Symptom Overview
Venous thromboembolism (VTE) can result in significant morbidity and mortality.1 The annual incidence of VTE in the general population is 117 per 100,000 (approximately 0.12%) patients. For patients diagnosed with cancer, the annual incidence of VTE is approximately 20%, and the risk for VTE in this patient population is estimated to be 7 times higher than in the general population.2-4 Multiple myeloma is associated with an even greater risk for VTE than other malignancies, with an incidence rate as high as 75% in association with certain therapies.5
Etiology
Advanced age, obesity, cardiac disease, chronic renal disease, diabetes, acute infection, immobilization, previous VTE, presence of a central venous catheter or pacemaker, surgery, and preexisting coagulopathy are known risk factors that have been associated with VTE in patients with and without cancer.2,6 Conditions unique to cancer that increase the risk for VTE among patients with cancer include hypercoagulability because of an increase in inflammatory markers, vessel wall injury caused by invading tumor or by surgical intervention, and stasis caused by hyperviscosity.6
Patients with multiple myeloma are at increased risk for VTE versus patients with other malignancies, because of disease-specific risk factors.4,6 Patients with multiple myeloma have been observed to be in a prothrombotic state, because of increased levels of von Willebrand factor and factor VIII. In addition, these patients have exhibited an increased incidence of acquired activated protein C resistance. Inflammatory cytokines, including interleukin-6, C-reactive protein, and tumor necrosis factor, are released during the disease process and may also play a role in multiple myeloma–induced VTE.
Furthermore, multiple myeloma therapies have been found to increase the incidence of VTE.4 Chief among these medication classes are the immunomodulatory drugs (IMiDs),5 which include thalidomide, lenalidomide, and pomalidomide. When combined with other agents, including dexamethasone and chemotherapy, the risk for IMiD-induced VTE increases further. By contrast, bortezomib, a proteasome inhibitor indicated for patients with multiple myeloma, has been associated with a lower VTE risk than the IMiDs, with VTE rates <5%.2 Use of erythropoiesis-stimulating agents as supportive care for patients with multiple myeloma has also been associated with an increased risk for VTE.7
Mitigating Thromboembolic Risk
Since the recognition of increased incidence of VTE among patients with multiple myeloma, researchers have looked for the most effective prophylaxis against this complication. Aspirin, warfarin, and low-molecular-weight heparin (LMWH) have been studied as thromboprophylaxis in patients with multiple myeloma who receive IMiDs.2-4
In a substudy of the GIMEMA clinical trial, investigators compared the use of LMWH (subcutaneous enoxaparin 40 mg daily) with oral low-dose aspirin (100 mg daily) and low-dose oral warfarin (1.25 mg by mouth daily) for reducing the risk for VTE in patients with multiple myeloma who receive various thalidomide-containing regimens.8 The investigators concluded that LMWH conferred the lowest risk for VTE among the 3 interventions, and low-dose warfarin conferred the highest risk; these results were not statistically significant.8
The aspirin 100-mg tablet is not available in the United States; therefore, the results of this study8 and the following studies9,10 are often used as a basis for recommending aspirin 81 mg as thromboprophylaxis in the United States.
In one study conducted in patients who received combination chemotherapy with a lenalidomide-containing regimen, investigators compared the efficacy of low-dose oral aspirin (100 mg daily) and subcutaneous LMWH (enoxaparin 40 mg daily) in decreasing the incidence of VTE.9 An interim analysis of this study showed that the incidence of VTE was similar between the 2 groups, and the difference was not statistically significant. At the final data analysis, no difference in efficacy was seen between aspirin and LMWH for thromboprophylaxis in patients with multiple myeloma.9 The investigators recommended the use of aspirin over LMWH,9 because of its lower cost, as well as reduced burden of treatment administration for patients.10
Other LMWHs, including dalteparin, have been used as thromboprophylaxis internationally at equivalent doses to enoxaparin 40 mg daily.2,11 Direct-acting oral anticoagulants have been used in clinical practice for the treatment of VTE in patients with cancer, but there are limited data on which to base recommendations regarding their use in the prophylactic setting.
