Immune effector cell therapy, such as chimeric antigen receptor (CAR) T-cell therapy, has become the newest treatment strategy for patients with relapsed or refractory B-cell lymphomas, including diffuse large B-cell lymphoma (DLBCL). Currently, 3 anti-CD19 CAR T-cell drugs are approved by the US Food and Drug Administration for the treatment of relapsed or refractory DLBCL, including tisagenlecleucel (Kymriah), axicabtagene ciloleucel (Yescarta), and lisocabtagene maraleucel (Breyanzi).1-3
These immunotherapy agents have demonstrated positive response rates for patients with chemorefractory DLBCL.4-7 However, significant adverse events have been reported with CAR T-cell therapies, ranging from cytopenia and infection to unique adverse events, such as cytokine release syndrome (CRS) and immune effector cell–associated neurotoxicity syndrome (ICANS).8,9
CRS and ICANS are the most notable adverse events associated with CAR T-cell therapy and require prompt recognition and proper management. CRS elicits the recruitment of several cytokines, including interleukin (IL)-6, interferon-γ, tumor necrosis factor–α, IL-1, IL-2, and IL-10, which lead to significant systemic inflammation.10
The exact pathophysiology of ICANS is unknown, but the proposed mechanism involves a disruption in the blood–brain barrier permeability as a result of systemic inflammation that leads to increased levels of circulating cytokines in the cerebrospinal fluid.11 Encephalopathy is the most notable symptom of ICANS; the American Society for Transplantation and Cellular Therapy is using the immune effector cell–associated encephalopathy (ICE) score for evaluating the severity of ICANS.11
Early identification and appropriate management are critical for the resolution of CRS and ICANS. Tocilizumab, a humanized IL-6 receptor antibody, is the mainstay for the management of CRS, but it is ineffective alone for the management of ICANS, because of its inability to cross the blood–brain barrier.12,13 For the treatment of ICANS, corticosteroids are recommended as first-line therapy.9 Clinical data in a murine model have previously demonstrated that IL-1 is a target to mitigate ICANS and CRS.13
In a study of adults with acute lymphoblastic leukemia, IL-1 levels were identified in samples of cerebrospinal fluid during severe neurotoxicity.14 Anakinra, an IL-1 receptor antagonist, is indicated for the treatment of rheumatoid arthritis, neonatal-onset multisystem inflammatory disease, and deficiency of IL-1 receptor antagonist and has been used for the treatment of secondary hemophagocytic lymphohistiocytosis and other disorders that are pathologically similar to CRS.15-17
In a single-center case series, Strati and colleagues demonstrated a possible steroid-sparing strategy with the use of anakinra for the treatment of ICANS, using a median anakinra dose of 100 mg daily for a median duration of 7 days.18
Antiepileptic drugs are a common treatment modality in patients with ICANS, and the choice of antiepileptic drug varies based on the patient’s seizure type, duration, and frequency. Levetiracetam is recommended for the management of seizures, and current guidelines, such as the Society for Immunotherapy of Cancer guidelines and the MD Anderson CAR T-Cell-Therapy-Associated Toxicity (CARTOX) Working Group’s recommendations, suggest the use of levetiracetam as prophylaxis in patients with CRS without ICANS.9,19
Other antiepileptic drugs may be used in patient-specific scenarios to treat seizures adequately. Current case reports and case series do not provide descriptions of the long-term management of antiepileptic therapy in patients with ICANS after receiving CAR T-cell therapy.18,20,21
Case Report
A 62-year-old white woman was initially diagnosed with the BCL2 gene mutation and the c-Myc transcription alteration associated with DLBCL, for which she received treatment with the R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) regimen for 6 cycles. After her completion of the R-CHOP regimen, imaging studies showed that the patient had primary refractory DLBCL. She then received treatment with rituximab, gemcitabine, and oxaliplatin for 4 cycles, but her disease continued to progress.
