Advanced and metastatic cancers have complicated and heterogenous molecular and biologic landscapes.1 Biologic heterogeneity exists between histologies, within the same diagnostic group, and even within individual patients.2 This diversity in genomic landscapes speaks to the need for customized combination treatments based on the genomic signature associated with each tumor. Combination therapy blocks multiple tumor growth pathways at once, thereby helping to improve the response to treatment and limit treatment resistance.1
Precision medicine is the science of providing an individualized treatment to each patient based on the genomic alterations involved in their cancer.3 A recent prospective, open-label precision medicine clinical trial administered off-label combinations of targeted therapies and immunotherapies to patients with advanced cancer or poor prognosis, based on their molecular testing results.4
The preliminary data from this clinical trial demonstrated a response rate of approximately 45% (≥30% decrease in the sum of the longest diameter of the target lesions compared with baseline per Response Evaluation Criteria in Solid Tumors version 1.1) in heavily pretreated patients with metastatic cancer who received therapy in the study and were well matched according to the molecular alterations in their tumors.3
The dosing of personalized combination therapy presents a challenge, given that clinical trial data are often lacking to guide the starting drug doses of novel combinations. With more than 300 oncology drugs currently approved by the US Food and Drug Administration (FDA), there are potentially close to 45,000 unique 2-drug combinations and 4.5 million 3-drug combinations. Finding the starting dose of a combination therapy that has minimal side effects and that can be titrated to an effective and tolerable dose is important.
We previously published a set of rules related to dosing of combination therapy based on literature reviews of clinical trials to help guide the starting doses of novel drug combinations.5-8 In the current study we sought to validate these starting doses with data from our precision medicine practice at Moores Cancer Center at the University of California San Diego (UCSD). The practice at our precision medicine clinic is to use molecular profiling information to provide a recommendation of 2 to 4 oncology therapies to be administered concomitantly, thereby blocking multiple tumor growth pathways.
The 3 agents that are among the most often administered targeted therapies in our precision medicine clinic are trametinib, a mitogen-activated protein kinase (MEK) inhibitor; palbociclib, a cyclin-dependent kinase (CDK)4/6 inhibitor; and everolimus, a mammalian target of rapamycin (mTOR) inhibitor. These agents were selected for our study because alterations in pathways that are targetable by these agents are among the most often seen in oncology; thus, these 3 agents are frequently used in precision medicine.
Trametinib is FDA-approved for the treatment of advanced or metastatic, BRAF mutation–positive melanoma, non–small-cell lung cancer, thyroid cancer, and solid tumors.9 Palbociclib is FDA-approved for the treatment of advanced, hormone receptor (HR)-positive, human epidermal growth factor receptor (HER)2-negative breast cancer.10 And everolimus is FDA-approved for the treatment of HR-positive, HER2-negative breast cancer, advanced renal-cell carcinoma, and progressing pancreatic neuroendocrine carcinoma.11
The aim of the current study was to determine safe, tolerable dosing of any one of these agents when combined with other drugs as part of novel combination therapy for advanced solid tumors at our institution.
Methods
This retrospective study included patients at Moores Cancer Center at UCSD who received novel combinations of oncology drugs as part of their advanced or metastatic cancer therapy. We defined novel combinations of oncology drugs as non–FDA-approved and non–standard-of-care combinations that include either trametinib, palbociclib, or everolimus.
Our retrospective analysis included patients from a clinical trial—the Personalized Cancer Therapy to Determine Response and Toxicity (PREDICT) study12—that was previously approved by the UCSD Institutional Review Board (IRB) and was performed in accordance with the UCSD IRB guidelines for data analysis and any investigational therapies for which the patients had provided consent. More than 6000 patients with advanced solid tumors were enrolled in the PREDICT study,12 and were then screened for our current retrospective analysis. (Deidentified data on these patients are available on request.)
Adults with advanced or metastatic cancer who were prescribed trametinib, palbociclib, or everolimus between December 2011 and July 2018 were identified through outpatient pharmacy prescriptions that had been ordered at UCSD, using our outpatient pharmacy prescription database. Physician, pharmacy, and nursing notes were reviewed in the electronic medical record (EMR) to determine if the patient started to use any of the drugs of interest, and if the patient was taking the drug as part of a novel oncology therapeutic combination.
