Melanoma remains a challenging disease. Although it represents less than 2% of all skin cancers, it accounts for the majority of skin cancer–related deaths.1 It is estimated that 76,100 new cases were diagnosed in 2014 and more than 9700 melanoma-related deaths occurred in the same year.2 Unfortunately, the incidence of melanoma continues to increase dramatically despite aggressive skin cancer awareness and prevention strategies. Advances in systemic therapies have improved outcomes for patients with unresectable or metastatic disease. However, the 5-year overall survival (OS) rate for patients with metastatic melanoma remains poor, at 16%.1
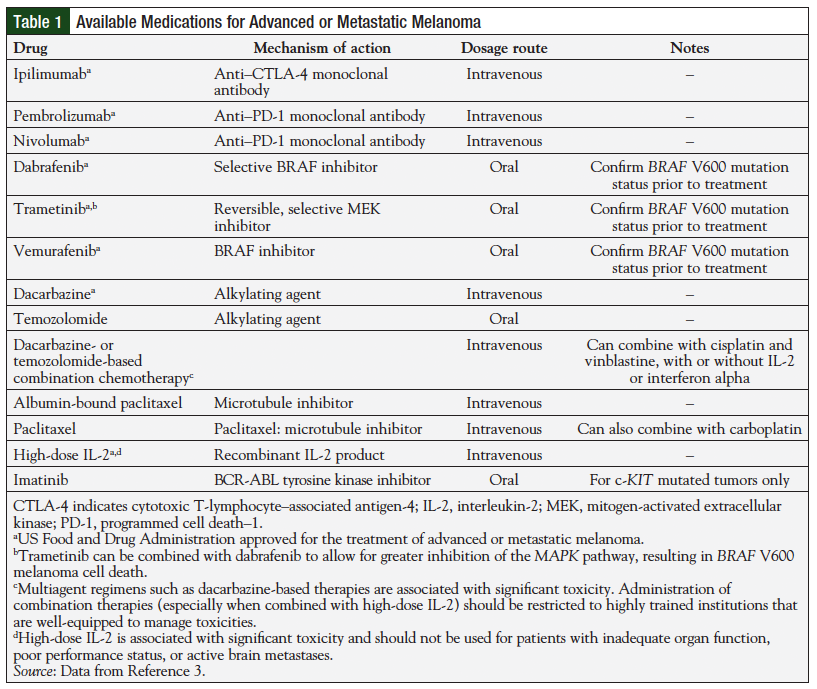
Cytokine-based immunotherapy is an important treatment strategy for patients with advanced melanoma requiring adjuvant therapy for stage III disease and for those with unresectable tumors. Moreover, the development of newer targeted therapies such as BRAF inhibitors and the immune checkpoint receptor inhibitor ipilimumab have further improved outcomes for these patients. Unfortunately, melanoma cells perpetually evolve to develop mechanisms to avoid immune recognition and antitumor effects.3-5 For this reason, limited subsequent treatment options exist for patients with advanced disease for whom previous immunotherapy has failed, such as interferon (IFN), interleukin-2 (IL-2), or the cytotoxic T-lymphocyte–associated antigen-4 (CTLA-4) receptor-blocking monoclonal antibody ipilimumab. Treatment options for advanced or metastatic disease, as recommended by the National Comprehensive Cancer Network, are listed in Table 1.6 Therefore, the development of additional therapeutic agents that target unique immune system receptors to promote anticancer immunity has become an important goal for this patient population.
