In the 21st century, a new paradigm for cancer management has evolved based on our understanding of tumor biology. The identification of numerous tumor-specific oncogenes and molecular markers has facilitated the development of targeted agents.1,2 These agents often target single receptors or mutations, intra- and extracellularly, that are involved in tumor growth, proliferation, and communication.
These more targeted agents are available as oral tyrosine kinase inhibitors (TKIs) or intravenous monoclonal antibodies that can target cellular markers, such as vascular endothelial growth factor (VEGF).1,2 The rationale supporting such targeted therapies entails improving the specificity of tumor cell death versus traditional cytotoxic chemotherapy, which targets all cells, malignant and healthy, that are undergoing rapid proliferation.1,2
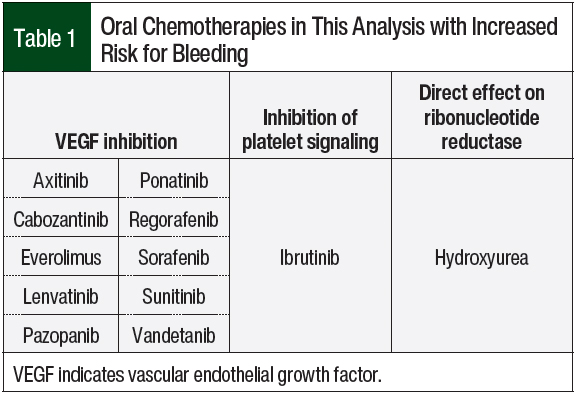
Such targeted therapies, which are often oral formulations, are designed to inhibit specific mutations or proteins that act as drivers or inhibitors of tumor growth. Inadvertently, because of unintended pharmacodynamic properties of such targeted oral chemotherapies, the medication may affect off-target proteins and cellular signaling pathways. Such off-target effects of multikinase inhibitors can result in a variety of unintended side effects, such as VEGF inhibition, which results in increased risks for bleeding and wound-healing complications.2-15 Ibrutinib has an off-target effect on platelet signaling via glycoprotein Ib, which results in the inhibition of platelet accumulation.16 Hydroxyurea has a direct effect on platelets through its inhibition of ribonucleotide reductase.17 These complications are detrimental and are compounded in the context of planned and unplanned surgical procedures.3-15 Currently, no formal guidelines have been developed for clinicians as to when to withhold oral chemotherapies before and after surgery. As a result, many protocols are based on institutional practices.18 Often such protocols on start and stop parameters are based on the biologic half-lives of each agent to allow for the pharmacokinetic extrapolation of drug excretion.3-15
Several small studies have evaluated VEGF oral chemotherapy hold times before and after surgery.19-21 In a small study of 18 patients receiving sorafenib, the medication was held for ≥7 days before surgery.19 This resulted in no wound dehiscence or other complications associated with wound healing; however, the study provided no insight into when treatment with sorafenib was restarted after surgery.19 In a small trial of 20 patients receiving sunitinib, 5 patients stopped taking the drug 5 days before surgery, and the remaining 15 patients continued the drug until the day before surgery.20 Treatment with sunitinib was not restarted postoperatively. This pilot study showed that no postoperative complications were related to when the medication was held.20 Furthermore, a review of regorafenib suggested holding the medication ≥4 weeks before surgery and to delay the reinitiation of regorafenib treatment until the wound is fully healed; however, the protocol did not differentiate between the recommendations for patients who have had major and minor surgical procedures.21
Despite the small number of trials evaluating the withholding and reinitiation times for such oral VEGF inhibitors, the pace of development and approval of such oral chemotherapies is growing rapidly. Fortunately, the prescribing information occasionally recommends general drug holding times, which are often based on the half-life of the medication, assuming that the drug will be cleared after 5 half-lives; for other drugs, the prescribing information acknowledges that the optimal time frame has not been determined.3,6-12,15,22 However, a limitation of such recommendations is that they do not differentiate between major and minor surgical procedures, which can play a role in therapy, because major surgeries are more likely to result in large-volume blood loss compared with minor surgeries.23 Major surgical procedures, such as a renal biopsy, are associated with a bleeding risk of ≥2%, whereas minor procedures, such as a central venous catheter removal, are associated with a bleeding risk of <2%.23
Given the paucity of data regarding the optimal withholding and reinitiation times for oral chemotherapy in patients having surgery, this study aims to describe such parameters for oral chemotherapy that can increase bleeding and their effect before and after minor surgical procedures. The primary objective of our study was to identify the current hold time and restart parameters for oral chemotherapy that were used by the health system. Geisinger has a pharmacist-driven oral chemotherapy clinic to assist with managing patients receiving oral oncolytic agents. Patients having surgery followed by the clinic are instructed to hold their oral chemotherapy and are documented in a progress note within the electronic health record (EHR). The secondary objectives included evaluating the risks for bleeding and wound-healing complications, as was documented in the EHR before and after the procedure, as well as whether these complications correlated with the oral chemotherapy medication being held or not being held before or after the surgical procedure.
