Cancer continues to be a significant cause of morbidity and mortality, with an estimated 1.9 million expected new cancer diagnoses and 609,820 projected cancer deaths in the United States in 2023.1 Despite many new advances in cancer therapies in the past few decades,2 traditional intravenous (IV) cancer therapy remains an essential component of numerous cancer treatment regimens. The use of cancer therapies requires the consideration of adverse events, interactions, and other patient-specific factors.3 Patients with cancer who receive therapy in the ambulatory care setting often have complications associated with treatment, but they are not monitored for 24 hours daily, such as patients who receive their treatment in the inpatient setting. Time and resources from the multidisciplinary oncology team are required to properly manage these drug-related adverse events to prevent emergency department visits or hospital admissions.4,5
Ambulatory care oncology pharmacists have significantly contributed to improvements in efficient healthcare and the outcomes of patients with cancer through medication reconciliation, medication error prevention, drug interaction assessment, and medication therapy management.6-10 Pharmacists also help with patient education on cancer therapy by filling healthcare gaps and optimizing the patients’ knowledge about their medications.11 Patients have responded positively to the presence of clinical pharmacists in ambulatory clinics, claiming that these services are worthwhile and helpful for managing their symptoms at home.12
The skill sets of oncology pharmacists add value to the care team through pretreatment education on cancer therapy, and they add value to the multidisciplinary approach through follow-up care.13-16 In recent years, pharmacists have established themselves as robust caregivers for follow-up on oral cancer therapies; pharmacists are particularly effective in this setting because of their knowledge of drug-related administration, adherence, and overall management.13-16 Financially, the addition of a pharmacist to the clinical setting allows for cost-savings while improving clinic operations and quality-based patient care.17
More recently, telehealth appointments have been used effectively in a variety of patient care areas. Telehealth, which has been defined as “the use of electronic information and telecommunications technologies to support or promote long-distance clinical health care, patient and professional health-related education, public health, and health administration,” has become increasingly used as a result of the COVID-19 pandemic.13,18,19 In the oncology setting, studies have shown the effectiveness of nurse practitioner–driven telehealth interventions for the early recognition of treatment-related symptoms in patients with cancer.20,21 Although pharmacists have used telehealth in other disease states and for the management of oral cancer therapies, there is a paucity of literature assessing the impact and value of an ambulatory clinic–based pharmacist in relation to pretreatment drug education and short-term follow-up for patients with cancer who are newly starting an IV treatment regimen.22
At the Abramson Cancer Center within the Hospital of the University of Pennsylvania, pharmacists play a key role in the initial management of patients who are newly starting IV treatments for cancer. This process begins with an in-person or telehealth cancer therapy education session led by 1 of 3 pharmacists. These pharmacists are primarily independent of the dispensing role and are responsible for supporting the ambulatory education program in partnership with a dedicated oncology nurse. These pharmacists also service the outpatient infusion center through order verification, covering staff pharmacists’ lunch and meeting times, and participating in rapid responses.
The cancer therapy education sessions primarily focus on explaining common drug-related adverse events and the management of side effects, addressing drug–drug interactions, and updating a patient’s medication list with a medication reconciliation. After the patient’s first treatment for cancer, the clinical pharmacist will perform a follow-up phone call about adverse events with the patient or caregiver within approximately 7 days. These calls aim to identify gaps in a patient’s supportive care plan and assist in early intervention and prompt adverse-event management. The pharmacists provide pharmacologic advice by counseling and reeducating patients on how to properly take their supportive-care medications, offering recommendations for over-the-counter drugs, and identifying when a patient’s supportive-care plan needs to be escalated to the primary oncology provider. The pharmacists also provide nonpharmacologic counseling for nutrition support, fatigue management, and informing and escalating referrals to the care team on behalf of the patient.
The objective of this study was to determine the clinical impact and financial value of a pharmacist-based IV cancer therapy education and early follow-up program at an academic medical center.
