Hypersensitivity reactions to drug therapy are an adverse event that typically occur within the first few hours after drug exposure. The symptoms of hypersensitivity reactions can include (but are not limited to) urticaria, angioedema, bronchospasm, and potentially anaphylaxis, which can be fatal.1 Hypersensitivity reactions can occur in the inpatient and outpatient settings, and in certain cases they warrant treatment with premedications, as well as patient monitoring.
Paclitaxel and carboplatin are frequently prescribed oncology drugs that induce hypersensitivity reactions and have therefore been associated with the use of test doses at the initiation of treatment.2 Paclitaxel, a semisynthetic anticancer agent that belongs to the drug class taxanes, is used for the treatment of a wide variety of solid-organ cancers.3 Paclitaxel is poorly water soluble and thus is formulated in a polyoxyethylated castor oil vehicle, which is often referred to as Cremophor EL.3 Reactions to paclitaxel may result from the drug itself or from the vehicle in which it is suspended.4
There is a wide range in the incidence of hypersensitivity reactions to paclitaxel treatment, extending from 1% to 41%.3 Paclitaxel-induced hypersensitivity reactions typically occur within the first 5 minutes of drug administration5; because of this, some practices use test doses to ensure drug tolerability before the infusion of the entire dose of paclitaxel. Nevertheless, the benefit of test doses is still unclear. A retrospective review of randomized studies has shown that test doses are ineffective and that they are associated with increased drug costs.6
Carboplatin belongs to the drug class platinum salts and is also used for the treatment of multiple types of solid-organ cancers. Hypersensitivity reactions in response to carboplatin treatment range from mild to severe, including anaphylaxis and death.7 The prevalence of hypersensitivity reactions with carboplatin is 2%.8 Contrary to most hypersensitivity reactions to other drugs, which occur during the first or second exposure, hypersensitivity reactions to carboplatin tend to occur after patients have undergone several courses of treatment, typically within the eighth exposure to the drug.9,10 To mitigate the risk for hypersensitivity reactions, intradermal skin tests for carboplatin have been used to identify the patients who are at risk for severe hypersensitivity reactions once they have accumulated exposure to carboplatin. Similar to paclitaxel, the data to support the use of skin testing for carboplatin are controversial, because it may not always accurately predict which patient will have a reaction.11
At our institution, we previously used test doses for paclitaxel and carboplatin. The test doses for paclitaxel were administered as a 1-mg slow intravenous push before dose 1, and the test doses for carboplatin were administered as a 0.02-mg intradermal skin test before dose number 7 and beyond, which was later reduced to 0.01 mg. In both cases, the patients were observed for the signs of a hypersensitivity reaction, and then the full dose of the chemotherapy was administered if the patient had no reaction. The test doses for these agents were removed in August 2018, and the impact of this change remained unknown. The goal of our study was to examine the safety of this change.
Methods
This retrospective, observational study was performed at UC Davis Health, a large academic medical center. All study procedures were approved by the Institutional Review Board at our institution. Adults aged ≥18 years were included in the study if they received their first dose of paclitaxel or any dose of carboplatin (based on when hypersensitivity reactions are expected for each drug) between June 2012 and June 2020.
Patients were excluded if they received a dose of paclitaxel or carboplatin in August 2018 to allow for cutover time, because this was when the removal of the test dose was implemented. Pregnant patients and prisoners were also excluded from data analysis.
We defined clinically significant hypersensitivity reactions as a reaction for which a patient warranted an incident report to be submitted, and we identified these patients using an internal risk module reporting system. Our search was performed by examining all adverse drug reaction reports to find entries containing the medications “paclitaxel” or “taxol” and “carboplatin.” The reports were then sorted by date to divide the patients into “test dose group” and “no test dose group” (henceforth, “preintervention group” and “postintervention group”).
Patients who had mild reactions that did not result in an incident report being filed were not included in this study.
Separately, an electronic medical record report was run to identify the total number of patients receiving paclitaxel or carboplatin during the study period. The numbers from these 2 reports were used to calculate the percentage of patients who had a hypersensitivity reaction. The prevalence of hypersensitivity reactions was compared between the preintervention and postintervention groups for carboplatin and for paclitaxel.