Methods
This single-center, retrospective cohort study was conducted at Geisinger, a rural academic medical center in Pennsylvania, via manual chart review. The study included patients who were aged >18 years who received an oral chemotherapy agent with an increased risk for bleeding (Table 1) and had a minor surgical procedure while receiving therapy between August 1, 2013, and July 31, 2018. Patients with cancer were excluded from the study if they did not receive an oral chemotherapy agent with an increased risk for bleeding and/or and if they did not have a minor surgical procedure while receiving therapy.
Based on the inclusion criteria, a list of patients was generated from the institution’s EHRs for individual chart review. The patients were allocated into 2 cohorts for evaluation: patients whose oral chemotherapy was held before having a minor surgical procedure and those whose oral chemotherapy was not held before surgery.
A chart review of the clinicians’ progress notes was performed to determine patients’ cancer diagnosis, the date that the oral chemotherapy was initiated, when the oral chemotherapy was stopped before the surgical procedure, the date of the surgical procedure, and the date that the oral chemotherapy was resumed after the surgical procedure. In addition, the progress notes were evaluated for the documentation of bleeding and wound-healing complications after surgery.
This study was performed in accordance with the requirements of the health system and was approval by the Geisinger Institutional Review Board.
Statistical Analysis
Descriptive statistics were used to analyze the patients’ demographic information. Categorical and nominal variables were described using percentages, medians, and standard deviations. Continuous variables were described using medians. Descriptive statistics were further stratified by whether the oral chemotherapy was held before the procedure. Chi-square (χ2) testing was used to compare the categorical variables between the arms. Statistical analysis was performed using IBM SPSS Statistics version 26 (IBM Corporation; Armonk, NY).
Results
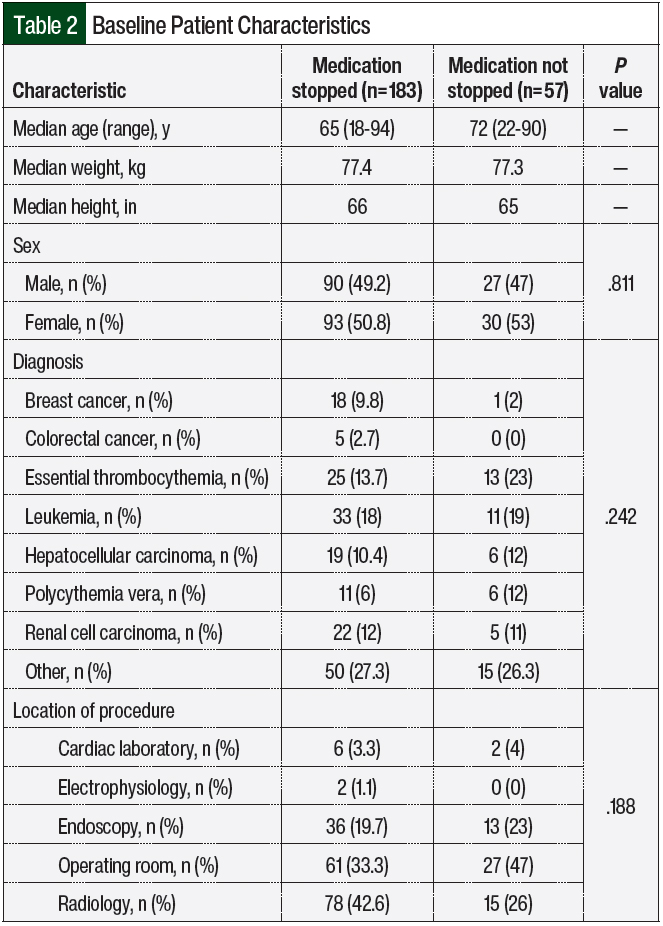
Of the 240 patients who met the specified inclusion criteria and were included in the final analysis, 183 (76.25%) stopped receiving oral chemotherapy before their procedure and 57 (23.75%) did not (Table 2). The median age was 65 years in the cohort whose oral chemotherapy was held versus 72 years in the cohort whose oral chemotherapy was not held. The distribution of sex was relatively balanced between the cohorts. The most common malignancy was leukemia, which comprised 33 (18%) patients in the group whose medication was held and 11 (19%) patients in the group whose medication was not held. The second most common diagnosis was essential thrombocythemia, which included 25 (13.7%) patients in the group whose medication was held and 13 (23%) patients in the group whose medication was not held.