Methods
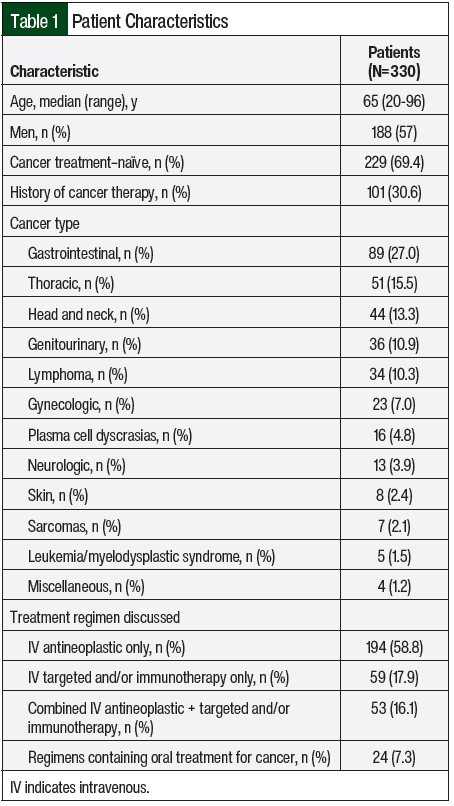
This single-arm, single-center, retrospective, observational study was conducted at the National Cancer Institute– and National Comprehensive Cancer Network–designated Abramson Cancer Center. Patients aged ≥18 years who newly started an IV cancer therapy within the infusion center between the dates of September 29, 2021, and January 14, 2022, and received education from an ambulatory oncology clinical pharmacist were included in this study. The data collection start date was September 29, 2021, because this is when the process began for the follow-up calls regarding adverse events. The clinical pharmacists support patients with a wide variety of malignancies who are receiving an array of cancer regimens (Table 1). Patients with breast cancer are not included in this study because the breast cancer nurse practitioner team specifically performed IV cancer therapy education for all their patients during the study period. This study was reviewed by and qualifies as quality improvement according to the University of Pennsylvania Institutional Review Board.
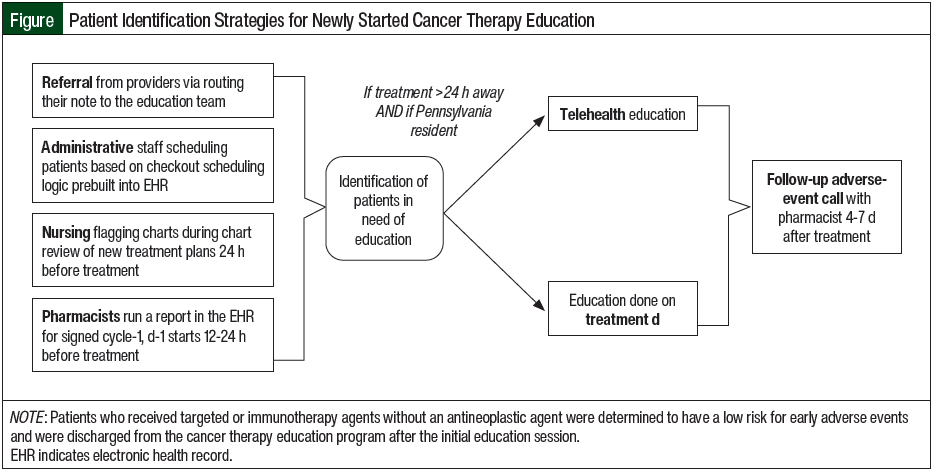
Patients who were beginning any cancer treatment were identified in a variety of ways (Figure). After the initial education and at the discretion of the pharmacist, the patients who received regimens that included an antineoplastic cancer agent or were otherwise expected to result in significant early adverse events were the primary ones to receive follow-up checks for adverse events approximately 4 to 7 days after treatment. Patients who received a targeted therapy or immunotherapy without an antineoplastic agent were determined to have a low risk for early adverse events and were discharged from the cancer therapy education program after the initial session. Each patient who was enrolled in the follow-up calls received 1 call after each cycle for the first 1 to 2 treatment cycles depending on their individual adverse events. Patients who did not answer or did not call back after the attempted call were scheduled to be seen during their next scheduled infusion visit.