Based on this finding, she was referred to our institution to consider whether she should receive CAR T-cell therapy or autologous hematopoietic stem-cell transplant. The decision was ultimately made to proceed with CAR T-cell therapy. Consequently, the patient received 1 cycle of polatuzumab, bendamustine, and rituximab as a bridge before the administration of CAR T-cell therapy. Her lymphodepletion regimen before the CAR T-cell infusion consisted of fludarabine 30 mg/m2 and cyclophosphamide 500 mg/m2 on days 5 to 3 before the infusion of axicabtagene ciloleucel.
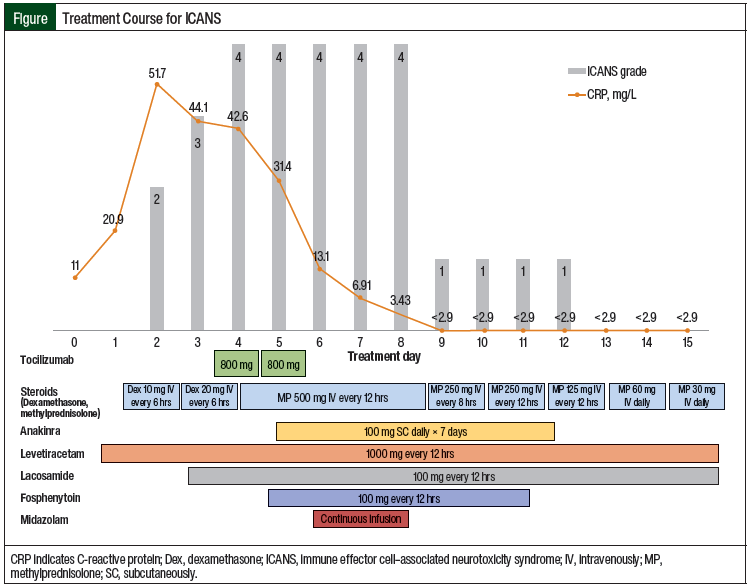
On day 1 after the axicabtagene ciloleucel infusion, the patient had a fever of 38.4°C that resolved with acetaminophen, correlating with grade 1 CRS. She started to receive antibiotics with cefepime and seizure prophylaxis with levetiracetam. Her ICE score remained 10, but her C-reactive protein (CRP) increased from baseline.
On day 2, her ICANS worsened to grade 2 and the electroencephalogram (EEG) results showed theta and delta slowing activity, without electrographic seizures. She began treatment with intravenous (IV) dexamethasone 10 mg every 6 hours.
On day 3, the patient started to have central nervous system irritability, along with ICANS progression to grade 3, although the CRS was well controlled. This prompted a dose-escalation of dexamethasone to 20 mg IV every 6 hours.
On day 4, her EEG showed generalized periodic epileptiform discharges, and she was placed on continuous EEG monitoring in the neurocritical intensive care unit (ICU). Her antiepileptic drugs were expanded to include levetiracetam and lacosamide. The seizures persisted despite the additional antiepileptic drugs, but the seizures subsequently resolved after she received a dose of lorazepam.
On the evening of day 4, the patient received her first dose of tocilizumab 8 mg/kg (800 mg) for the treatment of grade 1 CRS (ie, fever only) and was intubated for airway protection, because of her worsening mental status rather than the CRS-induced hypoxia. The steroid treatment was expanded to include methylprednisolone 500 mg every 12 hours. The patient remained seizure-free that night, although she was unable to complete the functions associated with the ICE score as of the evening of day 4.
On day 5, the patient was unarousable, with a Glasgow coma scale (GCS) of 7 and grade 1 papilledema. A second dose of tocilizumab 800 mg was administered for her persistent fever (ie, grade 1 CRS). Throughout the day, she became more arousable (GCS of 10), until she had nonconvulsive seizures that evening. Although the patient received a loading dose of fosphenytoin, she remained in a persistent, inducible seizure state, with elevated intracranial pressure.
Because of her persistent ICANS even while receiving high-dose steroids and aggressive supportive care, the patient was initiated treatment with anakinra at a dose of 100 mg, subcutaneously, once daily for 7 days. No dose adjustments were needed for renal dysfunction (dose adjustment is needed when creatinine clearance is <30 mL/min).