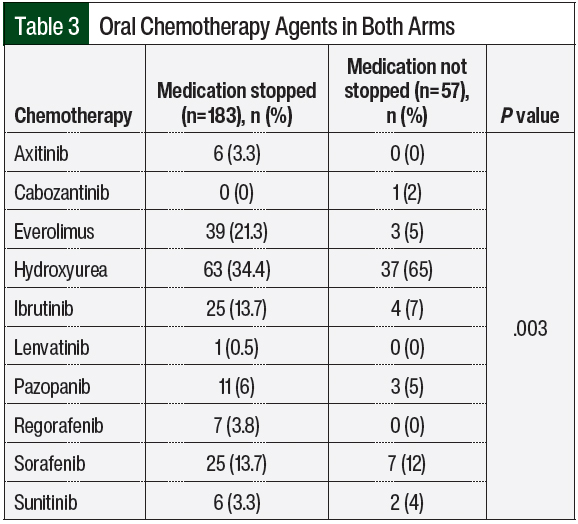
A total of 10 medications were included in the analysis, based on their ability to impact wound healing (Table 3). Hydroxyurea was the most-often received agent in both groups. In the cohort whose oral chemotherapy was held, 63 (34.4%) patients received hydroxyurea versus 37 (65%) patients in the group whose therapy was not held.
Esophagogastroduodenoscopy was the most common procedure in both groups, which accounted for 22 (12%) and 6 (11%) procedures in the groups that did and did not have the medication held before the procedure, respectively. No difference was seen in the types of procedures between the arms (P=.999). See Supplementary Appendix for a full list of the procedures.
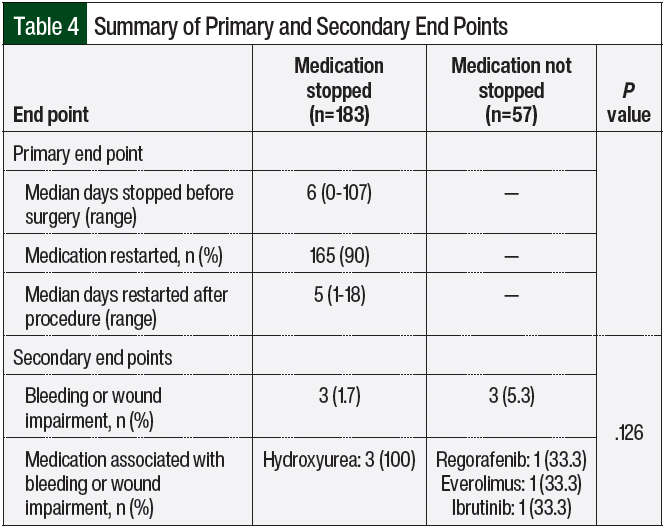
The median days in which oral chemotherapy was held before the procedure was 6 (range, 0-107 days). Ultimately, 165 (90%) patients restarted their medication after having a surgical procedure. The median time to restarting oral chemotherapy after the procedure was 5 days (range, 1-18; Table 4). Of note, of the 57 patients whose medication was not held, none were followed by the oral chemotherapy clinic.
