Soft-tissue sarcomas are a group of rare diseases with approximately 13,400 new cases diagnosed annually in the United States.1 Angiosarcomas are one subtype of soft-tissue sarcoma of vascular origin.2 Angiosarcomas have a heterogeneous presentation. The primary site of angiosarcomas can be visceral or cutaneous, and the natural history of these different angiosarcoma subtypes can vary. The underlying genomic drivers for these angiosarcoma subtypes vary as well.3
Cutaneous angiosarcoma represents less than 1% of all soft-tissue sarcomas.4 Cutaneous angiosarcomas can arise de novo, in the setting of previous radiation, or in the setting of lymphedema (Stewart-Treves syndrome).4
Similar to soft-tissue sarcomas as a whole, localized cutaneous angiosarcomas can be treated with a combination of surgery and radiation; neoadjuvant or adjuvant chemotherapy can be considered as well.5 Cutaneous angiosarcomas are typically markedly infiltrative through the dermis and deeper tissues.5 Imaging can underestimate the local extent of disease, and extensive wide local resection is often required.5,6
Surgical resection can be disfiguring, particularly for patients with cutaneous angiosarcoma of the head and neck.5 Alternative treatment options to resection for primary angiosarcoma are radiation and chemotherapy.6 External beam radiotherapy and brachytherapy are comparable radiation oncology treatment options for angiosarcoma.7
Many patients, however, have locally recurrent or metastatic angiosarcoma.8 Cutaneous angiosarcomas are often sensitive to chemotherapy, such as paclitaxel 80 mg/m2 weekly.9 The combination of paclitaxel plus other drugs has not been shown to be superior to paclitaxel monotherapy.10 Although effective, such treatments are not curative, and the use of chemotherapy for cutaneous angiosarcoma in the neoadjuvant and adjuvant settings has not been investigated in a randomized clinical trial. Overall, treatment options for angiosarcoma are limited, and new treatment strategies are required.
Immunotherapy has become a standard treatment for many cancer subtypes. No immunotherapy is yet approved by the US Food and Drug Administration (FDA) for the treatment of soft-tissue sarcomas. The SARC028 clinical trial showed that immunotherapy with a programmed cell death 1 (PD-1) and PD ligand 1 (PD-L1) inhibitor can result in responses in patients with soft-tissue sarcoma, particularly in patients with undifferentiated pleomorphic sarcoma and dedifferentiated liposarcoma.11
Treatment response in other sarcoma subtypes, such as alveolar soft part sarcoma and dedifferentiated chondrosarcoma, has been investigated in a small prospective study and retrospective case studies.12,13 In another retrospective case study, patients with angiosarcoma responded to immune checkpoint inhibitor therapy with a PD-1 inhibitor or a cytotoxic T-lymphocyte–associated protein 4 inhibitor; a partial response was observed in 5 (71%) of 7 patients at 12 weeks, with 4 ongoing responses at the time of the reporting.14 The 7 patients included 5 patients with cutaneous angiosarcoma, 1 with breast angiosarcoma, and 1 with radiation-associated breast angiosarcoma.14 This case study did not show any potential biomarkers for cutaneous angiosarcoma response to immunotherapy.14
The genomic features and tumor microenvironment for some angiosarcomas have been investigated, with one analysis showing that 50% of patients with head and neck cutaneous angiosarcomas had a high tumor mutation burden and ultraviolet A (UVA) mutation signature, as well as a high tumor inflammation signature.15 Angiosarcoma that responds to immune checkpoint inhibitor therapy with a PD-1 or PD-L1 inhibitor should be correlated with such potential biomarkers for response.
We present the case of a patient with recurrent cutaneous angiosarcoma who had a complete pathologic response to immune checkpoint inhibitor therapy. All permissions and informed consent required by law and by the Dana-Farber Cancer Institute/Harvard Cancer Center were obtained for this publication.