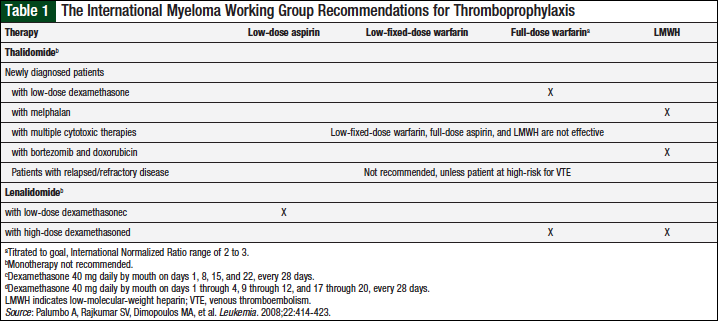
Guidelines from the International Myeloma Working Group (IMWG), the American Society of Clinical Oncology (ASCO), and the National Comprehensive Cancer Network (NCCN) have been published based on these findings. The IMWG has recommendations that are therapy specific (Table 1).11 The duration of prophylaxis therapy is not addressed.
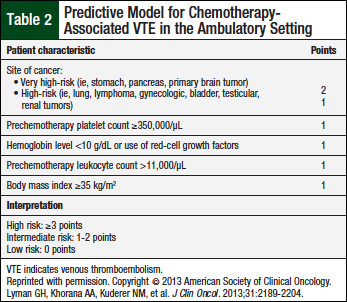
ASCO recommends the use of thromboprophylaxis for patients who receive thalidomide or lenalidomide in combination with cytotoxic chemotherapy and/or dexamethasone.12 For low-risk patients, low-dose aspirin or LMWH equivalent to enoxaparin 40 mg daily is appropriate (Table 2).
For high-risk patients, LMWH equivalent to enoxaparin 40 mg daily is recommended. Low-fixed-dose warfarin has been removed from this recommendation in the most recent ASCO guidelines.12 The duration of prophylaxis therapy is not addressed.
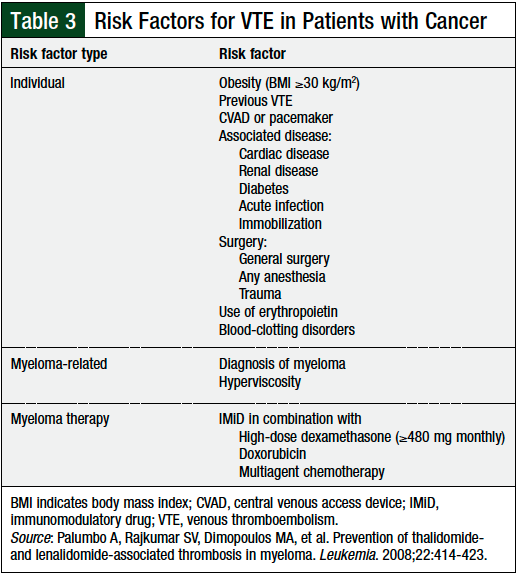
The NCCN guidelines recommend thromboprophylaxis for any patient with multiple myeloma who receives IMiDs, including pomalidomide.13 In patients with 0 to 1 individual or multiple myeloma risk factors identified by the NCCN, thromboprophylaxis with aspirin 81 mg to 325 mg daily is recommended (Table 3).
In the presence of ≥2 individual or multiple myeloma–related risk factors, prophylaxis with an LMWH equivalent to enoxaparin 40 mg daily or with full-dose warfarin is recommended. The duration of prophylaxis therapy is not addressed.
Recommendations for Practice
Patients at low- or intermediate-risk for chemotherapy-associated VTE (Table 2) should be prescribed aspirin 81 mg daily.12 Alternatively, the NCCN guidelines recommend that all patients with at least 1 risk factor, namely all patients with multiple myeloma, (Table 3) should be prescribed aspirin, 81 mg daily.13 Patients at high risk, or those with ≥2 risk factors for VTE should be prescribed enoxaparin 40 mg daily or an equivalent LMWH, or full-dose warfarin titrated to a goal, International Normalized Ratio range of 2 to 3.