There were 3 (5.3%) patients who had bleeding or a wound-healing complication in the cohort in which oral chemotherapy was not held before the procedure, of whom 1 patient each received regorafenib, everolimus, and ibrutinib. In the cohort of patients whose oral chemotherapy was held before their surgical procedure, 3 (1.7%) patients, who were all receiving hydroxyurea, had bleeding or a wound-healing complication (Table 4).
Discussion
To our knowledge, this is the largest review of hold times for oral chemotherapy agents before and after an invasive surgical procedure. This study includes a variety of targeted oral chemotherapy agents, comprising a large cohort of different multitargeted TKIs that inhibit VEGF. A large number of nonmultitargeted TKIs were also in our analysis, such as hydroxyurea and ibrutinib. In this analysis, the majority of patients were instructed to hold their oncolytic medication a median of 6 days before having a surgical procedure.
Of the patients whose oncolytic medication was held, 90% restarted their treatment after the surgical procedure (median time, 5 days after the procedure). This time period is within the 5 half-lives that many of the agents included in this study have, except for cabozantinib and sorafenib.3,6,7
Among 18 patients whose records cited reasons for not resuming therapy after surgery, the 2 reasons cited in the patients’ records for not resuming therapy after having surgery were disease progression (n=12; 66.7%) and medication intolerance (n=6; 33.3%). This analysis demonstrates that many patients whose medication is held appropriately restart their therapy if required and, based on the half-lives of the medications included in the analysis, are done after approximately 5 half-lives.
The patients’ baseline demographics were similar between the 2 cohorts. The most common oral agents received were hydroxyurea (41.6%), everolimus (17.5%), sorafenib (13.3%), and ibrutinib (12.1%). There was a larger percentage of patients with essential thrombocythemia who did not stop their medication before surgery than who stopped their medication (25% vs 13.7%, respectively). Furthermore, in patients with breast cancer, there were more patients whose medication was held before surgery than those whose medication was not held (9.8% vs 2%, respectively).
The incidence of bleeding in the overall population was low (2.5%). Although the bleeding rate was higher in the population that did not stop the medication before surgery than in those who did stop their medication before surgery, this was not statistically significant (P=.126).
Limitations
Our study had several limitations. Because this study was retrospective in nature, it had the potential to introduce documentation bias, although we used a standard data collection tool to help prevent this issue. In addition, manual chart review was required to identify the medication hold and restart dates, although we were unable to confirm in reality whether the patient had stopped and restarted the medication as instructed.
Because manual chart review was also used to assess the incidence of bleeding, cases may have been overlooked if they were not documented in the EHR. Because statistical power was not calculated for our study, the ability to truly detect a significant difference in the incidence of bleeding between the 2 arms may not have been achieved.
Conclusion
To our knowledge, this is one of the largest evaluations to assess the utility of holding oral chemotherapies that can impact wound healing, such as VEGF inhibitors, before and after surgical procedures. This review analyzed these medications using real-world data and demonstrated that the overall incidence of bleeding before and after minor surgical procedures is low, even when oral chemotherapy was not held before and after the procedure.
Based on our findings, the median medication hold times of 5 to 6 days before and after surgery are safe and effective in avoiding the risk for postoperative bleeding complications. Future prospective studies are needed to confirm these findings.
Author Disclosure Statement
Dr Andrick is an employee of Geisinger and has received a 1-year grant for VTE Research in Cancer Patients from the Hematology/Oncology Pharmacy Association. Dr Marjoncu, Dr Carmichael, and Dr Oxenberg have no conflicts of interest to report.
References
- Zondor SD, Medina PJ. Bevacizumab: an angiogenesis inhibitor with efficacy in colorectal and other malignancies. Ann Pharmacother. 2004;38(7-8):1258-1264.
- Hartmann JT, Haap M, Kopp HG, Lipp HP. Tyrosine kinase inhibitors–a review on pharmacology, metabolism and side effects. Curr Drug Metab. 2009;10(5):470-481.
- Nexavar (sorafenib) tablets, for oral use [prescribing information]. Bayer HealthCare Pharmaceuticals; July 2020. https://labeling.bayerhealthcare.com/html/products/pi/Nexavar_PI.pdf. Accessed April 4, 2023.
- Sutent (sunitinib malate) capsules, for oral use [prescribing information]. Pfizer Labs; September 2021. https://labeling.pfizer.com/showlabeling.aspx?id=607. Accessed April 4, 2023.