Patients who were receiving trametinib or everolimus in standard combinations—such as dabrafenib plus trametinib, everolimus plus fulvestrant, everolimus plus letrozole, or palbociclib plus letrozole—were excluded from the analysis.
Detailed information on dosing, schedule, dose reductions, start and stop dates of the drugs, other oncology drugs given in combination, and on adverse events was manually extracted from the EMR. For patients who continued receiving therapy, the data were censored on August 1, 2019, for the data analysis. Clinically significant serious adverse events were defined as any adverse events leading to hospitalization, dose reduction, dose interruption, the discontinuation of treatment, or grade ≥3 severity, according to the Common Terminology Criteria for Adverse Events v5.0 criteria.13
A safe and tolerable combination dose was defined as a dose of a single agent used in a novel combination that was tolerated for at least 1 month, with no clinically significant serious adverse events. Patients who were receiving therapy for less than 1 month, without drug-related side effects, or those who did not have clinically documented tolerability while receiving therapy were not evaluable for a safe and tolerable dose and were excluded from our analysis.
If no safe and tolerable dose was found, the lowest dose of the treatment administered was used for the summary statistics.
Statistical analysis was performed using SAS version 9.3 (SAS Institute; Cary, NC).
Results
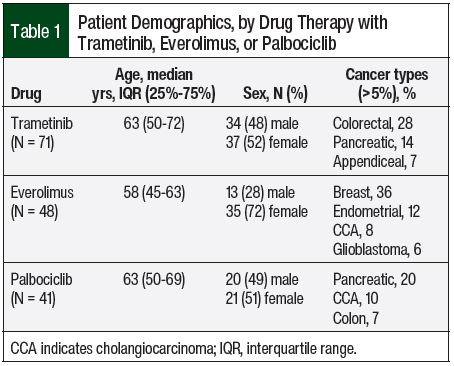
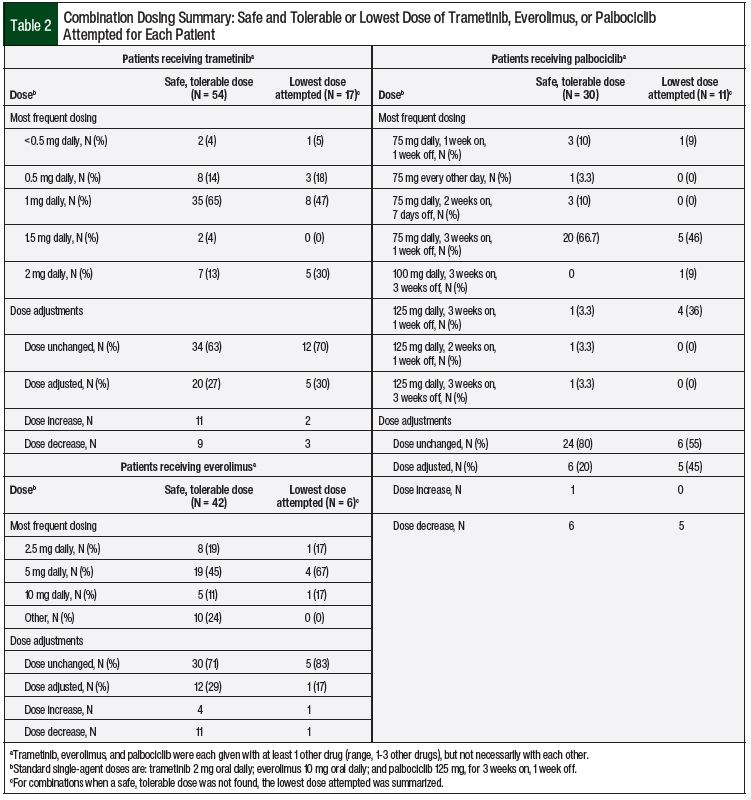
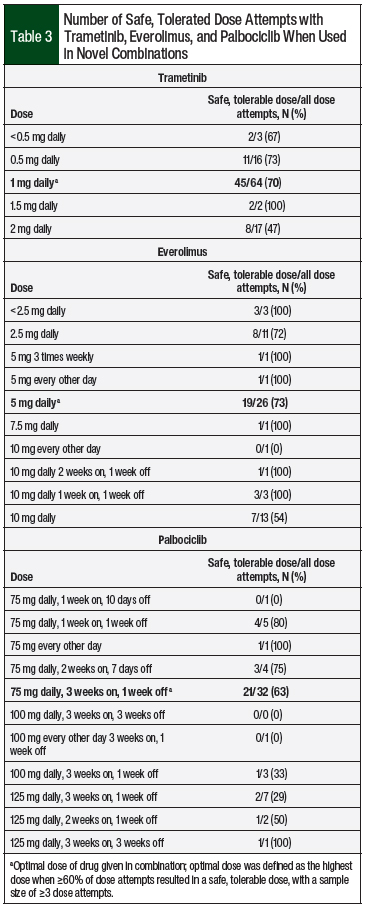
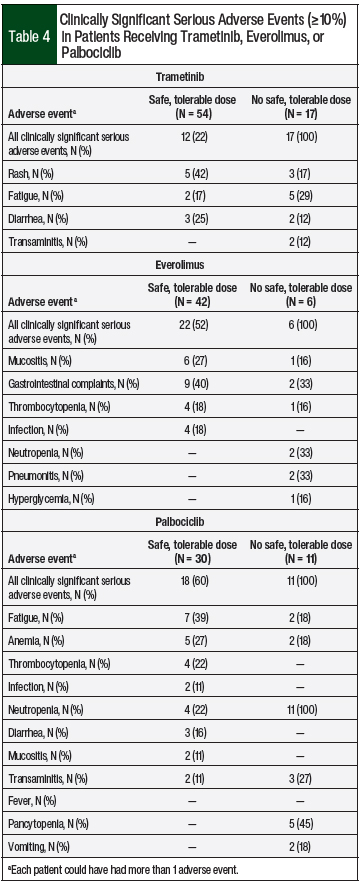
A total of 160 patients were evaluated for the study. The patients’ demographics, cancer type, and treatments are summarized in Table 1. A summary of the dosing for trametinib, everolimus, and palbociclib is provided in Table 2 and Table 3. The clinically significant serious adverse events seen with each of the 3 agents are listed in Table 4.
Trametinib
