The number of clinical trials conducted is continuing to expand, with approximately 419,487 registered studies on ClinicalTrials.gov as of June 2022.1 Interventional studies researching drugs or biologic therapies make up 172,517 of these studies, which is more than 40% of these clinical trials.1 With the expanding number and complexity of clinical trials, the need for increased pharmacist involvement has become apparent. Pharmacists’ role within the clinical research setting has continued to evolve, incorporating multiple key services to support clinical trials.
This is further highlighted by the Hematology/Oncology Pharmacy Association (HOPA) Investigational Drug Service Best Practice Standards and the American Society of Health-System Pharmacists (ASHP) guidelines.2,3 These guidelines emphasize the important roles of pharmacists and pharmacy technicians in the investigational drug research process.
In a national survey evaluating pharmacy services within the oncology clinical trial setting, a gap was identified between the services provided and the importance they held.4 The areas that were associated with high importance included medication reconciliation, investigator-initiated trial development, the assessment of patients’ adherence to oral chemotherapy as part of a study, and therapeutic drug monitoring. Although highly important, these services were limited as a result of barriers associated with financial resources, management priority, and pharmacist training. This survey demonstrated the importance of and the inconsistencies in understanding what investigational pharmacists do within the clinical trial setting.4
The HOPA Investigational Drug Service Best Practice Standards defined a role for the pharmacist within investigational drug services as participating in multiple operational duties.2 These standards focus on 4 key areas of investigational drug services: general best practices, best practices for prescribing investigational medications, best practices for dispensing investigational medications, and best practices for administering investigational medications. The investigational drug service general best practices range from developing policies and procedures for investigational drug management to creating a fee schedule for sponsored clinical trials. The best practices for prescribing investigational medications provide guidance related to protocol information availability to all essential pharmacy staff.2
Within the best practices for dispensing investigational medications, the areas of focus include facilitating education about investigational medications, creating policies and procedures related to the labeling and dispensing of investigational drugs, and conducting single- or double-blind studies. In addition, within the best practices for administering investigational medications, guidance is given related to conducting sponsored clinical trials.2 A more comprehensive list of these principles and practices for hematology and oncology pharmacists and their associates is provided in the HOPA Investigational Drug Service Best Practice Standards.2
Similar duties have been developed by the ASHP’s guidelines that are related to the management of investigational drugs.3 Although these guidelines provide a template for the development of an investigational drug service for all institutions,3 it continues to be a challenge for smaller facilities within the community setting to incorporate these standards, because of limited resources.
Cowell Family Cancer Center, a community cancer center located in northern Michigan, currently participates in multiple clinical trials through the Cancer Research Consortium of West Michigan, as well as in independently sponsored clinical trials. The center currently has 44 infusion chairs servicing approximately 80 to 100 patients daily.
Cowell Family Cancer Center has a dedicated research department, which manages all clinical trial patients. The research department is comprised of 8 staff members, including 3 research nurses, 3 data coordinators, 1 administrative assistant, and 1 director. In addition, the center has 8 oncologists, 2 of whom co-chair the research department, as well as 1 gynecologic oncologist and 4 radiation oncologists who engage in clinical trials.
The pharmacy department has 1 pharmacist and 1 technician who split their time between research and daily pharmacy tasks. Before the implementation of the investigational drug service, these roles were poorly defined within the pharmacy department, policies and procedures did not exist or were not updated to reflect HOPA Investigational Drug Service Best Practices Standards, and only a flat one-time fee was established to cover any and all pharmacy duties associated with a clinical trial. The increase in the number and complexity of clinical trials being conducted at Cowell Family Cancer Center has expanded the role of the pharmacy team.
The objective of this study was to evaluate the implementation of a formal investigational drug service at a community cancer center. The primary end point was to evaluate oncology provider and research staff satisfaction and the perception of pharmacists’ time commitment before and after the implementation of the service. We used a survey to evaluate providers and oncology research staff perception of pharmacist time commitment rather than actual time spent, because pharmacists’ time commitment historically has not been recorded, and a preimplementation period of 6 months to 1 year would be necessary to collect such data. The second primary end point was to evaluate the pharmacists’ and technicians’ confidence in providing investigational drug services before and after the implementation of such a service. The secondary end point was to measure the estimated financial benefit of the new fee schedule for sponsored trials compared with the flat fee.