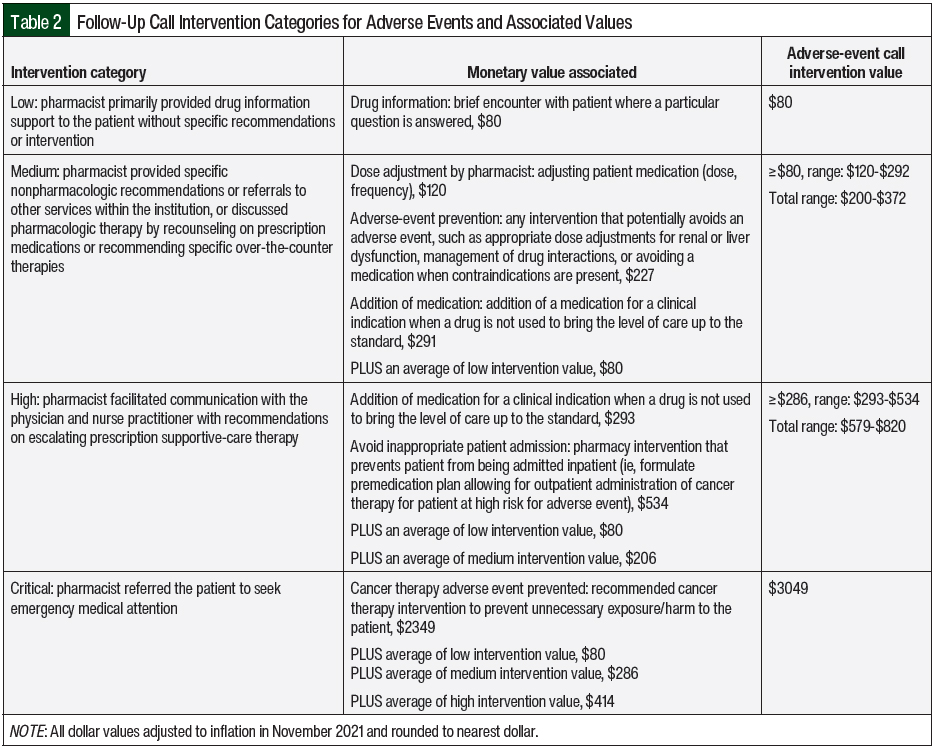
The initial education sessions and follow-up calls were tracked using the pharmacist iVent (intervention) tracking feature within the Epic electronic medical record and were documented as encounters visible for providers and patients. The iVent interface allowed for intuitive categorization of the significance of each pharmacist encounter. The pharmacists’ interventions were separated into the 4 intervention categories of low, medium, high, and critical. Low-level interventions included encounters in which the pharmacist primarily provided drug information to the patient without specific recommendations or intervention. Medium-level interventions included low-level interventions, but also encompassed an additional level in which the pharmacist provided specific nonpharmacologic recommendations, referrals to other services within the institution, or discussed pharmacologic therapy by reeducating the patient on prescription medications or recommending specific over-the-counter treatments. High-level interventions encompassed the services described in the low- and medium-level interventions with escalation to the physician or nurse practitioner for recommendations of adverse-event management with or without prescription supportive-care therapy on the patient’s behalf. Critical interventions were those that the pharmacist referred the patient to seek emergency medical attention.
Financial impact was assessed by using the estimated value of the pharmacist encounters for the specific interventions performed. The monetary values for these interventions were compiled from 2 studies that assessed pharmacists’ value in a similar outpatient setting with intervention values that were validated through literature and internal reviews.23,24 The values of the interventions in this study were determined by linking the cost associations from these articles to our prespecified intervention categories and adjusting for inflation. Because all new education follows a very similar process, the values of several key components of the pharmacist’s encounter were combined and averaged based on their monetary value. The new cancer therapy education included interventions on drug information ($20 per 10 minutes), medication reconciliation ($75 per 20 minutes), discharge prescription review ($320 per 15 minutes), and medication education ($308 per 30 minutes), which results in approximately $723.20 per 75 minutes of patient interaction within a cancer therapy education visit. Because the practice has an average new education encounter of 60 minutes, the value approve revision was adjusted and rounded to $578 per new education visit.