A total of 71 patients were included in the trametinib group. The majority (76%) of the patients in this cohort achieved a safe, tolerable dose of trametinib. Most patients did not have dose adjustments, even if no safe and tolerable dose was found. The majority (65%) of the patients achieving a safe, tolerable dose were dosed at 1 mg daily (Table 2).
Safe and tolerable doses of trametinib were achieved in 70% of patients who were administered 1 mg daily, and in 73% of patients who were administered 0.5 mg daily (Table 3).
Clinically significant adverse events were noted in 22% of the patients who achieved a safe and tolerable dose of trametinib, with rash, fatigue, and diarrhea being the most common adverse events (Table 4).
Appendix Table 1 summarizes the anticancer drugs used in combination with trametinib.
Everolimus
A total of 48 patients were included in the everolimus group (Table 1). The majority (88%) of the patients who received everolimus achieved a safe, tolerable dose. Most patients who received everolimus did not have dose adjustments, even if a safe, tolerable dose was not ascertained. A dose of 5 mg daily of everolimus was the most often (ie, 45% of patients) administered safe and tolerable dose (Table 2).
Safe and tolerable doses of everolimus were reported in 73% of the patients who were administered 5 mg daily, and in 72% of the patients who were administered 2.5 mg daily (Table 3).
Clinically significant adverse events were noted in 52% of patients who achieved a safe, tolerable dose, with mucositis, gastrointestinal symptoms, infection, and thrombocytopenia as the most frequent adverse events (Table 4).
Appendix Table 2 summarizes the anticancer drugs given in combination with everolimus.
Palbociclib
A total of 41 patients were included in the palbociclib group (Table 1). The majority (73% [30:41]) of the patients who received palbociclib achieved a safe, tolerable dose (Table 2). For patients who reached a safe, tolerable dose of palbociclib, 80% did not have dose adjustments compared with 55% of those who did not reach a safe, tolerable dose of palbociclib (Table 2).