The programmed cell death–1 (PD-1) receptor is an inhibitory receptor expressed by T-cells. Its primary ligand, PD-L1, is frequently overexpressed on melanoma cells to avoid T-cell recognition and evade the immune response and antitumor activity.4 Pembrolizumab (formerly MK-3475 and lambrolizumab) is the first anti–PD-1 monoclonal antibody approved by the US Food and Drug Administration (FDA) in September 2014 for the treatment of patients with unresectable or metastatic melanoma who have experienced disease progression following treatment with ipilimumab and, in BRAF–V600–mutated tumors, after treatment with a BRAF inhibitor such as dabrafenib or vemurafenib. This immune checkpoint inhibitor was granted accelerated approval based on preliminary data showing a high rate of sustained tumor regression, durability of response, and a manageable safety profile. Whether there is improvement in the survival rate or disease-related symptoms has not yet been established. Continued approval for this indication may be contingent on verification and description of clinical benefit in the confirmatory trials.7
Pharmacology and Pharmacokinetics
Cancer cells perpetually evolve to develop mechanisms to avoid immune cell recognition and subsequent antitumor functions.3 One such adaptation that has been discovered is the PD-1 receptor/PD-L1 and PD-L2 pathway. The PD-1 receptor is expressed by T-cells and is a key immune checkpoint receptor, which, when engaged by PD-L1 or PD-L2, exhibits a negative regulatory role.4 This negative feedback loop is essential for maintaining normal immune responses and limiting T-cell activity to protect normal cells during chronic inflammation.8 The PD-1 inhibitory receptor is expressed by T-cells, whereas PD-1 ligands are frequently expressed within the tumor microenvironment surrounding cancer cells and macrophages, in response to inflammation. Tumor cells that transgenically express PD-L1 demonstrated increased resistance to T-cell-mediated lysis in vitro and greater tumorigenesis in vivo compared with tumor cells that did not express PD-L1.9 Anti-PD-L1 antibody was sufficient to reverse these effects. In mice injected with tumor cells naturally expressing PD-L1, administration of anti-PD-L1 antibodies resulted in significant, albeit transient, suppression of tumor growth. Tumor growth was completely suppressed in PD-1-/- mice injected with tumor cells endogenously expressing PD-L1. Therefore, disruption of the PD-L1/PD-1 interaction could be an effective immunotherapeutic strategy.9 Furthermore, in mice with existing tumors, administration of anti-PD-1 and/or anti-PD-L1 antibodies resulted in reduced tumor burden and longer survival.10
Pembrolizumab is a highly selective humanized immunoglobulin G4 monoclonal antibody that binds to the PD-1 receptor on T-cells to block the interaction of the PD-1 ligand with its receptor. By blocking the inhibitory interaction between immune-surveillance T-cells and tumor cells, the cytotoxic function of tumor-specific T-cells can continue, potentially leading to decreased tumor proliferation.7
The pharmacokinetic profile of pembrolizumab was evaluated in clinical studies in patients who received doses of 1 to 10 mg/kg every 2 weeks or 2 to 10 mg/kg every 3 weeks. Steady-state drug concentrations were reached by 18 weeks with repeated dosing every 3 weeks. The peak concentration, trough concentration, and area under the curve at steady state of pembrolizumab increased proportionally according to dose in those who received 2 to 10 mg/kg every 3 weeks.7
The effects of various patient-specific parameters were evaluated in population pharmacokinetic analyses. No clinically important effect on the clearance of pembrolizumab was found between age (18-94 years), sex, or tumor burden. Moreover, no differences in pembrolizumab clearance were found for patients with mild (glomerular filtration rate [GFR] 60-89 mL/min/1.73 m2), moderate (GFR 30-59 mL/min/1.73 m2), or severe (GFR 15-29 mL/min/1.73 m2) renal impairment compared with those with normal renal function. Similarly, no clinically important differences were discovered among patients with mild hepatic impairment compared with those with normal hepatic function. Pembrolizumab has not been studied in patients with moderate or severe hepatic impairment.7
Clinical Studies
The FDA granted accelerated approval of pembrolizumab based on data from KEYNOTE-001, a large phase 1b clinical trial that evaluated pembrolizumab monotherapy in several types of solid tumors.7 The study was composed of a dose-escalation phase in patients with advanced solid tumors, followed by safety and efficacy evaluations in patients with melanoma and non–small-cell lung cancer (NSCLC); several of these evaluations are still ongoing.7,11,12 We will review the data reported for the melanoma cohorts of this trial.
