Acute lymphoblastic leukemia (ALL) is the most common pediatric malignancy.1 Current survival rates for pediatric ALL are approaching 90% in the United States.1,2 This has been accomplished by dose intensification, risk stratification, extended treatment duration, and central nervous system prophylaxis.1,2 Asparaginase, an enzyme responsible for catalyzing the hydrolysis of asparagine into aspartic acid and ammonia, remains an important component of therapy in pediatric patients with ALL. Asparagine is a nonessential amino acid in normal cells; however, it is essential for the survival and proliferation of lymphoblastic B-cells because of a dependence on exogenous sources required for protein, DNA, and RNA synthesis.3
All asparaginase agents are bacterially derived; L-asparaginase and polyethylene glycol asparaginase (pegaspargase) are derived from Escherichia coli, and Erwinia asparaginase is derived from Erwinia chrysanthemi.3 Despite the importance of asparaginase therapy in the management of ALL, several dose-limiting toxicities exist and require careful consideration.3 Currently, pegaspargase is the first-line option for treatment-naïve pediatric ALL patients.3
All asparaginase preparations, including pegaspargase and Erwinia asparaginase, can cause hypersensitivity reactions (HSRs) resulting from foreign protein immunogenicity.4 Other remarkable complications include pancreatitis, thrombosis, bleeding, hyperglycemia, hyperbilirubinemia, and liver dysfunction.3 The most common manifestation of asparaginase hypersensitivity is urticarial, although symptoms may range from local and transient erythema to systemic and life-threatening anaphylaxis.5 Other common hypersensitivity symptoms include induration, pain, bronchospasm, edema, pyrexia, emesis, and cutaneous reactions.4,6,7
Although the pathogenic pathway describing HSRs to asparaginase is not fully known, previous studies have demonstrated some involvement of immunoglobulin E antibodies and/or complement activation.5 Hypersensitivity to pegaspargase can be acute or delayed, and can occur after any dose administered throughout ALL therapy.4 Acute HSRs typically occur within 1 hour of drug administration and range from mild local irritation to complicated anaphylaxis.8
Although pegaspargase is less immunogenic than the native L-asparaginase agent, severe infusion reactions still occur frequently, with a recent meta-analysis showing an HSR rate of 23.5%.9 Historically, L-asparaginase was delivered via intramuscular (IM) injection.4,10-12 Previous studies showed significantly fewer systemic HSRs in patients receiving IM administration compared with those receiving intravenous (IV) administration.6,9,11-15
With native L-asparaginase, up to 65% of patients receiving IV administration had HSRs compared with only 20% of patients receiving IM administration.16,17 Consequently, these high rates of HSRs coupled with frequent dosing requirements of the nonpegylated formulation resulted in the United States adopting pegaspargase as first-line asparaginase therapy in 2006.3
Recent pegaspargase studies demonstrated that it can be given via IV administration without significant toxicity.6,13,18 The Children’s Oncology Group (COG) modified many of its ALL protocols to reflect this, and now IV administration is the preferred route in all pediatric ALL protocols. IV administration is a patient satisfier and improves treatment perceptions.9 Many patients dislike receiving multiple IM injections, and the alternative IV route is the more humane course of action, because it is associated with less pain and anxiety, along with fewer injections.9 After conversion from IM to IV pegaspargase, concerns regarding increased hypersensitivity rates with IV administration emerged.6,13
Severe HSR of pegaspargase via IV or IM route of administration necessitates substitution with antigenically distinct Erwinia asparaginase.19 Hypersensitivity to Erwinia asparaginase is generally not common, although still possible, and warrants careful monitoring.7,19 The treatment of these asparaginase reactions remains supportive care, with subsequent immune factor monitoring to determine the asparaginase activity.3,19-21 There is a relative paucity of data evaluating the safety and efficacy differences between IV and IM Erwinia asparaginase in pediatric patients with ALL.7,10,22
No recommendations are available regarding a preferred administration route for Erwinia asparaginase, although the majority of European oncology groups use the IV route, whereas North American groups prefer the IM route of administration.7 Vrooman and colleagues found IV Erwinia asparaginase to be safe and effective despite various pharmacokinetic differences, including possible subtherapeutic nadir serum asparaginase activity at 72 hours postdosing.10
