Fusariosis is an opportunistic fungal infection leading to significant morbidity and mortality in immunocompromised patients with cancer. More than 50 species of Fusarium have been identified, but not all are known to cause infections in humans.1,2 The most frequently isolated species reported in previous literature is Fusarium solani (50% of cases), which account for most cases of fusarial keratitis, followed by Fusarium oxysporum (approximately 20% of cases), which account for most cases of onychomycosis.2-4
Fusarium infections share many features with invasive aspergillosis and other mold infections, including in patients with prolonged neutropenia or corticosteroid use.2 The opportunistic nature of these infections leads to significant morbidity and mortality in the pediatric immunocompromised population.1
The overall incidence of fusariosis in patients undergoing hematopoietic stem-cell transplant (HSCT) is approximately 6 cases per 1000, and has been reported to occur in a trimodal distribution after transplant.2 The initial peak occurs immediately after transplant, in association with profound neutropenia, whereas the second and third peaks occur at an approximate median of 65 days.2,5,6 The second and third peaks result from profound T-cell immunodeficiency related to prolonged corticosteroid use for treatment of graft-versus-host disease.5,6 This suggests that innate immunity plays a major role in the defense mechanisms against mold infections and correlates with the higher death rates from fusariosis in patients receiving prolonged therapy with corticosteroids.7
The clinical manifestations often correlate with the degree of immune function.5 In individuals with some preservation of immune function, Fusarium infection may present as colonization of local regions, although approximately 40% to 60% of cases in immunocompromised hosts present as acute invasive or disseminated disease.1
Disease severity is also dependent on the entry site of the infection. The presentation of Fusarium infection reflects the entry site, most often with the presentations of keratitis or onychomycosis, but sinusitis, endophthalmitis, pneumonia, thrombophlebitis, fungemia, septic arthritis, osteomyelitis, or disseminated infection are also possible.2
The diagnosis of Fusarium infection requires identification and isolation of the pathogen, with consideration that many opportunistic molds may demonstrate resistance to antifungal agents.1 Two characteristic presentations that suggest the diagnosis of disseminated fusariosis in an immunocompromised patient are skin lesions and positive blood cultures for mold.2
Accurate cultures require isolation of several colonies from the same specimen or isolation of the same fungus from different specimens, because of the risk for contamination. However, confirmatory diagnosis of fusariosis may rely on histopathology. In many cases, the hyphae of Fusarium infection are similar to those of Aspergillus infection, but the finding of hyphae and yeastlike structures together is more suggestive of Fusarium infection.2 The clear limitations with these diagnostic methods may ultimately restrict the ability of providers to treat adequately these patients, which in turn limits favorable outcomes.
The mortality attributable to Fusarium infections in immunocompromised patients ranges from 50% to 70%, with prognosis directly correlated with the degree of persistent immunodeficiency.1 An analysis of 84 patients with hematologic disease showed a 30-day survival rate of 50% and a 90-day survival rate of 21% after invasive Fusarium diagnosis.7 Among the subgroup of HSCT recipients in this analysis, the 90-day survival rate was 13%, and the single poor predictor of outcome was persistent neutropenia.7
In addition, an analysis of 259 cases of fusariosis revealed that 100% of the patients who were persistently neutropenic most often because of cancer therapy had died. Among the entire cohort of patients in the study, those with disseminated infection also had a higher mortality rate compared with those who did not have disseminated infection (76% vs 39%, respectively; P <.0001).8 Considering these high mortality rates in the immunocompromised population,1,8 management of infection must begin promptly after diagnosis.
In general, patients with localized Fusarium infections benefit from surgical debridement of the wound, whereas patients with invasive or disseminated disease require immediate treatment with systemic therapy.2 Differing patterns of susceptibility make high-dose amphotericin B the treatment of choice for patients with F solani and F verticillioides, whereas the treatment of choice for other Fusarium species also includes voriconazole. Susceptibility testing should be performed to rule out resistance and confirm the most appropriate therapy for the individual patient.2
Although case series are available for preferred treatment, the optimal treatment of Fusarium infection remains unclear, suggesting that an escalation to combination, salvage, or adjunctive therapies may be warranted.9-13 Data relating to combination therapy for fusariosis are limited to case reports, but the use of amphotericin B in combination with caspofungin, voriconazole, posaconazole, or terbinafine has been documented.9-13
Adjunctive therapies, such as granulocyte-macrophage colony-stimulating factor and granulocyte infusions, are also documented in case reports14-16; but similar to combination therapy, no significant data exist to support routine use of adjunctive therapies.
