Ampullary carcinoma is a rare type of tumor that arises from the ampulla of Vater, otherwise known as the hepatopancreatic ampulla.1 The ampulla of Vater is anatomically characterized by the convergence of the pancreatic duct and common biliary duct, which then extends into the duodenum. Currently, ampullary carcinoma represents only 0.2% of all neoplasms in the gastrointestinal (GI) tract.1
Given the low incidence of ampullary carcinoma, a standard approach to treatment is not well-established and no clinical guidelines are available. Therefore, the current approach to treatment is often extrapolated from the setting of other GI tract malignancies.2 Current treatment modalities may include surgical resection, radiation, and chemotherapy, depending on the tumor stage, histology, and patient-specific considerations.2,3
The relatively recent introduction of immunotherapy to oncology has drastically changed the landscape of cancer care. This promising class of agents, which fundamentally act to restore the immune system’s ability to recognize malignancy, has produced a substantial evolution in the standards of care for myriad malignancy types.4,5 Immunotherapy agents act by targeting either the programmed-cell death-1 (PD-1) receptor or its respective ligand 1 (PD-L1).4
PD-1 is expressed endogenously on the surface of circulating T-cells. When activated by PD-L1, PD-1 is purported to regulate in a negative way the T-cell receptor signaling, ultimately blunting immune recognition and T-cell receptor–mediated cytotoxicity.4 In some cases, the tumor microenvironment exploits this negative feedback pathway by producing PD-L1 itself, thereby evading the host immune system.4
Pembrolizumab, a humanized monoclonal antibody that targets PD-1 directly, has shown good results in the treatment of many solid tumors. It has been approved by the US Food and Drug Administration (FDA) for many indications, and was the first anticancer treatment to be granted a tumor-agnostic indication for the treatment of solid tumors with microsatellite instability-high (MSI-H) or mismatch repair-deficient (dMMR) protein.6 In addition to MSI-H/dMMR, tumor-cell PD-L1 expression is often a predictor of response to immunotherapy agents such as pembrolizumab.5,7
Pembrolizumab has not been approved by the FDA for use in the setting of microsatellite-stable cholangiocarcinoma or ampullary carcinoma, but several studies have provided examples of PD-L1 expression in the setting of ampullary carcinoma.8,9 Furthermore, the preliminary findings of a prospective cohort study demonstrated modest benefit with pembrolizumab as a subsequent therapy for patients with advanced malignancy of the gallbladder or biliary tree and PD-L1 expression of at least 1%.10 In that study, the median progression-free survival was 1.5 months, the median overall survival was 4.3 months, and no grade 3 or 4 adverse events were reported with pembrolizumab use.10
We present a case of off-label pembrolizumab use that produced a durable response in a patient with PD-L1–positive, microsatellite-stable, metastatic ampullary carcinoma. The patient’s treatment benefit was not sustained with a planned treatment holiday, but disease control was regained with prompt resumption of pembrolizumab therapy.
To our knowledge, this is the first case report of a stop-and-go approach with pembrolizumab therapy in the ampullary carcinoma setting. Patient consent has been obtained for the purpose of this case report.
Case Presentation
A 75-year-old woman with relatively poor performance status presented to the clinic with a 5-month history of early satiety, weight loss, and obstructive jaundice. Further laboratory workup showed evidence of transaminitis, hyperbilirubinemia, and elevated lactate dehydrogenase enzyme levels. An endoscopic retrograde cholangiopancreatography demonstrated the presence of a 4-cm mass in the ampulla of Vater, with significant intrahepatic and extrahepatic biliary dilatation. Subsequent diagnostic imaging confirmed the absence of hepatic or pulmonary involvement. Her baseline carcinoembryonic antigen (CA) 19-9 at the time of diagnosis was 844 IU/mL.
The patient underwent a pancreaticoduodenectomy (Whipple procedure) for resectable disease approximately 4 weeks later. After resection, a pathology report described the primary tumor as a 2.4-cm, poorly differentiated, ampullary carcinoma with invasion of the common biliary duct, pancreatic duct, and 3 of 22 lymph nodes in proximity. No involvement of the gallbladder, liver, or pancreatic neck was noted.
At that time the tumor was staged as pT1pN1pMNa. An accompanying computed tomography (CT) scan of the chest, abdomen, and pelvis (C/A/P) also confirmed the absence of metastatic disease. Molecular profiling obtained at that time frame provided a sample insufficient to determine PD-L1 expression and MSI status. The patient received treatment with 5 cycles of adjuvant chemotherapy with gemcitabine. Her CA 19-9 level before starting gemcitabine therapy was measured at 368.6 IU/mL.
