In the United States, prostate cancer has the second highest rate of new cancer cases per year and is responsible for the third highest rate of cancer deaths annually, based on 2018 data (the most recent incidence data available).1 In a 2010 report on the incidence of cancer in US veterans, prostate cancer had the highest rate of all cancers, accounting for approximately 30% of all cancers among veterans.2
The treatment options for prostate cancer range from active surveillance to surgical intervention, radiation therapy, or medication therapy, depending on the severity of the disease.3 Androgens, such as testosterone and dihydrotestosterone, are thought to be the key drivers of prostate cancer.4
When the decision is made to manage prostate cancer with medications, the National Comprehensive Cancer Network (NCCN) guidelines recommend treatment with androgen-deprivation therapy (ADT) to achieve medical castration.3 Medical castration is defined as having a serum testosterone level of ≤50 ng/mL.5
In some patients, prostate cancer will progress and metastasize despite maintaining castrate levels of testosterone; in these cases, the disease is defined as metastatic castration-resistant prostate cancer (CRPC). Docetaxel had been the standard treatment for patients with metastatic CRPC until the introduction of 2 then-novel hormonal therapies, abiraterone, which was approved by the US Food and Drug Administration (FDA) in 2011, and enzalutamide, which was approved in 2012.
Abiraterone was first approved for the treatment of metastatic CRPC in the post-docetaxel setting based on a phase 3, randomized, placebo-controlled study that showed significantly improved overall survival versus docetaxel (14.8 months vs 10.9 months, respectively; hazard ratio [HR], 0.65; 95% confidence interval [CI], 0.54-0.77; P <.001).6
In a clinical trial of patients with metastatic CRPC, the use of abiraterone in the pre-docetaxel setting resulted in significant increases in the co-primary end points of radiographic progression-free survival (PFS) compared with placebo (16.5 months vs 8.3 months, respectively; HR, 0.53; 95% CI, 0.45-0.62; P <.001) and overall survival (median not reached vs 27.2 months, respectively; HR, 0.75; 95% CI, 0.61-0.93; P = .01).7
Shortly thereafter, enzalutamide was approved by the FDA based on similar results from 2 studies that showed improved overall survival with enzalutamide versus placebo in the post-docetaxel (18.4 months vs 13.6 months, respectively; HR, 0.63; 95% CI, 0.53-0.75; P <.001)8 and pre-docetaxel (median not reached vs 31 months, respectively; HR, 0.73; 95% CI, 0.63-0.85; P <.001)9 settings.
The current NCCN guidelines recommend the use of abiraterone or enzalutamide as first-line treatment options for metastatic CRPC, without a preference for one drug over the other.3
After disease progression with abiraterone or with enzalutamide treatment, it is less clear what the optimal course of treatment should be for metastatic CRPC. One strategy is to switch to the other drug that has not previously been tried among these 2 drugs. The current data suggest that some cross-resistance exists between abiraterone and enzalutamide, attenuating the response rates when the disease progresses after receiving one of these agents.10,11
Remaining unanswered issues include which sequence of these 2 agents is optimal, and what is the benefit of the second drug. Several retrospective studies have previously compared the sequence of abiraterone before enzalutamide (abiraterone-enzalutamide) and enzalutamide before abiraterone (enzalutamide-abiraterone), with mixed results.12-15 More recently, the first prospective, randomized clinical trial was published assessing the impact of different sequences of enzalutamide and abiraterone on treatment outcomes.16
To date, few studies have been published that have examined the sequencing of abiraterone and enzalutamide in the veteran population with metastatic CRPC.17-19 Veterans represent a unique population with complex psychosocial concerns that may affect their care relative to the civilian population.20 A cancer diagnosis may even be directly related to a veteran’s period of service in some cases.
Many veterans who served in the Vietnam War era might have been exposed to a tactical herbicide known as Agent Orange. Previous studies have shown that Agent Orange exposure is associated with an increased incidence of prostate cancer, and with the cancer being more aggressive.21,22 Furthermore, the rate of metastatic prostate cancer at diagnosis is higher within the veteran population than in nonveteran men.2
The goal of the present study was to evaluate the best sequencing of abiraterone and enzalutamide in the veteran population with metastatic CRPC.