A dose of 75 mg daily for 3 weeks on and 1 week off was the most often (approximately 67% of patients) administered safe and tolerable dose of palbociclib (Table 2). Safe, tolerable doses were achieved in 63% of the patients who were administered palbociclib 75 mg daily for 3 weeks on and 1 week off (Table 3).
Clinically significant adverse events were noted in 60% of the patients who achieved a safe, tolerable dose of palbociclib, with fatigue, anemia, thrombocytopenia, and neutropenia as the most frequent adverse events (Table 4).
Appendix Table 3 summarizes the anticancer drugs given in combination with palbociclib.
Combined Agents
When used in combination with another targeted agent, trametinib accounted for 61% of the safe, tolerable doses; everolimus accounted for 48% of the safe, tolerable doses; and palbociclib accounted for 73% of the safe, tolerable doses of the novel combinations (Appendix Figures 1A-1C).
Trametinib was added to 1 other agent in 39% of combination treatments and to 2 other agents in 41% of the combinations, everolimus was combined with 1 other agent in 69% of the combinations, and palbociclib was combined with 1 other agent in 49% of the combinations (Appendix Figures 2A-2C).
The majority of the patients achieved a safe, tolerable drug dose (trametinib, 76%; everolimus, 88%; palbociclib, 73%) (Table 2). Most of the patients did not have any dose adjustments, even if no safe, tolerable dose was found for the drug.
The majority of patients who achieved a safe, tolerable dose with 1 of the 3 agents received 1 mg daily of trametinib; 5 mg daily of everolimus; or 75 mg daily, for 3 weeks on, 1 week off of palbociclib (Table 3). These doses represent 50% of the standard doses for trametinib (ie, 2 mg oral daily) and everolimus (ie, 10 mg oral daily), and 60% of the standard dose for palbociclib (ie, 125 mg oral daily, for 3 weeks on, 1 week off).
Discussion
The dosing of novel combination therapies can be challenging and may require large dose reductions from the approved single-agent doses. We previously reviewed clinical trials data to determine the safe starting doses for novel drug combinations of targeted therapies and immunotherapies.5-8 Although standard dosing of oncology drug combinations has traditionally been guided by clinical trials data, a precision medicine approach requires a unique, individualized combination drug regimen that is specific to each patient’s molecular profile. Our previous investigations did not involve the applicability of these findings to clinical practice.
Trametinib, palbociclib, and everolimus are the 3 most frequently prescribed targeted agents in our precision medicine practice. These therapies are given in combination with a wide variety of other anticancer agents. Their respective targets—MEK, CDK4/6, and mTOR—represent important pathways for cancer-cell growth; thus, molecular alterations in these pathways are common in molecular profiling, and targeting these pathways is important for the treatment of a wide variety of cancers in a precision medicine approach.14-16 The effective dosing of each therapy in combination with other drugs is thus essential for an effective personalized treatment approach.
In the current study we describe safe and tolerable doses of trametinib, everolimus, and palbociclib when used in combination with a variety of other anticancer agents at our clinic. For the majority of patients, a safe combination dose was found. In cases where a safe dose was not found, the majority of treatment regimens were discontinued rather than attempting dose reductions.
Trametinib at 1 mg oral daily was a safe, tolerable dose in 70% of the patients, everolimus at 5 mg oral daily was safe and tolerable for 73% of patients, and palbociclib at 75 mg oral daily for 3 weeks on and 1 week off was safe and tolerable for 63% of patients (Table 3). These doses likely represent the optimal dosing for these agents when combined with other therapies.
It should be noted that neither our study nor previous literature reports the ability to administer everolimus in combination with trametinib at these doses.17 It is unclear if lowering the doses of these drugs when used in combinations to improve tolerability would affect efficacy; hence, additional investigation is needed.
We previously described that the recommended safe starting doses for small-molecule inhibitors that are administered in 2- and 3-drug combinations would require a dose reduction to achieve safe, tolerable doses.5,6,8 For everolimus, we previously suggested a safe starting dose of 30% of the standard single-agent dose when combined with another targeted agent.8 For 2-drug targeted combinations that include palbociclib or trametinib, we suggested a safe starting dose of 45% of the standard single-agent dose when combined with another small-molecule inhibitor.8
The findings in the current study suggest that everolimus and palbociclib will likely be tolerated at higher doses when prescribed in combination with other therapies in clinical practice.