As the patient neared completion of adjuvant chemotherapy, her CA 19-9 level had significantly increased, to 966.1 IU/mL. In response to biochemical evidence of disease progression, a positron-emission tomography (PET) scan demonstrated fludeoxyglucose avidity of the retrocrural and left-supraclavicular lymph nodes. An ultrasound-guided fine-needle aspiration biopsy of the left-supraclavicular lymph node confirmed the presence of metastatic carcinoma of biliary origin consistent with the primary malignancy.
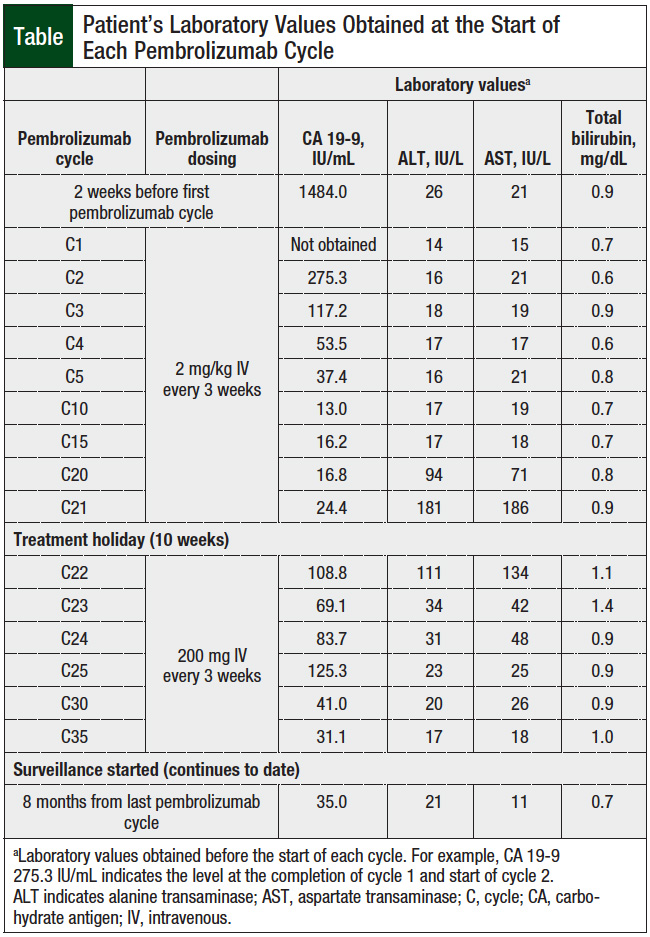
In response to this disease progression, palliative chemotherapy was promptly initiated for metastatic disease with the modified chemotherapy regimen of FOLFOX6 (leucovorin calcium [folinic acid], fluorouracil, and oxaliplatin). Repeated imaging with PET/CT scan performed on completion of 2 cycles of chemotherapy demonstrated disease progression in addition to new C7 vertebral body fludeoxyglucose avidity. The patient’s CA 19-9 level was 1484 IU/mL (Table), consistent with radiologic evidence of progressive disease.
With few therapeutic options because of the rare incidence of ampullary carcinoma, the patient’s treatment was switched to off-label pembrolizumab 2 mg/kg administered intravenously every 3 weeks. Because insurance authorization could not be obtained for the off-label use of pembrolizumab, a manufacturer assistance program provided help in obtaining pembrolizumab. On completion of 4 cycles of pembrolizumab therapy, restaging with a C/A/P CT scan demonstrated pseudo-progression of disease. This finding was inconsistent with the patient’s otherwise improving performance status and decreasing CA 19-9 level over time (from 1484.0 IU/mL 2 weeks before cycle 1 to 37.4 IU/mL by the start of cycle 5; Table).
Considering the patient’s clinical and biochemical response to the pembrolizumab therapy, we decided to continue therapy. On completion of 8 cycles of pembrolizumab, repeated imaging with a PET/CT scan demonstrated stable disease, with resolution of C7 vertebral body fludeoxyglucose avidity. The patient continued to receive pembrolizumab therapy for an additional 11 cycles, demonstrating stable disease on 2 separate occasions during this time frame. At this point (on completion of 19 cycles of pembrolizumab therapy), molecular profiling was repeated with an adequate tissue sample, revealing 35% PD-L1 expression on the primary tumor and microsatellite-stable status.
Because the patient maintained good disease control on completion of 21 cycles (approximately 1 year from the first cycle), we decided to hold therapy for 16 weeks to facilitate the patient’s personal travel plans. Approximately 8 weeks into this planned immunotherapy holiday, the patient reported worsening abdominal pain. Repeated imaging with a C/A/P CT scan indicated disease recurrence, and the patient’s CA 19-9 level had increased from 24.4 IU/mL at the beginning of cycle 21 to 108.8 IU/mL before instituting cycle 22 (Table).
