The use of oral medications in cancer treatment has increased considerably the past several years with the development of novel oral anticancer agents. The US Food and Drug Administration approximates that more than one-quarter of the 400 antineoplastic agents currently in the pipeline are oral therapies.1 Currently, a combination of both cytotoxic and small-molecule inhibitors are included as the mainstay of oral anticancer agents, but it is expected that the majority of newly developed oral agents will target specific receptors and cancer microenvironments.2 A final consensus statement on prescribing, monitoring, and patient education for oral anticancer agents was recently created by the American Society of Clinical Oncology (ASCO) and the Oncology Nursing Society (ONS), and was incorporated into the 2013 update of the ASCO/ONS Chemotherapy Administration Safety Standards.3
Oral anticancer therapy provides multiple benefits, including sophisticated mechanisms of action, genomic targeting, and improvement in patient quality of life. Patients are able to spend more time at home, and allowed the autonomy to administer medications at their own convenience.2 However, aspects of oral therapy that need to be addressed include adherence, side-effect management, and patient follow-up.1
Although many oral anticancer agents are generally well-tolerated by patients, they are not without side effects.1 Similar to their intravenous counterparts, oral anticancer medications have a narrow therapeutic index.4 This makes the potential for serious adverse events—including those from drug–drug interactions—a significant concern.4 Adherence is another potentially serious concern with oral anticancer agents. In a study by Noens and colleagues, patient nonadherence was associated with a suboptimal response to therapy.5 A collaborative agreement between physicians and patients addressing adherence should be established prior to prescribing an oral anticancer agent, and a process of assessing adherence should be maintained to ensure optimal benefit from therapy.
Cost is another concern because oral anticancer medications are one of the fastest growing expenses for third-party payers,6 with medications costing up to $10,000 per month per prescription.7 Medicare Part D changes have drastically transformed the historic reimbursement format for oral anticancer agents.1 Although Medicare Part D programs cover a significant portion of oral anticancer medication costs, copays can still be substantial for patients because of their high expense. Thus, financial burden could affect therapy decisions made by physicians and patients.1
Oral Anticancer Therapy Guidelines
In March 2013, members of ASCO and ONS revised their standards for safe chemotherapy administration.3 Documentation standards for patient chemotherapy medical records were included in these guidelines. These guidelines suggest that documentation include: patient comprehension regarding oral anticancer regimens, oral anticancer drugs, doses, and anticipated duration of therapy.3 Patient education guidelines suggest providing educational materials that are at an appropriate reading level for patients, as well as informing patients about possible side effects, how to contact their healthcare providers, and symptoms that would require a call.3 These guidelines were updated to address many of the safety issues regarding oral anticancer therapy use. These include ensuring adequate patient education regarding safe handling and disposal, addressing key barriers before initiation of oral anticancer therapy (eg, addressing financial concerns), screening for drug–drug interactions and performance of medication reconciliation at each clinic visit, and monitoring adherence to the oral anticancer therapy.3
In addition, in fall 2012, ASCO released an update to the Quality Oncology Practice Initiative (QOPI), which included 25 core measures.8 This update introduced guidelines for oral anticancer therapy, which are included in the Core 13 measures. Quality improvement guidelines include a documented plan for oral anticancer therapy consisting of: administration schedule, safe handling, start date, missed doses, food and drug interactions, side effects and toxicities, and clinic contact instructions. The initiative also suggests that medication adherence assessment be performed.8 Compliance with both the QOPI and ASCO/ONS standards is crucial to ensuring optimal patient care in this high-risk population. Need for an Outpatient Oral Anticancer Program.