To assess the cost-savings of the removal of the test dose strategy, we calculated the average number of test doses administered per drug annually using data from 2013 to 2017, because this is the time period for which we have a full year’s worth of data. The costs of the test doses were calculated using an estimate of 30 minutes of observation after the administration of the test dose and by using $1000 per hour of chair time. The equation we used is: (number of test doses/year) × (0.5 hours of chair time/dose) × ($1000/hour of chair time) = annual cost of test doses.
This same calculation was used in the preliminary analysis that led to the removal of test doses in 2018. The number calculated is the annual cost-savings since discontinuing the use of test doses.
The primary objective of this study was to compare the incidence of reported hypersensitivity reactions to carboplatin and paclitaxel before and after the removal of the test dose strategy in August 2018. The secondary outcome was to estimate the financial impact in terms of infusion chair time with the discontinuation of the test dose strategy. Our hypothesis was that the prevalence of clinically significant infusion-related reactions is the same before and after the intervention.
Statistical Analysis
We calculated that we would need 323 patients per group (646 patients per drug) to provide the study with 80% power using a noninferiority limit of 5%, at an overall 2-sided alpha level of 0.05. The primary end point—a comparison of the percentage of clinically significant infusion-related reactions in each group—was evaluated using a binary noninferiority test. P values of ≤.05 were considered statistically significant. Statistical analysis was performed using SAS software (SAS Institute, Inc; Cary, NC).
Results
Paclitaxel
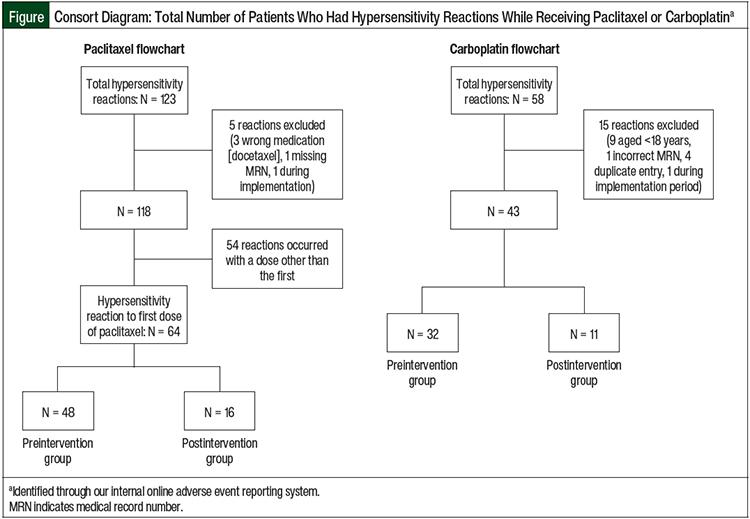
A total of 1091 patients received paclitaxel in the preintervention group versus 345 patients in the postintervention group. Of these administrations, 123 reactions were identified. Overall, 5 patients were excluded, primarily as a result of the incorrect medication being reported, which left 118 reactions. A total of 54 reactions were then excluded because the reactions were in response to doses other than the first dose, when a test dose would not have been used, which left a total of 64 infusion-related reactions to the first dose of paclitaxel. Of these reactions, 48 were in the preintervention group and 16 were in the postintervention group (Figure).
Carboplatin
In all, 1424 patients received carboplatin in the preintervention group, and 435 patients received carboplatin in the postintervention group. In all, 58 reactions were identified. A total of 15 patients were excluded, primarily because of being aged <18 years, which left 43 reactions to any dose of carboplatin. The reactions were calculated to have occurred in 32 patients in the preintervention group and 11 patients in the postintervention group (Figure).