- Inlyta (axitinib) tablets, for oral administration [prescribing information]. Pfizer Labs; October 2022. https://labeling.pfizer.com/ShowLabeling.aspx?id=759. Accessed April 4, 2023.
- Cabometyx (cabozantinib) tablets, for oral use [prescribing information]. Exelixis; February 13, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2022/208692Orig1s013lbl.pdf. Accessed April 4, 2023.
- Cometriq (cabozantinib) capsules, for oral use [prescribing information]. Exelixis; October 2020. www.cometriq.com/downloads/Cometriq_Full_Prescribing_Information.pdf. Accessed April 4, 2023.
- Lenvima (lenvatinib) capsules, for oral use [prescribing information]. Eisai; November 2022. www.lenvimahcp.com/-/media/Files/Lenvima/PDF/prescribing-information.pdf?&hash=6414cdb8-4d19-4d27-aa9a-051c959cd79d. Accessed April 4, 2023.
- Votrient (pazopanib) tablets, for oral use [prescribing information]. Novartis Pharmaceuticals Corporation; December 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2021/022465Orig1s033lbl.pdf. Accessed April 4, 2023.
- Iclusig (ponatinib) tablets, for oral use [prescribing information]. ARIAD Pharmaceuticals; February 2022. www.iclusig.com/hcp/sites/default/files/2022-10/ICLUSIG-Prescribing-Information.pdf. Accessed April 4, 2023.
- Stivarga (regorafenib) tablets, for oral use [prescribing information]. Bayer HealthCare Pharmaceuticals; December 2020. https://labeling.bayerhealthcare.com/html/products/pi/Stivarga_PI.pdf. Accessed April 4, 2023.
- Caprelsa (vandetanib) tablets, for oral use [prescribing information]. Genzyme Corporation; December 2022. https://products.sanofi.us/caprelsa/caprelsa.pdf. Accessed April 4, 2023.
- Afinitor (everolimus) tablets, for oral use/Afinitor Disperz (everolimus tablets for oral suspension) [prescribing information]. Novartis Pharmaceuticals Corporation; February 2022. www.novartis.com/us-en/sites/novartis_us/files/afinitor.pdf. Accessed April 4, 2023.
- Hydrea (hydroxyurea) capsules, for oral use [prescribing information]. Bristol-Myers Squibb; July 2021. https://packageinserts.bms.com/pi/pi_hydrea.pdf. Accessed April 4, 2023.
- Imbruvica (ibrutinib) capsules, tablets, for oral use/oral suspension [prescribing information]. Pharmacyclics, Janssen Biotech; August 2022. www.rxabbvie.com/pdf/imbruvica_pi.pdf. Accessed April 4, 2023.
- Wang J, Zhao A, Zhou H, et al. Risk of bleeding associated with ibrutinib in patients with B-cell malignancies: a systematic review and meta-analysis of randomized controlled trials. Front Pharmacol. 2020;11:580622.
- Ferrari A, Carobbio A, Masciulli A, et al. Clinical outcomes under hydroxyurea treatment in polycythemia vera: a systematic review and meta-analysis. Haematologica. 2019;104(12):2391-2399.
- Bodnar RJ. Anti-angiogenic drugs: involvement in cutaneous side effects and wound-healing complication. Adv Wound Care (New Rochelle). 2014;3(10):635-646.
- Zhang Y, Li Y, Deng J, et al. Sorafenib neoadjuvant therapy in the treatment of high risk renal cell carcinoma. PLoS One. 2015;10(2):e0115896.
- Hellenthal NJ, Underwood W, Penetrante R, et al. Prospective clinical trial of preoperative sunitinib in patients with renal cell carcinoma. J Urol. 2010;184(3):859-864.
- Krishnamoorthy SK, Relias V, Sebastian S, et al. Management of regorafenib-related toxicities: a review. Therap Adv Gastroenterol. 2015;8(5):285-297.
- Shah DR, Dholakia S, Shah RR. Effect of tyrosine kinase inhibitors on wound healing and tissue repair: implications for surgery in cancer patients. Drug Saf. 2014;37(3):135-149.
- Spyropoulos AC, Douketis JD. How I treat anticoagulated patients undergoing an elective procedure or surgery. Blood. 2012;120(15):2954-2962.