Of the combinations with unsafe doses, relatively few attempts were made to reduce the dose to find a safe dose. It is likely that physicians were concerned that a lower dose would no longer yield efficacy, and they opted to switch the regimen rather than lower the dose.
Administering doses at less than 100% of the single-agent doses has been evaluated in 2 studies.18,19 A single-institution study of 24 clinical trials suggested that patients receiving the lower doses of drugs used in phase 1 clinical trials did not fare worse than patients receiving a higher dose18; however, a separate study of 55 clinical trials sponsored by a single entity in multiple sites suggested that patients receiving higher doses had improved response rates and overall survival versus patients receiving lower doses.19
Thus, it is unclear if additional dose reductions of the drug combinations given in our current study would have resulted in decreased treatment efficacy. For a precision medicine approach, it is critical to evaluate each individual patient and each potential novel combination therapy that is using the recommended starting doses as a guideline.
Novel combination therapy approaches will require initial dose reductions of each agent in the combination to ensure patient safety while titrating to a dose that is effective and safe. Pharmacists play a critical role in helping to determine if further initial dose reductions are necessary, based on an individual patient’s end-organ function, potential drug–drug interactions, interactions with herbal supplements, and other comorbidities.
Limitations
The current study is limited by its retrospective nature; thus, information on adverse events was determined through a chart review of the physicians’ notes.
Because we investigated oral therapies, we were limited to determining dosing, medication start dates, and medication stop dates from the EMR notes and did not have detailed information available on patient adherence. In particular, poor adherence may alter the dosing conclusions that were reached in the current study.
Furthermore, although physician notes were used to determine which drug was likely to cause an adverse event, it can be difficult to determine which drug was responsible if 2 drugs in the combination had the potential to cause similar adverse events.
All the drug combination doses in this study were assessed together, without regard to the type of agents used in combination.
Although the majority of the study patients received a targeted agent in combination with trametinib, everolimus, or palbociclib, the patients who received immunotherapy might have had different adverse-event profiles that would need to be considered for safe starting doses. These rules may not hold for patients with significant comorbidities or clinically relevant organ dysfunction, because such patients were not represented in our data set.
Finally, this study also did not address the efficacy of the therapies when they were combined at reduced doses, or the potential mechanism of the adverse events.
Conclusion
Our findings show that novel drug combinations that include trametinib, palbociclib, or everolimus can be administered in safe and tolerable doses in a majority of patients.
Based on our findings, for a precision medicine approach that incorporates novel combinations with any of these 3 agents, we recommend starting doses of 1 mg oral daily for trametinib (50% of the standard 2-mg oral daily dose), 5 mg oral daily for everolimus (50% of the standard 10-mg oral daily dose), and 75 mg oral daily for 21 of 28 days for palbociclib (60% of the standard 125-mg oral daily dose for 21 of 28 days) when any of these agents is combined with another therapy.
The use of a combination that includes everolimus plus trametinib is not recommended, even at reduced doses.
Funding Source
This work was supported by the Joan and Irwin Jacobs philanthropic fund and National Cancer Institute grant P30 CA023100.
Author Disclosure Statement
Dr Nikanjam owns stock in Genentech; Dr Li is Employee of and owns stocks in Pfizer; Dr Sicklick receives Consultant fees from Deiphera, Aadi, and Grand Round, is Consultant to CureMatch, is Speaker for Deciphera, La-Hoffman Roche, Foundation Medicine, Merck, QED, Daiichi Sankyo, and owns stocks in Personalis; Dr Kato is Consultant to Medspace, Foundation Medicine, NeoGenomics, and CureMatch, is a Speaker for Chugai, Roche/Genentech, Bayer, and is on the Advisory Board of Pfizer; Dr Capparelli is Consultant to Merck and DSMB member of Melinta; Dr Kurzrock receives research funding from Biological Dynamics, Boehringer Ingelheim, Debiopharm, Foundation Medicine, Genentech, Grifols, Guardant, Incyte, Konica Minolta, Medimmune, Merck Serono, Omniseq, Pfizer, Sequenom, Takeda, and TopAlliance, and is Consultant to or on the Speaker’s Bureau or Advisory Board of many pharmaceutical companies; Dr Tinajero, Dr McGann, Dr Yang, and Dr Shen have no conflicts of interest to report.
