Chemotherapy-induced nausea and vomiting (CINV) is a severe adverse event that negatively affects patients’ quality of life (QOL).1,2 The reasons for CINV and hospitalization after chemotherapy include poor adherence to guidelines and not prescribing the appropriate outpatient antiemetic drugs, among others.3,4 CINV is frequently cited by patients as one of the most unpleasant and distressing adverse effects of chemotherapy.5 A cross-sectional study conducted by Lindley and colleagues demonstrated a significant decline in QOL in the patients who had emesis compared with those who did not.6
In another study, Martin and colleagues compiled and validated a questionnaire containing scales from the European Organization for Research and Treatment of Cancer QLQ-C30, the Morrow Assessment of Nausea and Emesis, and the Osoba Nausea and Emesis Module.2 The results showed that patients’ QOL significantly decreased as the number of CINV episodes increased.2 CINV is associated with declines in QOL and leads to substantial costs to the patient and the healthcare system as a result of unplanned office visits, hydration therapy, and hospitalizations.7,8
The emetic risk with chemotherapy is inherent to the specific drug regimen and often dictates the severity and duration of CINV.9 In addition, the specific patient characteristics and the efficacy of the antiemetic drug given before, during, and/or after the administration of chemotherapy influence the risk for CINV.10-12 Without a proactive approach, patients who are receiving chemotherapy are at risk for CINV, with or without severe dehydration, that requires medical attention.
The National Comprehensive Cancer Network (NCCN) and the American Society of Clinical Oncology (ASCO) guidelines on antiemesis recommend specific drug regimens based on the estimated risk for CINV with the administered chemotherapy.9,13 Despite the availability of these regularly updated guidelines, a study by Cohen and colleagues has shown that approximately 30% to 60% of patients who are receiving highly emetogenic drug regimens still have CINV while receiving routine prophylaxis.14
Adherence to antiemetic guidelines in practice is extremely variable, and the current literature suggests the need for clinicians’ review of clinical practice and implementation of change to optimize patient care.15 In 2014, the INSPIRE study showed that only approximately 57% of 1295 patients received guideline-consistent CINV prophylaxis, suggesting a continuing need to educate providers, pharmacists, and patients regarding the current guidelines and the options available for antiemetic prophylaxis.15
In 2020, Roeland and colleagues found that only 35% to 58% of clinicians had guideline adherence rates of >90% for highly emetogenic chemotherapy.16 Adherence to guidelines and the prevention of CINV can improve patients’ QOL, reduce healthcare costs, increase patients’ adherence to and tolerability of chemotherapy, and minimize anticipatory nausea and vomiting.16 As the medication experts, pharmacists are perfectly positioned to affect patients’ outcomes in this arena.
Pharmacists have sought out unique interventions in the hopes of mitigating CINV. In their 2014 study, Caracuel and colleagues compared 33 patients who had a pharmaceutical intervention, which consisted of antiemetic therapy recommendations and comprehensive patient education, and 69 patients who did not have the intervention.17 Their results demonstrated an approximate 15% absolute risk reduction in the incidence of CINV with the pharmacist intervention, despite the increased emetogenic risk of chemotherapy in the intervention group compared with the control group.17
In a more recent study, Hughes and colleagues concluded that the implementation of a pharmacist-run CINV protocol in which pharmacists completed all prior authorizations, prescribed and delivered medications, and counseled the patients led to complete response rates—defined as the absence of CINV at days 3 and 5 of the chemotherapy cycle—of >90% to antiemetic drugs in patients receiving highly emetogenic chemotherapy.18
From January 2020 through July 2020, Tampa General Hospital Cancer Center implemented a pharmacist-driven service to reduce the incidence of CINV. Through this pilot service, pharmacists identified patients who were starting their first cycle of moderately or highly emetogenic chemotherapy and placed a phone call within 48 hours after the infusion to assess CINV, patient adherence, and the efficacy of antiemetic drug regimens.