Case Report
A 56-year-old man presented to Dana-Farber Cancer Institute with a right scalp lesion. An initial pathology review at an outside institution provided a broad differential diagnosis, including a malignant neoplasm and atypical fibroxanthoma. A pathology review at Brigham and Women’s Hospital showed a morphologically high-grade angiosarcoma, with immunohistochemistry that was positive for CD31 and ERG confirming the diagnosis.
The initial 3.4-cm scalp lesion was excised, with negative deep and periosteal margins, although the circumferential margins were positive. A re-excision showed no evidence of residual tumor. Staging with a computed tomography (CT) scan of the chest, abdomen, and pelvis showed no evidence of metastatic disease. The patient underwent a split-thickness skin graft and flap reconstruction of the scalp. He also received adjuvant radiation therapy.
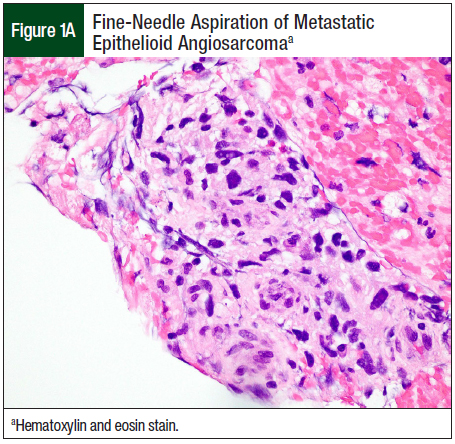
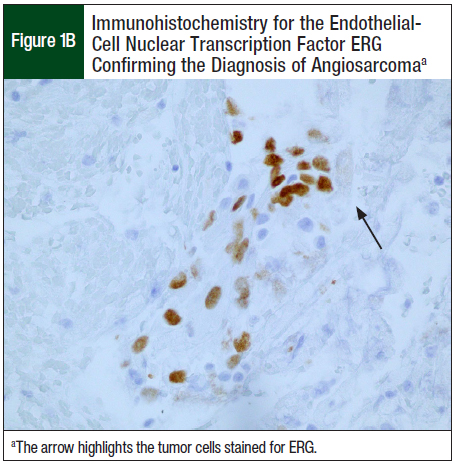
Ten months after the initial resection, the patient had right-face swelling. A CT scan of the face showed a 2.3-cm × 2.3-cm × 3.1-cm mass in the right parotid gland. Fine-needle aspiration showed metastatic epithelioid angiosarcoma (Figure 1A). Immunohistochemistry for the endothelial-cell nuclear transcription factor ERG confirmed the diagnosis of recurrent angiosarcoma (Figure 1B).
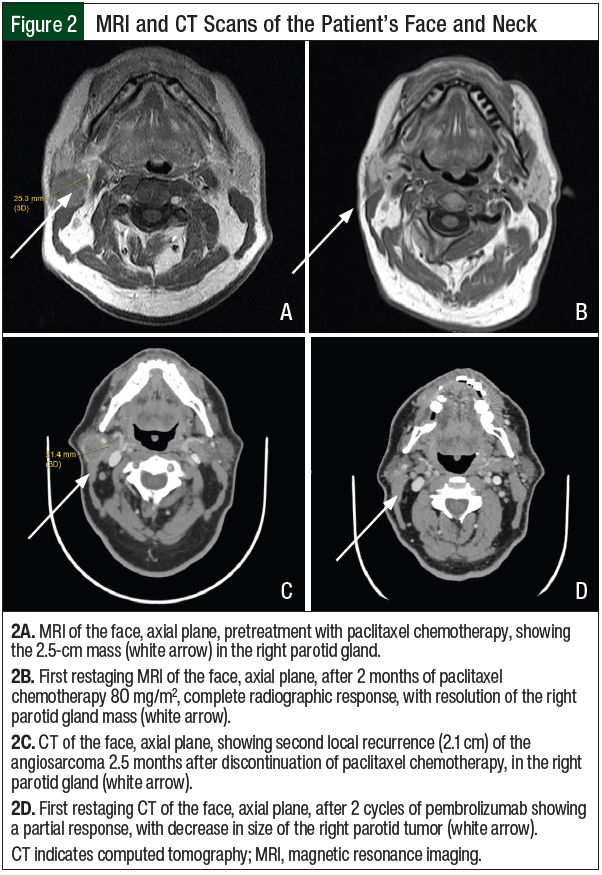
Repeated CT scan confirmed no evidence of other sites of metastatic disease. Pretreatment magnetic resonance imaging (MRI) scan of the face and neck showed a 2.5-cm tumor in the right parotid gland (Figure 2A). The patient received paclitaxel 80 mg/m2 and had a severe allergic reaction. He was seen in the allergy clinic for desensitization therapy. He then received weekly doses of paclitaxel 80 mg/m2 for 16 weeks, which was complicated by the development of grade 1 neuropathy. A restaging MRI after 2 months of paclitaxel therapy showed a radiographic complete response (Figure 2B). Although consolidative radiotherapy was planned, the patient could not tolerate the radiation oncology face mask because of his claustrophobia. Instead, he was placed on observation.