Methods
This study was a single-center, retrospective chart review of patients with metastatic CRPC who received treatment at VA Northeast Ohio Healthcare System between April 28, 2011, and October 31, 2019. A Veterans Information Systems and Technology Architecture medication report identified any patients within the VA Northeast Ohio Healthcare System who had filled at least 1 outpatient prescription for abiraterone and enzalutamide during the study period. We collected information from the patient charts within the electronic medical record (EMR) to determine if the patient met the study criteria for inclusion.
Patients included in the study were adult veterans aged ≥18 years with a diagnosis of metastatic CRPC who had received treatment with abiraterone and enzalutamide and received the first dose of the second sequential therapy by January 1, 2019, to allow time for adequate follow-up during the study period.
Patients were excluded from the study if they received any drug therapies for prostate cancer treatment (other than background ADT) in between or concurrently with abiraterone and enzalutamide. Patients were also excluded if they received treatment with abiraterone or enzalutamide outside of the VA Northeast Ohio Healthcare System.
The patients were divided into 2 groups based on the sequence of abiraterone and enzalutamide that they received. One group received abiraterone before enzalutamide and the second group received enzalutamide before abiraterone. The EMR was used for the patients’ baseline and outcomes data collection. The baseline information included the patient’s age at the index date (ie, the date of the first fill for the first sequential therapy), the Gleason score at diagnosis, the Eastern Cooperative Oncology Group performance status score within 30 days of the index date and the second-line therapy initiation, and the number and type of previous systemic therapies received.
This study was conducted in compliance with the Department of Veterans Affairs (VA) requirements and received VA Research and Development Committee approval. Based on its design, this study was determined to meet the criteria for Cleveland VA Institutional Review Board exemption.
The primary study outcome was combined PSA-PFS, which was defined as the sum of time from initiation of therapy with abiraterone or enzalutamide (as the first sequential drug) to PSA progression and initiation of the second sequential drug with abiraterone or enzalutamide to additional PSA progression. PSA progression was defined based on the Prostate Cancer Work Group 3 criteria, which is an increase of 25% (absolute increase of ≥2 ng/mL) in PSA from nadir, or death.5
The secondary outcomes included combined radiographic PFS, defined as the sum of time to radiographic progression (as determined by the radiologist interpretation in the medical record, or death) after receiving abiraterone and enzalutamide in sequence, and overall survival (defined as the time from initiation of the first therapy to death). The other secondary outcomes included the individual components of combined PSA-PFS and combined radiographic PFS, and medication adherence as determined by medication possession ratios (MPRs).23 The MPRs were capped at 1.0 to limit the skewing of numbers by patients filling medications early. We also collected information on any adverse events that led to treatment discontinuation.
Statistical Analysis
The primary outcome of combined PSA-PFS was assessed for between-group differences using a Kaplan-Meier survival analysis, with a log-rank test for significance. A priori power analysis was used for the Kaplan-Meier analysis. We determined that a sample size of 85 patients, with an alpha of .05 and a power of 80%, would be able to detect an estimated medium effect size of 0.39 (which was derived from the weighted average of previous studies).12-15
HRs were obtained from a univariate Cox proportional hazards analysis. The same statistical tests were used for the secondary outcomes.
For the combined PSA-PFS and combined radiographic PFS, the patients who had discontinued treatment because of adverse events were excluded from the statistical analysis. For overall survival, all patients were included, regardless of any therapy change because of adverse events or disease progression.
The MPR was assessed by a Mann-Whitney U test, because the data were not normally distributed. Baseline characteristics were assessed by a chi-square test or a Student’s t-test, where appropriate.
For all tests, P <.05 was considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics version 26 (IBM Corporation; Armonk, NY).
Results
A total of 109 patients with metastatic CRPC were identified at the VA Northeast Ohio Healthcare System who had received outpatient prescriptions for abiraterone and enzalutamide. Of those patients, 77 met the study criteria and were assigned to 1 of the 2 treatment groups.
