Allogeneic stem-cell transplant (SCT) is the standard of care for fit patients with intermediate- or high-risk myelodysplastic syndromes (MDS), intermediate- or high-risk acute myeloid leukemia (AML), or those with AML who have had a first or second complete response to therapy.1 A haploidentical transplant may be considered based on donor availability and the urgency for transplant.2
Ciurea and colleagues compared the use of haploidentical transplant and matched unrelated donor transplant in patients with AML.2 Patients received myeloablative and reduced-intensity conditioning regimens in the haploidentical transplant and matched unrelated donor cohorts. The 3-year overall survival rates were 45% in the haploidentical transplant group and 50% in the matched unrelated donor group that received myeloablative regimens (P = .38), and 46% and 44%, respectively (P = .71), in those who received reduced-intensity conditioning regimens.2
The 3-month rate of acute graft-versus-host disease (GVHD) in the myeloablative setting was significantly lower in the haploidentical transplant group than in the matched unrelated donor transplant group (16% vs 33%, respectively; P <.0001). Reduced-intensity regimens also showed similar results regarding acute GVHD in the haploidentical transplant group versus the unrelated donor group (19% vs 28%, respectively; P = .05).2
Traditionally, preparative regimens are administered on an inpatient basis because of the complexity of chemotherapy administration, the risk for severe neutropenia, and the need for isolation. In addition, the potential for acute adverse events, such as seizures or emetogenicity, associated with myeloablative regimens has also been an indication for inpatient administration.3 With proper outpatient clinical infrastructure, preparative regimens have been transitioned safely to the outpatient setting to decrease the overall cost of care, with a potential increase in patient satisfaction.4-6
In a nonrandomized, prospective cohort study, 132 patients underwent bone marrow transplant, 17 of whom were based in the outpatient setting. This study showed a significant decrease in the length of inpatient hospital days in the outpatient group versus the inpatient group (22 days vs 47 days, respectively; P <.001). The total charges for outpatient-based bone marrow transplant were 34% lower than for inpatient transplant ($104,540 vs $158,780; P = .056).4
Allogeneic SCT preparative regimens administered in the outpatient setting are primarily reported with nonmyeloablative regimens.5,7 A retrospective study of 1037 patients receiving nonmyeloablative preparative regimens in the outpatient setting before allogeneic SCT demonstrated the feasibility of outpatient administration.7 Throughout the transplant course, 47% of patients remained in the outpatient setting for the entire transplant, with a reported median hospital length of stay of 6 days in the entire patient population. The most common cause of hospitalization was infection. The study results showed a higher 5-year nonrelapse mortality rate in patients who had 1 hospitalization in the first 100 days than in patients who completed their treatment as an outpatient (26% vs 13%, respectively; P <.0001).7
Although several studies have examined the use of nonmyeloablative regimens in the outpatient setting, few have directly compared outpatient versus inpatient allogeneic SCT.7,8 Guru Murthy and colleagues compared patients who received allogeneic SCT with a reduced-intensity regimen in the outpatient setting (N = 35) versus the inpatient setting (N = 116).5 In the first 100 days, approximately 51.5% of the patients in the outpatient cohort did not require hospitalization; the 1-year nonrelapse mortality rate was 3.2% for the outpatient cohort and 10.8% for the inpatient cohort. The 1-year progression-free survival (PFS) was 63.6% and 64.4% for the outpatient and inpatient groups, respectively (P = .39), with overall survival rates of 82.8% and 73.8%, respectively (P = .93). This study also reported on cost-effectiveness, and noted a median cost of $247,334 for outpatient allogeneic SCT versus $339,621 for inpatient allogeneic SCT at 100 days after transplant (P <.001).5
In addition to a significant decrease in cost, the transition of transplants to the outpatient setting has also resulted in increased patient quality-of-life (QOL) scores. Summers and colleagues conducted a QOL study in patients undergoing autologous SCT using the Functional Assessment of Cancer Therapy-Bone Marrow Transplantation scale.6 The outpatient cohort had higher QOL scores than the inpatient cohort, indicating statistically increased emotional well-being through days 12 to 16 (P <.001) and through day 30 after the transplant (P = .01). Patients in the outpatient cohort reported a 12% higher global QOL rating than the inpatient group.6
Studies demonstrating the feasibility of outpatient administration of myeloablative preparative regimens are limited. Solomon and colleagues demonstrated the safety of outpatient administration of myeloablative preparative regimens in 100 patients undergoing allogeneic SCT with myeloablative busulfan-containing regimens or total body irradiation.3 Through day 30 after allogeneic SCT, 77% of patients were admitted to the hospital for further management, with a median length of hospital stay of 12 days (range, 1-44 days). At 4 years, the PFS rate was 48%, the overall survival rate was 50%, the nonrelapse mortality rate at 100 days was 10%, and the nonrelapse mortality rate at 6 months was 15%.3
Our study aimed to demonstrate that administering myeloablative and reduced-intensity regimens in the outpatient setting is as feasible as the administration of such regimens in the inpatient setting.