On day 6, the patient continued to have multifocal myoclonus and ictal stimulus-induced rhythmic, periodic, or ictal discharges. She started treatment with an infusion of midazolam and continued to receive levetiracetam, lacosamide, fosphenytoin, and methylprednisolone. The midazolam infusion continued until she woke up on day 8, but she still had inducible seizures on tactile stimuli. The patient received a bolus dose of 3% saline to keep her sodium levels at >145 mmol/L.
On day 9 (day 5 of anakinra treatment), the patient’s seizure activity started to improve. She was extubated, and the EEG was discontinued on day 10. Treatment with fosphenytoin was stopped, and she was transferred out of the neurocritical ICU on day 11.
The patient’s methylprednisolone treatment was decreased to 250 mg every 8 hours starting on day 9, with plans for rapid tapering that was completed on day 15. She completed anakinra treatment on day 11.
On day 12, the patient’s ICE score was 9, and she was unable to write a standard sentence, although the score improved to 10 on day 13. She had a stable course for the remainder of her hospitalization, and she was discharged on day 22, after her absolute neutrophil count recovery.
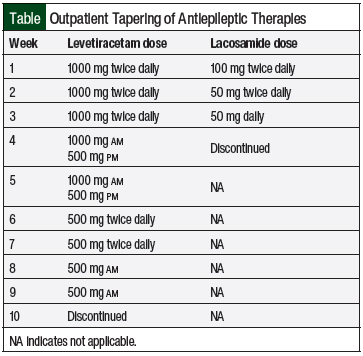
A summary of the patient’s treatment course for ICANS is outlined in the Figure. At discharge, the patient was prescribed lacosamide and levetiracetam. She followed up with the neurology department for the tapering of the antiepileptic drug regimen (Table).
At her 6-month follow-up, a positron emission tomography (PET) scan showed that she had a hypermetabolic iliac nodal mass (Deauville score, 4-5), with decreasing hypermetabolic components of the central fluid component; this was classified as a partial metabolic response.
The 9-month follow-up PET scan showed a stable hypermetabolic iliac nodal mass (Deauville score, 5). This node was biopsied, and the pathology report identified necrotic cells, so the patient was classified as having stable disease in remission.
A 1-year follow-up PET scan also showed a stable hypermetabolic iliac nodal mass (Deauville score, 5), and the patient was classified as having stable disease in remission. The patient is still undergoing active surveillance and has not had any seizures since discharge.
Discussion
ICANS is a known adverse event associated with CAR T-cell therapy and requires prompt recognition and management to ensure positive patient outcomes. ICANS occurs in approximately 64% of patients with DLBCL who receive axicabtagene ciloleucel.5 A phase 2 clinical trial has shown that the median onset of ICANS is on day 5 to 9 after axicabtagene ciloleucel infusion, with a median duration of 17 days.5
ICANS should be suspected when any mental status change occurs in a patient within the first 2 to 3 weeks after receiving an infusion of CAR T-cell therapy. First-line treatment for grade ≥2 ICANS is dexamethasone.9,19 Methylprednisolone is recommended for the treatment of grade 4 ICANS.9,19
Based on our clinical experience, dexamethasone may be considered a preferred corticosteroid compared with methylprednisolone for patients with ICANS, because of the increased blood–brain barrier penetration that results from differences in plasma protein binding. However, no clinical data are available that have evaluated the efficacy outcomes between different corticosteroids in relation to ICANS.22,23
At high doses, corticosteroids have lymphodepleting effects.24 Given the theoretical risk to negate the therapeutic effect of CAR T-cell therapies, the early use of corticosteroids should be implemented cautiously. Scarce clinical data exist that demonstrate the negative impact of corticosteroids on the efficacy of CAR T-cell therapy, including overall survival, progression-free survival, and overall response rates.25
Because of the progression of neurologic symptoms consistent with worsening ICANS on day 3, our patient immediately started treatment with dexamethasone and antiepileptic drugs. She received approximately 113 mg of dexamethasone equivalent, daily, for the 11-day treatment period (ie, cumulative dexamethasone equivalent of approximately 1240 mg). This high dose of cumulative dexamethasone equivalent is slightly higher than that in the cases presented by Strati and colleagues that had a median cumulative dexamethasone dose of 273 mg (range, 0-1344 mg).18
Despite aggressive standard-of-care treatment with high-dose glucocorticoids, our patient continued to demonstrate consistent and prolonged epileptiform changes on EEG, which led to the decision to initiate treatment with anakinra.