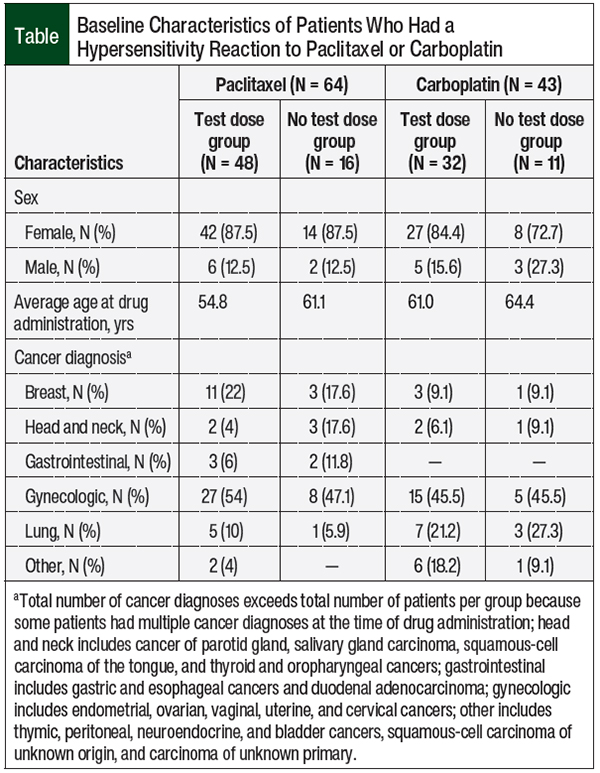
The baseline characteristics of the patients who had hypersensitivity reactions were similar across the groups (Table). The average patient age was 58 years in the paclitaxel group and 63 years in the carboplatin group. A large majority of patients who had hypersensitivity reactions were female (88% for paclitaxel, 79% for carboplatin). The most frequently reported cancers were gynecologic cancers, with a 51% prevalence rate in the paclitaxel group and a 46% prevalence rate in the carboplatin group. Other common tumor types included breast, head and neck, gastrointestinal, and lung cancers.
We used a noninferiority margin of 5% to conduct a noninferiority analysis of the postintervention group compared with the preintervention group. The incidence rates of hypersensitivity reactions to paclitaxel were 4.4% in the preintervention group and 4.7% in the postintervention group (P <.0001). The incidence rates of hypersensitivity reactions for carboplatin were 2.3% in the preintervention group and 2.5% in the postintervention group (P <.0001). These P values indicate strong evidence of noninferiority of the postintervention group compared with the preintervention group.
To assess for the cost-savings, we calculated an average of 235 test doses of paclitaxel annually and 357 test doses of carboplatin annually.
Discussion
In this study, the use of test dosing strategies before 2018 did not affect the incidence of the reported hypersensitivity reactions in the patients who were receiving carboplatin or paclitaxel. Furthermore, as a result of removing the test doses for these 2 drugs, we estimated an annual cost-savings of $295,800 since the implementation of this change. The cost-savings for paclitaxel was calculated as: 235 test doses/year × 0.5 hours of chair time × $1000/hour of chair time = $117,500. The same equation was used for carboplatin, resulting in $178,300 saved.
In addition to finding no difference in the incidence of hypersensitivity reactions between the preintervention and postintervention groups, the severity of hypersensitivity reactions and the management of these reactions were unchanged between the groups as well.
Before and after implementing the strategy change, the patients who had a clinically significant hypersensitivity reaction would have their infusion of paclitaxel or carboplatin stopped, were given hypersensitivity reaction medications (ie, diphenhydramine, methylprednisolone, and/or famotidine), and were observed for the resolution of symptoms. If the symptoms resolved, the patients were rechallenged with the medication, based on the provider’s assessment. The patients with more serious anaphylactic reactions were given epinephrine and were sent to the emergency department.
In a prospective study by Stanford and colleagues, patients were randomized to receive their full dose of paclitaxel or docetaxel with or without a test dose to evaluate the value of test doses for these medications.4 The patients in that study received a 1-mg intravenous test dose of paclitaxel or docetaxel, which is the same dose the patients received in our study. Their study showed an overall incidence of hypersensitivity reactions of 6.5%, with no significant difference between the 2 groups. These findings are consistent with the results of our study.4
There are data to support the negative predictive value of carboplatin skin testing.7,12 In a study by Zanotti and colleagues, a negative skin test accurately predicted the absence of a hypersensitivity reaction in 166 of 168 cases.12 It is important to note that the intradermal test dose used in their study was 0.02 mL of undiluted carboplatin based on the patient’s full dose, with test doses ranging from 100 to 240 mcg.
Our institution previously used 20-mcg intradermal skin tests, but later decreased the doses to 10 mcg because of skin scarring with the higher dose. The lack of predictive value seen with our institution’s carboplatin intradermal skin tests may be a result of the doses used.