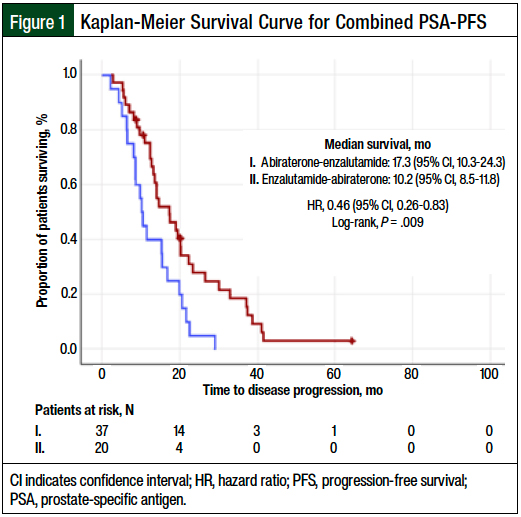
The baseline characteristics of both groups were similar, except for Gleason score at diagnosis (Table 1). A greater percentage of patients in the abiraterone-before-enzalutamide group had a Gleason score of 8 to 10 at diagnosis, indicating worse disease severity at baseline.
Most patients had no previous systemic therapy (excluding ADT) for metastatic CRPC. In patients who did receive previous systemic therapy, docetaxel was the most often used drug. The prescribed doses of abiraterone and enzalutamide were concordant with the normal starting doses of the 2 medications, with most patients receiving abiraterone at a dose of 1000 mg daily (94.8%) and enzalutamide at a dose of 160 mg daily (97.4%).
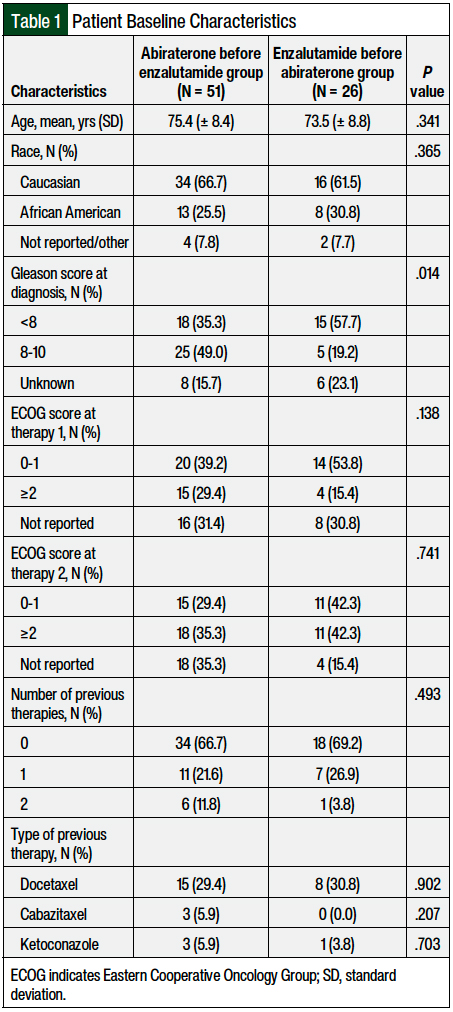
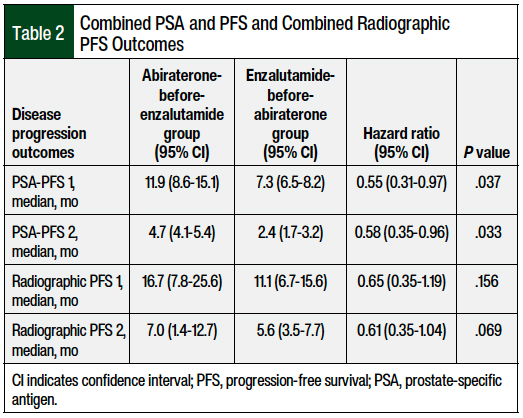
A total of 57 patients had an evaluable combined PSA-PFS (37 patients in the abiraterone-before-enzalutamide group and 20 in the enzalutamide-before-abiraterone group). In a Kaplan-Meier analysis, the median combined PSA-PFS was 17.3 months (95% CI, 10.3-24.3 months) for the abiraterone-before-enzalutamide group and 10.2 months (95% CI, 8.5-11.8 months) for the enzalutamide-before-abiraterone group (log-rank P = .009). The difference was significantly longer in the abiraterone-before-enzalutamide group than in the other group, with an unadjusted Cox regression (HR, 0.46; 95% CI, 0.26-0.83; Figure 1). For each component of the combined primary outcome of PSA-PFS, the abiraterone-before-enzalutamide group had a significantly longer time to progression than the other group (Table 2).