Methods
We conducted a retrospective, comparative, single-center chart review of patients who received myeloablative or reduced-intensity preparative regimens for allogeneic SCT and for haploidentical transplant in the outpatient setting versus the inpatient setting between January 1, 2015, and August 1, 2019.
The primary end point of this study was to compare the incidence of 100-day nonrelapse mortality in patients with AML or MDS who received myeloablative or reduced-intensity regimens for allogeneic SCT in the outpatient setting versus the inpatient setting. The secondary end points included hospital length of stay, 100-day PFS, time to absolute neutrophil count (ANC) and platelet engraftment, time to first neutropenic fever, the incidence of acute GVHD, and the number of hospitalizations before day 100 after allogeneic SCT.
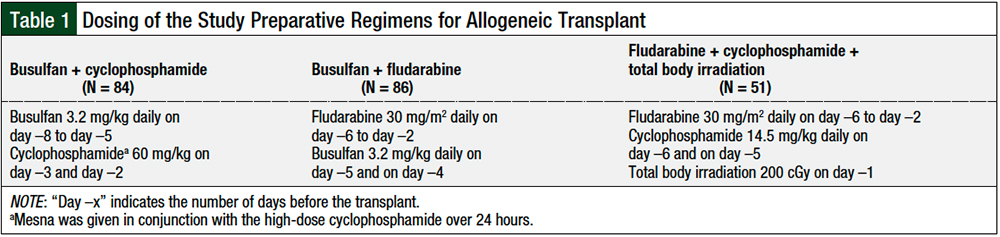
Patients were aged ≥18 years and had a diagnosis of AML or MDS. The outpatient and inpatient groups received similar preparative regimens of myeloablative busulfan plus cyclophosphamide, reduced-intensity busulfan plus fludarabine, or fludarabine plus cyclophosphamide and total body irradiation for haploidentical transplant (Table 1). Patients were excluded if they had previously had an autologous transplant or were diagnosed with undifferentiated acute leukemia, acute promyelocytic leukemia, or myelofibrosis.
At the University of Kansas Health System, the outpatient bone marrow transplant clinic is located 1.5 miles from our main hospital, with 24-hour on-call coverage, a triage call center, and designated clinical treatment algorithms to guide supportive care therapy. Per institutional protocol, all patients were admitted to the hospital the day of stem-cell administration. Patients were eligible to receive their preparative regimens in the outpatient setting, based on several factors, including age, performance status, and caregiver support.
Patients who received their preparative regimen in the inpatient setting were admitted to the hospital on the first day of the preparative regimen: 8 days before transplant for busulfan plus cyclophosphamide, 6 days before transplant for fludarabine plus cyclophosphamide and total body irradiation, and 6 days before transplant for busulfan plus fludarabine. GVHD prophylaxis and supportive care did not differ between the 2 groups.
All data were evaluated using chi-square test or Student’s t-test for parametric data. A Fisher’s exact test or Wilcoxon rank-sum test was used for nonparametric data. Alpha was set a priori at 0.05. Statistical analysis was performed using IBM SPSS Statistics Version 20.0 (IBM; Armonk, NY).
Results
In our analysis, a total of 221 patients underwent allogeneic SCT, with the underlying malignancies of AML (N = 161) and MDS (N = 60). A total of 89 patients received the preparative regimen as an outpatient and 132 received the preparative regimen as an inpatient. In the outpatient group, 15 (16.9%) patients received busulfan plus cyclophosphamide, 51 (57.3%) received busulfan plus fludarabine, and 23 (25.8%) received fludarabine plus cyclophosphamide and total body irradiation.