A strength of this study is that we met the power to detect statistical significance. Because of this, we can say with a high level of confidence that the intervention of removing test doses for paclitaxel and carboplatin did not lead to a difference in the number of patients with hypersensitivity reactions.
Limitations
Some limitations of this study are that it was conducted at a single center, and the retrospective nature of this study. We chose to define clinically significant hypersensitivity reactions as reactions that required an incident report to be filed.
Because our institution was going through an educational period regarding the proper administration of epinephrine during our study period, we thought that capturing reactions through the use of rescue medications, including epinephrine, might skew our data between the groups. Because of this, the number of hypersensitivity reactions reported depended on the nurses reporting the reaction.
Although this method may not have captured all the data, we do not think that there are any differences in the reporting of hypersensitivity reactions before 2018 and after 2018.
Finally, our baseline characteristics are only reflective of patients who had hypersensitivity reactions, not of the entire population of patients who received carboplatin and paclitaxel. Because of this, we cannot say with certainty that these characteristics are true of the rest of the population. Perhaps some of these characteristics are higher in our patients with hypersensitivity reaction than in patients who do not have such reactions, and could be correlated with the increased likelihood of having hypersensitivity reactions. However, given the cancer types (see Table) that carboplatin and paclitaxel are used for, these characteristics would likely reflect the larger population of patients who receive these 2 drugs.
Conclusion
The removal of our institution’s test dosing strategy for paclitaxel and carboplatin resulted in cost-savings and did not affect the prevalence of reported hypersensitivity reactions. Adding a universal test dose policy would provide minimal benefit, while increasing cost.
We believe that this study is generalizable to other institutions that use test doses for these 2 drugs and are considering the removal of these test doses.
Author Disclosure Statement
Dr McCann and Dr Steele have no conflicts of interest to report.
References
- Aun MV, Kalil J, Giavina-Bianchi P. Drug-induced anaphylaxis. Immunol Allergy Clin North Am. 2017;37:629-641.
- Boulanger J, Boursiquot JN, Cournoyer G, et al. Management of hypersensitivity to platinum- and taxane-based chemotherapy: CEPO review and clinical recommendations. Curr Oncol. 2014;21:e630-e641.
- Paclitaxel injection [prescribing information]. Hospira; April 2021. https://labeling.pfizer.com/ShowLabeling.aspx?id=4559. Accessed November 9, 2022.
- Stanford BL, Shah SR, Ballard EE, et al. A randomized trial assessing the utility of a test-dose program with taxanes. Curr Med Res Opin. 2005;21:1611-1616.
- Markman M, Kennedy A, Webster K, et al. Paclitaxel-associated hypersensitivity reactions: experience of the gynecologic oncology program of the Cleveland Clinic Cancer Center. J Clin Oncol. 2000;18:102-105.
- Henry A, Charpiat B, Perol M, et al. Paclitaxel hypersensitivity reactions: assessment of the utility of a test-dose program. Cancer J. 2006;12:237-245.
- Leguy-Seguin V, Jolimoy G, Coudert B, et al. Diagnostic and predictive value of skin testing in platinum salt hypersensitivity. J Allergy Clin Immunol. 2007;119:726-730.
- Carboplatin injection [prescribing information]. Hospira; August 2021. https://labeling.pfizer.com/showlabeling.aspx?format=PDF&id=4379. Accessed November 9, 2022.
- Markman M, Kennedy A, Webster K, et al. Clinical features of hypersensitivity reactions to carboplatin. J Clin Oncol. 1999;17:1141-1145.
- Sliesoraitis S, Chikhale PJ. Carboplatin hypersensitivity. Int J Gynecol Cancer. 2005;15:13-18.
- Makrilia N, Syrigou E, Kaklamanos I, et al. Hypersensitivity reactions associated with platinum antineoplastic agents: a systematic review. Met Based Drugs. 2010;2010:207084. doi: 10.1155/2010/207084.
- Zanotti KM, Rybicki LA, Kennedy AW, et al. Carboplatin skin testing: a skin-testing protocol for predicting hypersensitivity to carboplatin chemotherapy. J Clin Oncol. 2001;19:3126-3129.