Ibrutinib is a Bruton tyrosine kinase (BTK) inhibitor used for the treatment of a variety of lymphoid malignancies, as well as chronic graft-versus-host disease (GVHD).1 The BTK pathway is involved in B-cell proliferation and survival, as well as macrophage receptor–mediated phagocytosis.2 Individuals with congenital loss of BTK expression have an increased risk for infections.1 Recently, data have emerged suggesting a risk for fungal infections in patients receiving ibrutinib; however, no published data currently exist on concomitant use of ibrutinib and antifungal prophylaxis or safety and efficacy outcomes of this combination therapy.2-6 It is unknown which, if any, antifungals are prescribed in clinical practice for patients who are receiving ibrutinib therapy.
Azole antifungals are widely used for the prevention and treatment of fungal infections and are inhibitors of cytochrome P450 3A (CYP3A).7 Ibrutinib is a CYP3A substrate; therefore, it poses the potential for drug interactions when used in combination with azoles.8 The prescribing information for ibrutinib has specific recommendations for dose adjustments when used concomitantly with voriconazole, posaconazole, and other moderate or severe CYP3A inhibitors.8
In addition to the lack of data on the prescribing practices for antifungal prophylaxis in patients who are receiving ibrutinib therapy, the data are scant on details of antifungal prophylaxis in published reports on ibrutinib-associated fungal infections.3 Therefore, it is unknown if clinical outcomes differ for patients who are receiving ibrutinib therapy and antifungal prophylaxis versus those who are receiving ibrutinib without antifungal prophylaxis.
Our study assessed whether patients who were receiving ibrutinib at our Georgia Cancer Center also received azole prophylaxis. For patients receiving azole prophylaxis, we assessed whether appropriate dose adjustment was made in accordance with the prescribing information; whether dose adjustment of ibrutinib affected the efficacy; and whether dose adjustment affected the safety, including the risk for having fungal infections.
Methods
In this retrospective, single-center chart review approved by the local investigational review board, we assessed patients aged ≥18 years with documentation of receiving ibrutinib (ie, medication discoverable through insurance fill history, physician documentation of the patient taking the medication). The chart review included patients receiving treatment at the center between November 1, 2013, and October 31, 2018.
The primary outcome was the rate of azole prophylaxis in patients receiving ibrutinib. Secondary outcomes for all patients included the rate of ibrutinib and/or azole discontinuation resulting from adverse events associated with either agent, the rate of fungal infections, the time to cancer progression, the time from ibrutinib initiation to death, and the reason for death.
Additional outcomes for patients who received azole prophylaxis included appropriateness of ibrutinib dose adjustment and the duration of concomitant exposure to ibrutinib and the azole. In patients who had a fungal infection, the rate of mortality due to fungal infection was determined. Descriptive statistics were used for all outcomes.
Results
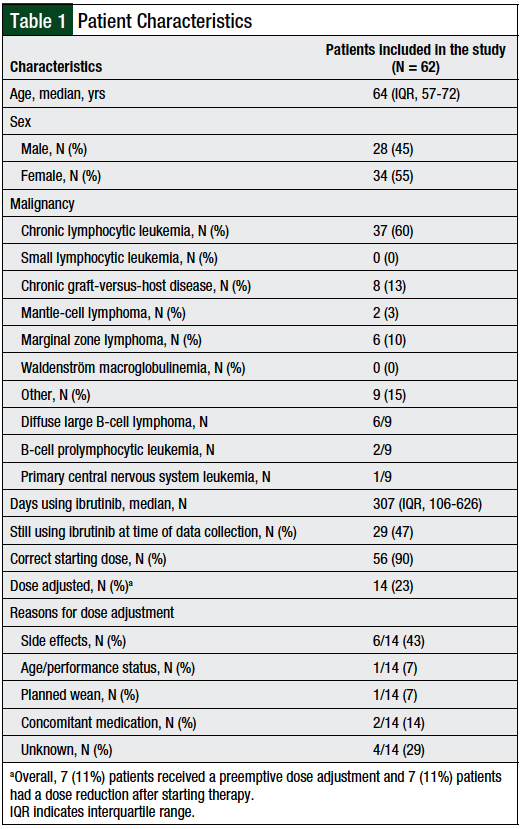
We identified a total of 64 patients using a report of ibrutinib prescriptions. Of these, 2 patients were excluded because they had no documentation of receipt of the drug. For the 62 evaluated patients, the median age was 64 years (interquartile range [IQR], 57-72 years), with a female predominance (N = 34; 55%). Patient characteristics are outlined in Table 1.