Methods
This single-center, observational study was approved by the Institutional Review Board. The study was conducted at Cowell Family Cancer Center, a community cancer center in Traverse City, MI, to evaluate the impact of the implementation of a new investigational drug service that incorporated the core components of the HOPA Investigational Drug Service Best Practice Standards from October 2020 through March 2021.2
Before the implementation of this new service, a gap analysis was performed by the study team to evaluate the then-current pharmacy practices, and how they aligned with HOPA Investigational Drug Service Best Practice Standards. This evaluation resulted in the identification of multiple areas for improvement to create the new investigational drug service. Of the 4 best practice areas outlined in the HOPA Investigational Drug Service Best Practice Standards, the general best practices and those related to dispensing investigational medications were identified as key areas of improvement in building the new service at our center.
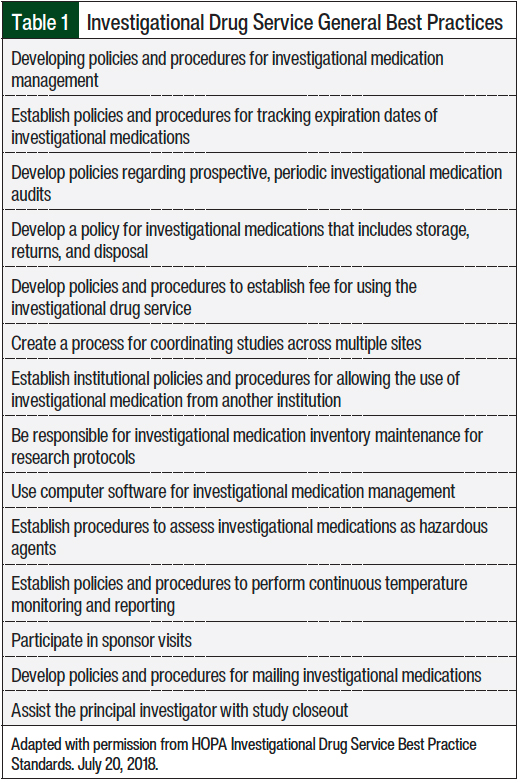
Table 1 outlines the many general best practice suggestions for an investigational drug service. Of these, the first best practice listed in the HOPA standards—the development of policies and procedures for investigational medication management—was widely applied to build our new service.2
The completed tasks included the creation of several standard operating procedures related to the preparation and dispensing of investigational medications and the development of a fee schedule for the use of the investigational drug service to replace our then-current $1500 flat fee for sponsored trials. Specifically, standard operating procedures were created for medication inventory maintenance for research protocols; tracking expiration dates of investigational medications; and the storage, return, and disposal of investigational medications.
These standard operating procedures were created using guidance from HOPA Investigational Drug Service Best Practice Standards,2 aligned with institutional-specific policies and procedures, and validated by the research department at our center.
Next, best practices were implemented for dispensing investigational medications (Table 2). The key policies and standard operating procedures related to investigational drug dispensing included facilitating the dissemination of investigational medication information, ensuring adequate investigational medication order review and verification, establishing dispensing and labeling requirements for oral investigational medications, and creating procedures to dispense investigational medication for single- or double-blind studies.
The standard operating procedures related to the dissemination of investigational medication and the creation of a drug accountability record form (DARF) can be found in the Appendix (available at the top of the article).
The standard operating procedures for the new service were developed and implemented over a 6-month period. HOPA Investigational Drug Service Best Practices Standards for Dispensing Investigational Medications was evaluated first. The key policies and procedures that were chosen to be evaluated and updated were related to the dissemination of investigational medication information and creating a standard operating procedure for dispensing investigational medications.
Weekly education sessions included a handout providing instruction on how to perform each task, as well as an example document demonstrating a correctly filled-out report or label. From these education sessions, standard operating procedures were created and were integrated into read and sign competencies for pharmacists and technicians to reference and to complete.