The primary goal of our study was to evaluate the impact of a pharmacist intervention in reducing the incidence of CINV through a telehealth pharmacist-led service model to further add to the existing body of literature the value of pharmacist intervention, and taking it one step further by assessing the potential reduction in the incidence of emergency department or clinic visits resulting from CINV within 30 days of chemotherapy infusion. The secondary goals were to describe the impact of CINV on a patient’s QOL and to analyze our institution’s adherence to national antiemesis guidelines.
Methods
This single-center, retrospective cohort study was conducted at Tampa General Hospital Cancer Center in Florida. Tampa General Hospital is a 1041-bed nonprofit, tertiary, research and academic medical center and is the region’s only safety-net hospital. The population at Tampa General Hospital Cancer Center includes patients with solid tumors as well as hematologic cancers. Within the cancer center, our oncology pharmacists play an active role in the multidisciplinary care of patients. Their daily tasks extend beyond order verification and medication preparation to include direct patient interviews to assess adverse reactions and to recommend therapy modifications to the oncologists, as deemed appropriate. Permission to access protected health information data was granted by the Office of Clinical Research at Tampa General Hospital, and this study was subsequently approved by the Institutional Review Board at the University of South Florida.
Patients were included in the study if they were aged ≥18 years and were receiving newly initiated moderately or highly emetogenic parenteral chemotherapy during the study period from January through July 2020. The emetogenicity of chemotherapy was determined based on the NCCN’s antiemesis guidelines version 1.2021,12 which did not change significantly from the 2019 version to the 2020 version. Pediatric patients, patients receiving low emetogenic chemotherapy, pregnant patients, and patients who were only receiving oral chemotherapy were excluded from the study.
To assess the impact of the pharmacist-led service model, the cohort of patients who had a follow-up with a pharmacist between January 1, 2020, and July 31, 2020, were compared with a historical cohort of patients who were identified by a query of the electronic health record (EHR) for having received a cycle-1, day-1 chemotherapy infusion between January 1, 2019, and July 31, 2019, and who met the study’s inclusion criteria.
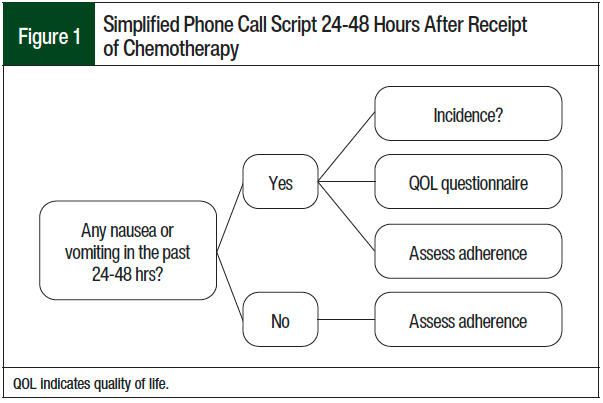
During this pilot service model, an EHR query was used to identify patients who received a cycle-1, day-1 infusion in the previous 48 hours. The patients identified through the EHR who met the inclusion criteria were screened for the exclusion criteria before a phone call was attempted 24 to 48 hours after the patients had received chemotherapy. Patients were excluded from the study if no contact was made within 48 hours of chemotherapy initiation after 2 phone call attempts. Patients who answered the phone call 24 to 48 hours after receiving an infusion were assessed by the pharmacist for the incidence of CINV, the patient’s adherence to the antiemetic drug regimen, and the efficacy of the antiemetic drug regimen (Figure 1).