Patient 1
A 6-year-old male with stage IV neuroblastoma had undergone autologous HSCT as part of his consolidation therapy. The presence of transplant-associated thrombotic microangiopathy complicated his second autologous HSCT.
As part of his therapy for transplant-associated thrombotic microangiopathy, the patient received multiple doses of eculizumab, further increasing the degree of his immunosuppression. He had several central access devices, including a double-lumen broviac catheter and a hemodialysis catheter, because of his worsening renal function.
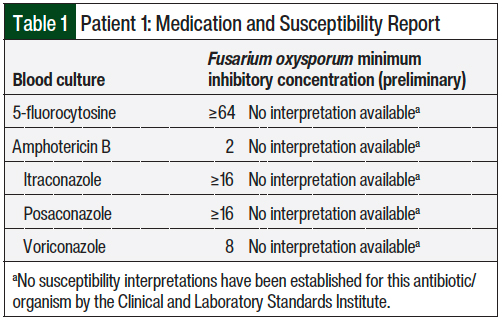
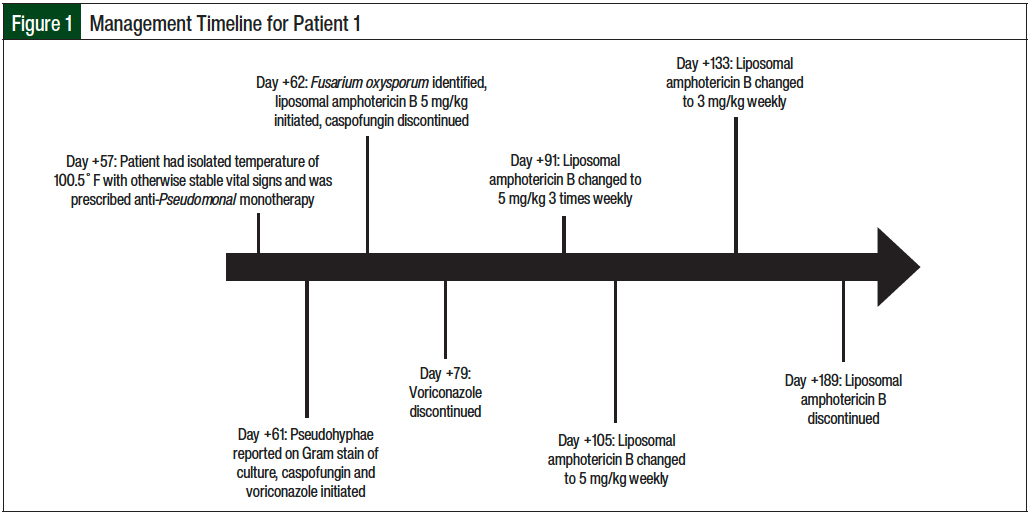
On day 57 after the second HSCT, the patient had an isolated temperature of 100.5˚F, with otherwise stable vital signs, and he was initiated treatment with anti-Pseudomonal monotherapy. The patient remained clinically stable, with no additional fevers, until blood cultures from 1 lumen of the broviac catheter showed pseudohyphae on the Gram stain on day 61 after the second transplant. F oxysporum was identified, and the culture and susceptibility report demonstrated his condition was highly resistant to several antifungal agents (Table 1).
Repeated blood cultures collected after the identification of the Fusarium species showed positive results on 2 additional cultures 5 and 6 days after the initial positive culture. Furthermore, the first and the second additional cultures grew F oxysporum 7 and 8 days, respectively, later. All positive blood cultures were collected before the patient started therapy with liposomal amphotericin B (AmBisome) and before the removal of the broviac catheter.
We swiftly initiated treatment of F oxysporum when the culture came back. After the initial doses of caspofungin and voriconazole on day 61 and a review of the available literature, the patient started treatment with the combination of liposomal amphotericin B and voriconazole (Figure 1).