Chronic lymphocytic leukemia (CLL) was the most common diagnosis with an approved indication for ibrutinib therapy among the study patients (N = 37; 60%). Other diagnoses with approved indications for ibrutinib included chronic GVHD (N = 8; 13%),
marginal zone lymphoma (N = 6; 10%), mantle-cell lymphoma (N = 2; 3%), and several diagnoses with no US Food and Drug Administration (FDA)-approved indications (N = 9; 15%).
These 9 diagnoses with no FDA-approved indications included diffuse large B-cell lymphoma (N = 6), B-cell prolymphocytic leukemia (N = 2), and primary central nervous system lymphoma (N = 1). All the patients had received ibrutinib therapy for a median of 307 days (IQR range, 106-626 days), and 29 (47%) patients were still receiving ibrutinib therapy at the time of study data collection.
In addition, 7 (11%) patients had preemptive dose reductions and 56 (90%) patients started therapy with an FDA-approved dose for their specific disease. The reasons for incorrect dose reductions included poor performance status (N = 1; 2%), concomitant use of a moderate CYP3A4 inhibitor that did not require a dose adjustment according to the prescribing information (N = 1; 2%), and no documented reason for initiating a lower dose (N = 4; 6%).
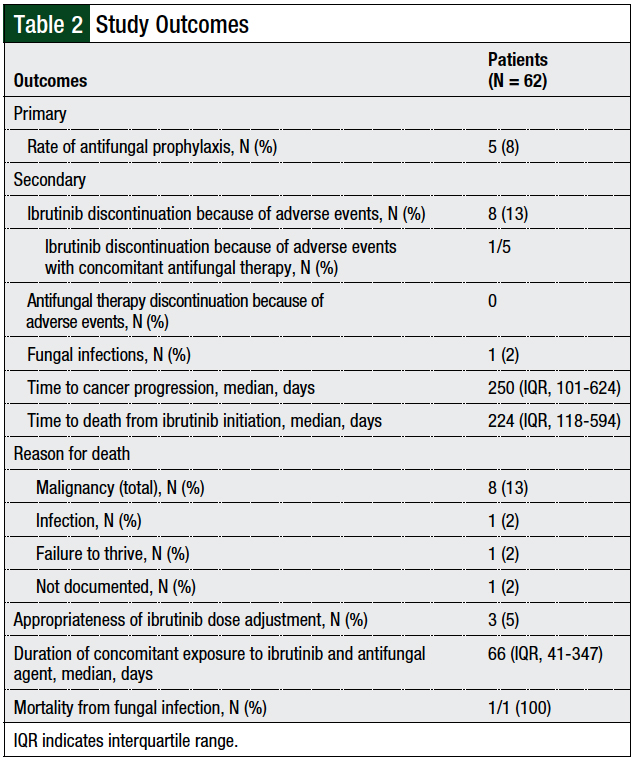
In all, 5 (8%) patients were receiving concomitant azole prophylaxis (Figure 1), which included voriconazole (N = 1), fluconazole (N = 1), posaconazole (N = 1), and isavuconazonium sulfate (N = 2; Figure 2). The primary and secondary treatment outcomes are described in Table 2.
Of the 5 patients who received prophylactic azoles, 3 (60%) had appropriate ibrutinib dose adjustments. The median duration of concomitant exposure to ibrutinib and prophylaxis azoles was 66 days (IQR, 41-347 days). Of the 5 patients receiving azole prophylaxis, 1 patient died because of the underlying malignancy and another because of failure to thrive; the remaining 3 patients were alive at the time of study data collection. The median time to follow-up from the start of ibrutinib therapy for these 5 patients was 82 days (IQR, 41-383 days).
A total of 8 (13%) patients discontinued ibrutinib therapy because of adverse events, including 1 of the 5 patients receiving a concomitant azole. The reasons for ibrutinib discontinuation included bleeding events (N = 3; 38%), thrombocytopenia (N = 2; 25%), skin rash, atrial fibrillation, and gastrointestinal intolerance (N = 1; 12% each). No patients in the study discontinued the azole prophylaxis because of an adverse event.
In the overall study population, only 1 (2%) patient had a fungal infection while receiving ibrutinib; this patient subsequently died from the infection after receiving antifungal treatment. A computed tomography (CT) scan of the sinuses identified the infection, but the patient’s condition was too unstable to undergo debridement and a culture had not been collected to identify a pathogen. The patient had not received concomitant antifungal prophylaxis.