A pharmacologic comparison of available asparaginase agents is provided in Table 1.20,21 At the time of this study, data comparing HSR rates between IV and IM asparaginase agents in pediatric patients with ALL were scant. Since converting from IM to IV administration of pegaspargase, our institution identified anecdotal evidence suggesting a disparity in rates of HSRs based on route of administration. This is consistent with recent findings from other institutions.6,11,13,14
For example, Petersen and colleagues showed a significant difference in rates of HSRs between IV and IM pegaspargase (19.5% vs 10.7%, respectively; P = .028).13 A similar report comparing rates of HSRs showed a significant difference between IV and IM administration (36% vs 9%, respectively; P = .019).6 This study had a relatively small cohort, with only 11 total patients receiving IV pegaspargase.6 Finally, a large retrospective study evaluated 6 COG trials with a combined 16,534 patients.8 This analysis provides the largest collection of pegaspargase data for IV and IM administration. From 2003 through 2015, these COG trials showed that IV pegaspargase elicited fewer HSRs than IM administration (3.2% vs 5.4%, respectively; P <.0001).8
Unfortunately, the rates of Erwinia asparaginase hypersensitivity based on route of administration were not evaluated, and the data remain sparse. One large compassionate-use study evaluated the safety profile for IV and IM Erwinia asparaginase.22 The majority (91%) of patients received IM Erwinia asparaginase, and the rates of grade 3 or 4 HSRs were 6.9% and 2.8% in patients receiving IV and IM Erwinia asparaginase, respectively.22 Furthermore, this study showed patients receiving Erwinia asparaginase had nausea (2.4%) and vomiting (3%) infrequently.22
The primary objective of our study was to identify whether the route of asparaginase administration alters the rate of HSRs. In addition, we evaluated whether patients had HSRs during different treatment phases of ALL, based on the administration route, and if the route of administration altered the incidence or severity of other adverse effects in pediatric patients with ALL.
Methods
Our single-center, retrospective study was reviewed and approved by the Institutional Review Board at a tertiary academic medical center. All patients receiving pegaspargase or Erwinia asparaginase between January 1, 2008, and September 30, 2015, were considered for evaluation. Inclusion criteria for the study were individuals receiving pegaspargase or Erwinia asparaginase for a confirmed ALL diagnosis. We did not have an age limit; because this is a children’s hospital, the vast majority of patients are young. We were more interested in the rates of reactions in all of our patients, so we left the age limit open. The study excluded any individuals who received native L-asparaginase drug or the investigational drug EZN-2285 during the course of their treatment.
Eligible patients were cross-matched with patients who had a documented pegaspargase or Erwinia asparaginase allergy in the electronic medical record (EMR). The EMR query also extracted specific data points, including total asparaginase doses, date of allergy, and last date of asparaginase therapy.
The primary study outcome was to evaluate the incidence of HSRs in patients receiving IV or IM pegaspargase or Erwinia asparaginase. Secondary outcomes measured the rates of adverse drug events such as nausea, vomiting, or hospitalization based on route of administration. Nausea and vomiting were identified using administered antiemetic therapy as a surrogate marker. The administration route was evaluated to determine if it played a role in the severity of HSR in patients receiving asparaginase agents. Finally, we assessed the timing (ie, induction therapy, consolidation therapy, or postconsolidation therapy) of HSRs and evaluated which pegaspargase dose resulted in HSR.
All patients who had an HSR were evaluated using Common Terminology Criteria for Adverse Events (CTCAE) version 4.0 to determine the severity of the hypersensitivity reaction.23
A chi-square analysis was used to evaluate the incidence of hypersensitivity reactions between pegaspargase routes of administration. The Erwinia asparaginase groups were evaluated using a Fisher’s exact test. Treatment effects across the primary and secondary outcomes were evaluated with a 2-sided significance level, set a priori at 0.05. No adjustments for multiple comparisons were made.