References
- Wheler J, Lee JJ, Kurzrock R. Unique molecular landscapes in cancer: implications for individualized, curated drug combinations. Cancer Res. 2014;74:7181-7184.
- Gerlinger M, Rowan AJ, Horswell S, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366:883-892. Erratum in: N Engl J Med. 2012;367:976.
- Sicklick JK, Kato S, Okamura R, et al. Molecular profiling of cancer patients enables personalized combination therapy: the I-PREDICT study. Nat Med. 2019;25:744-750.
- Study of molecular profile-related evidence to determine individualized therapy for advanced or poor prognosis cancers (I-PREDICT). NLM identifier: NCT02534675. Updated February 18, 2022. https://clinicaltrials.gov/ct2/show/NCT02534675. Accessed January 16, 2023.
- Nikanjam M, Liu S, Kurzrock R. Dosing targeted and cytotoxic two-drug combinations: lessons learned from analysis of 24,326 patients reported 2010 through 2013. Int J Cancer. 2016;139:2135-2141.
- Nikanjam M, Liu S, Yang J, Kurzrock R. Dosing three-drug combinations that include targeted anti-cancer agents: analysis of 37,763 patients. Oncologist. 2017;22:576-584.
- Nikanjam M, Patel H, Kurzrock R. Dosing immunotherapy combinations: analysis of 3,526 patients for toxicity and response patterns. Oncoimmunology. 2017;6:e1338997. doi: 10.1080/2162402X.2017.1338997.
- Liu S, Nikanjam M, Kurzrock R. Dosing de novo combinations of two targeted drugs: towards a customized precision medicine approach to advanced cancers. Oncotarget. 2016;7:11310-11320.
- Mekinist (trametinib) tablets, for oral use. [Prescribing information.] Novartis. June 2022. www.novartis.com/us-en/sites/novartis_us/files/mekinist.pdf. Accessed January 20, 2023.
- Ibrance (palbociclib) tables/capsules, for oral use. [Prescribing information.] Prizer. December 2022. https://labeling.pfizer.com/ShowLabeling.aspx?id=12921. Accessed January 20, 2023.
- Afinitor (everolimus) tables, for oral use. [Prescribing information.] Novartis. www.novartis.com/us-en/sites/novartis_us/files/afinitor.pdf. Accessed January 20, 2023.
- Study of Personalized Cancer Therapy to Determine Response and Toxicity (UCSD PREDICT). NLM identifier: NCT02478931. Updated April 5, 2021. https://clinicaltrials.gov/ct2/show/NCT02478931. Accessed January 16, 2023.
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. November 27, 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf. Accessed January 20, 2023.
- Du Q, Guo X, Wang M, et al. The application and prospect of CDK4/6 inhibitors in malignant solid tumors. J Hematol Oncol. 2020;13:41. doi: 10.1186/s13045-020-00880-8.
- Dhillon AS, Hagan S, Rath O, Kolch W. MAP kinase signalling pathways in cancer. Oncogene. 2007;26:3279-3290.
- Ilagan E, Manning BD. Emerging role of mTOR in the response to cancer therapeutics. Trends Cancer. 2016;2:241-251.
- Tolcher AW, Bendell JC, Papadopoulos KP, et al. A phase IB trial of the oral MEK inhibitor trametinib (GSK1120212) in combination with everolimus in patients with advanced solid tumors. Ann Oncol. 2015;26:58-64.
- Jain RK, Lee JJ, Hong D, et al. Phase I oncology studies: evidence that in the era of targeted therapies patients on lower doses do not fare worse. Clin Cancer Res. 2010;16:1289-1297.
- Gupta S, Hunsberger S, Boerner SA, et al. Meta-analysis of the relationship between dose and benefit in phase I targeted agent trials. J Natl Cancer Inst. 2012;104:1860-1866.