The efficacy of anakinra for the treatment of steroid-refractory ICANS has not been well-defined outside of case reports and case series.18,20,21 In the case series by Strati and colleagues of 8 patients who received axicabtagene ciloleucel and had grade ≥3 ICANS, the management strategy consisted of high-dose corticosteroids and anakinra, with a median of 100 mg (range, 50-200 mg) daily dose of anakinra, for a median of 7 days (range, 1-7).18 Anakinra was started at a median of 12 days after the infusion of axicabtagene ciloleucel.18
The indication for anakinra treatment was hemophagocytic lymphohistiocytosis in 2 of the 8 patients, and refractory ICANS in the other 6 patients.18 Of the 6 patients with DLBCL, 3 (50%) had a documented response to anakinra. Of these 6 patients, 4 had documented recurrence of adverse events; 5 of the 6 patients died within 90 days of CAR T-cell infusion (1 from pneumonia, 2 from progressive disease, 1 from hemophagocytic lymphohistiocytosis, and 1 from intracerebral hemorrhage). No information related to adverse events associated with anakinra treatment was provided in this case series.18 Each case had a different time to the presentation of ICANS (range, days 3-7), different dosing and treatment duration of corticosteroids, and received anakinra at varying stages of the treatment course (range, day 6-41).18
In addition, Wehrli and colleagues reported on the use of anakinra in 14 patients with steroid-refractory ICANS who received CAR T-cell infusion with axicabtagene ciloleucel or with tisagenlecleucel.20 The patients received a median cumulative dose of dexamethasone of 297 mg (range, 26-1228 mg). Anakinra was initiated at a median of 8.5 days after the CAR T-cell infusion and was given at a dose of 100 mg to 200 mg, subcutaneously, daily for up to 3 days. In 9 of the 14 (64%) patients, the time to the reduction of peak ICANS or refractory CRS was within 1 day of a patient receiving the last dose of anakinra.20
In our patient, treatment with anakinra was started on day 5 and was given for 7 days. Anakinra was initiated earlier in our patient than in the previous cases discussed in the studies by Strati and colleagues and by Wehrli and colleauges,18,20 because her neurologic adverse events continued to progress as a result of a lack of response to current therapy. Based on the recommended dosing of anakinra in its prescribing information,15 our patient’s normal renal function, and the previous case series by Strati and colleagues,18 we used once-daily dosing for 7 days (ie, days 5-11 after CAR T-cell infusion).
The dosing strategy used in the cases discussed by Wehrli and colleagues was similar to ours, but a notable difference from our case was a shorter duration of anakinra use.20 In our patient, we did not extend the duration of anakinra use past 7 days, given the paucity of literature supporting its safety and efficacy for prolonged treatment courses. In addition, our patient began to show neurologic improvement that was associated with the termination of seizure activity and an improvement in her ICE scores.
After the initiation of treatment with anakinra, our patient’s ICANS showed improvement by day 8, and she had complete resolution of ICANS by day 13, with anakinra treatment completed on day 11 post–CAR T-cell infusion. This was similar to the case series by Wehrli and colleagues, which showed resolution within 1 day of the last dose of anakinra.
It is likely that the resolution of ICANS in our patient may be attributed to several treatment modalities, including supportive care, antiepileptic drugs, high-dose corticosteroids, and anakinra. Our patient had no adverse events from the use of anakinra and remained in remission and seizure-free at the 1-year follow-up visit, although she did not receive antiepileptic drugs since 10 weeks after receiving CAR T-cell therapy.