Results
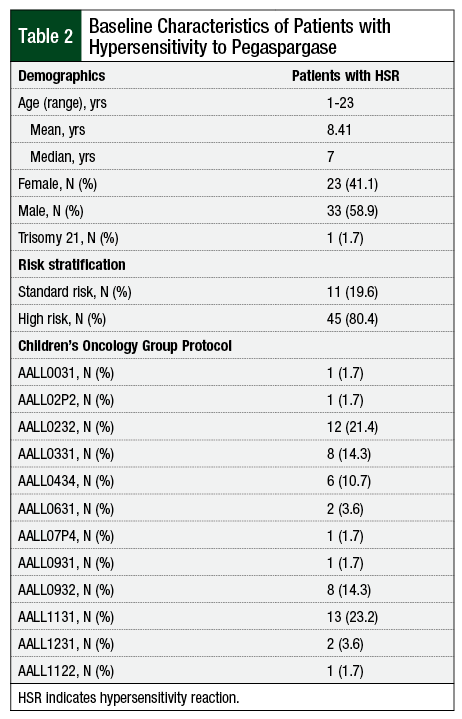
A total of 368 patients who received pegaspargase and 31 patients who received Erwinia asparaginase were deemed eligible for the study. Overall, 199 patients received only IV pegaspargase and 138 patients received only IM pegaspargase. The remaining 31 patients received both IV and IM pegaspargase during the course of their treatment (Table 2).
Grade 3 and 4 HSRs occurred in 18.6% of patients receiving IV and 13.8% receiving IM (P = .241) pegaspargase (Table 3). Among patients who received IV and IM pegaspargase, the hypersensitivity rate was 9.7%. Of all patients with a documented HSR, 33 (59%) were male.
Furthermore, 39% (13) of male and 39% (9) of female patients received IM pegaspargase, whereas 70% (23) of male and 74% (17) of female patients received IV pegaspargase at some point in their treatment. Overall, the mean age of patients treated with pegaspargase was 8.3 years (range, 1-23 years; median, 7). Patients treated with IM pegaspargase were an average age of 9.2 years (range, 1-23 years; median, 7), whereas patients treated with the IV formulation were an average age of 8.1 years (range, 1-19 years; median, 7).
A significant increase in antiemetics use was detected in patients receiving IV Erwinia asparaginase compared with IM Erwinia asparaginase (P <.001) or pegaspargase (P = .003). Only 28 (7%) of the total 440 Erwinia asparaginase IM doses required antiemetics, whereas 28 (36.4%) of the 77 IV Erwinia asparaginase doses required antiemetic therapy.
We evaluated the timing of grade 3 and grade 4 HSRs relative to the phase of treatment to determine when patients were at the highest risk for reacting to an asparaginase drug. In both groups, a majority of the HSRs occurred in the consolidation phase of therapy; 43 of 56 reactions occurred during consolidation. The second scheduled pegaspargase dose resulted in HSRs in 36 patients and was by far the most common dose responsible for infusion-related reactions (64.3% of HSRs).
We also evaluated hospitalization rates. A total of 26 patients were hospitalized as a result of HSRs, and 6 patients were already admitted when anaphylaxis occurred. Nearly 61% of the patients who reacted had an HSR to IM pegaspargase that required hospitalization compared with only 39% of those receiving IV pegaspargase. Hospital admission did not correlate with a specific route of administration (P = .324) or a total number of pegaspargase doses (P = .163). Of the patients with an HSR to IV or IM pegaspargase, 5 (13.5%) and 7 (36.8%), respectively, were documented as grade 4 (P = .241). Hospital admission was significantly related to the grade of the HSR: 34% (15 of 44) of patients were admitted after a grade 3 reaction compared with 92% (11 of 12) of patients who had a grade 4 reaction (P = .001).
Discussion
Asparaginase therapy is one of the most important components of the treatment of pediatric patients with ALL; however, many important clinical questions remain. Although IV administration is preferred by patients, the rate of HSR may be higher, if less severe, than with the traditional IM route of administration. Unlike other retrospective studies,6,8,13 our study did not find a significant difference between routes of administration and the rates of HSR to pegaspargase. Additional studies are warranted to assess the rates and severity of adverse reactions in a prospective manner.