The safety of pembrolizumab was evaluated in a nonrandomized expansion cohort of patients with advanced melanoma, at 3 dose levels: 2 mg/kg every 3 weeks, 10 mg/kg every 3 weeks, and 10 mg/kg every 2 weeks.13 One hundred thirty-five patients were enrolled in this cohort, including 48 who had previous exposure to ipilimumab. Most adverse events were grade 1 or 2. Fatigue (30%), rash (21%), pruritus (21%), diarrhea (20%), and myalgia (12%) were the most frequently reported. Grade 3 or 4 adverse events occurred in 13% of patients, including renal failure, aminotransferase elevations, and hypothyroidism. Pneumonitis occurred in 6 (4%) patients and was considered related to the study medication in all 6; however, all cases were low grade. Not surprisingly, patients who received the highest dose (10 mg/kg every 2 weeks) had the highest incidence of adverse events (23%; vs 9% and 4% in the groups that received 2 mg/ kg or 10 mg/kg every 3 weeks, respectively).13
Response rate was assessed as a secondary end point. Across all dosing cohorts, the overall response rate (ORR) was 38%, and the median duration of response was not reached at the time of analysis (median follow-up, 11 months).13 Although the majority of responses were seen at 12 weeks with the first scheduled imaging, some were seen several weeks later. In 1 patient, treatment response was observed after 48 weeks. Prior exposure to immunotherapy, including ipilimumab, did not impact the response rate.13
The acceptable safety profile led to the formation of an additional expansion cohort to evaluate efficacy in patients with advanced melanoma who experienced progression following ipilimumab exposure or, in cases of BRAF V600 mutation, following a BRAF or MEK inhibitor (or both).12 In this open-label study, 173 patients were randomly assigned to receive pembrolizumab 2 mg/kg or 10 mg/kg every 3 weeks until disease progression, unacceptable toxicity, protocol violation, or withdrawal of consent occurred. Excluded from participation were patients with a history of serious immune-related adverse events and those whose ipilimumab-related adverse events had not resolved to grade 0 or 1. ORR was the primary end point, and progression-free survival (PFS), OS rate, and response duration were secondary end points. The majority (64%) of enrollees had stage M1c, and more than one-third had elevated concentrations of lactate dehydrogenase at baseline,12 both of which are poor prognostic indicators.13
After the median follow-up period of 8 months, the ORR was 26% for both treatment arms, and median duration of response had not been reached in either arm.12 Although disease progression was the most common reason for treatment discontinuation, 42% of patients were still receiving treatment at the time of analysis. Most responses were noted at approximately 12 weeks after treatment initiation, but 1 patient responded 36 weeks after starting therapy. The median PFS periods and 1-year OS rates were 22 weeks and 58% for the 2-mg/kg arm and 14 weeks and 63% for the 10-mg/kg arm, respectively.12 Patients with BRAF V600 mutations had a slightly lower response rate (19%), but this patient population was small (n = 26). Common adverse effects were similar to those reported in the safety analysis. Grade 3 or 4 adverse effects were reported for 12% of patients and included fatigue, autoimmune hepatitis, rash, and pancreatitis. As in the safety cohort, pneumonitis occurred, but in only 1 patient; the condition was high grade.12
Adverse Effects
Overall, pembrolizumab was very well tolerated in patients with advanced melanoma, including those who were heavily pretreated and had received immunotherapy previously. Only 4% to 9% of patients required drug discontinuation because of treatment-related adverse effects.14 The most common adverse effects were low grade and typically dermatologic or gastrointestinal in nature. Diarrhea, fatigue, rash, pruritus, constipation, nausea, decreased appetite, and cough were reported for ≥20% of patients.15
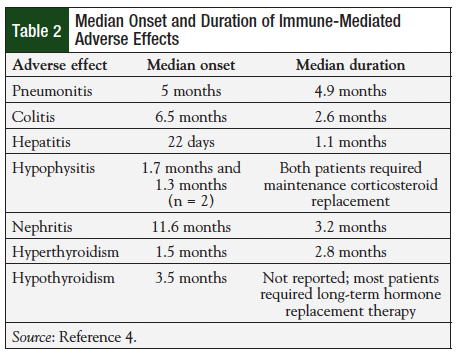
Due to the immune-modulating mechanism of action of pembrolizumab, adverse effects that could be considered immune-related were of special interest.12 Several adverse effects were thought to be immune-mediated, including pneumonitis, colitis, hepatitis, hypophysitis, nephritis and renal failure, hyper- and hypothyroidism, exfoliative dermatitis, uveitis, myositis, pancreatitis, hemolytic anemia, and adrenal insufficiency.7,15 All occurred infrequently, the most common being pneumonitis, which was reported for 2.9% of patients.7 The median onset and duration of immune-mediated adverse effects are shown in Table 2. The severity of immune-mediated adverse effects was typically mild, with grade 3 or higher colitis, hepatitis, and pneumonitis occurring in 0.5%, 0.2%, and 0.2% of patients, respectively.7 No deaths related to pembrolizumab treatment have been reported.