Given the potentially wide range of clinical activities that could encapsulate the prespecified intervention categories, we used a range of values to present each specific intervention category. Because the advanced intervention categories naturally include more actions with more meaningful interventions, the average value of the range from previous intervention categories was added for the more advanced intervention categories. For example, a high-level intervention would have its own range, with the addition of the average of the ranges for the low- and medium-level category interventions (Table 2).
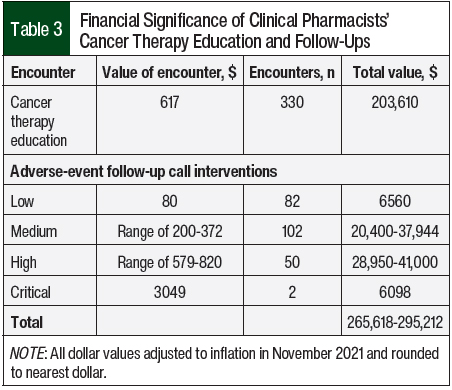
The Consumer Price Index inflation calculator was used to calculate the adjusted values of these interventions, with an estimated date of November 2021 compared with the original articles’ published dates.23,24 These values were then rounded to the nearest whole dollar. This resulted in values for each encounter of $617 for new education, $80 for low-impact adverse-event checks, a range of $200 to $372 for medium-impact adverse-event checks, a range of $579 to $820 for high-impact adverse-event checks, and $3049 for critical-impact adverse-event checks (Table 3).
Results
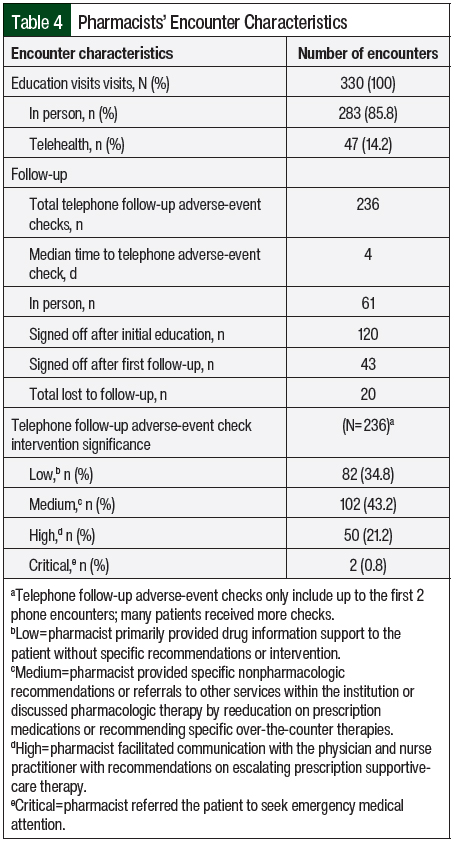
From September 29, 2021, to January 14, 2022, a total of 330 patients from the Abramson Cancer Center infusion suite were included in this study (Table 1). The patients’ median age was 65 years, with a majority (57%) being men. The patients had a wide variety of cancers, with the most common being gastrointestinal (27%). Other main cancer sites included thoracic (15.5%), head and neck (13.3%), genitourinary (10.9%), lymphoma (10.3%), and gynecologic (7%). Most patients were receiving first-line treatment, with 69.4% being cancer treatment–naïve, and 30.6% having previously received cancer therapy. Most patients (58.8%) received IV antineoplastic chemotherapy alone. Approximately 17.9% of the patients received IV targeted and/or immunotherapy alone, 16.1% of patients received treatment with combination IV chemotherapy and IV targeted therapy and/or immunotherapy, and 7.3% of patients received a regimen containing an oral treatment for cancer (Table 1).