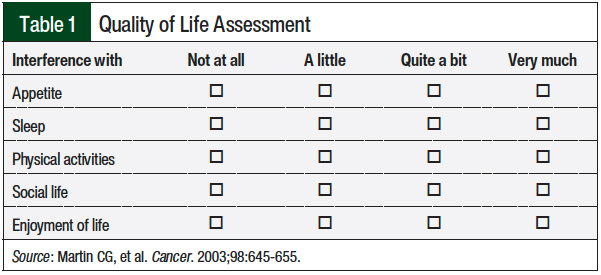
Patients who were reached in the initial phone call were placed on a list to receive a second phone call 72 to 96 hours after receiving an infusion. The second phone call followed the same structure as the first phone call (Figure 1), with the addition of follow-up for any changes made in the initial phone call. For patients who reported having CINV, the pharmacist recorded the incidence, followed by the administration of a validated QOL questionnaire adapted from Martin and colleagues’ study (Table 1).2 The pharmacists inquired how CINV affected the patient’s appetite, sleep, physical activities, social life, and enjoyment of life, using a 4-point scale of not at all, a little, quite a bit, and very much (Table 1).
To assess antiemetic drug adherence, patients were asked if they picked up their prescription(s), and how they were taking the medication(s). In addition, the pharmacist’s time rendering services was measured by charting the number of units (reported in 15-minute increments) that was spent on each phone call (eg, 2 units of service equal 30 minutes). This information was charted as a telephone encounter by the pharmacist in the patient’s EHR.
The primary outcome was a composite of the incidence of emergency department visits, hospital admissions, or infusion center appointments for CINV, with or without dehydration, within 30 days of chemotherapy initiation. To identify the primary outcome incidence, the patients’ charts were retrospectively reviewed for additional encounters beyond the scheduled infusions within the 30 days after the initial chemotherapy administration. If the additional encounter was an emergency department visit, inpatient admission, or outpatient infusion center visit, the discharge summary for the encounter was analyzed for the primary diagnosis.
Patients were considered to have the primary outcome if the primary diagnosis on discharge was CINV, or if they had a primary diagnosis of dehydration with CINV as a secondary diagnosis. The secondary end points included the initial antiemetic drug administration and prescribing practices for patients who started moderately or highly emetogenic chemotherapy compared with the national guidelines,9,13 and the impact of CINV on the patient’s QOL.
Antiemetic drug regimens were considered to be adherent to the national guidelines if the drugs administered aligned with the drug classes in the NCCN-recommended treatment options.9 For example, highly emetogenic chemotherapy requires at least 3 drug classes in the following combinations: (1) a neurokinin 1 (NK1) receptor antagonist plus a selective serotonin receptor antagonist (5-HT3) and dexamethasone, with or without olanzapine, or (2) a 5-HT3 receptor antagonist (palonosetron, specifically) plus dexamethasone and olanzapine.9 The effects of CINV on a patient’s QOL were assessed based on the results of the validated patient questionnaire described earlier (Table 1). We analyzed the primary composite end point for statistical significance using a chi-square (X2) test within Microsoft Excel, with a predetermined alpha value of 0.05. The baseline characteristics and secondary end points were analyzed using descriptive statistics, as well as X2 tests.
Results
Between January 2020 and July 2020, 81 patients were eligible to receive pharmacist follow-up. Of these 81 patients, pharmacists were able to contact 68 (84%) patients within 48 hours of the administration of cycle-1, day-1 chemotherapy. Of these 68 patients, 30 (44.1%) patients were reached for a second follow-up call 72 to 96 hours after receiving cycle-1, day-1 chemotherapy.
In the historical control cohort, 70 patient charts between January 2019 through July 2019 were identified by the EHR as having a cycle-1, day-1 chemotherapy infusion and were reviewed for potential inclusion in the study. Of these 70 patients, 43 (61.4%) met the study’s inclusion criteria. The most common reasons for study exclusion were receipt of a low emetogenic chemotherapy regimen (N = 12; 17.1%); not receiving true cycle-1, day-1 chemotherapy (ie, patient had received the chemotherapy regimen before: N = 7; 10%); and initiating chemotherapy as an inpatient (N = 4; 5.7%).