Intravenous (IV) liposomal amphotericin B was dosed at 5 mg/kg every 24 hours for the first 4 weeks of treatment. Oral voriconazole was initially dosed at 9 mg/kg every 12 hours. We monitored the drug therapy closely, with the goal of attaining target trough levels of >2 mcg/mL. After 1 week of therapy, a voriconazole trough was <0.1 mcg/mL, and the dose was subsequently increased by approximately 33%, to 12 mg/kg orally every 12 hours.
After another week of therapy, the voriconazole trough level was again less than the desired range, at 0.6 mcg/mL. With consultation from the infectious diseases service, we made the decision to discontinue voriconazole because the patient continued to do well clinically, in conjunction with the high minimal inhibitory concentration of voriconazole observed in the F oxysporum susceptibilities report.
After receiving 4 weeks of liposomal amphotericin B therapy, the patient slowly transitioned to a prophylactic regimen, as shown in Figure 1. The prophylactic liposomal amphotericin B therapy was discontinued once the patient’s absolute CD4 count was more than 200/µL on 2 separate occasions. The patient remains clinically well and free from any complications of the F oxysporum fungemia.
The patient retained the hemodialysis catheter throughout the entire antifungal course, as a result of his need for renal replacement therapy. He underwent whole body imaging using computed tomography (CT) and magnetic resonance imaging (MRI). The only positive finding on the CT and MRI was maxillary sinus disease, although the results were not indicative of fungal disease.
No skin lesions were observed on the patient throughout the disease course. An ophthalmologic examination was negative for fungal disease. After extensive workup, no occult source of fungal infection beyond the fungemia was noted, and the patient remained clinically stable, with a temperature of ≤100.4°F.
Patient 2
Our second case report of an invasive Fusarium infection involves a 13-year-old male (weight, 103 kg) with high-risk B-cell acute lymphoblastic leukemia (ALL) who was undergoing induction chemotherapy. During induction therapy, the patient received a combination of pegaspargase, daunorubicin, vincristine, and twice-daily prednisone. On day 21 after starting the induction therapy, the patient reported 2 skin lesions, approximately 1 cm in diameter, with erythematous borders and nodular centers.
The skin lesions were on his right forearm and left shoulder and were painful to palpation. At the time of the new skin findings, the patient had an absolute neutrophil count of 0 cells/µL, had remained afebrile in the past 24 hours, and was receiving oral fluconazole prophylaxis. Subsequently, we consulted the dermatology department, which recommended obtaining bacterial, viral, and fungal cultures on the skin lesions via a punch biopsy.
We also obtained cultures of the peripheral and central lumens. The patient began to receive IV liposomal amphotericin B, dosed at 3 mg/kg every 24 hours, and IV anti-Pseudomonal therapy was added. At this time, we consulted the pediatric infectious disease service, and after further investigation, we found out that the patient had gone on a camping trip 3 weeks before his diagnosis of B-cell ALL. The infectious disease team recommended the addition of an expanded fungal workup, including fungal complement fixation studies, fungal immunodiffusion cryptococcocal antigen, urine histoplasmosis antigen, serum galactomannan, and a universal polymerase chain reaction (PCR) of the tissue biopsy.
Three days after the punch biopsy, branching septate hyphae were revealed, which were highly suspicious for Aspergillus infection, and anti-Pseudomonal therapy was discontinued. Five days after the biopsy, scant growth of Fusarium species was detected. Two additional lesions were then biopsied: one at the superior aspect of the right eyebrow and one at the upper lateral aspect of the thigh.
A chest CT was obtained, which showed right upper-lobe pulmonary opacities, suspected to be of fungal origin. Given the constellation of skin and lung involvement in the setting of worsening clinical status, an oral voriconazole loading dose of 400 mg twice daily was administered for 2 doses.
Subsequently, a maintenance regimen of oral voriconazole 300 mg twice daily was added, in combination with IV liposomal amphotericin B, while awaiting the susceptibilities report. In addition, granulocyte colony-stimulating factor was administered, and all intravenously accessed ports were removed, because of concerns for biofilm formation. One week after the biopsy, the universal PCR of the initial skin lesion was identified as F solani.