We modeled the clinical trial fee schedule after an unpublished national survey conducted by a University Health System Consortium, based on personal communication. This survey assessed 34 different health systems conducting clinical trials and evaluated each fee schedule, the size of the health system, and the location of the health system. Based on the most common fee charged, the health systems’ size, and the location, we created a fee schedule for Cowell Family Cancer Center.
We implemented fees related to study start-up, annual maintenance, monitoring, randomization, study closure, and chemotherapy dispensing. This new fee schedule was then applied to sponsored clinical trials conducted at our institution during the 2019-2020 fiscal period to determine the difference in potential fees, because the previous fee schedule was a one-time flat-rate fee of $1500 per clinical trial.
After incorporating the new fee schedule into the 4 sponsored clinical trials that were previously open, the average total fee per study was $3500, based on the monetary charges of study setup, annual maintenance, monitoring, randomization, closure, and chemotherapy dispensing. When incorporating the new fee schedule, an increase of $2000 per patient per clinical trial was estimated.
To evaluate all these best practice changes before and after the implementation of the new service, surveys were developed for the pharmacy staff and the oncology research department. Oncology providers and oncology research staff were asked to assess the new investigational drug service through 2 surveys—a satisfaction survey and a survey assessing the perception of pharmacist time commitment. The survey questions were developed by a team of pharmacists, including the current investigational drug service pharmacist, a PGY2 pharmacy oncology resident, and the clinical manager for pharmacy and infusion services.
All the survey data analyses for the primary outcome were evaluated using Stata 15 (StataCorp; College Station, TX), with the ordinal data analysis assessed via Kruskal-Wallis rank test and interval-level analyses assessed with a matched-pairs t-test for nonnormally distributed variables. Descriptive statistics were used for the secondary outcome of measuring the financial benefit of a new fee schedule for sponsored trials.
Results
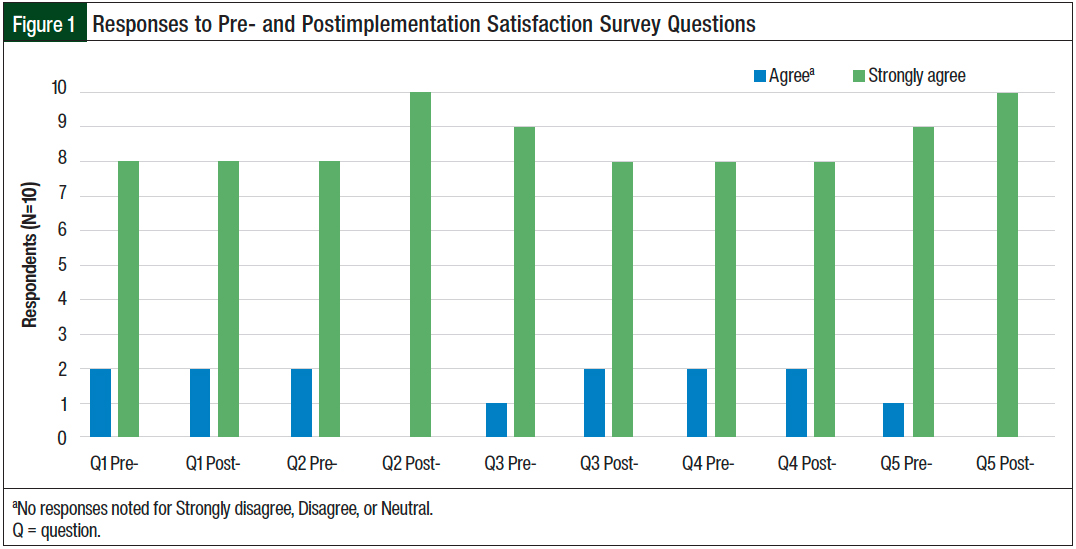
The satisfaction survey used a 5-point Likert-based scale ranging from strongly disagree to strongly agree to evaluate the overall satisfaction with pharmacy services provided in the setting of clinical trials (Table 3). This survey involved 5 questions that were developed based on the National Survey of comprehensive pharmacy services provided in cancer clinical trials.4
The 5 questions were developed based on performance and importance related to clinical trials and were related to pretrial implementation support, trial implementation support, medication therapy management, and miscellaneous support. Figure 1 illustrates the responses to the satisfaction survey questions pre- and postimplementation.