Need for an Outpatient Oral Anticancer Program
In 2007, Weingart and colleagues published a survey evaluating oral anticancer therapy safety practices at cancer centers in the United States.9 They concluded that most organizations had no required elements for prescribing oral anticancer therapy. Furthermore, there was considerable inconsistency with patient education, and little infrastructure to support adherence to treatment.9 Although pharmacists were widely available at most centers, the majority of patients receiving oral anticancer agents were never counseled by a pharmacist, and pharmacy services were underutilized. Because of the lack of safeguards to support safe oral anticancer therapy use, it is not surprising that respondents at 10 (20%) of 49 of the centers self-reported that a serious adverse event related to oral anticancer therapy had occurred in the past year.9
Providing patients with thorough and accurate information about their anticancer regimens is of utmost importance. 2 There are numerous opportunities to improve quality in the prescribing and monitoring of patients receiving oral anticancer medications. It is imperative that patients have an opportunity to address their concerns about side effects, cost of medications, and medication adherence strategies.
As we discussed earlier, the financial barriers of oral anticancer therapy warrant patient advocacy and support in ensuring reasonable access based on drug cost. Opportunities exist for guaranteeing that patients are able to receive their medications in a timely fashion, including taking an active role in medication acquisition through obtaining prior authorizations, copay assistance, navigation of limited distribution programs, and other patient support services.
Early study results are becoming available regarding the benefits of oral anticancer therapy programs.10,11 In a recent study, Dr Wong and colleagues demonstrated that the use of an oral chemotherapy management clinic resulted in early identification of interventions needed for oral anticancer patients.11 These interventions may, in turn, result in decreased rates of adverse effects, nonadherence, and medication errors.
Methods
Implementation at UMCCC
There are multiple opportunities to enhance patientcare with the use of an oral anticancer therapy program. These include standardization of medication and adherence education, improving methods for medication acquisition, and prompt side-effect management.
Prior to the implementation of the oral anticancer therapy program at the University of Michigan Comprehensive Cancer Center (UMCCC), a meeting was held with key stakeholders involved in the prescribing, dispensing, and financial assessment of oral anticancer therap within UMCCC. This group consisted of oncology physicians representing patients with genitourinary cancers, head and neck cancers, sarcomas, melanomas, brain tumors, gynecologic cancers, gastrointestinal cancers, lung cancer, breast cancer, and hematologic malignancies. In addition to the physicians, our group included a pharmacist lead, clinical nurse practitioners, and members from UMCCC administration, nursing administration, and our Cancer Symptom Management & Supportive Care Program. Through these meeting discussions, program goals were finalized and included creating useful educational materials, streamlining medication acquisition, and standardizing medication and adherence education, medication review, and drug–drug interaction screening and documentation. We discussed our communication plan, and a consistent process for documenting the above information. We also acknowledged that the initial point of contact with the patient happens during their oncologist visit. Therefore, we structured our program to arm the clinics with necessary tools, including educational documents and information about the program, the program’s goals, and program contact information, while we planned to provide support for the patient after he or she had left the clinic setting. We designed our program with the goal of consistent communication between the patient and healthcare team in mind. Educational documents, including patient self-care instructions, were created by our pharmacy team, reviewed by clinic providers, and posted online for ease of accessibility by all healthcare providers.
Providers were educated on the oral anticancer therapy program services described below, and patient education was developed to describe what services were to be provided to the patient and the future contact they would receive. This program went live in May 2012. Referral to the oral anticancer therapy program occurs automatically for all patients initiated on an oral anticancer therapy via a report generated from our electronic ordering system. In addition, initial prescriptions are encouraged to be routed through the University of Michigan (UM) Cancer Center Pharmacy for coverage assessment. The prescriber may also refer other patients receiving oral anticancer therapy to the pharmacy for follow up as needed.