Most of the patients were taught in the infusion center (85.8%), with approximately 14.2% of patients receiving their cancer therapy education through an initial telehealth visit ahead of their first infusion (Table 4). A total of 236 follow-up adverse-event management calls were completed, but the numbers of patients who did not answer the call or who did not call back were not available. Of the calls, 82 (34.8%) were low significance, 102 (43.2%) were medium significance, 50 (21.2%) were high significance, and 2 (0.8%) were critical significance (Table 4). Of the 2 critically significant calls, 1 was a referral to the emergency department to rule out myocardial infarction after a pharmacist called and the patient had chest pain. The other critically significant call was a referral to the emergency department when the pharmacist called and the patient had shortness of breath. In the emergency department, the patient was diagnosed with pulmonary emboli, pleural effusion, and pericardial effusion, which were all managed appropriately.
In terms of financial value, the combined 330 initial cancer therapy education visits brought a total of $203,610 of value (Table 3). Low-impact and critical-impact adverse-event calls brought total values of $6560 and $6098, respectively. The medium-impact adverse-event calls brought a range of $20,400 to $37,944 of value. The high-impact adverse-event calls brought a range of $28,950 to $41,000 of value. Overall, during the 3-month assessment, the clinical pharmacists accounted for $265,618 to $295,212 of value (Table 3).
Discussion
As the burden of cancer care weighs on the United States, it is vital to develop and implement novel approaches to guide patient-centered care.1 Ambulatory oncology pharmacists can play a unique role in the delivery of medication information and supportive-care management for patients receiving outpatient cancer therapy.6-11 Although pharmacists have established their place in the continuum of outpatient oral oncolytic drug management, there is a gap of direct involvement of these trained professionals with IV drugs for patients with cancer, an area with many unmet needs.8 Because of their distinct understanding of medication properties, pharmacists are an ideal member of the healthcare team to provide cancer therapy education and early supportive care follow-up.25 In addition, pharmacists have a broad range of clinical understanding; this study alone included patients with more than 12 distinct categories of malignancies. Furthermore, the clinical pharmacists provided education on complex multiagent regimens, patients with language barriers, and patients aged 20 to 96 years.
Only patients receiving IV antineoplastic chemotherapy agents received pharmacist-driven follow-up calls, because they were expected to have immediately addressable drug-related adverse events. Follow-up phone calls by the pharmacists within the first week of therapy allowed for the early identification and mitigation of drug-related adverse events, which permitted quick intervention and symptom management by the pharmacist or a referral to the physician or nurse practitioner for further prescription-strength pharmacologic intervention. This was beneficial in that the patient received immediate attention instead of waiting for the next scheduled physician and nurse practitioner visit, and it allowed for more meaningful time spent during provider visits for matters unrelated to symptom management.
Furthermore, critical interventions allowed an opportunity for fast escalation of potentially fatal adverse events from cancer therapy in patients who may not have been identified until their next infusion. In our study, we identified 2 critical interventions. Neither of the critical-intervention patients had reached out to the team regarding their adverse events; the phone call from the pharmacist allowed for swift identification of potentially life-threatening adverse events with immediate recommendations to seek emergency medical attention.
This study focused only on early follow-up for adverse events, with most patients being signed off the education service within the first 3 cycles of cancer therapy. Clinical pharmacists’ follow-up for later cycles of IV cancer therapy may also prove beneficial in this patient population. There is a potential for clinical pharmacists to manage long-term adverse events related to antineoplastic agents, as well as the specific management of complications associated with IV targeted drugs and immunotherapies. For example, moving forward, pharmacists may be beneficial in the management of hypertension after treatment with bevacizumab or steroid management for immunotherapy-related adverse events. These potential scenarios were not included in this study, and they are not yet a part of the current workflow.