Similar to another retrospective study,13 we found a significant difference in HSR rates between male (60.7%) and female (39.3%) pegaspargase recipients; however, males are at an increased risk for leukemia, and therefore comprise a larger percentage of the patient population. It would be worthwhile to analyze the proportion of males and females with ALL who had an adverse reaction to pegaspargase therapy.
Our study findings suggest that IM Erwinia asparaginase may be significantly more tolerable than the IV administration with regard to emetogenicity. The mechanism behind higher rates of nausea and vomiting with IV administration is not fully understood and requires additional research, but it is perhaps related to a faster rate of asparagine depletion and secondary production of ammonia, a chemical known to elicit nausea and vomiting at high levels.8,24-26
Our finding of fewer hospitalizations in patients who received IV pegaspargase may be attributable to the ability of the healthcare team to intervene at the first sign of allergic reaction and stop the infusion. Quick intervention in cases of suspected hypersensitivity allows the provider to reduce overall pegaspargase exposure and potentially minimize the severity of the HSR. With IM pegaspargase administration, the patient receives the full dose and therefore may have a more profound and prolonged immune response.
Several important factors should be kept in mind when considering IV versus IM pegaspargase administration. For patients with an HSR to pegaspargase, subsequent IM or IV Erwinia asparaginase treatment is required.4 Currently, no long-acting formulation exists; therefore, 6 doses are required for each scheduled pegaspargase administration.21 This can result in up to 50 doses of Erwinia asparaginase, depending on the protocol and at which phase of treatment the reaction occurred. This is a notable burden for patients and their families, because each dose must be administered in the hospital or infusion clinic. In addition, Erwinia asparaginase is a high-cost medication, with potential costs exceeding $100,000 for many patients.15,27
Until 2015, Erwinia asparaginase was only administered via the IM route, and some patients would require up to 3 separate injections per Erwinia asparaginase dose.21 Many patients and providers were dissatisfied with this route and selected IV Erwinia asparaginase over IM administration, despite potentially reduced asparaginase activity at 72 hours.21
Limitations
The potential limitations to this study include varying hypersensitivity grading practices as a result of transitioning from CTCAE version 3.0 to version 4.0 during the study period. The investigators, however, independently graded the HSRs using the criteria in CTCAE version 4.0 based on descriptions found in the EMR. The CTCAE grading change likely resulted in higher rates of reported reactions in our study. This could be particularly important for the rates of HSR with IV administration, because a change to the CTCAE criteria involved the addition of “infusion interruption,”23 which was common in patients with an adverse reaction to IV pegaspargase.
In addition, healthcare professionals at our institution were hypervigilant with the introduction of IV pegaspargase as a result of their previous experience with IV L-asparaginase and its increased rate of complications compared with the IM route of administration.12,28 This heightened caution could have led to increased reporting and apprehension.
Our study only evaluated clinically overt hypersensitivity, and therefore cannot address the development of antiasparaginase neutralizing antibodies and subclinical hypersensitivity, which is known as silent inactivation with pegaspargase or with Erwinia asparaginase. Silent inactivation lacks clinical symptoms and results from neutralizing antidrug antibodies, leading to asparaginase inactivity and suboptimal asparagine depletion in an estimated 8% to 29% of patients.29,30 Patients with silent inactivation continue to receive an asparaginase agent that is ineffective,31 whereas those with clinically overt pegaspargase hypersensitivity can proceed to antigenically distinct Erwinia asparaginase. Therapeutic drug monitoring surrounding asparaginase activity continues to gain popularity as we move to more individualized treatment plans.30
Furthermore, surrogate markers for chemotherapy-induced nausea and vomiting (CINV) were used to evaluate emetogenicity.
Finally, our study only included 31 patients who received Erwinia asparaginase, and each of them received an average of 14 doses. This is a small sample size, but we believe that CINV resulting from IV Erwinia asparaginase is clinically significant and merits further evaluation.
Conclusion
Asparaginase agents are a vital component of pediatric ALL treatment, and the route of administration may have important implications. Pegaspargase will continue to be given via the IV route of administration for pediatric patients with ALL on COG protocols at our institution. Because no specific recommendation exists regarding the preferred route for Erwinia administration, many institutions allow the parent, caregiver, and/or patient to decide the route of administration.