The manufacturer recommends administration of corticosteroids for grade 2 or higher immune-mediated pneumonitis, colitis, hepatitis, hypophysitis, and nephritis.7 Isolated hypothyroidism may be managed with thyroid hormone replacement if indicated, but corticosteroids are recommended for hypo- or hyperthyroidism of grade 3 or higher. The majority of patients in the KEYNOTE-001 trial who required high-dose corticosteroid treatment (≥ 40 mg prednisone or equivalent daily) for immune-mediated adverse events were eventually able to taper off the corticosteroids and restart pembrolizumab therapy.7 Treatment discontinuation is recommended for grade 3 or higher pneumonitis or nephritis, and for grade 4 colitis, hypophysitis, hyperthyroidism, or hypothyroidism. Pembrolizumab's pregnancy category is D, and breastfeeding during pembrolizumab treatment is not recommended.7
Although the tolerability of pembrolizumab appears to be excellent, it should be noted that available data are limited to a phase 1 clinical study, and experience with this agent is minimal. In addition to being cognizant of the expected adverse effects noted herein, clinicians must be attentive to unexpected and previously unreported adverse effects that may occur when pembrolizumab is used outside the clinical trial setting.
Drug Interactions
Drug interactions with pembrolizumab have not been formally studied or established.7
Dosage and Administration
The recommended dosage of pembrolizumab is 2 mg/kg administered intravenously over 30 minutes.7 Findngs have not shown that a higher dose, specifically 10 mg/kg, is superior to 2 mg/kg in terms of safety or efficacy (see Clinical Studies section).12 Therefore, no maximum dose has been established. Pembrolizumab should be repeated every 3 weeks until the disease progresses or until intolerable toxicity occurs.7
In patients with hepatotoxicity whose aspartate aminotransferase (AST) or alanine transaminase (ALT) is greater than 3 to 5 times the upper limit of normal (ULN), or whose total bilirubin exceeds 1.5 to 3 times the ULN, treatment should be withheld and therapy resumed only if toxicity recovers to grade 1 or lower.7 In patients with hepatotoxicity and AST or ALT greater than 5 times the ULN, or with total bilirubin exceeding 3 times the ULN, therapy should be permanently discontinued. Although the manufacturer recommends dose modification or treatment discontinuation for nephritis, there are no renal-related adjustment guidelines based on laboratory values such as serum creatinine or creatinine clearance.7 Additional conditions prompting dose withholding or discontinuation, as recommended by the manufacturer, are listed in Table 3.7
Future Implications
Much progress has been made in the treatment of advanced and metastatic melanoma with the advent of targeted immunotherapy agents such as pembrolizumab and other agents that modulate PD-1 and PD-L1. These medications may improve clinical efficacy for patients with melanoma and may have better toxicity profiles than older, nontargeted chemotherapy and immunotherapy regimens. Future research should focus on assessing which patients may derive the most benefit from pembrolizumab as well as utilizing combination therapy with other immune checkpoint inhibitors and/or oral targeted agents. Several clinical trials are ongoing to further define the role of pembrolizumab in the treatment of advanced melanoma. In addition to the ongoing KEYNOTE trial, pembrolizumab is currently being evaluated in various combinations with ipilimumab, trametinib/dabrafenib, and pegylated interferon. The benefits of this PD-1 inhibitor may extend to the treatment of other solid tumor types, and further evaluation is warranted.