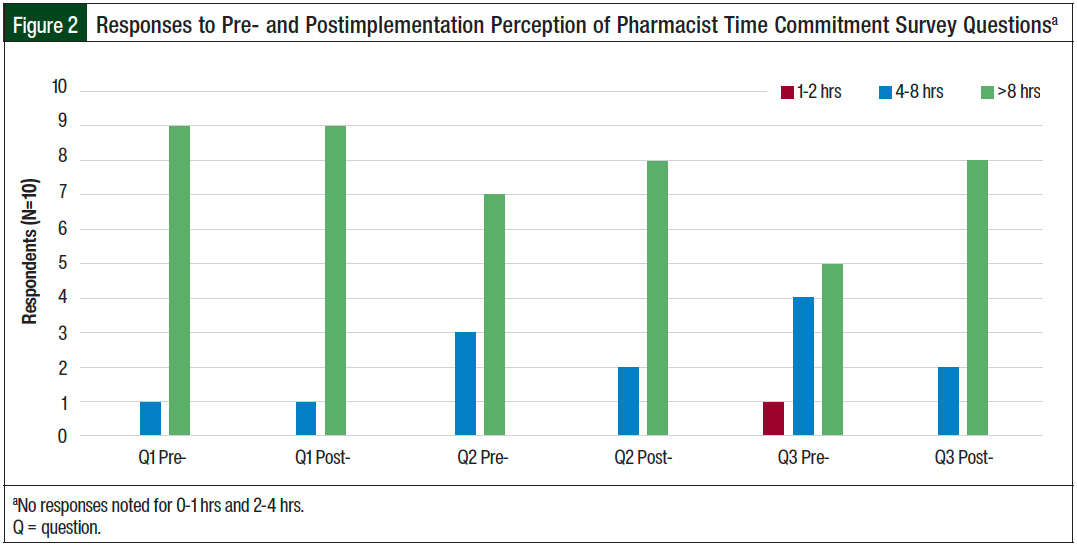
The survey questions for time commitment were formulated to assess the average time needed to open a new clinical trial, manage a clinical trial, and close a clinical trial, separating it into 5 intervals, which included 0 to 1 hour, 1 to 2 hours, 2 to 4 hours, 4 to 8 hours, and more than 8 hours, and were developed from discussions with the 2 pharmacists who provide these services at Cowell Family Cancer Center (Table 4). Figure 2 illustrates the responses to the commitment survey questions pre- and postimplementation.
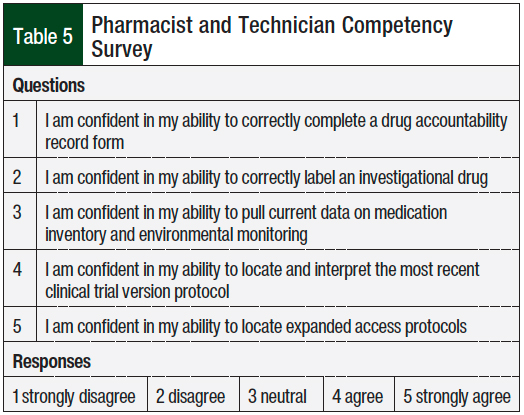
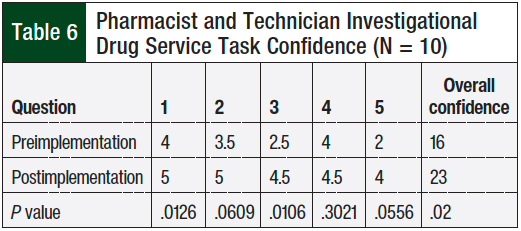
Because education was provided as part of the implementation, pharmacists and technicians were assessed before and after implementation on their ability to complete investigational drug service tasks confidently and correctly. This survey had a 5-point Likert scale of strongly disagree to strongly agree before and after the implementation of the formal investigational drug service (Table 5). A total of 5 questions were developed to evaluate the pharmacists’ and technicians’ ability to complete specific investigational drug service tasks confidently and correctly (Table 6). Of these tasks, the ability to complete a DARF correctly, label investigational drugs appropriately, pull environmental monitoring data, locate and interpret clinical trial protocols, and locate expanded access protocols were assessed.