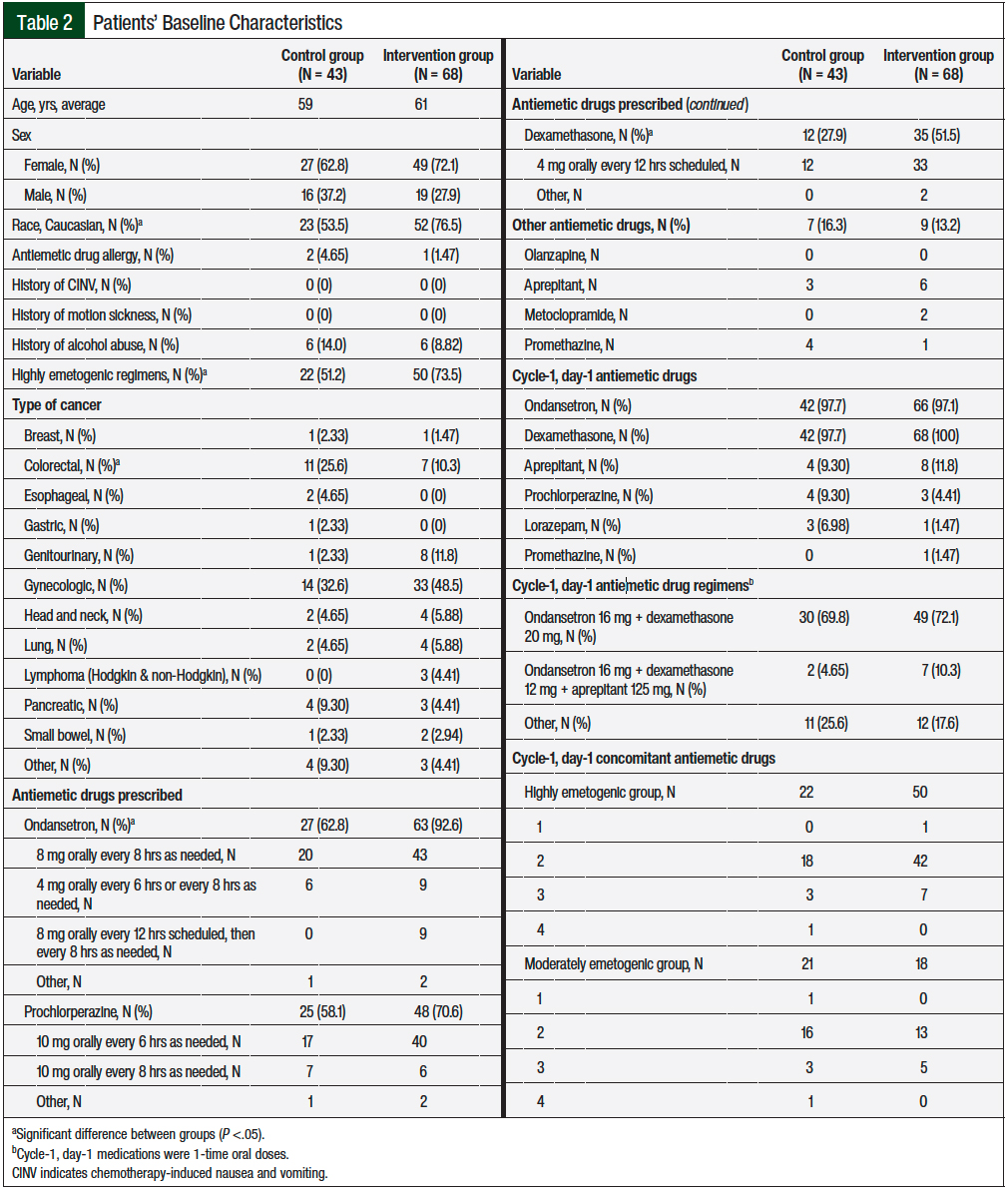
The baseline demographics of the intervention group and the control group were similar overall, but with a few notable differences (Table 2). The average patient age was 60 years (standard deviation, 11.7 years). The cancer types were similar in the 2 groups, but significantly more patients had colorectal cancer in the control group than in the intervention group (N = 11 [25.6%] vs N = 7 [10.3%]; P = .033).