Two and a half months after the completion of paclitaxel chemotherapy, the patient had a recurrent right-face mass. A CT scan of the face showed a 2.1-cm recurrent right parotid mass (Figure 2C). Staging CT scans did not show evidence of additional metastatic disease.
Targeted next-generation sequencing was performed using OncoPanel.16 The targeted next-generation sequencing showed a tumor mutational burden of 17.5 mut/Mb, as well as a UVA signature with a high number of C>T transitions. Therefore, the patient received the PD-1 inhibitor pembrolizumab, 200 mg every 3 weeks. The tumor responded after 2 cycles of pembrolizumab treatment, resulting in an approximate 50% radiographic partial response from 2.1 cm to 1.3 cm (Figure 2D).
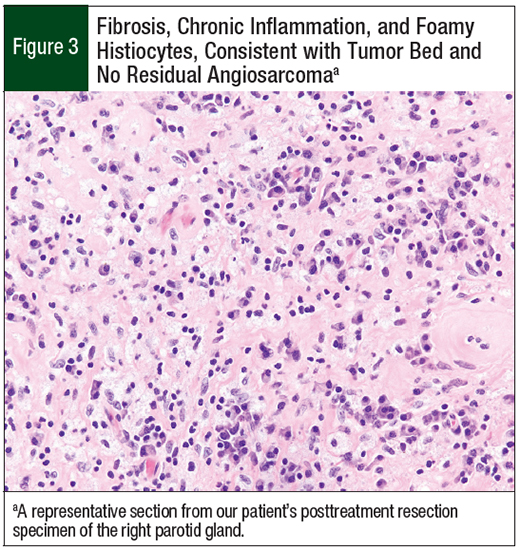
After 4 cycles of pembrolizumab treatment, the patient underwent right parotidectomy. This showed a 1.4-cm area of hyalinized fibrosis, hemosiderin-laden macrophages, and foreign-body giant-cell reaction, with no evidence of residual tumor, consistent with a 100% pathologic response (Figure 3). In addition, 13 lymph nodes were negative for angiosarcoma.
The patient completed 18 cycles of pembrolizumab treatment over 1 year; he was then observed for 11 months, with staging every 3 months. Since surgery, he has had no evidence of disease for at least 20 months, and has had negative surveillance CT scans. The patient is currently free of disease.