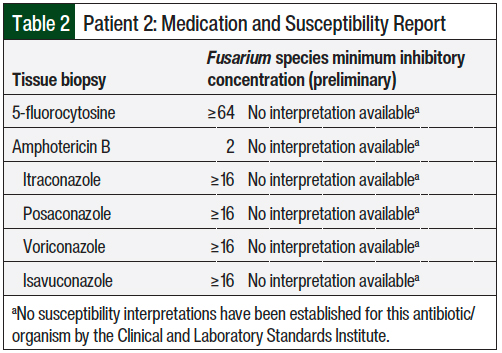
A voriconazole serum trough concentration—which was obtained on the fifth day of the double antifungal coverage—was 2.2 mcg/mL. Therefore, it was recommended that the patient should continue using the current regimen. Two weeks after biopsy of the initial skin lesion, the susceptibilities of the Fusarium species returned, showing a minimum inhibitory concentration associated with each antifungal agent (Table 2), and double coverage was continued.
The voriconazole serum trough concentration, which was tested again on the tenth day of therapy, was 1.3 mcg/mL; this is below the desired therapeutic reference range: serum trough concentrations of more than 2.2 mcg/mL are associated with improved clinical outcomes.17 As a result, we increased the voriconazole dose to 400 mg twice daily.
After consultation with an additional infectious disease specialist, who specializes in invasive fungal infections, we increased the liposomal amphotericin B to a daily dose of 7.5 mg/kg, which is higher than the recommended dosing strategy; however, pharmacokinetic evidence shows that daily doses of up to 10 mg/kg are safe in pediatric patients.18
During the third week of antifungal therapy, the patient’s clinical status began to improve, as indicated by the hemodynamic stability and improved neutrophil count. The infectious disease service then recommended discontinuing voriconazole therapy and continuing with liposomal amphotericin B as monotherapy. The results of repeated blood cultures, chest CT, and ophthalmologic examination were negative for invasive fungal processes.
The patient continued to receive liposomal amphotericin B 7.5 mg/kg daily for 7 months, at which point his serum creatinine level began to increase gradually. In response, the daily dose of amphotericin B was decreased to 5 mg/kg. The patient was gradually transitioned to a liposomal amphotericin B prophylactic regimen, as shown in Figure 2.
The patient was transitioned to the maintenance phase for the management of B-cell ALL and, therefore, will not be receiving intensive chemotherapy. Prolonged periods of neutropenia are not anticipated in the maintenance phase; therefore, our goal is to continue with liposomal amphotericin B prophylactic therapy until the completion of the patient’s antileukemic therapy.
Discussion
Although fungal disease may be difficult to detect, prompt diagnosis is essential to ensure adequate treatment in a timely manner. The slow growth of Fusarium species results in clinical manifestations that may be present for several days before the identification of a culprit.
In our first patient, it took 3 days after the recognition of the skin lesions to isolate a fungal element, and several additional days to recognize F solani as the specific causative organism. The patient remained clinically stable throughout the initial 5 days of culture growth, and therefore we did not initiate antifungal therapy until the suspicion for F oxysporum emerged in blood cultures, with no focal source noted.
For patient 2, we started treatment with liposomal amphotericin B swiftly after the discovery of new skin lesions. Prompt identification of a species allowed our team to initiate combination therapy with liposomal amphotericin B and voriconazole. It took several days from that point to determine the susceptibilities for this specific organism, a limitation that delayed the time to de-escalation to a single agent.
Unlike the majority of fungi and bacteria, Fusarium species do not have standardized minimum inhibitory concentration breakpoints designated by the Clinical and Laboratory Standards Institute (CLSI) or the European Committee on Antimicrobial Susceptibility (EUCAST).19 The lack of uniform minimum inhibitory concentrations for Fusarium species poses a challenge to providers when determining the susceptibility of Fusarium strains to various antifungal agents.