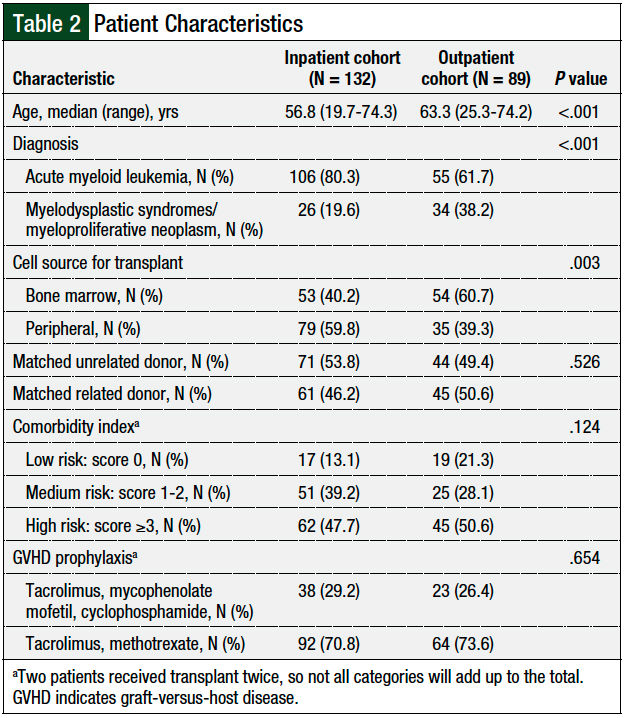
In the inpatient group, 69 (52.3%) patients received busulfan plus cyclophosphamide, 35 (26.5%) received busulfan plus fludarabine, and 28 (21.2%) received fludarabine plus cyclophosphamide and total body irradiation. The patients’ median age was 63.3 years (25.3-74.2 years) in the outpatient group and 56.8 years (19.7-74.3 years) in the inpatient group (P <.001).
Of note, there was a significant difference (P = .003) in graft sources between the 2 groups. More patients in the outpatient group received a bone marrow graft (60.7% vs 40.2%), and more patients in the inpatient group received peripheral stem cells (59.8% vs 39.3%; Table 2). There were no significant differences noted between the 2 groups in regard to the diagnosis or GVHD prophylaxis.
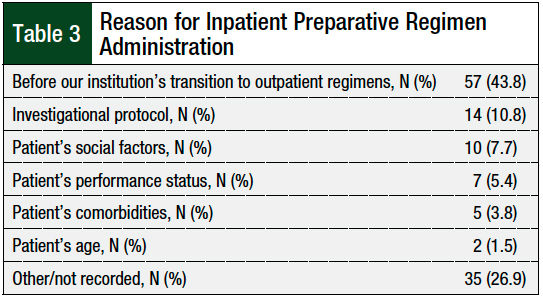
Of note, patients with a high (>3) comorbidity index score did not differ between the 2 groups, signifying that this did not preclude patients from receiving their preparative chemotherapy on an outpatient basis (50.6% for the outpatient group vs 47.7% for the inpatient group; P = .124; Table 2). The rationale for patients receiving inpatient preparative chemotherapy is shown in Table 3.
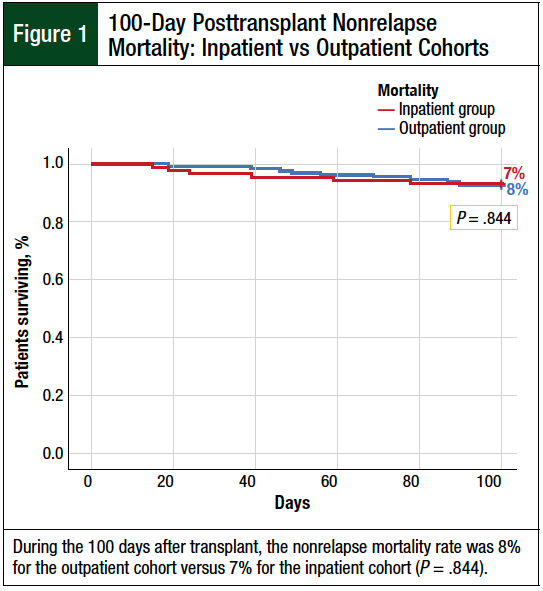
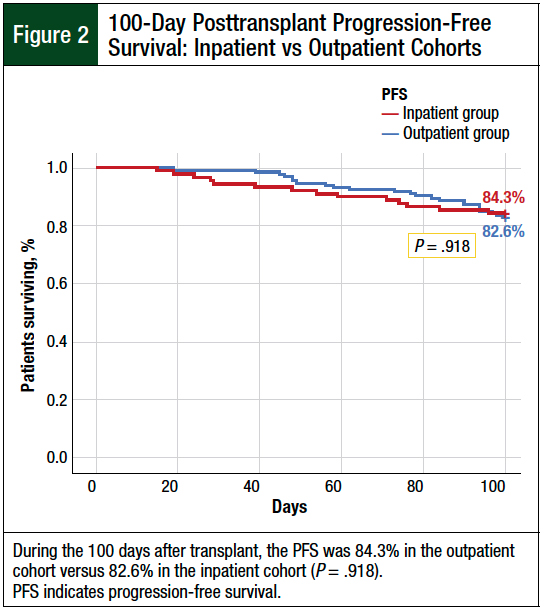
The 100-day nonrelapse mortality rate was not significantly different between the outpatient and inpatient groups (8% vs 7%, respectively; P = .844; Figure 1). The PFS at 100 days was 84.3% for the outpatient group versus 82.6% for the inpatient group (P = .918; Figure 2).