Overall, 12 (19%) patients had disease progression, which occurred at a median of 250 days (IQR, 101-624 days) from the initiation of ibrutinib therapy. The reasons for death included underlying malignancy (N = 8; 13%), failure to thrive (N = 1; 2%), and 1 undocumented cause. Patients died a median of 224 days (IQR, 118-594 days) from the time of ibrutinib initiation.
Discussion
Several published studies have described the incidence of invasive fungal infections in patients receiving ibrutinib.1,3-6 Varughese and colleagues evaluated serious infections in patients with lymphoid cancer who received ibrutinib; they found that 16 (4%) of 378 patients had fungal infections, including invasive aspergillosis, Pneumocystis jirovecii pneumonia, and cryptocococcis.3 By contrast, Visentin and colleagues conducted a retrospective chart review of 795 patients with CLL and found that only 11 (1.4%) patients had an invasive fungal infection after a median follow-up of 8.5 years.4
In patients who are receiving ibrutinib therapy, the time to onset of fungal infections is variable and has been reported to range from less than 1 month and up to 3 years after the initiation of ibrutinib therapy.2,5
Ghez and colleagues performed a multicenter survey in France and identified 33 patients who had invasive fungal infections while receiving ibrutinib.6 The median time to diagnosis of the infection was 3 months (range, 1-30 months), with 28 (85%) of the 33 cases occurring within 6 months and 20 (61%) of the 33 cases occurring within 3 months.6 These findings suggest that patients receiving ibrutinib are at a high risk for fungal infections, particularly at the beginning of treatment.
Of note, only 1 of these studies addressed whether patients received antifungal prophylaxis, but the authors did not comment about the duration of the antifungal prophylaxis or the timing of initiation or discontinuation of ibrutinib therapy.3 Some of the patients in these studies who had an invasive fungal infection had baseline characteristics that are known to increase the risk for a fungal infection.
Chamilos and colleagues reported 41 cases of invasive fungal infection in patients receiving ibrutinib.1 All these patients were receiving treatment for a hematologic malignancy, 9 (22%) of these patients were reported to have received concomitant corticosteroids, and 5 (12%) patients were reported to have received previous treatment with fludarabine.
Varughese and colleagues reported that 5 (31%) of 16 patients who had an invasive fungal infection had received concomitant corticosteroids and 3 (19%) patients had previously received fludarabine,3 and Ghez and colleagues reported that 4 (12%) of 33 patients with an invasive fungal infection received concomitant corticosteroids, but they did not report the number of patients who had received a purine analog.6
To our knowledge, to date, only 1 study has evaluated the concomitant use of azole medications and ibrutinib. Cummins and colleagues assessed 8 patients who are receiving concomitant isavuconazonium sulfate and ibrutinib for adverse events. Patients were receiving a varied dose of ibrutinib, including 140 mg (N = 3), 280 mg (N = 1), dose reduction to 280 mg after receiving the full dose (420 mg) for 4 days (N = 1), and no dose adjustments (N = 3).7
According to the prescribing information, ibrutinib 280 mg daily was the recommended dose adjustment.8 In the study by Cummins and colleagues, 1 patient had paroxysmal atrial fibrillation with an ibrutinib dose of 140 mg, and ibrutinib was later discontinued.8 The patient subsequently died from cancer progression 218 days after a fungal infection diagnosis with Aspergillus fumigatus. One patient had worsening thrombocytopenia while receiving 140 mg, and ibrutinib was discontinued. Another patient died 92 days after fungal infection diagnosis presumably because of cancer progression. In addition, 2 patients had transient corrected QT prolongation; however, the ibrutinib dose was not reported.7 The small sample size makes it difficult to extrapolate the safety of concomitant use of ibrutinib with isavuconazonium sulfate or the impact of ibrutinib dose modifications on the time to disease progression.
Currently, no guidelines exist regarding routine antifungal prophylaxis concomitant with ibrutinib therapy. In our study, all 5 patients who received azole prophylaxis were receiving ibrutinib for chronic GVHD, and the azole was initiated before the initiation of ibrutinib. Of these 5 patients, 3 were started with the appropriate initial ibrutinib dose. According to the prescribing information of ibrutinib, for the indication of chronic GVHD, concomitant mild and moderate CYP3A inhibitors do not require an ibrutinib dose reduction (from 420 mg), whereas concomitant voriconazole does require ibrutinib dose adjustment.8 One patient who was prescribed isavuconazonium sulfate prophylaxis received a dose reduction of ibrutinib which is not recommended.8 The patient who received voriconazole did not have an ibrutinib dose reduction.