Although minimum inhibitory concentration values are obtained through the microbiology lab, interpretation of the results is based on limited published literature that evaluates in vitro findings with in vivo data. Understanding of local laboratory testing is important, because minimum inhibitory concentration values can vary between CLSI and EUCAST based on the specific method for determining the minimum inhibitory concentration.20
A recently published study indicates a good correlation between minimum inhibitory concentration breakpoints based on the CLSI and the EUCAST for amphotericin B, voriconazole, and posaconazole among Fusarium species. In that study, amphotericin B minimum inhibitory concentrations ranged from 0.25 to 8 mcg/mL, whereas voriconazole and posaconazole minimum inhibitory concentrations ranged from 0.25 to 32 mcg/mL.19
With information obtained in our 2 case reports relating to antifungal susceptibilities, the next challenge at our institution was related to the removal of the indwelling catheter. For Candida and Aspergillus infections, the Infectious Diseases Society of America recommends the removal of indwelling catheters when feasible and when the indwelling catheter is related to the cause of infection.21,22 No guidelines exist, however, regarding Fusarium infections and line removal. Published case reports describing successful treatment of Fusarium infections include systemic antifungal therapy in addition to the removal of indwelling access devices.9-13
As noted, our patient 1 had a broviac and hemodialysis catheter at the time of diagnosis. The positive cultures for Fusarium were collected from the broviac catheter, and this device was promptly removed on confirmation of infection. The hemodialysis catheter remained for the duration of treatment, because of the patient’s continued need for hemodialysis. When faced with decisions surrounding removal of indwelling catheters, it is necessary to assess the patient’s clinical status and the risk versus benefit associated with removal of indwelling lines.
Additional consideration must be given to each patient’s preexisting comorbid conditions when selecting appropriate antifungal treatment plans. For example, our 2 patients had some degree of renal dysfunction throughout their treatment course, which discouraged the use of nephrotoxic medications as part of their antifungal treatment plans. Close coordination with our nephrology team was necessary to ensure that both patients could tolerate the aggressive supportive care and hydration necessary to administer amphotericin B agents. The use of the liposomal form of this agent helped to minimize the damage caused to their already tenuous renal function.
Preventing infections after HSCT remains the cornerstone for the management of these patients; however, in patients with a history of Fusarium infections, a prophylactic medication regimen is required. Based on our institution’s experience, prophylactic therapy should be selected according to Fusarium strain susceptibilities, and in some cases, the preferred agent may be an IV agent.
The resistant Fusarium strain in patient 1 made liposomal amphotericin B the preferred prophylactic agent. Liposomal amphotericin B does not have standard prophylactic dosing, but 2 case reports have reported successful management with varying dosing schemas.23,24
After discussion with our infectious disease consultants, each of our 2 patients was initially transitioned to 3 times weekly dosing, followed by an interval and a dose wean, as shown in Figure 2. During the prophylactic period, both patients were monitored for signs of reinfection. Patient 1 discontinued therapy once his CD4 function was more than 200/µL on 2 separate occasions, whereas patient 2 is continuing to receive weekly liposomal amphotericin B.
The treatment of Fusarium infections in immunosuppressed patients presents many clinical challenges for clinicians. Difficulty in prompt detection and diagnosis and nonstandardized susceptibility breakpoints are factors contributing to the high mortality rates in this population. After Fusarium infection stabilization, the management of prophylactic therapies remains a crucial hurdle to overcome to prevent infection relapse or progression.
Conclusion
The 2 complex patients discussed here outline management strategies for working through common clinical hurdles encountered with the treatment of Fusarium infection in patients with cancer, illuminating the need for further published guidance surrounding the management of this patient population. Our 2 cases demonstrate the safety and efficacy of using combination liposomal amphotericin B with voriconazole for the management of invasive Fusarium infections in the pediatric immunocompromised population. Future studies are needed to assess the specific dose, frequency, and duration of combination antifungal therapy in this context.
Author Disclosure Statement
Dr Elder, Dr Harwood, Dr Gulley, Dr Infanti, and Dr Holt have no conflicts of interest to report.
References
- Tortorano AM, Richardson M, Roilides E, et al; for the European Society of Clinical Microbiology and Infectious Diseases Fungal Infection Study Group and European Confederation of Medical Mycology. ESCMID and ECMM joint guidelines on diagnosis and management of hyalohyphomycosis: Fusarium spp., Scedosporium spp. and others. Clin Microbiol Infect. 2014;20(suppl 3):27-46.
- Nucci M, Anaissie E. Fusarium infections in immunocompromised patients. Clin Microbiol Rev. 2007;20:695-704.
- Dóczi I, Gyetvai T, Kredics L, Nagy E. Involvement of Fusarium spp. in fungal keratitis. Clin Microbiol Infect. 2004;10:773-776.
- Ninet B, Jan I, Bontems O, et al. Molecular identification of Fusarium species in onychomycoses. Dermatology. 2005;210:21-25.
- Shoham S, Levitz SM. The immune response to fungal infections. Br J Haematol. 2005;129:569-582.
