urothelial carcinoma of bladder in the past several years. This is the beginning of precision medicine for the treatment of patients with this type of malignancy. As more genomic mutations and other biomarker targets specific to urothelial cancer are discovered, new targeted therapies will likely be approved for the treatment of patients with this type of cancer.
Author Disclosure Statement
Dr Stevich-Heemer has no conflicts of interest to report.
References
- National Cancer Institute SEER program. Cancer stat facts: bladder cancer. https://seer.cancer.gov/statfacts/html/urinb.html. Accessed June 14, 2021.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7-30.
- DeGeorge KC, Holt HR, Hodges SC. Bladder cancer: diagnosis and treatment. Am Fam Physician. 2017;96:507-514.
- Xu Y, Huo R, Chen X, Yu X. Diabetes mellitus and the risk of bladder cancer: a PRISMA-compliant meta-analysis of cohort studies. Medicine (Baltimore). 2017;96:e8588.
- Amin MB, Edge S, Greene F, et al, eds. AJCC Cancer Staging Manual. 8th ed. New York: Springer International Publishing; 2017.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Bladder Cancer. Version 3.2021. Accessed May 4, 2021.
- Alexandrov LB, Nik-Zainal S, Wedge DC, et al. Signatures of mutational processes in human cancer. Nature. 2013;500:415-421. Erratum in: Nature. 2013;502:258.
- Cancer Genome Atlas Research Network. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature. 2014;507:315-322.
- US Food and Drug Administration. FDA approves new, targeted treatment for bladder cancer. May 18, 2016. www.fda.gov/news-events/press-announcements/fda-approves-new-targeted-treatment-bladder-cancer. Accessed January 28, 2020.
- US Food and Drug Administration. FDA limits the use of Tecentriq and Keytruda for some urothelial cancer patients. July 5, 2018. www.fda.gov/drugs/resources-information-approved-drugs/fda-limits-use-tecentriq-and-keytruda-some-urothelial-cancer-patients. Accessed January 28, 2020.
- Tecentriq (atezolizumab) injection, for intravenous use [prescribing information]. Genentech; April 2021. www.gene.com/download/pdf/tecentriq_prescribing.pdf. Accessed July 22, 2021.
- Rosenberg JE, Hoffman-Censits J, Powles T, et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet. 2016;387:1909-1920.
- Powles T, Durán I, van der Heijden MS, et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet. 2018;391:748-757. Erratum in: Lancet. 2018;392:1402.
- Galsky MD, Arranz Arija JÁ, Bamias A, et al; for the IMvigor130 study group. Atezolizumab with or without chemotherapy in metastatic urothelial cancer (IMvigor130): a multicentre, randomised, placebo-controlled phase 3 trial. Lancet. 2020;395:1547-1557.
- US Food and Drug Administration. Nivolumab for treatment of urothelial carcinoma. February 2, 2017. www.fda.gov/drugs/resources-information-approved-drugs/nivolumab-treatment-urothelial-carcinoma. Accessed January 28, 2020.
- Opdivo (nivolumab) injection, for intravenous use [prescribing information]. Bristol-Myers Squibb Company; May 2021. https://packageinserts.bms.com/pi/pi_opdivo.pdf. Accessed July 22, 2021.
- Sharma P, Retz M, Siefker-Radtke A, et al. Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate 275): a multicentre, single-arm, phase 2 trial. Lancet Oncol. 2017;18:312-322.
- US Food and Drug Administration. Durvalumab (Imfinzi). May 1, 2017. www.fda.gov/drugs/resources-information-approved-drugs/durvalumab-imfinzi. Accessed January 28, 2020.
- AstraZeneca. Voluntary withdrawal of Imfinzi indication in advanced bladder cancer in the US. February 22, 2021. www.astrazeneca.com/media-centre/press-releases/2021/voluntary-withdrawal-imfinzi-us-bladder-indication.html. Accessed May 5, 2021.
- US Food and Drug Administration. FDA grants accelerated approval to avelumab for urothelial carcinoma. May 9, 2017. www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-avelumab-urothelial-carcinoma. Accessed January 28, 2020.
- Bavencio (avelumab) injection, for intravenous use [prescribing information]. EMD Serono; November 2020. www.emdserono.com/us-en/pi/bavencio-pi.pdf. Accessed June 14, 2021.