Discussion
The response of soft-tissue sarcoma to standard chemotherapy in the metastatic setting can be limited. The standard first-line therapy is doxorubicin alone or doxorubicin in combination with ifosfamide.17 The objective response rates vary from 10% to 20% for doxorubicin alone to 20% to 40% for doxorubicin plus ifosfamide.18,19 These 2 studies are limited by the inclusion of all soft-tissue sarcomas and the lack of subtype-specific evaluation of response.18,19
Angiosarcomas have been noted clinically to have a higher response rate to chemotherapy, although the overall response rates in the ANGIOTAX (ie, paclitaxel) and ANGIOTAX II (ie, paclitaxel and bevacizumab) clinical trials were 18% and 17%, respectively,9,10 which is similar to first-line treatment with doxorubicin. The 4-month progression-free survival rate in the ANGIOTAX study was 45%.9 The mean time to disease progression in the ANGIOTAX II study was 5.06 months.10 Based on the results of the ANGIOTAX study, the National Comprehensive Cancer Network guidelines recommend paclitaxel as a preferred first-line treatment option for angiosarcomas.17
In the study by Florou and colleagues, the response rate to treatment with an immune checkpoint inhibitor in 7 patients with angiosarcoma was 71%, and the progression-free survival rate was 57% at a median of 3.4 months.14 This response rate was higher than that reported in patients with angiosarcoma who received standard chemotherapy,9,10 although admittedly the sample size was small14; the progression-free survival was comparable. The results of the study by Florou and colleagues14 suggest that immunotherapy is worth exploring for the treatment for angiosarcomas.
Still remains to be explored is the combination of oral targeted therapy, such as vascular endothelial growth factor inhibitors (eg, pazopanib), or systemic chemotherapy plus immunotherapy, in patients with angiosarcomas. Given the known response of microtubule inhibitors in angiosarcoma, this type of therapy would be an ideal systemic chemotherapy to combine with immunotherapy. The adverse events associated with these agents are not necessarily overlapping, and therefore could potentially be tolerable.
Such combinations of immunotherapy and chemotherapy could be explored in future clinical trials. For example, a phase 2 clinical trial, which was presented at the Connective Tissue Oncology Society virtual meeting in 2021, is exploring the role of eribulin combined with pembrolizumab for the treatment of leiomyosarcoma, liposarcomas, and other sarcomas, which could include angiosarcoma.20,21 In addition, a clinical trial currently enrolling patients (NCT04339738) is examining the combination of nivolumab plus paclitaxel or cabozantinib for the treatment of angiosarcoma.22
The treatment course of pembrolizumab 200 mg every 3 weeks for a responding patient (for all tumors) was initially 1 year. This treatment course has since been extended to 2 years for responding patients.23,24 Our patient received pembrolizumab for 1 year, with no evidence of local or distant disease recurrence. He was given the option to continue treatment with pembrolizumab for a total of 2 years, but he chose to discontinue pembrolizumab after 1 year and 18 cycles.
The length of therapy with immune checkpoint inhibitor in patients with sarcoma has not been adequately studied. The standard length of immune checkpoint therapy should also be applied to soft-tissue sarcomas, and should be adjusted on a per-patient basis.
The ideal timing of immunotherapy in the treatment of soft-tissue sarcoma, including angiosarcoma, is being investigated.25 It has not yet been elucidated whether the timing of immunotherapy for the treatment of metastatic angiosarcomas can influence the probability of response. Would earlier treatment in the metastatic setting be beneficial? Does the burden of disease influence the response? These questions remain open. The SARC032 clinical trial is investigating the role of neoadjuvant pembrolizumab in undifferentiated pleomorphic sarcoma and liposarcomas, specifically pleomorphic and dedifferentiated liposarcomas.25 This is the first study to examine immunotherapy in the neoadjuvant or adjuvant setting of soft-tissue sarcomas. If the results are encouraging, the strategy should then be examined in other types of sarcoma, including patients with angiosarcomas. As in other cancer types, standard chemotherapy could be combined with immunotherapy to further increase the chance of response, improved progression-free survival, and better overall survival.26
There is a long history of using immunotherapy for soft-tissue sarcoma, although to date the use of immunotherapy strategies in soft-tissue sarcoma has been limited.27 The potential biomarkers related to immunotherapy include mismatch repair deficiency, which rarely occurs in soft-tissue sarcomas,28 and a high mutational load. An additional marker is the presence of PD-L1 staining, although this is not a proved biomarker in soft-tissue sarcomas.29 The FDA has approved the use of a PD-1 or a PD-L1 inhibitor in tumors with mismatch repair deficiency and/or high mutational load (>10 mut/Mb), regardless of tumor type.30-32
Approximately 50% of head and neck cutaneous angiosarcomas have a high mutational load, as well as a UVA damage signature.15 Therefore, immunotherapy may be a reasonable strategy in patients with these tumor types. However, it requires investigation to determine what the best biomarker is for response in cutaneous angiosarcoma. Tumor inflammation signature score may also be a predictor of response.15
Conclusion
Cutaneous angiosarcoma is a sarcoma subtype that may have a propensity to respond to immunotherapy with a PD-1 and/or a PD-L1 inhibitor. Our patient case exemplifies the excellent response to immunotherapy alone that can be achieved in patients with cutaneous angiosarcoma. This response could potentially be predicted by a high mutational load or a UVA signature, both of which are common in patients with cutaneous angiosarcomas. Treatment with immunotherapy in cutaneous angiosarcomas should be investigated further in a prospective clinical trial.