Initial staffing of the oral anticancer therapy program consisted of 1 full-time equivalent (FTE) oncology clinical pharmacist coordinating with pre-established financial support groups within UMCCC. The position was justified by the growing oral oncolytic population, increasing time spent by clinic providers coordinating prescription fills and financial assistance, and an anticipated increase in revenue because of UM Cancer Center Pharmacy prescription fills. One year into the program, we justified the addition of 1 FTE oral anticancer therapy financial coordinator, and the use of UM’s specialty pharmacy for support. This further streamlined the process and allowed for additional financial support through the program rather than directly from each clinic provider. In addition to the oncology pharmacist, we use nursing expertise and pharmacy student and resident support for patient education and medication reconciliation when available. Our nurse lead for the program reached out to the UMCCC clinics for the first 6 months after we went live, received ongoing feedback regarding the educational documents, and reminded nursing staff of the available resources. The pharmacy student we relied on was an intern who worked with our program for a year. She was trained on the oral anticancer therapy program, the educational documents, and workflow of the program. Her phone calls were monitored by the clinical pharmacy specialist, and, after competency was determined, all notes were reviewed prior to posting the final note in the electronic medical record (EMR). The oncology clinical pharmacist was often working in the same office as the student, but for times when they were not working together, the pharmacist was available for any questions via pager. Oncology residents were trained similarly, with continuous exposure to the clinic occurring during their residency. The oral anticancer therapy program assists with approximately 100 to 120 new patients monthly.
Services Provided by Outpatient Oral Anticancer Therapy Program
Medication Acquisition
To avoid delays in treatment initiation, the oral anticancer program streamlines the medication acquisition process. Patients are encouraged, but not required, to at least have their first prescription filled through the Cancer Center Pharmacy to facilitate optimal communication with the pharmacists and nurses in the UMCCC clinics. We currently capture and maintain approximately 60% of prescriptions. Regardless of whether the prescription is filled through the Cancer Center Pharmacy, the oral anticancer therapy program’s financial coordinator triages and assesses medication access for all patients. For limited distribution medications, or when insurance coverage dictates medication acquisition via a specific or specialty pharmacy, the patient financial coordinator relays this information to the patient and prescriber to facilitate access. If there is a medication coverage issue, the financial coordinator provides assistance with prior authorizations, copay assistance, and other patient assistance programs for UMCCC. This progress is documented in the patient’s EMR and includes final patient access information for future prescribing. Any delays in therapy are shared with the patient and provider via the EMR messaging system, and documentation in the medical record.
The recent partnership between UMCCC and UM’s specialty pharmacy has greatly expanded the medication acquisition services of the oral anticancer therapy program. UMCCC patients now have the option to receive medications in the mail, thereby increasing access. In addition, the partnership with the specialty pharmacy has enhanced the assessment of adherence measures through prescription refill monitoring, phone reminders, and access to dispensing data. We observed the success of the transplantation specialty pharmacy program within our institution, and are hopeful to see similar outcomes.12 In addition, we are hopeful that this collaboration will further benefit our ability to receive certain limited distribution medications while maintaining consistent communication with the patient and provider.
Patient Education
Standard patient education documents for oral anticancer agents were developed by our pharmacy team and approved by the UMCCC providers and patient education coordinator; the literacy target was a sixth grade reading level. Currently, educational documents only exist in English. These materials include information on administration and safe handling, actions to take if a dose is missed, potential drug and food interactions, UMCCC-specific information for patient self-management of side effects, and thresholds for when to call the UMCCC clinic. At the time of medication pick up, our Cancer Center Pharmacy ensures that patients received this information in the clinic, and, if not, provides the patient with appropriate documents and education. If patients do not pick up medications from the Cancer Center Pharmacy, the oral anticancer therapy program will mail or e-mail the patients educational materials, depending on patient preference.