In December 2014, the FDA approved a second PD-1 inhibitor, nivolumab, for the treatment of unresectable or metastatic melanoma and disease progression following treatment with ipilimumab and, in cases of BRAF V600 mutation, with a BRAF inhibitor. In March 2015, nivolumab was approved for the treatment of patients with metastatic squamous NSCLC following progression during or after platinum-based chemotherapy. Accelerated approval for the melanoma indication was based on a multicenter open-label trial in which 370 patients with unresectable or metastatic melanoma were randomly assigned (2:1) to receive nivolumab 3 mg/kg every 2 weeks or the investigator's choice of chemotherapy (dacarbazine or carboplatin plus paclitaxel) every 3 weeks.16 ORR for the 120 patients included in the efficacy analysis was 32%, and 4 responses were complete. At the time of analysis, 13 patients were experiencing ongoing responses after 6 months of treatment. The most common adverse events reported for nivolumab-treated patients were rash (21%), pruritus (19%), cough (17%), upper respiratory tract infection (11%), and peripheral edema (10%).16 At this time, it is challenging to compare the 2 available PD-1 inhibitors in terms of efficacy and safety because no head-to-head clinical trial exists. Dose frequency should also be considered in overall cost estimations. As data for clinical trials mature and these agents are used outside of clinical trials, differences between these drugs may become more apparent. Numerous clinical trials are ongoing to evaluate the benefits of pembrolizumab or nivolumab for treatment of other advanced tumors, including renal-cell carcinoma, NSCLC, prostate cancer, and breast cancer.
Conclusion
Advanced or metastatic melanoma often carries a grim prognosis, especially given the limited number of effective treatment options. FDA approval of the anti-PD-1 monoclonal antibody pembrolizumab represents an important step in the treatment of advanced and metastatic melanoma. Pembrolizumab is an additional treatment option for patients whose disease did not respond successfully to ipilimumab and, if BRAF-mutated, a BRAF inhibitor such as dabrafenib or vemurafenib. Results of clinical trials suggest that some patients can achieve a high rate of tumor regression while experiencing minimal therapy-related toxicity. Although the majority of patients experienced only grade 1 or 2 adverse events, prompt evaluation and treatment should be given to patients with possible immune-mediated toxicities because of the potentially serious nature of these adverse effects. Due to the promising early results experienced with PD-1 inhibitors in melanoma, clinical trials are ongoing to evaluate pembrolizumab in combination with other therapies in the treatment of melanoma and other solid tumors.
Author Disclosure Statement
The authors reported no conflicts of interest.
Dr Baxley and Dr Huddleston are Clinical Pharmacists, Stephenson Cancer Center, University of Oklahoma Health Sciences Center, Oklahoma City, OK. Dr Johnson is Staff Pharmacist, Norman Regional Health System, Norman, OK.
References
1. American Cancer Society. Cancer facts & figures 2014. www.cancer.org/acs/ groups/content/@research/documents/webcontent/acspc-042151.pdf. Published 2014. Accessed June 16, 2015.
2. Siegel R, Ma J, Zou Z, Jemal A. Cancer Statistics, 2014. CA Cancer J Clin. 2014;64:9-29.
3. Ishida Y, Agata Y, Shibahara K, Honjo T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992;11:3887-3895.
4. Freeman GJ, Long AJ, Iwai Y, et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med. 2000;192:1027-1034.
5. Dong H, Strome SE, Salomao DR, et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med. 2002;8:793- 800.
6. National Comprehensive Cancer Network. Melanoma. Version 3.2015. www. nccn.org/professionals/physician_gls/pdf/melanoma.pdf. Published March 11, 2015. Accessed June 17, 2015.
7. Keytruda [package insert]. Whitehouse Station, NJ: Merck Sharp & Dohme Corp; 2014.
8. Liang SC, Latchman YE, Buhlmann JE, et al. Regulation of PD-1, PD-L1, and PD-L2 expression during normal and autoimmune responses. Eur J Immunol. 2003;33:2706-2716.
9. Iwai Y, Ishida M, Tanaka Y, et al. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci U S A. 2002;99:12293-12297.
10. Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264.
11. Patnaik A, Kang SP, Rasco D, et al. Phase I study of pembrolizumab (MK-3475; anti-PD-1 monoclonal antibody) in patients with advanced solid tumors. Clin Cancer Res. 2015;21:4286-4293.
12. Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384:1109-1117.
13. Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
14. Ribas F, Hodi FS, Kefford R, et al. Efficacy and safety of the anti-PD-1 monoclonal antibody MK-3475 in 411 patients (pts) with melanoma (MEL). J Clin Oncol. 2014;32(18 Suppl):LBA9000.
15. Supplement to: Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014; published online July 15.
16. Opdivo [package insert]. Lawrence Township, NJ: Bristol-Myers Squibb Company; 2014.