Author Disclosure Statement
Dr Nathenson is currently an employee of GlaxoSmithKline; Dr Hornick is a consultant to Aadi Biosciences and TRACON Pharmaceuticals.
References
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33. Erratum in: CA Cancer J Clin. 2021;71:359.
- WHO Classification of Tumours Editorial Board, ed. Soft Tissue and Bone Tumours. Lyon, France: International Agency for Research on Cancer; 2020. WHO Classification of Tumours, 5th ed, vol 3.
- Painter CA, Jain E, Tomson BN, et al. The Angiosarcoma Project: enabling genomic and clinical discoveries in a rare cancer through patient-partnered research. Nat Med. 2020;26:181-187.
- Conic RRZ, Damiani G, Frigerio A, et al. Incidence and outcomes of cutaneous angiosarcoma: a SEER population-based study. J Am Acad Dermatol. 2020;83:809-816.
- Guadagnolo BA, Zagars GK, Araujo D, et al. Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck. 2011;33:661-667.
- Mitra D, Devlin PM, Buzurovic I, et al. Clinical outcomes following high-dose-rate surface applicator brachytherapy for angiosarcoma of scalp and face. J Contemp Brachytherapy. 2021;13:172-178.
- Mitra D, Pei Y, Buzurovic I, et al. Angiosarcoma of the scalp and face: a dosimetric comparison of HDR surface applicator brachytherapy and VMAT. Sarcoma. 2020;2020:7615248. doi: 10.1155/2020/7615248.
- Lahat G, Dhuka AR, Lahat S, et al. Outcome of locally recurrent and metastatic angiosarcoma. Ann Surg Oncol. 2009;16:2502-2509.
- Penel N, Nguyen Bui B, Bay JO, et al. Phase II trial of weekly paclitaxel for unresectable angiosarcoma: the ANGIOTAX study. J Clin Oncol. 2008;26:5269-5274.
- Bui N, Kamat N, Ravi V, et al. A multicenter phase II study of Q3 week or weekly paclitaxel in combination with bevacizumab for the treatment of metastatic or unresectable angiosarcoma. Rare Tumors. 2018;10:2036361318771771. doi: 10.1177/2036361318771771.
- Tawbi HA, Burgess M, Bolejack V, et al. Pembrolizumab in advanced soft-tissue sarcoma and bone sarcoma (SARC028): a multicentre, two-cohort, single-arm, open-label, phase 2 trial. Lancet Oncol. 2017;18:1493-1501. Errata in: Lancet Oncol. 2017;18:e711; Lancet Oncol. 2018;19:e8.
- Brahmi M, Vanacker H, Dufresne A. Novel therapeutic options for alveolar soft part sarcoma: antiangiogenic therapy, immunotherapy and beyond. Curr Opin Oncol. 2020;32:295-300.
- Kostine M, Cleven AHG, de Miranda NFCC, et al. Analysis of PD-L1, T-cell infiltrate and HLA expression in chondrosarcoma indicates potential for response to immunotherapy specifically in the dedifferentiated subtype. Mod Pathol. 2016;29:1028-1037.