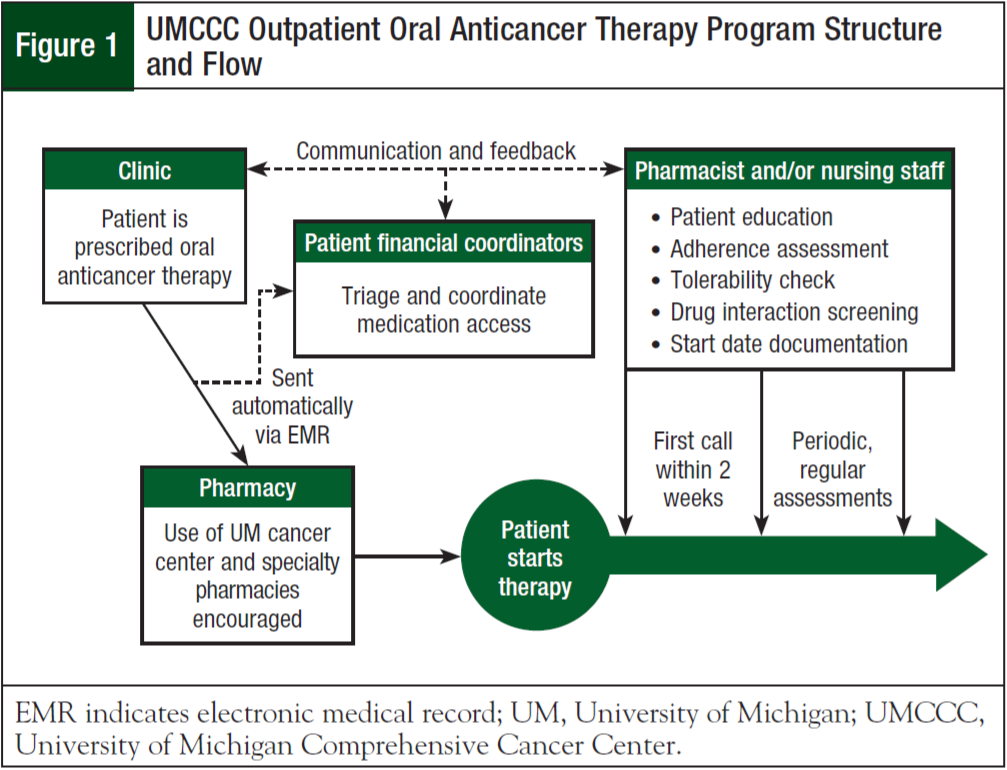
Subsequently, pharmacists and/or nurses contact patients within 10 days of enrollment into the program. During the initial phone call, medication education is reiterated and adherence is reinforced. Medication reconciliation is also performed at this time. The average time for counseling sessions is 30 minutes, and does not typically vary between healthcare professionals. After the medication reconciliation is complete, the pharmacist performs an additional drug–drug interaction screening, and contacts the provider if any clinically significant drug–drug interactions are noted. Documentation of all aspects of this interaction, including oral anticancer therapy start date, is recorded in the patient’s EMR. Any side effects, toxicities, or issues with the medication noted by the patient are documented, and the provider is made aware through the outpatient EMR system and/ or a phone call or e-mail. A follow-up phone call with a pharmacist and/or nurse is then scheduled to periodically monitor each patient for adherence and tolerability, to reinforce education, and to screen for drug–drug interactions. These follow-up calls typically occur at 3- to 6-month intervals, but may occur sooner based on potential problems identified in the initial phone call. Figure 1 illustrates the current structure and flow of the UMCCC Outpatient Oral Anticancer Program.
The most common agents and associated diagnoses seen in the UMCCC outpatient oral anticancer therapy program are lenalidomide (multiple myeloma), abiraterone and enzalutamide (prostate cancer), capecitabine (colorectal, esophageal, breast, and neuroendocrine malignancies), temozolomide (central nervous system and neuroendocrine malignancies), cyclophosphamide multiple myeloma, and several other malignancies), and the BCR-ABL tyrosine kinase inhibitors, dasatinib, imatinib, nilotinib, and ponatinib (chronic myelogenous leukemia or acute lymphoblastic leukemia). The most common insurance plans are Medicare Part D and Blue Cross and Blue Shield.