Future management strategies for severe CRS and refractory ICANS could focus on mitigation and prevention. Park and colleagues published the first prospective study evaluating the use of anakinra for the prevention of severe CRS and ICANS in 31 adults receiving CD19-directed CAR T-cells.21 The early initiation of anakinra at a dose of 100 mg, subcutaneously, every 12 hours, up to a maximum dose of 100 mg every 6 hours, demonstrated a reduction in severe CRS and ICANS and required reduced doses of tocilizumab and corticosteroids. No differences were seen among the patients in terms of overall disease response in this small cohort, which demonstrates that the early initiation of anakinra is safe and effective.21
Based on its relatively short half-life (4-6 hours), more frequent than once daily dosing of anakinra may provide a novel pharmacokinetic approach to enhancing its effects.15 Long-term prospective studies are needed to evaluate the safety of early IL-1 inhibition in patients receiving CD19-directed CAR T-cell therapy.
The management of seizures in patients with grade ≥3 ICANS depends on the seizure type, duration, and frequency. In our patient, multiple antiepileptic drugs were used to manage her refractory seizures. Phenytoin levels were drawn daily and remained therapeutic during the course of her treatment (ie, treatment goal: total phenytoin level of 10-20 mcg/mL). Fosphenytoin was discontinued when our patient’s ICE scores improved and in the absence of signs of continued epileptic changes.
Lacosamide treatment was weaned over a 3-week period at our patient’s discharge from the hospital. She continued treatment with levetiracetam throughout the duration of her hospital stay, and it was to be weaned over the 2 months after her discharge.
Long-term use of antiepileptic drugs in patients with ICANS is not well-defined. In our patient, a slow, prolonged tapering was used to prevent any additional seizure-like activity in the months after her discharge from the hospital.
The lack of serum IL-1 level monitoring is a limitation of this case, but the use of biomarker modeling in such patients is still novel and not well-defined. Other case reports have provided serum levels of various inflammatory cytokines and other biomarkers as indicators of response to anakinra.18,21 Our patient did not have such laboratory tests ordered, although we did order sequential CRP tests, and those levels increased and decreased with the patient’s worsening and improving ICANS, respectively.
In addition, given the variety of treatments used to mitigate the patient’s neurologic adverse events, we are unable to make a direct correlation between anakinra and the resolution of ICANS. It is reasonable to conclude that anakinra is a safe adjunct treatment option to corticosteroids and supportive care (ie, antiepileptic drugs) to combat refractory ICANS.
Conclusion
Given the limited available literature regarding the use of anakinra for the management of ICANS associated with axicabtagene ciloleucel treatment, this case report provides insight regarding a patient’s outcomes after receiving early treatment with anakinra for steroid-refractory ICANS.
Appropriate supportive care, including the use of antiepileptic therapy, in patients with clinical evidence of seizures should be managed according to the patient’s symptoms and local guidelines. Based on our case report, slow tapering of antiepileptic medications may provide a viable strategy for the long-term management of ICANS associated with CAR T-cell therapy.
Author Disclosure Statement
Dr Randolph is on the Speaker’s Bureau of Bristol Myers Squibb; Dr Wells, Dr Holman, and Dr Marjoncu have no conflicts of interest to report.
References
- Kymriah (tisagenlecleucel) suspension for intravenous infusion [prescribing information]. Novartis; May 2022. www.novartis.com/us-en/sites/novartis_us/files/kymriah.pdf. Accessed January 6, 2023.
- Yescarta (axicabtagene ciloleucel) suspension for intravenous infusion [prescribing information]. Kite Pharma; November 2022. www.gilead.com/-/media/files/pdfs/medicines/oncology/yescarta/yescarta-pi.pdf. Accessed January 6, 2023.
- Breyanzi (lisocabtagene maraleucel) suspension for intravenous infusion [prescribing information]. Juno Therapeutics, a Bristol-Myers Squibb Company; June 2022. https://packageinserts.bms.com/pi/pi_breyanzi.pdf. Accessed January 6, 2023.
- Schuster SJ, Bishop MR, Tam CS, et al; for the JULIET investigators. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380:45-56.
- Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377:2531-2544.
- Abramson JS, Palomba ML, Gordon LI, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396:839-852.