Considering the patient’s worsening clinical status and the evidence of disease progression, pembrolizumab therapy was promptly resumed 10 weeks from the last cycle, at a dose of 200 mg, administered intravenously every 3 weeks. Within 1 cycle of restarting pembrolizumab therapy, the patient reported a significant improvement in abdominal pain, and her CA 19-9 level decreased to 69.1 IU/mL at the end of cycle 22 (Table).
The patient received an additional 14 cycles (for approximately 11 months) of pembrolizumab therapy before surveillance for stable disease was initiated. Currently, the patient continues to be monitored and remains in stable disease. Her CA 19-9 levels remain within normal limits (at 35 IU/mL, most recently by February 2020), and the patient continues to report excellent symptom control.
Discussion
This case report describes a patient with 35% PD-L1 expression, microsatellite-stable, metastatic ampullary carcinoma who had stable disease with pembrolizumab therapy in the third line. Although a PD-L1 expression of 35% may explain this patient’s positive response to pembrolizumab therapy, it is interesting that such a durable response was maintained in a patient with microsatellite-stable status. Of note, the patient had rapid disease progression when a treatment holiday was initiated, after having received more than 12 months of pembrolizumab therapy at that point.
On resumption of pembrolizumab therapy, clinical disease control was regained in this patient, raising the question whether pembrolizumab should be stopped when used in the setting of metastatic ampullary carcinoma.
According to the most recent National Comprehensive Cancer Network guidelines for the management of hepatobiliary cancer, pembrolizumab may be continued until disease progression or until toxicity in the setting of metastatic or unresectable hepatobiliary cancer.11 However, these guidelines specifically note the lack of evidence for use of pembrolizumab in this setting.11
It can be argued that pembrolizumab be continued until disease progression or toxicity in the setting of metastatic ampullary carcinoma, based on the example of our patient, who had disease recurrence during her pembrolizumab holiday. However, considering the expenses and adverse events associated with immunotherapy, the question arises whether a temporary holiday in immunotherapy is appropriate in this setting.
Conclusion
Given the absence of studies investigating the use of immunotherapy in the setting of metastatic ampullary carcinoma, the optimal timeline for immunotherapy use is not yet defined. The findings from this case report may provide preliminary insights on the potential role of immunotherapy for the third-line treatment of metastatic ampullary carcinoma. Further studies are warranted to confirm these findings.
Author Disclosure Statement
Dr Seyedkazemi owns stocks in AbbVie, Pfizer, Immunogen, Immunomedics, Dynavax, Celldex, and SXT. Dr Kundranda is a consultant to Bayer and has received research funding from Celgene and Curonix. Dr Heers and Dr Tanner have no conflicts of interest to report.
References
- Ahn DH, Bekaii-Saab T. Ampullary cancer: an overview. Am Soc Clin Oncol Educ Book. 2014;34:112-115.
- Ghosn M, Kourie HR, Rassy EE, et al. Where does chemotherapy stand in the treatment of ampullary carcinoma? A review of literature. World J Gastrointest Oncol. 2016;8:745-750.
- Zheng-Pywell R, Reddy S. Ampullary cancer. Surg Clin North Am. 2019;99:357-367.
- Blank C, Mackensen A. Contribution of the PD-L1/PD-1 pathway to T-cell exhaustion: an update on implications for chronic infections and tumor evasion. Cancer Immunol Immunother. 2007;56:739-745.
- He Y, Liu ZX, Jiang ZH, Wang XS. Identification of genomic features associated with immunotherapy response in gastrointestinal cancers. World J Gastrointest Oncol. 2019;11:270-280.
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Whitehouse Station, NJ: Merck & Co; April 2020.
- Herbst RS, Soria JC, Kowanetz M, et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature. 2014;515:563-567.
- Sideras K, Biermann K, Yap K, et al. Tumor cell expression of immune inhibitory molecules and tumor‐infiltrating lymphocyte count predict cancer‐specific survival in pancreatic and ampullary cancer. Int J Cancer. 2017;141:572-582.
- Saraggi D, Galuppini F, Remo A, et al. PD-L1 overexpression in ampulla of Vater carcinoma and its pre-invasive lesions. Histopathology. 2017;71:470-474.
- Kang J, Yoo C, Jeong JH, et al. Efficacy and safety of pembrolizumab in patients with PD-L1 positive advanced biliary tract cancer (BTC): a prospective cohort study. J Clin Oncol. 2019;37(15_suppl):Abstract 4082.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Hepatobiliary Cancers. Version 3.2020. June 1, 2020. www.nccn.org/professionals/physician_gls/pdf/hepatobiliary.pdf. Accessed June 29, 2020.