Among the historical control group, 22 (51.2%) patients received highly emetogenic drug regimens compared with 50 (73.5%) patients in the intervention group (P = .017). Significantly more patients were prescribed ondansetron for discharge in the intervention group than in the control group (N = 63 [92.6%] vs N = 27 [62.8%]; P <.001) and a similar pattern was seen with dexamethasone (N = 35 [51.5%] vs N = 12 [27.9%]; P = .014).
No patients in either group had a documented history of CINV or motion sickness. In the control group, 2 patients had documented intolerances to ondansetron, and in the intervention group 1 patient had an unspecified promethazine allergy. Each group had 6 patients with documented histories of alcohol abuse.
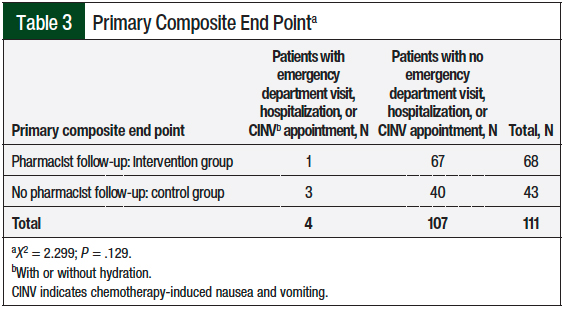
In the control cohort, 3 (7%) patients had the composite primary end point of an emergency department visit, hospital admission, or infusion center appointment for CINV, with or without dehydration, within 30 days of chemotherapy initiation compared with 1 (1.5%) patient in the intervention group (P = .129; Table 3). In the overall study population, 71% of the patients received the antiemetic drug regimen of ondansetron 16 mg orally once plus dexamethasone 20 mg orally once before their chemotherapy initiation on cycle 1, day 1, regardless of chemotherapy emetogenicity.
In 60% of the patients, the antiemetic drugs administered on cycle 1, day 1 were inconsistent with the NCCN’s guideline recommendations.9 Nearly all (92.5%) inconsistencies were attributable to the use of a 2-drug antiemetic regimen instead of a 3-drug regimen for patients receiving highly emetogenic chemotherapy. Ondansetron was the most prescribed prophylactic outpatient antiemetic drug (N = 90; 81.1%), followed by prochlorperazine (N = 73; 65.8%).
In addition, 14% of the patients were administered or prescribed aprepitant, and none of the patients were administered or prescribed olanzapine. Furthermore, none of the patients received fosaprepitant, because that drug was not on the hospital’s formulary.
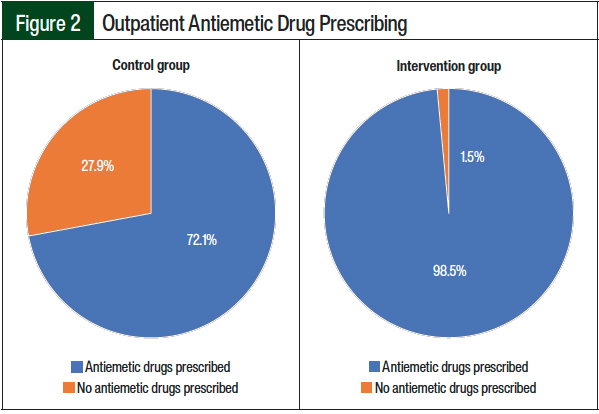
A significant difference was seen in the number of patients who did not have outpatient antiemetic drugs prescribed in the control group compared with the intervention group (N = 12; 27.9% vs N = 1; 1.5%; P <.000024; Figure 2). For QOL outcomes, a total of 21 (30.9%) patients reported CINV in the 48- or 96-hour phone call, and 59% of these patients also reported interference with appetite, 27% reported sleep interference, 27% reported physical activity disruption, 18% reported social life interruption, and 41% reported disruption of life enjoyment.