The only patient who had an invasive fungal infection during ibrutinib therapy was not receiving azole prophylaxis. The patient had a diagnosis of CLL and had an infection 7 months after the initiation of ibrutinib therapy. The diagnosis of a fungal infection was based on a CT scan of the sinuses, showing an inflammatory process with a significant lytic component highly suspicious for invasive fungal sinusitis, and a serum Beta-D-glucan assay showed a positive result after the patient died. Liposomal amphotericin B was prescribed for treatment, but the patient subsequently died from sepsis because of the infection 224 days from ibrutinib initiation.
There is a paucity of data regarding the efficacy and safety outcomes for patients who are receiving ibrutinib therapy with concomitant antifungal prophylaxis compared with patients who are receiving ibrutinib without prophylaxis. With only 5 patients in our study receiving concomitant azole prophylaxis, meaningful comparisons between the groups cannot be done. Future studies are required to answer this critical question.
Limitations
A retrospective chart review requires heavy reliance on adequate physician documentation. In this study, documentation of the cause of death, the reason for preemptive dose reduction, and concomitant medications used at the time of ibrutinib prescribing was missing from the medical record for multiple patients.
In addition, several patients were lost to follow-up, limiting the longitudinal assessment. As an academic referral center, the most common reason for patients lost to follow-up was the patients returning to their local oncologist. Those who were lost to follow-up were censored at the time of the last documentation in the medical record.
We also did not capture data on whether a patient had fungal infection onset during ibrutinib therapy, discontinued ibrutinib because of an adverse event, had cancer progression, or died beyond follow-up in our system.
Finally, the small sample size of 62 patients limits the generalizability to the overall patient population of those receiving ibrutinib therapy.
Conclusion
Our retrospective chart review did not show a significantly increased rate of invasive fungal infections associated with the use of ibrutinib therapy, despite a low rate of azole prophylaxis; therefore, routine use of azole prophylaxis with ibrutinib may not be warranted unless the patient has additional risk factors that necessitate antifungal prophylaxis. Healthcare providers should continue to prescribe prophylactic azoles according to current indication-specific guideline recommendations for ibrutinib (ie, chronic GVHD, secondary prophylaxis). Further studies are needed to determine potential differences, if any, in safety and efficacy outcomes in patients receiving concomitant azoles and ibrutinib therapy.
Author Disclosure Statement
Dr Daniels, Dr Gandhi, Dr Bryan, and Dr Clemmons have no conflicts of interest to report.
References
- Chamilos G, Lionakis MS, Kontoyiannis DP. Call for action: invasive fungal infections associated with ibrutinib and other small molecule kinase inhibitors targeting immune signaling pathways. Clin Infect Dis. 2018;66:140-148.
- Baron M, Zini JM, Belval TC, et al. Fungal infections in patients treated with ibrutinib: two unusual cases of invasive aspergillosis and cryptococcal meningoencephalitis. Leuk Lymphoma. 2017;58:2981-2982.
- Varughese T, Taur Y, Cohen N, et al. Serious infections in patients receiving ibrutinib for treatment of lymphoid cancer. Clin Infect Dis. 2018;67:687-692.
- Visentin A, Gurrieri C, Imbergamo S, et al. Epidemiology and risk factors of invasive fungal infections among 795 patients with chronic lymphocytic leukemia from the Padua University. Blood. 2016;128:2527.
- Kreiniz N, Bejar J, Polliack A, Tadmor T. Severe pneumonia associated with ibrutinib monotherapy for CLL and lymphoma. Hematol Oncol. 2018;36:349-354.
- Ghez D, Calleja A, Protin C, et al; for the French Innovative Leukemia Organization (FILO) CLL group. Early-onset invasive aspergillosis and other fungal infections in patients treated with ibrutinib. Blood. 2018;131:1955-1959.
- Cummins KC, Cheng MP, Kubiak DW, et al. Isavuconazole for the treatment of invasive fungal disease in patients receiving ibrutinib. Leuk Lymphoma. 2019;60:527-530.
- Imbruvica (ibrutinib) capsules/tablets, for oral use [prescribing information]. Sunnyvale, CA: Pharmacyclics; Horsham, PA: Janssen Biotech; April 2020.