- Locke FL, Miklos DB, Jacobson CA, et al; for all ZUMA-7 investigators and contributing Kite members. Axicabtagene ciloleucel as second-line therapy for large B-cell lymphoma. N Engl J Med. 2022;386:640-654.
- Brudno JN, Kochenderfer JN. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood. 2016;127:3321-3330.
- Maus MV, Alexander S, Bishop MR, et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune effector cell-related adverse events. J Immunother Cancer. 2020;8:e001511. doi: 10.1136/jitc-2020-001511.
- Teachey DT, Lacey SF, Shaw PA, et al. Identification of predictive biomarkers for cytokine release syndrome after chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Cancer Discov. 2016;6:664-679.
- Lee DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25:625-638.
- Actemra (tocilizumab) injection, for intravenous or subcutaneous use [prescribing information]. Genentech; June 2022. www.gene.com/download/pdf/actemra_prescribing.pdf. Accessed January 6, 2023.
- Giavridis T, van der Stegen SJC, Eyquem J, et al. CAR T cell–induced cytokine release syndrome is mediated by macrophages and abated by IL-1 blockade. Nat Med. 2018;24:731-738.
- Santomasso BD, Park JH, Salloum D, et al. Clinical and biological correlates of neurotoxicity associated with CAR T-cell therapy in patients with B-cell acute lymphoblastic leukemia. Cancer Discov. 2018;8:958-971.
- Kineret (anakinra) for injection, for subcutaneous use [prescribing information]. Swedish Orphan Biovitrum AB; December 2020. www.kineretrx.com/pdf/Full-Prescribing-Information-English.pdf. Accessed January 6, 2023.
- Bami S, Vagrecha A, Soberman D, et al. The use of anakinra in the treatment of secondary hemophagocytic lymphohistiocytosis. Pediatr Blood Cancer. 2020;67:e28581. doi: 10.1002/pbc.28581.
- Hines MR, von Bahr Greenwood T, Beutel G, et al. Consensus-based guidelines for the recognition, diagnosis, and management of hemophagocytic lymphohistiocytosis in critically ill children and adults. Crit Care Med. 2022;50:860-872.
- Strati P, Ahmed S, Kebriaei P, et al. Clinical efficacy of anakinra to mitigate CAR T-cell therapy–associated toxicity in large B-cell lymphoma. Blood Adv. 2020;4:3123-3127.
- Neelapu SS, Tummala S, Kebriaei P, et al. Chimeric antigen receptor T-cell therapy—assessment and management of toxicities. Nat Rev Clin Oncol. 2018;15:47-62.
- Wehrli M, Gallagher K, Chen YB, et al. Single-center experience using anakinra for steroid-refractory immune effector cell-associated neurotoxicity syndrome (ICANS). J Immunother Cancer. 2022;10:e003847. doi: 10.1136/jitc-2021-003847.
- Park JH, Sauter CS, Palomba ML, et al. A phase II study of prophylactic anakinra to prevent CRS and neurotoxicity in patients receiving CD19 CAR T cell therapy for relapsed or refractory lymphoma. Blood. 2021;138(suppl 1):96.
- Dexamethasone sodium phosphate injection, solution [prescribing information]. General Injectables and Vaccines; December 2022. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=cf63670b-258a-411f-a9d4-5e7664d3cbb0&type=display. Accessed January 6, 2023.
- Solu-Medrol (methylprednisolone sodium succinate for injection, USP) [prescribing information]. Pfizer; October 2021. https://labeling.pfizer.com/ShowLabeling.aspx?id=873. Accessed January 6, 2023.
- Aston WJ, Hope DE, Cook AM, et al. Dexamethasone differentially depletes tumour and peripheral blood lympocytes and can impact the efficacy of chemotherapy/checkpoint blockade combination treatment. Oncoimmunology. 2019;8:e1641390. doi: 10.1080/2162402X.2019.1641390.
- Sun Z, Xun RD, Liu MS, et al. The association between glucocorticoid administration and the risk of impaired efficacy of axicabtagene ciloleucel treatment: a systematic review. Front Immunol. 2021;12:646450. doi: 10.3389/fimmu.2021.646450.