In this pilot service model, between January 2020 and July 2020 pharmacists spent a total of 27.6 hours on the phone with patients, with the initial call contributing approximately 19.2 total hours. The average amount of pharmacist time spent was 16.9 minutes per phone call.
Discussion
In our study, oncology pharmacists identified a significant number of patients who were not prescribed prophylactic outpatient antiemetics for delayed CINV through a pharmacist-led follow-up service model. Our pharmacists’ active involvement in the multidisciplinary care for patients at Tampa General Hospital Cancer Center allowed for the prompt resolution of absent prescriptions and increased patient access to medications for delayed CINV, as demonstrated by the significant increase in prescribed ondansetron and dexamethasone in the intervention group (Table 2).
Prechemotherapy antiemetic drug administration practices are important to consider. Antiemetic drug selection should be based on the emetic risk of therapy, the patient’s previous receipt of antiemetic drugs, and patient-specific factors. Known patient risk factors that increase the likelihood of CINV include female sex, younger age, a history of CINV, little or no previous alcohol use, being prone to motion sickness, and anxiety about or high expectation of treatment-related nausea.9 Our patients were nearly free of baseline risk factors, except that the majority of patients were female (Table 2).
The NCCN and the ASCO guidelines define highly emetogenic chemotherapy as more than 90% frequency of emesis and moderately emetogenic chemotherapy as more than 30% to 90% frequency of emesis.9,13 The NCCN guidelines recommend 3 category 1 treatment options for acute (ie, ≤24 hours) and delayed (ie, >24 hours) emesis prevention for moderately and highly emetogenic parenteral chemotherapy,9 and ASCO has similar recommendations that are further stratified by specific agents.13 According to these guidelines, a highly emetogenic chemotherapy requires a 3- or a 4-drug antiemetic drug regimen on day 1 of treatment, whereas patients receiving moderately emetogenic agents can be given a 2-drug antiemetic regimen.9,13
Our institution’s most often administered cycle-1, day-1 antiemetic drug regimen is ondansetron 16 mg orally once plus dexamethasone 20 mg orally once. This is an appropriate treatment regimen for moderately emetogenic agents, albeit a higher dose of dexamethasone than the recommended 12 mg, but we did not qualify the dexamethasone dose as guideline-inconsistent, given a footnote in the NCCN guidelines citing that higher dexamethasone doses may be considered, especially in the absence of an NK1 receptor antagonist.9
However, this 2-drug regimen of ondansetron and dexamethasone is insufficient for highly emetogenic agents, according to the NCCN guidelines.9 Tampa General Hospital’s antiemesis drug guideline’s adherence rate of 40% by clinicians is similar to previously reported rates that ranged from 35% to 58% of clinicians whose adherence rates were more than 90%.15,16
Of the 4 patients (3 in the control group and 1 in the intervention group) who had the primary composite end point of an emergency department visit, hospital admission, or infusion center appointment for CINV, with or without dehydration, within 30 days of chemotherapy initiation, 2 received moderately emetogenic chemotherapy and 2 received highly emetogenic chemotherapy. The former 2 patients received guideline-consistent premedication antiemetic drugs and outpatient antiemetic drugs. The latter 2 patients received a 2-drug antiemetic drug regimen on cycle 1, day 1, which was inconsistent with the NCCN guidelines.9 In addition, neither of the 2 patients who received highly emetogenic chemotherapy received guideline-consistent outpatient antiemetic drugs, and 1 of these patients was not prescribed any antiemetic drugs for delayed CINV.