Pharmacists are optimally positioned to help in this process by discussing the risks and benefits of IV and IM Erwinia asparaginase with parents, caregivers, patients, and other relevant healthcare professionals. IV Erwinia asparaginase reduces the number of patient injections but may lead to higher rates of CINV. Erwinia asparaginase delivered via IV or IM route elicits low rates of HSRs. Future studies should evaluate the mechanism and significance of IV administration on CINV rates and patient satisfaction.
Our single-center study did not show a significant difference in the rates of HSRs between IV and IM pegaspargase. Although IV administration resulted in a higher rate of hypersensitivity, patients who had an HSR required fewer hospital admissions and noted a milder reaction than those receiving IM administration. Erwinia asparaginase had exceedingly low HSR rates, with only 1 patient having a documented reaction. Interestingly, antiemetic use increased markedly in patients who received IV Erwinia asparaginase versus IM administration, warranting further evaluation.
Author Disclosure Statement
Dr Halford and Dr Park have no conflicts of interest to report.
References
- Hunter SP, Mullighan CG. Acute lymphoblastic leukemia in children. N Engl J Med. 2015;373:1541-1552.
- Hunger SP, Lu X, Devidas M, et al. Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: a report from the Children’s Oncology Group. J Clin Oncol. 2012;30:1663-1669.
- Kawedia JD, Rytting ME. Asparaginase in acute lymphoblastic leukemia. Clin Lymphoma Myeloma Leuk. 2014;14(suppl):S14-S17.
- van der Sluis IM, Vrooman LM, Pieters R, et al. Consensus expert recommendations for identification and management of asparaginase hypersensitivity and silent inactivation. Haematologica. 2016;101:279-285.
- Soyer OU, Aytac S, Tuncer A, et al. Alternative algorithm for L-asparaginase allergy in children with acute lymphoblastic leukemia. J Allergy Clin Immunol. 2009;123:895-899.
- Pidaparti M, Bostrom B. Comparison of allergic reactions to pegasparaginase given intravenously versus intramuscularly. Pediatr Blood Cancer. 2012;59:436-439.
- Pieters R, Hunger SP, Boos J, et al. L-asparaginase treatment in acute lymphoblastic leukemia: a focus on Erwinia asparaginase. Cancer. 2011;117:238-249.
- Burke MJ, Devidas M, Maloney K, et al. Severe pegaspargase hypersensitivity reaction rates (grade ≥3) with intravenous infusion vs. intramuscular injection: analysis of 54,280 doses administered to 16,534 patients on children’s oncology group (COG) clinical trials. Leuk Lymphoma. 2018;59:1624-1633.
- Hasan H, Shaikh OM, Rassekh SR, et al. Comparison of hypersensitivity rates to intravenous and intramuscular PEG-asparaginase in children with acute lymphoblastic leukemia: a meta-analysis and systematic review. Pediatr Blood Cancer. 2017;64:81-88.
- Vrooman LM, Kirov II, Dreyer ZE, et al. Activity and toxicity of intravenous Erwinia asparaginase following allergy to E. coli-derived asparaginase in children and adolescents with acute lymphoblastic leukemia. Pediatr Blood Cancer. 2016;63:228-233.
- MacDonald T, Kulkarni K, Bernstein M, et al. Significantly higher incidence of allergic reactions for intravenous peg-asparaginase as compared to intramuscular peg-asparaginase in children with high risk acute lymphoblastic leukemia. Pediatr Blood Cancer. 2014;61(Suppl 2):S171.
- Nesbit M, Chard R, Evans A, et al. Evaluation of intramuscular versus intravenous administration of L-asparaginase in childhood leukemia. Am J Pediatr Hematol Oncol. 1979;1:9-13.
- Petersen WC Jr, Clark D, Senn SL, et al. Comparison of allergic reactions to intravenous and intramuscular pegaspargase in children with acute lymphoblastic leukemia. Pediatr Hematol Oncol. 2014;31:311-317.