- US Food and Drug Administration. FDA approves avelumab for urothelial carcinoma maintenance treatment. July 1, 2020. www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-avelumab-urothelial-carcinoma-maintenance-treatment. Accessed July 26, 2021.
- Patel MR, Ellerton J, Infante JR, et al. Avelumab in metastatic urothelial carcinoma after platinum failure (JAVELIN Solid Tumor): pooled results from two expansion cohorts of an open-label, phase 1 trial. Lancet Oncol. 2018;19:51-64. Erratum in: Lancet Oncol. 2018;19:e335.
- US Food and Drug Administration. Pembrolizumab (Keytruda): advanced or metastatic urothelial carcinoma. May 19, 2017. www.fda.gov/drugs/resources-information-approved-drugs/pembrolizumab-keytruda-advanced-or-metastatic-urothelial-carcinoma. Accessed January 28, 2020.
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck & Co; May 2021. www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf. Accessed June 14, 2021.
- Bellmunt J, de Wit R, Vaughn DJ, et al; for the KEYNOTE-045 investigators. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med. 2017;376:1015-1026.
- Balar AV, Castellano D, O’Donnell PH, et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): a multicentre, single-arm, phase 2 study. Lancet Oncol. 2017;18:1483-1492.
- Powles T, Loriot Y, Gschwend JE, et al. KEYNOTE-361: phase 3 trial of pembrolizumab ± chemotherapy versus chemotherapy alone in advanced urothelial cancer. Eur Urol Suppl. 2018;17:e1147-e1148. Abstract 801.
- US Food and Drug Administration. FDA approves first targeted therapy for metastatic bladder cancer. April 12, 2019. www.fda.gov/news-events/press-announcements/fda-approves-first-targeted-therapy-metastatic-bladder-cancer. Accessed January 28, 2020.
- Balversa (erdafitinib) tablets, for oral use [prescribing information]. Janssen Products; April 2020. www.janssenlabels.com/package-insert/product-monograph/prescribing-information/BALVERSA-pi.pdf. Accessed June 14, 2021.
- Haugsten EM, Wiedlocha A, Olsnes S, Wesche J. Roles of fibroblast growth factor receptors in carcinogenesis. Mol Cancer Res. 2010;8:1439-1452.
- Loriot Y, Necchi A, Park SH, et al; for the BLC2001 study group. Erdafitinib in locally advanced or metastatic urothelial carcinoma. N Engl J Med. 2019;381:338-348.
- Padcev (enfortumab vedotin-ejfv) for injection, for intravenous use [prescribing information]. Astellas Pharma/Seagen; March 2021. https://astellas.us/docs/PADCEV_label.pdf. Accessed June 14, 2021.
- US Food and Drug Administration. FDA grants accelerated approval to enfortumab vedotin-ejfv for metastatic urothelial cancer. December 19, 2019. www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-enfortumab-vedotin-ejfv-metastatic-urothelial-cancer. Accessed January 28, 2020.
- Rosenberg JE, O’Donnell PH, Balar AV, et al. Pivotal trial of enfortumab vedotin in urothelial carcinoma after platinum and anti-programmed death 1/programmed death ligand 1 therapy. J Clin Oncol. 2019;37:2592-2600.
- US Food and Drug Administration. FDA grants accelerated approval to sacituzumab govitecan for advanced urothelial cancer. April 13, 2021. www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-sacituzumab-govitecan-advanced-urothelial-cancer. Accessed May 5, 2021.
- Trodelvy (sacituzumab govitecan-hziy) for injection, for intravenous use [prescribing information]. Immunomedics; April 2021. www.gilead.com/-/media/files/pdfs/medicines/oncology/trodelvy/trodelvy_pi.pdf. Accessed June 14, 2021.
- US Food and Drug Administration. FDA approves pembrolizumab for BCG-unresponsive, high-risk non-muscle invasive bladder cancer. January 8, 2020. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-pembrolizumab-bcg-unresponsive-high-risk-non-muscle-invasive-bladder-cancer. Accessed January 28, 2020.
- ClinicalTrials.gov. Study of pembrolizumab (MK-3475) in participants with high risk non-muscle invasive bladder cancer (MK-3475-057/KEYNOTE-057). https://clinicaltrials.gov/ct2/show/NCT02625961?term=02625961&draw=2&rank=1. Accessed January 28, 2020.