Results
Provider Satisfaction
Eight months after the implementation of the UMCCC oral anticancer therapy program, physicians and their nurse practitioners and/or physician assistants were surveyed using Qualtrics, an online survey tool. A reminder e-mail was sent 2 to 3 weeks after the initial e-mail. The goal of this survey was to get feedback about satisfaction with the rollout of the program, communication and documentation, and the services provided to patients. The survey design was based on a 5-point Likert scale, with answers ranging from strongly agree to strongly disagree, and also included yes/no and short answer questions. Survey questions were developed from previous trials examining patient satisfaction with pharmacy services, and included additional questions specific to our unique program.13,14
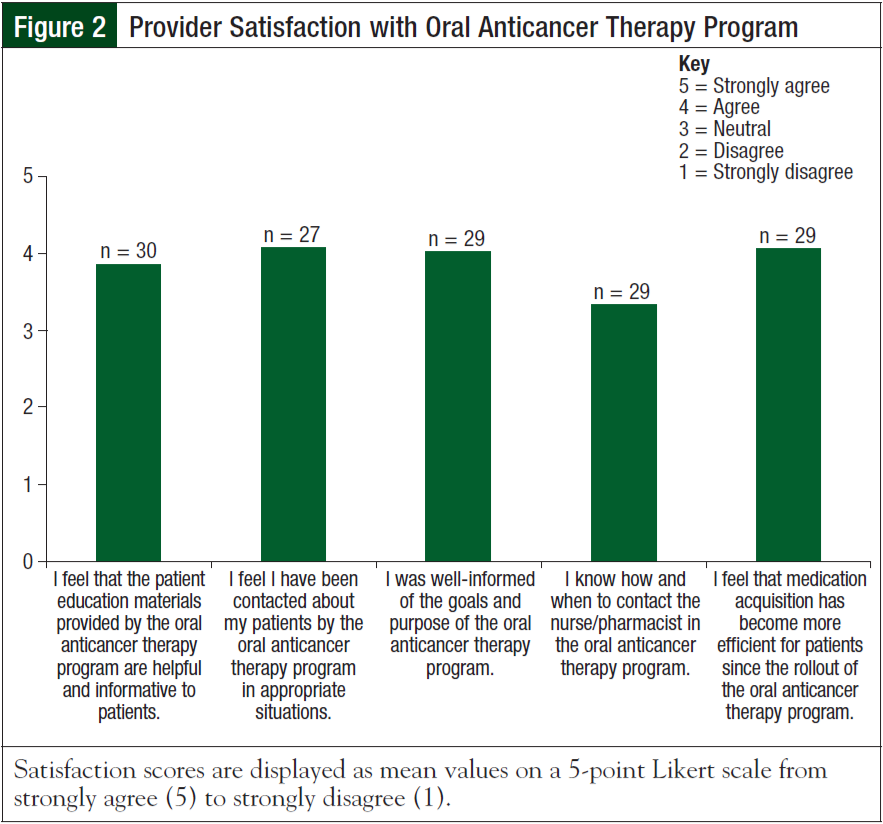
Thirty of the 61 surveys were completed with a response rate of 49.2%. Of the 30 providers who responded, 14 were physicians, 6 were physician assistants, and 10 were nurse practitioners. Providers were divided on what they felt was the most important aspect of the program: 33% felt that aiding in acquisition of medications was most important, 30% felt that drug– drug interaction screening was most important, 27% felt that reinforcement of medication education was most important, and only 10% felt that patient follow-up was most important. Although the majority of providers felt that they were well-informed about the rollout of the program, 5 providers disagreed or strongly disagreed that they understood how to contact the oral anticancer therapy clinic (Figure 2).
Importantly, a majority of the providers felt that the medication education tools were helpful to patients (Likert scale mean [standard deviation], 3.9 [0.9]; median, 4). Sixty percent (18/30) of providers had read the information documented in the EMR by the oral anticancer therapy program; among the 18 providers, 16 (89%) found it to be useful. Overall, providers were highly satisfied with the services of the oral anticancer therapy program. In response to the results of this survey, the goals of this program will be reinforced to providers, and contact information will be reiterated.