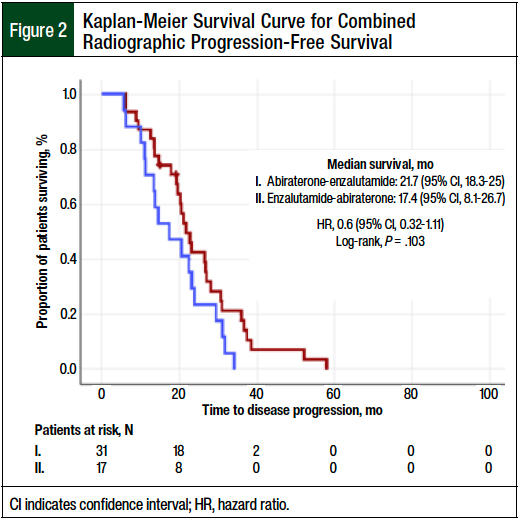
For the combined radiographic PFS, 48 patients had complete evaluable radiographic data (31 patients in the abiraterone-before-enzalutamide group and 17 in the enzalutamide-before-abiraterone group). The median combined radiographic PFS was 21.7 months (95% CI, 18.3-25 months) in the abiraterone-before-enzalutamide group and 17.4 months (95% CI, 8.1-26.7 months) in the other group, a nonsignificant difference (HR, 0.6; 95% CI, 0.32-1.11; log-rank P = .103; Figure 2). The components of the combined radiographic PFS were also not significantly different between the 2 cohorts (Table 2).
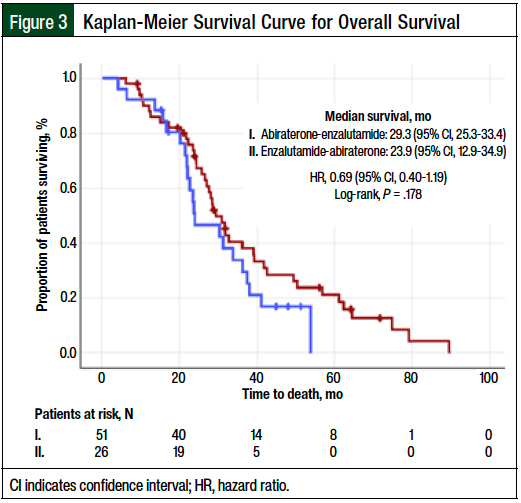
All 77 patients were evaluable for overall survival (51 patients in the abiraterone-before-enzalutamide group and 26 in the second group). The overall survival was not significantly different between the 2 groups (abiraterone before enzalutamide: median, 29.3 months; 95% CI, 25.3-33.4 months; enzalutamide before abiraterone: median, 23.9 months; 95% CI, 12.9-34.9 months; HR, 0.69; 95% CI, 0.4-1.19; log-rank P = .178; Figure 3).
Overall, medication adherence, as measured by MPR, was very high. The median MPR for abiraterone was 98% (interquartile range [IQR], 82.6%-100%) and for enzalutamide was 98.3% (IQR, 90.2%-100%). No significant differences were found in the MPR of the 2 drugs between the 2 treatment sequences (P = .402 for abiraterone’s MPR between the treatment sequence groups, and P = .567 for enzalutamide’s MPR between the treatment sequence groups).