The financial analysis showed significant value for the use of pharmacists in this setting. The estimated pharmacist’s salary in Pennsylvania for this role is $92,000 to $128,000. With our current staffing of up to 3 full-time equivalents, this results in a total pharmacy cost of $276,000 to $384,000 annually. Given that this calculation does not include benefits and other nonsalary compensation, we assumed the total cost for 3 full-time equivalents to be $384,000 annually. Over 3 months, the clinical pharmacists showed a potential value of $265,618 to $295,212. Extrapolated to 1 year, this would be equivalent to a range of $1,062,472 to $1,180,848. Given the involvement and integration of the pharmacist team beyond the cancer therapy education program, it is anticipated that the true value of these pharmacists would be closer to the high end of the range, which would result in up to $1,180,848 of value from the 3 clinical pharmacists. Accounting for the upper limit of the pharmacists’ salary compared with the value demonstrated, the pharmacists were concluded to result in $796,848 of additional value to the institution as a group over the course of 1 calendar year. Given the expected expansion of this program to support more patients at satellite sites and further deepen the impact from each clinical pharmacist, it is likely that this value will continue to grow.
Limitations
This study had several limitations. With the current workflow of identifying patients, patients may not be identified for initial pharmacist education, which may decrease the overall number of patients included. The process for scheduling patients for follow-up is also limited to manual addition by the pharmacist, which increases the risk for patients being lost to follow-up as a result of human error. This is currently being addressed by the information technology team to better streamline the identification of patients who are cancer therapy–naïve or who are changing treatment to ensure timely education.
Furthermore, the infusion center total numbers and staffing were affected by the COVID-19 Omicron outbreak, which may have also resulted in a lower number of patients included than a standard 3-month time frame.
The financial analysis was extrapolated from specific interventions to broad intervention categories. In future assessments, we plan to identify a specific disease state and regimen where we can validate the exact clinical and financial impacts of our interventions versus a historic control in terms of hospitalizations, disease state outcomes, and adverse events. This will allow us to determine clear-cut interventions to give a definitive value of our encounters as opposed to the ranges presented in this study.
We also plan to implement a system to track patient-reported outcomes through our patient portal. This will provide an easily accessible way to analyze the impact of pharmacists, as well as patients’ quality of life. This study demonstrates the potential for significant growth and the implementation of clinical pharmacists in the ambulatory oncology setting.
Conclusion
The use of ambulatory oncology clinical pharmacists for initial IV cancer education and early follow-up provides clinical and financial value. Pharmacists can provide significant value to the healthcare organization through medication therapy management, detailed cancer therapy education, and supportive-care adverse-event management follow-up, with a potential to provide up to $796,848 in additional value to the institution over 1 calendar year.
Acknowledgments
We would like to thank the leaders of the Hospital of the University of Pennsylvania and the Abramson Cancer Center for affording our teams the time and resources to define a quality improvement project at the Perelman Center for Advanced Medicine to improve the lives of our patients. We would specifically like to acknowledge Donna Capozzi for providing leadership to inspire the project and Daniel Rubin for his invaluable assistance with data reporting.
Author Disclosure Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- American Cancer Society. Cancer Facts & Figures 2023. 2023. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf. Accessed May 1, 2023.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-674.
- American Cancer Society. Chemotherapy side effects. May 1, 2020. www.cancer.org/treatment/treatments-and-side-effects/treatment-types/chemotherapy/chemotherapy-side-effects.html. Accessed May 1, 2023.
- Read H, Ladds S, Rhodes B, et al. The impact of a supplementary medication review and counselling service within the oncology outpatient setting. Br J Cancer. 2007;96:744-751.
- McKenzie H, Hayes L, White K, et al. Chemotherapy outpatients’ unplanned presentations to hospital: a retrospective study. Support Care Cancer. 2011;19:963-969.
- Iihara H, Ishihara M, Matsuura K, et al. Pharmacists contribute to the improved efficiency of medical practices in the outpatient cancer chemotherapy clinic. J Eval Clin Pract. 2012;18:753-760.
- Lustig A. Mediation error prevention by pharmacists-an Israeli solution. Pharm World Sci. 2000;22:21-25.