Although the primary end point was not statistically significant, our study provides a notable finding for our institution. The 40% guideline-adherence rate and lack of NK1 receptor antagonist use highlighted the disparity between our clinical practice and the national guidelines. Despite the long-time presence of NK1 receptor antagonists in the antiemesis guidelines, we speculate that the reasons for their omission were provider preference, cost concern, or the underappreciation of CINV risk associated with highly emetogenic chemotherapy. These findings underscore the need for pharmacists’ intervention to increase our institution’s adherence to national guidelines.
Limitations
This study has several limitations. The first limitation involved identifying the patients for this pilot service. As mentioned earlier, an EHR query was used to identify the patients with a cycle-1, day-1 infusion in the previous 48 hours for the pharmacist to call. Although it was our intent to run the report daily and capture every patient who was newly starting moderately or highly emetogenic chemotherapy over a 7-month period, this was not always possible because of holidays, weekends, busy days, and sick days. In addition, for patients who received follow-up, inclusion in the study was dependent on reaching the patient via phone; this was not always possible because of the inability to leave voicemails (eg, full voicemail boxes), unreturned calls from patients, or incorrect contact information.
The retrospective nature of this study is another limitation. The evaluation of certain end points was difficult because of our reliance on chart documentation.
Furthermore, the methodology used to select patients for the historical control group was not ideal, although the baseline characteristics were similar in the 2 groups. Ideally, every patient from the intervention group would have a matched historical control based on demographics, type of cancer, and chemotherapy regimen. The lack of matching in our study might have introduced bias or might have confounded the results. Significantly more patients in the control group had colorectal cancer, naturally placing them at a higher risk for nausea and vomiting compared with the intervention group. Also, if a patient was admitted to an outside hospital, this information would not be known and ultimately was absent from our study.
A major unforeseen issue with our study was the presence of the COVID-19 global pandemic. Knowledge and fear of the pandemic spread to our geographic region starting in March 2020 and worsened throughout the remainder of the study period (the pilot concluded in July 2020). During the pandemic, emergency department visits and hospital admissions were inherently fewer than in previous years, based on the public’s general avoidance of healthcare, except for extreme emergencies. According to a Centers for Disease Control and Prevention report, by the end of June 2020 a staggering 41% of US adults had delayed or avoided medical care as a result of concerns related to COVID-19.19
Moreover, the COVID-19 pandemic was likely a confounding factor for our study, because it was not possible to distinguish if the lack of an emergency department visit, hospital admission, or infusion center appointment in 2020 was the result of a pharmacist’s intervention or simply healthcare avoidance secondary to the pandemic. A consequence of the global pandemic was a shift from in-person visits to telehealth via phone or video calls, but this transition did not hinder the pharmacists’ abilities to intervene.
Conclusion
The results of this study clearly show a positive impact of pharmacist-led follow-up calls and demonstrate the value of a pharmacist’s active role to the multidisciplinary team for patients who are initiating moderately or highly emetogenic chemotherapy. This new pharmacist-led service model was well received by patients and led to meaningful interventions. Our institution plans to integrate this service model into our cancer center’s standard of care, and this model could be adopted by other institutions.
An additional finding was our inconsistency with the current guidelines regarding the receipt of antiemetic drug regimens before moderately or highly emetogenic chemotherapy infusions, further adding to the existing body of literature on poor guideline adherence. As a result, Tampa General Hospital Cancer Center’s pharmacists are leading an antiemetic drug class review and subsequent update to all chemotherapy treatment plans to reflect the current antiemesis guidelines.
Author Disclosure Statement
Dr McCabe, Dr Al-Bahou, Dr Koontz, Dr Linderbeck, Dr Verbosky, and Dr Prieto have no conflicts of interest to report.
References
- Rao KV, Faso A. Chemotherapy-induced nausea and vomiting: optimizing prevention and management. Am Health Drug Benefits. 2012;5(4):232-240.