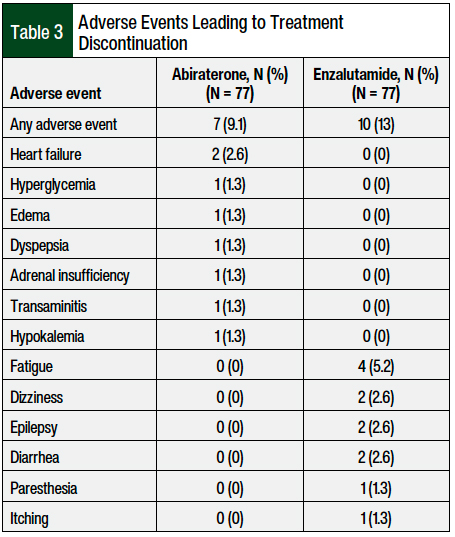
Overall, the rates of patients who had adverse events leading to treatment discontinuation were 9.1% for abiraterone and 13% for enzalutamide (Table 3). Each medication had unique adverse events. The only adverse event with more than 1 occurrence with abiraterone was heart failure (N = 2). The most common adverse event leading to treatment discontinuation in the enzalutamide group was fatigue (N = 4), followed by dizziness, epilepsy, and diarrhea (N = 2 each; Table 3).
Discussion
Among veterans with metastatic CRPC, receiving the sequence of abiraterone before enzalutamide was associated with a significantly longer combined PSA-PFS than receiving the sequence of enzalutamide before abiraterone. No significant difference was observed in combined radiographic PFS or in overall survival between the 2 groups, but this could be a result of the limited sample size and power of the study.
The 2 study groups were well-matched at baseline, with the exception of the Gleason score. The abiraterone-before-enzalutamide group had a greater proportion of patients with a higher histopathologic grade at the time of diagnosis, which resulted in the potential to skew the results in favor of enzalutamide before abiraterone, because these patients could be expected to have more rapid disease progression. The results showed that despite this baseline imbalance, the patients in the abiraterone-before-enzalutamide group had a greater combined PSA-PFS than the patients in the enzalutamide-before-abiraterone group, which enhances the interpretation of the results.
Previous retrospective studies have examined the optimal sequencing of abiraterone and enzalutamide in patients with metastatic CRPC.12-14 A study by Maughan and colleagues included 81 patients with metastatic CRPC who either previously received or did not receive chemotherapy at Johns Hopkins Comprehensive Cancer Center and received abiraterone and enzalutamide in sequence.12 The combined clinical or radiographic PFS results were significantly better with the abiraterone-before-enzalutamide sequence over enzalutamide before abiraterone in a multivariable propensity-weighted model (19.5 months vs 13 months, respectively; HR, 0.37; 95% CI, 0.22-0.64; P <.001).12
In a retrospective study, Terada and colleagues examined a cohort of patients with metastatic CRPC from the Maughan and colleagues study, but also included patients from Kyoto University Hospital in Japan, for a total of 113 patients.12 The combined PSA-PFS was significantly longer in the abiraterone-before-enzalutamide group than in the enzalutamide-before-abiraterone group (37.9 months vs 24.7 months, respectively; HR, 0.56; 95% CI, 0.41-0.76; P <.001).13
In a study by Miyake and colleagues, 108 chemotherapy-naïve patients with metastatic CRPC received sequential treatment with abiraterone before enzalutamide or with enzalutamide before abiraterone. The results were similar to those of the previously mentioned studies,11,12 with the outcome of combined PSA-PFS favoring abiraterone before enzalutamide versus enzalutamide before abiraterone (18.4 months vs 12.8 months, respectively; HR, 0.44; 95% CI, 0.37-0.81; P = .009).14
Our study’s results were consistent with the results of the studies showing a longer combined PSA-PFS with the sequence of abiraterone before enzalutamide versus enzalutamide before abiraterone.13,14 The results of Maughan and colleagues also favored the abiraterone-before-enzalutamide sequence versus enzalutamide before abiraterone, although a slightly different end point of combined radiographic and clinical progression was used in that study.12 When these studies assessed the individual lines of therapy, a significant difference between the 2 sequences was only seen with the time to progression while receiving second-line therapy.12-14
By contrast, our study’s results showed a significant difference in the time to progression for the first- and second-line therapies in favor of the abiraterone-before-enzalutamide sequence versus enzalutamide before abiraterone.