Patient Satisfaction
The design of the patient survey was also based on a 5-point Likert scale, with answers ranging from strongly agree to strongly disagree. The survey also included yes/no and short answer questions. The survey and study were approved by the Institutional Review Boards of the University of Michigan Medical School. Similar to the provider survey, questions were derived from previous trials examining patient satisfaction with pharmacy services, and made specifically for the UMCCC outpatient oral anticancer therapy program.13,14
In January 2013, patients received surveys mailed to them with prepaid return envelopes. All patients who had been enrolled in the program up until this point were sent surveys; thus, the amount of time that had passed between their first medication fill and/or education and survey varied from patient to patient, providing a global, time-independent view of patient satisfaction with the UMCCC outpatient oral anticancer therapy program. Surveys were mailed to 161 patients and 41 were returned with a 25.5% response rate.
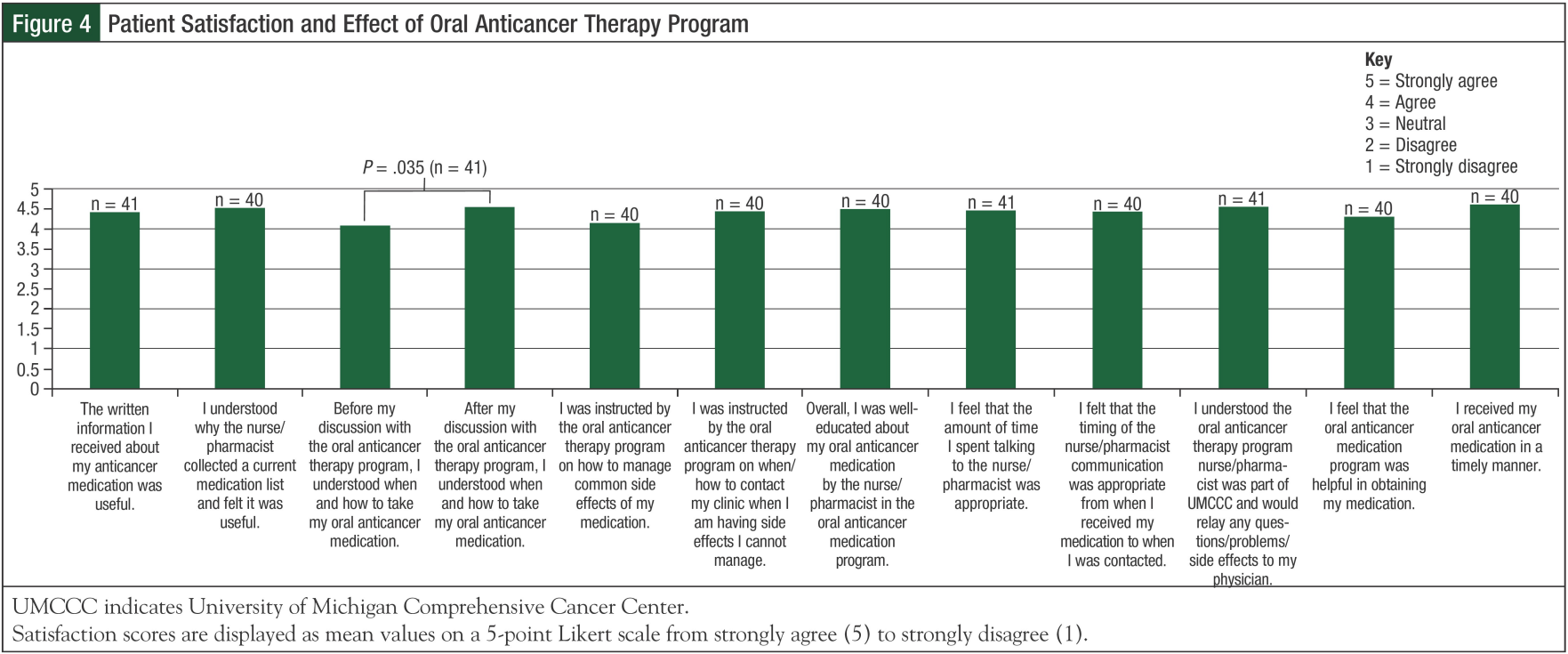
Patients were asked about the 3 main pillars of the oral anticancer therapy program: medication education, communication, and medication acquisition. The majority of patients received their medication education documents in the clinic from the physician (23 [56%]), or at the UM Cancer Center Pharmacy (26 [63%]), and their verbal medication education from physicians (33 [80%]) or nurses (22 [54%]) in the clinic, or the pharmacist at the Cancer Center Pharmacy (14 [34%]). Although every patient should have received verbal medication education from the pharmacist or nurse at the outpatient oral anticancer clinic, only 11 (27%) patients identified this in their surveys. Through our patient education, 56.9% of patients learned new information about their medication (Figure 3), and, prior to this education, 22% of patients neither agreed nor strongly agreed that they knew how and when to take their oral anticancer medication (Figure 4). However, this number dropped to 5% after provision of education by the oral anticancer program. On a 5-point Likert scale, the mean score for this statement was significantly higher after provision of education (4.5 vs 4.1; P = .035).