Thromboprophylaxis should be started at the initiation of IMiD therapy and continued until completion of IMiD therapy. For many patients who receive long-term maintenance with lenalidomide or pomalidomide, this means lifelong thromboprophylactic therapy. For patients who receive pomalidomide and who start LMWH therapy, the risk for VTE can be reevaluated after 4 months of anticoagulant therapy; they may be able to transition to prophylaxis with low-dose aspirin.14
The risks for thromboprophylaxis and anticoagulation must be weighed against the potential benefits for all patients, specifically those who are at a high risk for bleeding.13
For obese patients who are taking an LMWH such as enoxaparin, anti–factor Xa levels should be monitored to minimize toxicity and prevent bleeding.13 In addition, the NCCN recommends that patients with a creatinine clearance of <30 mL/min receive a lower dose, 30 mg daily, of enoxaparin. Enoxaparin is not approved by the US Food and Drug Administration for use in patients undergoing dialysis. Anti–factor Xa levels may be followed more closely for patients undergoing dialysis, or the use of an alternative agent (ie, full-dose warfarin) may be explored. Providers should refer to their institution-specific guidelines and reference ranges for monitoring anti–factor Xa levels.
Patients given low-fixed-dose or full-dose warfarin should be screened for pertinent drug–drug and drug–food interactions; a notable interaction exists between dexamethasone and warfarin, necessitating close monitoring and dose adjustments as needed.
Conclusion
In the past few years, the treatment of multiple myeloma has grown to encompass more effective therapies. However, those therapies, including IMiDs and erythropoiesis-stimulating agents, also add VTE risk to a disease that is already associated with an increased incidence of VTE. To provide protection against VTE in this patient population, low-dose aspirin, LMWH (eg, enoxaparin), and warfarin can be used.
In the future, pending further study, other therapies, such as direct-acting oral anticoagulants, may be used as thromboprophylaxis.
Author Disclosure Statement
Dr Gerardo has no conflicts of interest to report.
Correction
In the print version of this article, the source for Table 3 was incorrect. This has been corrected online and in the PDF.
References
- Silverstein MD, Heit JA, Mohr DN, et al. Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med. 1998;158:585-593.
- Leebeek FW. Update of thrombosis in multiple myeloma. Thromb Res. 2016;140(Suppl 1):S76-S80.
- Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293:715-722.
- Crowley MP, Eustace JA, O’Shea SI, Gilligan OM. Venous thromboembolism in patients with myeloma: incidence and risk factors in a “real-world” population. Clin Appl Thromb Hemost. 2014;20:600-606.
- Falanga A, Marchetti M. Venous thromboembolism in the hematologic malignancies. J Clin Oncol. 2009;27:4848-4857.
- Kristinsson SY. Thrombosis in multiple myeloma. Hematology Am Soc Hematol Educ Program. 2010;2010:437-444.
- Palmaro A, Rougé-Bugat ME, Gauthier M, et al. Real-life practices for preventing venous thromboembolism in multiple myeloma patients: a cohort study from the French health insurance database. Pharmacoepidemiol Drug Saf. 2017;26:578-586.
- Palumbo A, Cavo M, Bringhen S, et al. Aspirin, warfarin, or enoxaparin thromboprophylaxis in patients with multiple myeloma treated with thalidomide: a phase III, open-label, randomized trial. J Clin Oncol. 2011;29:986-993.
- Palumbo A, Cavallo F, Yehuda DB, et al. A prospective, randomized study of melphalan, prednisone, lenalidomide (MPR) versus melphalan (200 Mg/M2) and autologous transplantation (Mel200) in newly diagnosed myeloma patients: an interim analysis. Blood. 2009;114:Abstract 350.
- Larocca A, Cavallo F, Bringhen S, et al. Aspirin or enoxaparin thromboprophylaxis for patients with newly diagnosed multiple myeloma treated with lenalidomide. Blood. 2012;119:933-939; quiz 1093.
- Palumbo A, Rajkumar SV, Dimopoulos MA, et al. Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia. 2008;22:414-423.
- Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31:2189-2204.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Cancer-Associated Venous Thromboembolic Disease. Version 1.2016. July 22, 2016. www.nccn.org/professionals/physician_gls/pdf/vte.pdf. Accessed May 13, 2017.
- Dimopoulos MA, Leleu X, Palumbo A, et al. Expert panel consensus statement on the optimal use of pomalidomide in relapsed and refractory multiple myeloma. Leukemia. 2014;28:1573-1585.