- Florou V, Rosenberg AE, Wieder E, et al. Angiosarcoma patients treated with immune checkpoint inhibitors: a case series of seven patients from a single institution. J Immunother Cancer. 2019;7:213. doi: 10.1186/s40425-019-0689-7. Erratum in: J Immunother Cancer. 2019;7:285.
- Chan JY, Lim JQ, Yeong J, et al. Multiomic analysis and immunoprofiling reveal distinct subtypes of human angiosarcoma. J Clin Invest. 2020;130:5833-5846.
- Garcia EP, Minkovsky A, Jia Y, et al. Validation of OncoPanel: a targeted next-generation sequencing assay for the detection of somatic variants in cancer. Arch Pathol Lab Med. 2017;141:751-758.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Soft Tissue Sarcoma. Version 2.2022. May 17, 2022.
- Tap WD, Wagner AJ, Schöffski P, et al; for the ANNOUNCE investigators. Effect of doxorubicin plus olaratumab vs doxorubicin plus placebo on survival in patients with advanced soft tissue sarcomas: the ANNOUNCE randomized clinical trial. JAMA. 2020;323:1266-1276.
- Judson I, Verweij J, Gelderblom H, et al; for the European Organisation and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. Doxorubicin alone versus intensified doxorubicin plus ifosfamide for first-line treatment of advanced or metastatic soft-tissue sarcoma: a randomised controlled phase 3 trial. Lancet Oncol. 2014;15:415-423.
- A Phase II Study of Eribulin and Pembrolizumab in Soft Tissue Sarcomas. NLM identifier: NCT03899805. Updated May 25, 2022. https://clinicaltrials.gov/ct2/show/NCT03899805. Accessed September 1, 2022.
- Nathenson MJ, Choy E, Mazzola E, et al. Phase II study of eribulin and pembrolizumab in patients with metastatic soft tissue sarcoma. Abstract 32. Presented at the Connective Tissue Oncology Society virtual annual meeting; November 10-13, 2021.
- Testing the Addition of Nivolumab to Chemotherapy in Treatment of Soft Tissue Sarcoma. NLM identifier: NCT04339738. Updated September 1, 2022. https://clinicaltrials.gov/ct2/show/NCT04339738. Accessed September 2, 2022.
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck & Co; August 2022. www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf. Accessed September 2, 2022.
- Fradet Y, Bellmunt J, Vaughn DJ, et al. Randomized phase III KEYNOTE-045 trial of pembrolizumab versus paclitaxel, docetaxel, or vinflunine in recurrent advanced urothelial cancer: results of >2 years of follow-up. Ann Oncol. 2019;30:970-976.
- A Randomized Trial of Pembrolizumab & Radiotherapy Versus Radiotherapy in High-Risk Soft Tissue Sarcoma of the Extremity (SU2C-SARC032). NLM identifier: NCT03092323. Updated March 28, 2022. https://clinicaltrials.gov/ct2/show/NCT03092323. Accessed September 2, 2022.
- Uppaluri R, Campbell KM, Egloff AM, et al. Neoadjuvant and adjuvant pembrolizumab in resectable locally advanced, human papillomavirus-unrelated head and neck cancer: a multicenter, phase II trial. Clin Cancer Res. 2020;26:5140-5152.
- Nathenson MJ, Conley AP, Sausville E. Immunotherapy: a new (and old) approach to treatment of soft tissue and bone sarcomas. Oncologist. 2018;23:71-83.
- Doyle LA, Nowak JA, Nathenson MJ, et al. Characteristics of mismatch repair deficiency in sarcomas. Mod Pathol. 2019;32:977-987.
- D’Angelo SP, Shoushtari AN, Agaram NP, et al. Prevalence of tumor-infiltrating lymphocytes and PD-L1 expression in the soft tissue sarcoma microenvironment. Hum Pathol. 2015;46:357-365.
- US Food and Drug Administration. FDA approves pembrolizumab for adults and children with TMB-H solid tumors. June 16, 2020. www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-adults-and-children-tmb-h-solid-tumors. Accessed September 2, 2022.
- Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357:409-413.
- Asaoka Y, Ijichi H, Koike K. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;373:1979.