Because self-management of side effects and recognition of the need for clinical intervention are paramount in the setting of oral anticancer therapy, it is noteworthy that the majority of patients self-reported that the oral anticancer therapy program provided them with this critical knowledge. In addition, patients found the written information useful, and felt well-educated by the oral anticancer program, overall. The majority of patients believed that the timing of, and amount of time spent on, the initial phone call from the oral anticancer therapy program was appropriate.
Medication acquisition and cost are additional barriers that may adversely affect management of patients receiving oral anticancer medications. In fact, 27% (11/41) of patients surveyed self-reported that they had difficulty filling their medication. Fortunately, 80% (32/41) of patients felt that the oral anticancer therapy program was helpful in obtaining the medication, and 93% (38/41) felt that they received their medication in a timely manner. With regard to medication cost, 68% (27/40) of the ora anticancer medications cost patients more than $100 to obtain. In these cases, 59% (16/27) of patients reported receiving practical assistance from UMCCC to obtain the medication. Therefore, because of the strong support from the patient financial coordinators of the oral anticancer therapy program, in addition to other assistance measures, medication acquisition was streamlined and access to high-cost therapies was improved. Limitations of this survey include that it was not validated, that there is a lack of a retrospective comparison cohort of patients who did not receive UMCCC oral anticancer therapy program services, and the lack of 100% survey participation. Also, the inherent nature of a survey-based study creates the possibility for response bias. Results from this survey indicate that patient satisfaction overall is positive; however, further attention can be paid to informing patients about how to self-manage side effects, and additional education on adherence tools can be addressed.
Future Directions
Additional quality care end points are currently being assessed from the implementation of this program, and they include: (1) adherence to anticancer therapy, (2) adverse reactions and drug–drug interactions with oral anticancer therapies, and (3) financial outcomes of the service with regard to added revenue from prescription fills. Future considerations for research include assessing the impact of the program on patient knowledge and comfort with their cancer therapy, as well as communication and documentation shared between the patient’s primary care and specialty physicians.
Conclusion
Although oral anticancer medications can provide significant benefits to patients, a robust system for ensuring the safety and efficacy of these medications is necessary. The decision to use oral anticancer medications requires detailed discussions with the patient and the entire oncology team, as well as ongoing support and side-effect management from healthcare providers. Oral anticancer therapy programs that rely on the collaboration of a multidisciplinary group, including pharmacy, nursing, and financial support experts, are able to provide the services necessary for the optimal treatment of patients with cancer. Overall, the implementation of the UMCCC oral anticancer therapy program has been well-accepted, and has garnered both patient and provider satisfaction. Future studies are needed to assess the additional outcomes of outpatient oral anticancer programs
Author Disclosure Statement
The authors reported no conflicts of interest.
Acknowledgments
Dr Koselke, Dr Walker, Ms Allore, and Dr Mackler performed this work during affiliation with the University of Michigan Comprehensive Cancer Center.