Matsubara and colleagues examined 97 chemotherapy-naïve patients with metastatic CRPC who received sequential abiraterone before enzalutamide or enzalutamide before abiraterone from several institutions in Japan.15 No significant difference was seen in the median combined PFS (defined as radiographic and
PSA progression) between the abiraterone-before-enzalutamide and enzalutamide-before-abiraterone groups (11.1 months vs 9.04 months, respectively; HR, 0.71; 95% CI, 0.46-1.08; P = .105), although there was a numerical difference in favor of abiraterone before enzalutamide.15
Matsubara and colleagues proposed that the different results were because their study only included chemotherapy-naïve patients, had a more equal sample size in both sequence groups, and used an outcome incorporating radiographic and PSA data.15 Our study also differs on these points, which may explain the differences in the results. Nonetheless, the accumulation of evidence from retrospective studies, including the present study, favors the sequence of abiraterone before enzalutamide.
Since completing our retrospective study, the first prospective, randomized clinical trial was published examining the impact of abiraterone and enzalutamide sequencing on disease progression.16 Khalaf and colleagues enrolled 202 patients with newly diagnosed metastatic CRPC who were randomly assigned in a 1:1 ratio to abiraterone plus prednisone or to enzalutamide as first-line therapy, until PSA progression occurred, which required crossover to the other group.
The results showed that the time to the second PSA progression was longer in the abiraterone-before-enzalutamide group than in the enzalutamide-before-abiraterone group (median, 19.3 months vs 15.2 months, respectively; HR, 0.66; 95% CI, 0.45-0.97; P = .036). In addition, the PSA response (>30% decrease in PSA from baseline) rates to second-line therapy were higher with second-line enzalutamide (36%) than with abiraterone (4%; P <.0001). The overall survival was not significantly different between the 2 groups.16
Although combined PSA-PFS was not measured in this study, these results add support for the optimal sequence of abiraterone before enzalutamide, because enzalutamide in the second-line setting was associated with longer time to PSA progression and retained some PSA response rates after previous abiraterone exposure.16
The evidence cited here indicates that enzalutamide compared with abiraterone retains greater activity in the second-line setting, leading to a greater combined PSA-PFS when assessing the entire sequence of therapies. One hypothesis of why variation in the sequencing of these agents affects outcomes is related to the mechanisms of resistance with these agents.10,11 Some resistance mechanisms are common between abiraterone and enzalutamide, which may explain the overall blunted response rate seen with their sequencing.10,11 These common mechanisms include the upregulation of the androgen receptor as a result of gene amplification, and androgen receptor splice variants that are constitutively active without regard for ligand binding.24
Other mechanisms of resistance are unique to each drug. Abiraterone is an inhibitor of cytochrome (CY) P-450 isoform 17A1 (CYP17A1), which decreases the conversion of androgen precursors to testosterone and dihydrotestosterone.25 The inhibition of CYP17A1 can increase the levels of upstream steroid precursors, which in turn may activate the androgen receptor in the presence of mutations to bypass CYP17A1 blockade effectively.25 Two such mutations are the L701H mutation, which leads to the androgen receptor activation by glucocorticoids, and the T878A mutation, which leads to activation by progesterone.24
Enzalutamide, a high-affinity androgen receptor antagonist and blocker of androgen receptor nuclear translocation, also has unique resistance mechanisms.25 These include the F876L mutation, which causes an antagonist to agonist switch for enzalutamide, and a bypass pathway with glucocorticoid receptor induction and expression of androgen receptor gene targets.24
Unique resistance mechanisms may explain why some patients respond to second-line abiraterone or enzalutamide treatment after initial disease progression with one of these agents, and why enzalutamide has demonstrated a longer time to second PSA progression than abiraterone. Our study also showed a significant difference in the time to first PSA progression in favor of the abiraterone-before-enzalutamide sequence, which could be related to some mechanism other than cross-resistance.