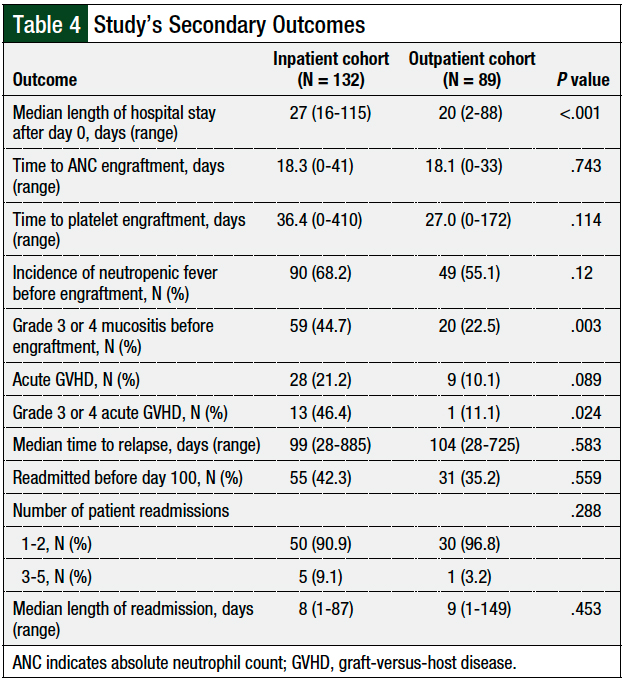
All secondary outcomes are outlined in Table 4. The median length of hospital stay within the first 100 days after day 0 (ie, the day of the transplant) was lower in the patients who received their preparative regimen in the outpatient setting versus the inpatient setting (20 days vs 27 days, respectively; P <.001; Table 4).
The times to ANC and platelet engraftment were similar between the groups (Table 4). A total of 3 patients in our study did not meet the definition of ANC engraftment (1 outpatient and 2 inpatients; P = .645), and 14 patients did not meet platelet engraftment criteria (6 outpatients and 8 inpatients; P = .838). The median time to relapse was 104 days in the outpatient cohort and 99 days in the inpatient cohort (P = .583; Table 4).
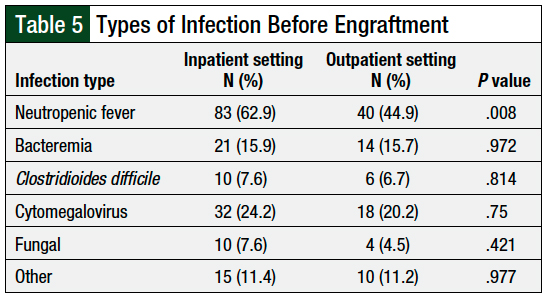
The incidence of neutropenic fever in the first 100 days was 55.1% in the outpatient group versus 68.2% in the inpatient group (P = .12), and the incidence of acute GVHD was 10.1% versus 21.2%, respectively (P = .089). There was a significant increase in the rates of neutropenic fever in the outpatient cohort compared with the inpatient cohort (44.9% vs 62.9%, respectively; P = .008; Table 5).
Of all patients with acute GVHD and mucositis, 46.4% of patients in the inpatient cohort versus 11.1% of the outpatient cohort had grade 3 or 4 acute GVHD (P = .024), and 44.7% versus 22.5% of patients in these cohorts, respectively, had grade 3 or 4 mucositis (P = .003; Table 4).