- Abbott LS, Zakova M, Shaikh F, et al. Allergic reactions associated with intravenous versus intramuscular pegaspargase: a retrospective chart review. Paediatr Drugs. 2015;17:315-321.
- MacDonald T, Kulkarni K, Bernstein M, Fernandez CV. Allergic reactions with intravenous compared with intramuscular pegaspargase in children with high-risk acute lymphoblastic leukemia: a population-based study from the Maritimes, Canada. J Pediatr Hematol Oncol. 2016;38:341-344.
- Place AE, Stevenson KE, Vrooman LM, et al. Intravenous pegylated asparaginase versus intramuscular native Escherichia coli L-asparaginase in newly diagnosed childhood acute lymphoblastic leukaemia (DFCI 05-001): a randomized, open-label phase 3 trial. Lancet Oncol. 2015;16:1677-1690.
- Veerman AJ, Kamps WA, van den Berg H, et al. Dexamethasone-based therapy for childhood acute lymphoblastic leukaemia: results of the prospective Dutch Childhood Oncology Group (DCOG) protocol ALL-9 (1997-2004). Lancet Oncol. 2009;10:957-966.
- Douer D, Yampolsky H, Cohen LJ, et al. Pharmacodynamics and safety of intravenous pegaspargase during remission induction in adults aged 55 years or younger with newly diagnosed acute lymphoblastic leukemia. Blood. 2007;109:2744-2750.
- Salzer WL, Asselin B, Supko JG, et al. Erwinia asparaginase achieves therapeutic activity after pegaspargase allergy: a report from the Children’s Oncology Group. Blood. 2013;122:507-514.
- Oncaspar (pegaspargase) injection, for intramuscular or intravenous use [prescribing information]. Westlake Village, CA: Baxalta; May 2015.
- Erwinaze (asparaginase Erwinia chrysanthemi) for injection, intramuscular or intravenous use [prescribing information]. Palo Alto, CA: Jazz Pharmaceuticals; March 2016.
- Plourde PV, Jeha S, Hijiya N, et al. Safety profile of asparaginase Erwinia chrysanthemi in a large compassionate-use trial. Pediatr Blood Cancer. 2014;61:1232-1238.
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. May 28, 2009. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/Archive/CTCAE_4.0_2009-05-29_QuickReference_8.5x11.pdf. Accessed October 10, 2018.
- Häberle J, Boddaert N, Burlina A, et al. Suggested guidelines for the diagnosis and management of urea cycle disorders. Orphanet J Rare Dis. 2012;7:32.
- Nussbaum V, Lubcke N, Findlay R. Hyperammonemia secondary to asparaginase: a case series. J Oncol Pharm Pract. 2016;22:161-164.
- Jörck C, Kiess W, Weigel JF, et al. Transient hyperammonemia due to L-asparaginase therapy in children with acute lymphoblastic leukemia or non-Hodgkin lymphoma. Pediatr Hematol Oncol. 2011;28:3-9.
- Tong WH, van der Sluis IM, Alleman CJ, et al. Cost-analysis of treatment of childhood acute lymphoblastic leukemia with asparaginase preparations: the impact of expensive chemotherapy. Haematologica. 2013;98:753-759.
- Beaupin LK, Bostrom B, Barth MJ, et al. Pegaspargase hypersensitivity reactions: intravenous infusion versus intramuscular injection-a review. Leuk Lymphoma. 2017;58:766-772.
- Salzer W, Bostrom B, Messinger Y, et al. Asparaginase activity levels and monitoring in patients with acute lymphoblastic leukemia. Leuk Lymphoma. 2018;59:1797-1806.
- Tong WH, Pieters R, Kaspers GJ, et al. A prospective study on drug monitoring of PEGasparaginase and Erwinia asparaginase and asparaginase antibodies in pediatric acute lymphoblastic leukemia. Blood. 2014;123:2026-2033.
- Panosyan EH, Seibel NL, Martin-Aragon S, et al. Asparaginase antibody and asparaginase activity in children with higher-risk acute lymphoblastic leukemia: Children’s Cancer Group Study CCG-1961. J Pediatr Hematol Oncol. 2004;26:217-226.