Dr Koselke is an Oncology Clinical Pharmacist, Indiana University Health Ball Memorial Cancer Center, Muncie. Dr Marini is a Clinical Pharmacist, Inpatient Hematology/Oncology, University of Michigan Health System, Ann Arbor. Dr Kraft is a Clinical Pharmacist, Hematology/Oncology, University of Michigan Comprehensive Cancer Center, and Clinical Assistant Professor, University of Michigan College of Pharmacy, Ann Arbor. Dr Walker is a Nurse Practitioner, McKenzie Health System, Sandusky, MI. Ms Allore is a registered nurse in the University of Michigan Medical Observation Unit, University of Michigan Health System, Ann Arbor. Dr Mackler is an Oncology Clinical Pharmacist, Michigan Oncology Quality Consortium at the University of Michigan Comprehensive Cancer Center, and Adjunct Clinical Associate Professor, University of Michigan College of Pharmacy, Ann Arbor.
References
- Weingart SN, Brown E, Bach PB, et al. NCCN task force report: oral chemotherapy. J Natl Compr Canc Netw. 2008;6(suppl 3):S1-S14.
- Halfdanarson TR, Jatoi A. Oral cancer chemotherapy: the critical interplay between patient education and patient safety. Curr Oncol Rep. 2010;12:247-252.
- Neuss MN, Polovich M, McNiff K, et al. 2013 updated American Society of Clinical Oncology/Oncology Nursing Society Chemotherapy Administration Safety Standards including standards for the safe administration and management of oral chemotherapy. J Oncol Pract. 2013;9(suppl 2):5s-13s.
- Chan A, Tan SH, Wong CM, et al. Clinically significant drug-drug interactions between oral anticancer agents and nonanticancer agents: a Delphi survey of oncology pharmacists. Clin Ther. 2009;31(pt 2):2379-2386.
- Noens L, van Lierde MA, De Bock R, et al. Prevalence, determinants, and outcomes of nonadherence to imatinib therapy in patients with chronic myeloid leukemia: the ADAGIO study. Blood. 2009;113:5401-5411.
- Tschida SJ, Aslam S, Lal LS, et al. Outcomes of a specialty pharmacy program for oral oncology medications. Am J Pharm Benefits. 2012;4:165-174.
- Red Book Online. Greenwood Village, CO: Truven Health Analytics. www.micro medexsolutions.com/micromedex2/librarian/ND_T/evidencexpert/ND_PR/evidenc expert/CS/9228A9/ND_AppProduct/evidencexpert/DUPLICATIONSHIELD SYNC/149B89/ND_PG/evidencexpert/ND_B/evidencexpert/ND_P/evidencexpert/ PFActionId/redbook.FindRedBook?navitem=topRedBook&isToolPage=true. Accessed June 18, 2015.
- Neuss M, Gilmore TR, Kadlubek P. Tools for measuring and improving the quality of oncology care: the Quality Oncology Practice Initiative (QOPI) and the QOPI certification program. Oncology (Williston Park). 2011;25:880, 883, 886-887.
- Weingart SN, Flug J, Brouillard D, et al. Oral chemotherapy safety practices at US cancer centres: questionnaire survey. BMJ. 2007;334(7590):407.
- MacLeod A, Branch A, Cassidy J, et al. A nurse-/pharmacy-led capecitabine clinic for colorectal cancer: results of a prospective audit and retrospective survey of patient experiences. Eur J Oncol Nurs. 2007;11:247-254.
- Wong SF, Bounthavong M, Nguyen C, et al. Implementation and preliminary outcomes of a comprehensive oral chemotherapy management clinic. Am J Health Syst Pharm. 2014;71:960-965.
- Hlubocky JM, Stuckey LJ, Schuman AD, et al. Evaluation of a transplantation specialty pharmacy program. Am J Health Syst Pharm. 2012;69:340-347.
- Flanagan P, Kainth S, Nissen L. Satisfaction survey for a medication management program: satisfaction guaranteed? Can J Hosp Pharm. 2013;66:355-360.
- Moczygemba LR, Barner JC, Brown CM, et al. Patient satisfaction with a pharmacist- provided telephone medication therapy management program. Res Social Adm Pharm. 2010;6:143-154.