On completion of the preimplementation survey, education sessions were created and were presented for each question at the weekly meetings.
For the primary end points of satisfaction and the perception of pharmacists’ time commitment, 10 research department staff (2 oncology providers and 8 oncology research staff) participated in the surveys before and after the implementation of the investigational drug service. No statistical differences were observed in either survey (Figures 1 and 2).
A total of 10 members of the pharmacy department, including 5 technicians and 5 pharmacists, participated in the second primary end point surveys—confidence in providing the new service before and after the implementation of the investigational drug service. A significant overall improvement in pharmacy staff confidence was identified from the pre- to the postimplementation survey, with a total median score of 16 versus 23 of a total possible score of 25 (P = .02; Table 6).
Questions 1 and 3 showed a meaningful difference. The median score for question 1, which was related to pharmacists’ and technicians’ ability and confidence to correctly complete a DARF, improved from a median score of 4 to 5 of a total possible 5 (P = .0126). The median score for question 3, which was related to confidence in pulling current data on medication inventory and environmental monitoring, improved from 2.5 to 4.5 of a total possible score of 5 (P = .0106). When analyzed individually, the results of the other 3 questions in the pre- and postimplementation surveys were not different.
Discussion
Multiple significant improvements in the pharmacy team survey scores were seen after the implementation of a formal investigational drug service in our community cancer center. The scores improved significantly overall, specifically with respect to tasks involving completing the DARF and data collection for medication inventory and environmental monitoring. Before the creation of the formal service, the pharmacy team members had an annual requirement to complete a repetitive questionnaire about how to fill out a DARF, but they were not instructed on or provided with a formal standard operating procedure.
During the study, all team members were educated as a group, were remediated individually if questions arose, and were made aware of the central file location for this standard operating procedure for reference. In addition, knowledge related to medication inventory and environmental monitoring were previously limited to 2 clinical pharmacists and 1 technician. The education provided in the study was again universal and provided a standard operating procedure and a central file location to help team members gain confidence in these processes.
Conversely, surveys given to providers and research staff did not show a difference in the perception of time spent by pharmacy staff or satisfaction with pharmacy services related to investigational drugs (Figures 1 and 2). One possible reason for this is that the preimplementation survey results for the 2 items were very high on the Likert scale, making it improbable to see a statistical difference on the postimplementation survey.
A discussion of how to limit the subjectivity of survey questions occurred when the questions were written; however, the existing provider and research staff relationships with the pharmacy department were strong and the time commitment of the pharmacy team was not routinely shared, so the survey question responses were likely affected by these factors. As mentioned, the actual time commitment by the pharmacists was deemed too difficult to measure before and after the implementation of the service, because this was not historically recorded and would take 6 months to 1 year to collect. This timeline did not align with the service implementation and thus a survey-based analysis was used.
Publications describing the implementation of formal investigational drug services within the community setting are unavailable to our knowledge, despite the important role of these centers in oncology clinical trials. As far as we know, this is the first study to implement and evaluate an investigational drug service at a community cancer center.
Despite a lack of dedicated pharmacy resources, we were able to establish a formal investigational drug service using the key core components of the HOPA Investigational Drug Service Best Practice Standards. Although not all core components of the standards were evaluated, many were implemented, including the development of standard operating procedures for creating and completing a clinical trial DARF for oral and intravenous chemotherapy; defining the pharmacy technician’s role within the investigational drug service; and creating standard operating procedures for investigational drug dispensing, study maintenance information, study design and drug information, and the dissemination of information relevant to the nursing staff.