- van Leeuwen RWF, Jansman FGA, van den Bemt PMLA, et al. Drug–drug interactions in patients treated for cancer: a prospective study on clinical interventions. Ann Oncol. 2015;26:992-997.
- Chun DS, Faso A, Muss HB, et al. Oncology pharmacist-led medication reconciliation among cancer patients initiating chemotherapy. J Oncol Pharm Pract. 2020;26:1156-1163.
- Jones LK, Greskovic G, Grassi DM, et al. Medication therapy disease management: Geisinger’s approach to population health management. Am J Health Syst Pharm. 2017;74:1422-1435.
- McKee M, Frei BL, Garcia A, et al. Impact of clinical pharmacy services on patients in an outpatient chemotherapy academic clinic. J Oncol Pharm Pract. 2011;17:387-394.
- Crespo A, Tyszka M. Evaluating the patient-perceived impact of clinical pharmacy services and proactive follow-up care in an ambulatory chemotherapy unit. J Oncol Pharm Pract. 2017;23:243-248.
- Bohnenkamp C, Martin GA, DuBois L, et al. Outcomes of pharmacist-provided telehealth services in an ambulatory oncology clinic during the COVID-19 pandemic. J Hematol Oncol Pharm. 2021;11(5):256-263.
- Muluneh B, Schneider M, Faso A, et al. Improved adherence rates and clinical outcomes of an integrated, closed-loop, pharmacist-led oral chemotherapy management program. J Oncol Pract. 2018;14:e324-e334.
- Patel JM, Holle LM, Clement JM, et al. Impact of a pharmacist-led oral chemotherapy-monitoring program in patients with metastatic castrate-resistant prostate cancer. J Oncol Pharm Pract. 2016;22:777-783.
- Holle LM, Puri S, Clement JM. Physician–pharmacist collaboration for oral chemotherapy monitoring: insights from an academic genitourinary oncology practice. J Oncol Pharm Pract. 2016;22:511-516.
- Lee GW, Mathur AD, Andrick BJ, et al. Pharmacist value-added to neuro-oncology subspecialty clinics: a pilot study uncovers opportunities for best practices and optimal time utilization. J Oncol Pharm Pract. 2020;26:1937-1941.
- Segal EM, Alwan L, Pitney C, et al. Establishing clinical pharmacist telehealth services during the COVID-19 pandemic. Am J Health Syst Pharm. 2020;77:1403-1408.
- American Pharmacists Association. Telehealth. 2020. www.pharmacist.com/Practice/Practice-Resources/Telehealth. Accessed May 16, 2023.
- Yajima C, Bowe C, Barber D, Dains J. Telehealth interventions for supportive management and early recognition of treatment-related symptoms in patients with hematologic malignancies. J Adv Pract Oncol. 2021;12:835-849.
- Ysebaert L, Larcher M, Compaci G, et al. Oncology nurse phone calls halve the risk of reduced dose intensity of immunochemotherapy: results of the randomized FORTIS study in chronic lymphocytic leukemia. Ann Hematol. 2019;98:931-939.
- Niznik JD, He H, Kane-Gill SL. Impact of clinical pharmacist services delivered via telemedicine in the outpatient or ambulatory care setting: a systematic review. Res Social Adm Pharm. 2018;14:707-717.
- Virani A, Schlei Z, Gleason C, et al. Impact of an oncology clinical pharmacist specialist in an outpatient multiple myeloma clinic. Clin Lymphoma Myeloma Leuk. 2020;20:e543-e546.
- Woolley AB, Berds CA IV, Edwards RA, et al. Potential cost avoidance of pharmacy students’ patient care activities during advanced pharmacy practice experiences. Am J Pharm Educ. 2013;77:164.
- Mackler E, Segal EM, Muluneh B, et al. 2018 Hematology/Oncology Pharmacist Association best practices for the management of oral oncolytic therapy: pharmacy practice standard. J Oncol Pract. 2019;15:e346-e355.