Cytomegalovirus infection within the first 100 days was similar between the outpatient and inpatient groups (20.2% vs 24.2%, respectively; P = .75). Other types of infections that occurred before engraftment are shown in Table 5.
A multiple regression analysis showed that cytomegalovirus infection was the only risk factor associated with increased mortality (P = .012).
The readmission rates before day 100 were not significantly different between the outpatient group and the inpatient group (35.2% vs 42.3%, respectively; P = .559; Table 4). The most common reason for readmission for all patients was infection (N = 58), followed by acute GVHD (N = 50). The most common cause of infection in our patients was neutropenic fever. The median length of hospital stay for a readmission was 9 days in the outpatient group versus 8 days in the inpatient group (P = .453).
The mean total cost in the outpatient setting was $516,316.64 versus $671,414.66 in the inpatient setting (P = .004).
Discussion
To our knowledge, this is the first study that directly compares these preparative regimens in inpatient and outpatient cohorts and that demonstrates that myeloablative and reduced-intensity regimens can be safely transitioned to the outpatient setting. Solomon and colleagues conducted a study of myeloablative regimens administered in the outpatient setting and included a wide variety of disease states and preparative regimens.3 That study reported a 100-day nonrelapse mortality of 10% and a median posttransplant hospital length of stay of 15 days at day 100, results that are comparable with our analysis. Of the 100 patients included in that study, 87 completed their preparative regimens in the outpatient clinic.3
Notably, the outpatient infusion clinic was connected to the hospital in the study by Solomon and colleagues, whereas in our study the outpatient infusion clinic was located approximately 1.5 miles from the acute care hospital.
In general, there are no set parameters for who receives an allogeneic transplant as an inpatient or an outpatient at our institution. Most frequently, a patient will receive the transplant as an inpatient because of a lack of transportation for the daily clinic visits.
The study by Guru Murthy and colleagues included matched inpatient and outpatient cohorts who received reduced-intensity conditioning regimens.5 Their study demonstrated similar 1-year nonrelapse mortality rates (10.8% vs 3.2%, respectively) and 1-year PFS (64.6% vs 63.6%, respectively) for the inpatient and outpatient cohorts.5 In addition, the reason for hospital readmission and the readmission rates were similar to those seen in our study.
In our analysis, the patients’ mean age was higher in the outpatient group than in the inpatient group, signifying that age did not preclude outpatient preparative conditioning administration. PFS and nonrelapse mortality were not significantly different between the 2 groups.
We anticipate that with a larger cohort of patients, the rates of nonrelapse mortality and PFS would be comparable. We performed a multiple regression analysis for overall mortality risk factors, and the only risk factor that was associated with increased mortality was cytomegalovirus infection.
Our institution’s transition to outpatient preparative regimens began in December 2015, with the busulfan plus fludarabine regimen. This was soon followed by the fludarabine plus cyclophosphamide regimen and total body irradiation in September 2016, and then, by the transition of myeloablative busulfan plus cyclophosphamide regimen in October 2017. As a result of this later transition in 2017, more patients with AML in our analysis received busulfan plus cyclophosphamide preparative chemotherapy in the inpatient unit than in an outpatient setting. There was no difference in the comorbidity index score between the groups. High-risk patients based on transplant comorbidity index were not admitted to the hospital more frequently to receive their preparative regimen (50.6% for outpatients vs 47.7% for inpatients; P = .124). The median length of hospital stay after day 0 was significantly less in the outpatient preparative regimen group by approximately 7 days (P <.001).
Preparative regimens are on average administered over 7 days. Therefore, administering regimens in the outpatient setting with admission on day 0 could correlate to significant cost-savings to an institution. Our analysis demonstrated that the transition from the inpatient setting to the outpatient setting results in significant cost-savings (P = .004). On average, the outpatient cost was $374,938.05 versus an inpatient cost of $671,414.66, providing an average cost-savings of $155,098.02 (P = .001). Reimbursement was also similarly favorable for outpatient administration versus inpatient administration.
The time to engraftment was similar between the 2 treatment groups; however, there was a higher incidence of mucositis and acute GVHD in the inpatient group than in the outpatient group. We hypothesize that this increased incidence is related to a higher percentage of busulfan plus cyclophosphamide administration in the inpatient setting versus the outpatient setting.