Policies and procedures were also created for continuous temperature monitoring and reporting so that all pharmacy staff could access and report this information. Labels were developed specifically for investigational agents to align with language outlined by the US Food and Drug Administration. Finally, standard operating procedures were created related to locating and interpreting expanded drug access protocols.
Financial support can be a barrier at a community cancer center, and having the opportunity to increase profit through a new fee schedule can potentially allow for additional revenue to help fund resources within the clinical trial setting. Before implementing the formal investigational drug service, a single start-up fee was used at our center, and no annual fees related to maintenance, monitoring, randomization, closure, and chemotherapy dispensing were established. This new fee schedule may increase the income potential for a clinical trial that is open for a long time, enrolls many patients, and in turn requires more pharmacy resources. This opportunity for increased revenue also helps to justify the time that a pharmacist takes to be a part of the investigational drug service.
Although the results related to the nursing and provider survey were not significant, the implementation process of the formal investigational drug service provided opportunities to educate these research team members on the many changes made to create the service, as well as the time commitment required by the pharmacy.
Improvements within the pharmacy department were seen with the newly developed standard operating procedures created during the implementation of the new service. These will now serve as resources for training and compliance with HOPA Investigational Drug Service Best Practice Standards within the pharmacy department.
Finally, the new fee schedule provides an opportunity for additional fees versus the previous flat fee, which may aid in providing future resources for the department.
Limitations
Our results should be considered together with the study limitations. These results reflect the practices of a single community cancer center with a small sample size. In addition, we used a survey-based evaluation to gather data from oncology providers, research staff, and the pharmacy team.
Furthermore, our survey-based results are specific to our center and may not be externally valid to other community-based cancer centers. The benefit of the education and procedures was shown at our center, but other centers attempting to replicate all or portions of this study will need to consider their own needs carefully and the methods for assessing the implementation of such a service. Data about actual pharmacist and/or technician time before and after the implementation of an investigational drug service and records of actual fees paid after implementation are less subjective, but may take significant time to accrue in a smaller community center.
One issue that arose during our study was education compliance. One pharmacist and a technician were unavailable for the in-person education because of scheduling. This led to the pharmacist and the technician relying on the read-and-sign education and asking questions if issues arose.
In addition, there might have been some bias in the preimplementation survey by the research staff. These staff members had a pre-existing strong positive relationship with the pharmacy team, which might have made ranking any presurvey question lower on the Likert scale more difficult. We attempted to address this by making the survey anonymous, but bias may still exist because of the low number of participants.
Conclusion
The results of this study demonstrate improved confidence in the pharmacists’ and technicians’ abilities about standard operating procedures of a new formal investigational drug service, as well as the positive potential financial impact of a new standardized fee schedule for sponsored clinical trials. Our findings show that oncology providers’ and research staff’s satisfaction remained high after the implementation of the formal investigational drug service, and the perception of time commitment did not change after the new service implementation. In addition, the new fee schedule may help pharmacy departments generate new revenues when participating in sponsored clinical trials. Overall, our study demonstrates the feasibility to create a new formal investigational drug service using HOPA Investigational Drug Service Best Practice Standards guidelines at a community cancer center.
Author Disclosure Statement
Dr Zwart, Dr Cloud, Dr Kogelman, and Dr Boomershine have no conflicts of interest to report.
References
- ClinicalTrials.gov. Trends, charts, and maps. Updated June 26, 2022. https://clinicaltrials.gov/ct2/resources/trends. Accessed July 19, 2022.
- Hematology/Oncology Pharmacy Association. HOPA Investigational Drug Service Best Practice Standards. July 20, 2018. www.hoparx.org/images/hopa/resource-library/guidelines-standards/HOPA16_IDS_Guidelines.reviewed_2018.pdf. Accessed July 22, 2022.
- Kay SC, Luke DG, Tamer HR. ASHP guidelines for the management of investigational drug products. Am J Health Syst Pharm. 2018;75:561-573.
- Khandoobhai A, Poi M, Kelley K, et al. National survey of comprehensive pharmacy services provided in cancer clinical trials. Am J Health Syst Pharm. 2017;74(suppl 2):S35-S41.