Future treatment decisions may ultimately rely on biomarkers to be able to choose the ideal therapy. One particular biomarker of interest is androgen receptor splice variant 7 messenger RNA, which has been associated with a lack of response to abiraterone and enzalutamide treatment.26 In addition, an ongoing clinical trial is investigating circulating tumor DNA as a way to predict therapy efficacy with enzalutamide or with docetaxel after exposure to abiraterone.27
Limitations
Our study had several limitations as a retrospective review. First, the potential for missing data or gaps in patient follow-up in the EMR limits the ability to calculate some of the progression outcomes accurately. These outcomes are dependent on the measurement of a time to an event, which can only be determined once a patient has laboratory test results or scans. For patients with long follow-up gaps, we cannot rule out that disease progression occurred at an earlier point.
In addition, outcomes such as determining radiographic progression and adverse events were limited to the extent of interpretation and documentation of the treating physician at the time.
Furthermore, we cannot definitively state any true cause-and-effect relationship based on the results, but merely potential associations.
Finally, the study’s small sample size, especially for the enzalutamide-before-abiraterone treatment sequence, increases the chance of a type II error, and the differences observed between the groups could be the result of not having a large enough study population. Nevertheless, this retrospective review does reflect real-world medical practice; there will always be differing treatment practices among providers, and for various reasons, patients may not have laboratory tests and scans at precise intervals.
Conclusion
Our findings show that in veterans with metastatic CRPC, abiraterone followed by enzalutamide has a longer combined PSA and PFS than enzalutamide followed by abiraterone. Because of the potential for cross-resistance between these medications, consideration should be given to other alternative treatments after disease progression, such as taxane chemotherapy, although optimal sequencing of the entire spectrum of agents needs further research.
Because abiraterone, enzalutamide, and other androgen receptor axis inhibitors are used earlier in the course of prostate cancer treatment, the investigation of optimal sequencing becomes increasingly important.
Additional research is needed for the optimal treatment of patients with metastatic CRPC who have disease progression after receiving first-line therapies. When patients with metastatic CRPC receive sequential treatment with abiraterone and enzalutamide, the sequence of abiraterone followed by enzalutamide may need to be considered based on the results of our study and the results of previous studies.
Author Disclosure Statement
Dr Newman, Dr Gordon, Dr Nock, Dr Burant, and Dr Wenzell have no conflicts of interest to report.
References
- Centers for Disease Control and Prevention. Cancer statistics at a glance: leading cancer cases and deaths, all races and ethnicities, male and female, 2018. United States Cancer Statistics Data Visualizations Tool. June 2020. https://gis.cdc.gov/Cancer/USCS/DataViz.html. Accessed September 22, 2020.
- Zullig LL, Sims KJ, McNeil R, et al. Cancer incidence among patients of the U.S. Veterans Affairs health care system: 2010 update. Mil Med. 2017;182:e1883-e1891.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Prostate Cancer. Version 2.2022. November 30, 2021. www.nccn.org/professionals/physician_gls/pdf/prostate.pdf. Accessed January 20, 2022.
- Dai C, Heemers H, Sharifi N. Androgen signaling in prostate cancer. Cold Spring Harb Perspect Med. 2017;7:a030452. doi: 10.1101/cshperspect.a030452.
- Scher HI, Morris MJ, Stadler WM, et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the Prostate Cancer Clinical Trials Working Group 3. J Clin Oncol. 2016;34:1402-1418.
- de Bono JS, Logothetis CJ, Molina A, et al; for the COU-AA-301 investigators. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
- Ryan CJ, Smith MR, de Bono JS, et al; for the COU-AA-302 investigators. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2013;368:138-148. Erratum in: N Engl J Med. 2013;368:584.
- Scher HI, Fizazi K, Saad F, et al; for the AFFIRM investigators. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187-1197.
- Beer TM, Armstrong AJ, Rathkopf DE, et al; for the PREVAIL investigators. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424-433.