Readmissions before day 100 after the initial stay for transplant and the average length of hospital readmissions were similar in both groups. Infection was the primary cause of readmission through day 100, but was higher in the inpatient group than in the outpatient group.
Neutropenic fever occurred more frequently in the inpatient group and could be associated with the larger number of patients who received busulfan plus cyclophosphamide in the inpatient setting. The incidence of bacteremia, Clostridioides difficile, fungal infections, and cytomegalovirus infections did not differ between the groups; thus, outpatient administration of preparative regimens did not result in an increase in infectious complications.
The outpatient administration of preparative regimens requires a strong outpatient infrastructure for patient management. The ambulatory clinic in our institution is operational 7 days a week, including holidays, and has a robust patient triage system to support patients receiving chemotherapy as an outpatient. Logistically, our clinic uses ambulatory infusion pumps for hydration or for medications such as mesna. Beyond the operational logistics, it requires a large multidisciplinary team working together to ensure the safety of patients. This large team includes physicians, nurses, nurse practitioners, pharmacists, social workers, financial analysts, and psychologists.
Pharmacists are involved in many aspects of the preparation process for transplant, including building the preparative chemotherapy regimen into electronic medical records, determining supportive care medications, reviewing the patient’s medication list to minimize drug interactions, patient counseling, and immunosuppression monitoring after transplant. In addition, pharmacists are involved with pharmacokinetic dose adjustments for medications, such as busulfan, because our busulfan pharmacokinetics are done in the outpatient clinic.
Limitations
This study has several limitations, including its retrospective design, single-center design, and the small sample size.
We also did not take into consideration patient satisfaction and QOL data. The outpatient administration of preparative regimens allows patients to spend more time out of the hospital, which has been shown by Summers and colleagues to increase patient satisfaction and QOL.6
Although our 2 study cohorts were well-balanced in terms of age and comorbidity index score, there was a possibility of selection bias, such as potentially fewer fit patients with factors outside of the comorbidity index score who are being admitted more often.
Conclusion
Myeloablative and reduced-intensity regimens received in the outpatient setting demonstrated 100-day nonrelapse mortality rates and PFS that are comparable with the rates seen in those received in the inpatient setting. This indicates that administration of transplant-related preparatory regimens in the outpatient setting can be done safely, and at a significant cost-savings compared with the inpatient setting. Prospective, randomized studies are needed to further confirm the results of our analysis.
Author Disclosure Statement
Dr Pardo, Dr Eddy, Dr Grauer, and Dr McGuirk have no conflicts of interest to report. Dr Mahmoudjafari has participated in the Advisory Board of Omeros, Genentech, Legend Biotech, and Celgene.
References
- Majhail NS, Farnia SH, Carpenter PA, et al. Indications for Autologous and Allogeneic Hematopoietic Cell Transplantation: Guidelines from the American Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2015;21:1863-1869.
- Ciurea SO, Zhang MJ, Bacigalupo AA, et al. Haploidentical transplant with posttransplant cyclophosphamide vs matched unrelated donor transplant for acute myeloid leukemia. Blood. 2015;126:1033-1040.
- Solomon SR, Matthews RH, Barreras AM, et al. Outpatient myeloablative allo-SCT: a comprehensive approach yields decreased hospital utilization and low TRM. Bone Marrow Transplant. 2010;45:468-475.
- Rizzo JD, Vogelsang GB, Krumm S, et al. Outpatient-based bone marrow transplantation for hematologic malignancies: cost saving or cost shifting? J Clin Oncol. 1999;17:2811-2818.
- Guru Murthy GS, Hari PN, Szabo A, et al. Outcomes of reduced-intensity conditioning allogeneic hematopoietic cell transplantation performed in the inpatient versus outpatient setting. Biol Blood Marrow Transplant. 2019;25:827-833.
- Summers N, Dawe U, Stewart DA. A comparison of inpatient and outpatient ASCT. Bone Marrow Transplant. 2000;26:389-395.
- Granot N, Storer BE, Cooper JP, et al. Allogeneic hematopoietic cell transplantation in the outpatient setting. Biol Blood Marrow Transplant. 2019;25:2152-2159.
- Petersen SL, Madsen HO, Ryder LP, et al. Haematopoietic stem cell transplantation with non-myeloablative conditioning in the outpatient setting: results, complications and admission requirements in a single institution. Br J Haematol. 2004;125:225-231.