- Martin CG, Rubenstein EB, Elting LS, et al. Measuring chemotherapy-induced nausea and emesis: psychometric properties of a quality of life questionnaire. Cancer. 2003;98:645-655.
- Aapro M. CINV: still troubling patients after all these years. Support Care Cancer. 2018;26(suppl 1):S5-S9.
- Check DK, Basch EM. Appropriate use of antiemetics to prevent chemotherapy-induced nausea and vomiting. JAMA Oncol. 2017;3:307-309.
- Griffin AM, Butow PN, Coates AS, et al. On the receiving end V: patient perceptions of the side effects of cancer chemotherapy in 1993. Ann Oncol. 1996;7:189-195.
- Lindley CM, Hirsch JD, O’Neill CV, et al. Quality of life consequences of chemotherapy-induced emesis. Qual Life Res. 1992;1:331-340.
- Craver C, Gayle J, Balu S, Buchner D. Clinical and economic burden of chemotherapy-induced nausea and vomiting among patients with cancer in a hospital outpatient setting in the United States. J Med Econ. 2011;14:87-98.
- Burke TA, Wisniewski T, Ernst FR. Resource utilization and costs associated with chemotherapy-induced nausea and vomiting (CINV) following highly or moderately emetogenic chemotherapy administered in the US outpatient hospital setting. Support Care Cancer. 2011;19:131-140.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Antiemesis. Version 1.2021. December 23, 2020. www.nccn.org/professionals/physician_gls/pdf/antiemesis.pdf. Accessed February 1, 2021.
- De Mulder PHM, Seynaeve C, Vermorken JB, et al. Ondansetron compared with high-dose metoclopramide in prophylaxis of acute and delayed cisplatin-induced nausea and vomiting: a multicenter, randomized, double-blind, crossover study. Ann Intern Med. 1990;113:834-840.
- Pater J, Slamet L, Zee B, et al. Inconsistency of prognostic factors for post-chemotherapy nausea and vomiting. Support Care Cancer. 1994;2:161-166.
- Osoba D, Zee B, Pater J, et al; for the Quality of Life and Symptom Control Committees of the National Cancer Institute of Canada Clinical Trials Group. Determinants of postchemotherapy nausea and vomiting in patients with cancer. J Clin Oncol. 1997;15:116-123.
- Hesketh PJ, Kris MG, Basch E, et al. Antiemetics: ASCO Guideline update. J Clin Oncol. 2020;38:2782-2797. Errata in: J Clin Oncol. 2020;38:3825; J Clin Oncol. 2021;39:96.
- Cohen L, de Moor CA, Eisenberg P, et al. Chemotherapy-induced nausea and vomiting—incidence and impact on patient quality of life at community oncology settings. Support Care Cancer. 2007;15:497-503.
- Gilmore JW, Peacock NW, Gu A, et al. Antiemetic guideline consistency and incidence of chemotherapy-induced nausea and vomiting in US community oncology practice: INSPIRE study. J Oncol Pract. 2014;10:68-74.
- Roeland EJ, Ruddy KJ, LeBlanc TW, et al. What the HEC? Clinician adherence to evidence-based antiemetic prophylaxis for highly emetogenic chemotherapy. J Natl Compr Canc Netw. 2020;18:676-681.
- Caracuel F, Baños Ú, Herrera MD, et al. Influence of pharmaceutical care on the delayed emesis associated with chemotherapy. Int J Clin Pharm. 2014;36:287-290.
- Hughes DM, Vose RL, Shah B, Jhaveri R. Pharmacist-run chemotherapy-induced nausea and vomiting management protocol improves patient outcomes and benefits physician workflow. J Hematol Oncol Pharm. 2020;10(4):190-196.
- Czeisler MÉ, Marynak K, Clarke KEN, et al. Delay or avoidance of medical care because of COVID-19–related concerns—United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1250-1257.