- Azad AA, Eigl BJ, Murray RN, et al. Efficacy of enzalutamide following abiraterone acetate in chemotherapy-naive metastatic castration-resistant prostate cancer patients. Eur Urol. 2015;67:23-29.
- Loriot Y, Bianchini D, Ileana E, et al. Antitumour activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide (MDV3100). Ann Oncol. 2013;24:1807-1812.
- Maughan BL, Luber B, Nadal R, Antonarakis ES. Comparing sequencing of abiraterone and enzalutamide in men with metastatic castration-resistant prostate cancer: a retrospective study. Prostate. 2017;77:33-40.
- Terada N, Maughan BL, Akamatsu S, et al. Exploring the optimal sequence of abiraterone and enzalutamide in patients with chemotherapy‐naïve castration‐resistant prostate cancer: the Kyoto‐Baltimore collaboration. Int J Urol. 2017;24:441-448.
- Miyake H, Hara T, Tamura K, et al. Comparative assessment of efficacies between 2 alternative therapeutic sequences with novel androgen receptor-axis-targeted agents in patients with chemotherapy-naïve metastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 2017;15:e591-e597.
- Matsubara N, Yamada Y, Tabata KI, et al. Abiraterone followed by enzalutamide versus enzalutamide followed by abiraterone in chemotherapy-naive patients with metastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 2018;16:142-148.
- Khalaf DJ, Annala M, Taavitsainen S, et al. Optimal sequencing of enzalutamide and abiraterone acetate plus prednisone in metastatic castration-resistant prostate cancer: a multicentre, randomised, open-label, phase 2, crossover trial. Lancet Oncol. 2019;20:1730-1739.
- Leuva H, Sigel K, Zhou M, et al. A novel approach to assess real-world efficacy of cancer therapy in metastatic prostate cancer. Analysis of national data on Veterans treated with abiraterone and enzalutamide. Semin Oncol. 2019;46:351-361.
- Goldberg H. ASCO GU 2020: Overall survival in men with chemotherapy-naïve metastatic castration-resistant prostate cancer receiving bicalutamide followed by enzalutamide or abiraterone. www.urotoday.com/conference-highlights/asco-gu-2020/asco-gu-2020-prostate-cancer/119328-asco-gu-2020-overall-survival-in-men-with-chemotherapy-naive-metastatic-castration-resistant-prostate-cancer-receiving-bicalutamide-followed-by-enzalutamide-or-abiraterone.html. Accessed June 14, 2022.
- Freedland SJ, De Hoedt AM, DerSarkissian M, et al. Veterans Affairs prostate cancer treatment sequencing (VAPCaTS): a real-world evidence study of men with metastatic prostate cancer. Urol Pract. 2021;8:112-118.
- Olenick M, Flowers M, Diaz VJ. US veterans and their unique issues: enhancing health care professional awareness. Adv Med Educ Pract. 2015;6:635-639.
- Ansbaugh N, Shannon J, Mori M, et al. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119:2399-2404.
- Chamie K, deVere White RW, Lee D, et al. Agent Orange exposure, Vietnam War veterans, and the risk of prostate cancer. Cancer. 2008;113:2464-2470.
- Lam WY, Fresco P. Medication adherence measures: an overview. Biomed Res Int. 2015;2015:217047.
- Buttigliero C, Tucci M, Bertaglia V, et al. Understanding and overcoming the mechanisms of primary and acquired resistance to abiraterone and enzalutamide in castration resistant prostate cancer. Cancer Treat Rev. 2015;41:884-892.
- Giacinti S, Bassanelli M, Aschelter AM, et al. Resistance to abiraterone in castration-resistant prostate cancer: a review of the literature. Anticancer Res. 2014;34:6265-6269.
- Antonarakis ES, Lu C, Wang H, et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med. 2014;371:1028-1038.
- Prostate Cancer Treatment Optimization via Analysis of Circulating Tumour DNA (PROTRACT). NLM identifier: NCT04015622. Updated January 21, 2022. https://clinicaltrials.gov/ct2/show/NCT04015622. Accessed January 24, 2022.