- Nucci M, Marr KA, Queiroz-Telles F, et al. Fusarium infection in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2004;38:1237-1242.
- Nucci M, Anaissie EJ, Queiroz-Telles F, et al. Outcome predictors of 84 patients with hematologic malignancies and Fusarium infection. Cancer. 2003;98:315-319.
- Nucci M, Anaissie E. Cutaneous infection by Fusarium species in healthy and immunocompromised hosts: implications for diagnosis and management. Clin Infect Dis. 2002;35:909-920.
- Uemura S, Tamura A, Yamamoto N, et al. Successful combination therapy of liposomal amphotericin B and caspofungin for disseminated fusariosis in a pediatric patient with acute lymphoblastic leukemia. Pediatr Infect Dis J. 2018;37:e251-e253.
- Liu JY, Chen WT, Ko BS, et al. Combination antifungal therapy for disseminated fusariosis in immunocompromised patients: a case report and literature review. Med Mycol. 2011;49:872-878.
- Inano S, Kimura M, Iida J, Arima N. Combination therapy of voriconazole and terbinafine for disseminated fusariosis: case report and literature review. J Infect Chemother. 2013;19:1173-1180.
- Neuburger S, Massenkeil G, Seibold M, et al. Successful salvage treatment of disseminated cutaneous fusariosis with liposomal amphotericin B and terbinafine after allogeneic stem cell transplantation. Transplant Infect Dis. 2008;10:290-293.
- Raad II, Hachem RY, Herbrecht R, et al. Posaconazole as salvage treatment for invasive fusariosis in patients with underlying hematologic malignancy and other conditions. Clin Infect Dis. 2006;42:1398-1403.
- Chu S, McCormick TS, Lazarus HM, et al. Invasive fungal disease and the immunocompromised host including allogeneic hematopoietic cell transplant recipients: improved understanding and new strategic approach with sargramostim. Clin Immunol. 2021;228:108731. doi: 10.1016/j.clim.2021.108731.
- Lewis R, Hogan H, Howell A, Safdar A. Progressive fusariosis: unpredictable posaconazole bioavailability, and feasibility of recombinant interferon-gamma plus granulocyte macrophage-colony stimulating factor for refractory disseminated infection. Leuk Lymphoma. 2008;49:163-165.
- Kadri SS, Remy KE, Strich JR, et al. Role of granulocyte transfusions in invasive fusariosis: systematic review and single-center experience. Transfusion. 2015;55:2076-2085.
- Miyakis S, van Hal SJ, Ray J, Marriott D. Voriconazole concentrations and outcome of invasive fungal infections. Clin Microbiol Infect. 2010;16:927-933.
- Seibel NL, Shad AT, Bekersky I, et al. Safety, tolerability, and pharmacokinetics of liposomal amphotericin B in immunocompromised pediatric patients. Antimicrob Agents Chemother. 2017;61:e01477-16. doi: 10.1128/AAC.01477-16.
- Al-Hatmi AMS, Normand AC, Ranque S, et al. Comparative evaluation of Etest, EUCAST, and CLSI methods for amphotericin B, voriconazole, and posaconazole against clinically relevant Fusarium species. Antimicrob Agents Chemother. 2017;61:e01671-16. doi: 10.1128/AAC.01671-16.
- Al-Hatmi AMS, Curfs-Breuker I, de Hoog GS, et al. Antifungal susceptibility testing of Fusarium: a practical approach. J Fungi (Basel). 2017;3:19. doi:10.3390/jof3020019.
- Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62:e1-e50.
- Patterson TF, Thompson GR III, Denning DW, et al. Practice guidelines for the diagnosis and management of aspergillosis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;63:e1-e60.
- El Cheikh J, Castagna L, Wang L, et al. Once-weekly liposomal amphotericin B for prophylaxis of invasive fungal infection after graft-versus-host disease in allogeneic hematopoietic stem cell transplantation: a comparative retrospective single-center study. Hematol Oncol Stem Cell Ther. 2010;3:167-173.
- Busca A, D’Ardia S, Terruzzi E, et al. Secondary antifungal prophylaxis with liposomal amphotericin B (AmB-L) in patients receiving allogeneic hematopoietic stem cell transplantation (HSCT). Blood. 2007;110:4972.