Presenting Author: Katelyn Toeniskoetter, PharmD, BCOP, Dana-Farber Cancer Institute, Boston, MA
BACKGROUND: The potent, reversible HER2 tyrosine kinase inhibitor sevabertinib (BAY 2927088)1 has demonstrated manageable safety and antitumor activity in patients with advanced non–small-cell lung cancer (NSCLC) with HER2-activating mutations.
OBJECTIVE: To report the safety and efficacy data from 2 cohorts of the ongoing, open-label, multicenter phase 1/2 SOHO-01 trial (NCT05099172).
METHODS: Patients with advanced NSCLC and HER2-activating mutations were enrolled and received oral sevabertinib 20 mg twice daily. Patients in expansion/extension cohort D had disease progression after receiving ≥1 systemic therapy and were naïve to HER2-targeted therapy; patients in expansion cohort F had not received any systemic therapy for locally advanced or metastatic disease. Safety (MedDRA, version 27.1 and Common Terminology Criteria for Adverse Events, version 5.0) was the primary end point; antitumor activity (RECIST, version 1.1) was a key secondary end point.
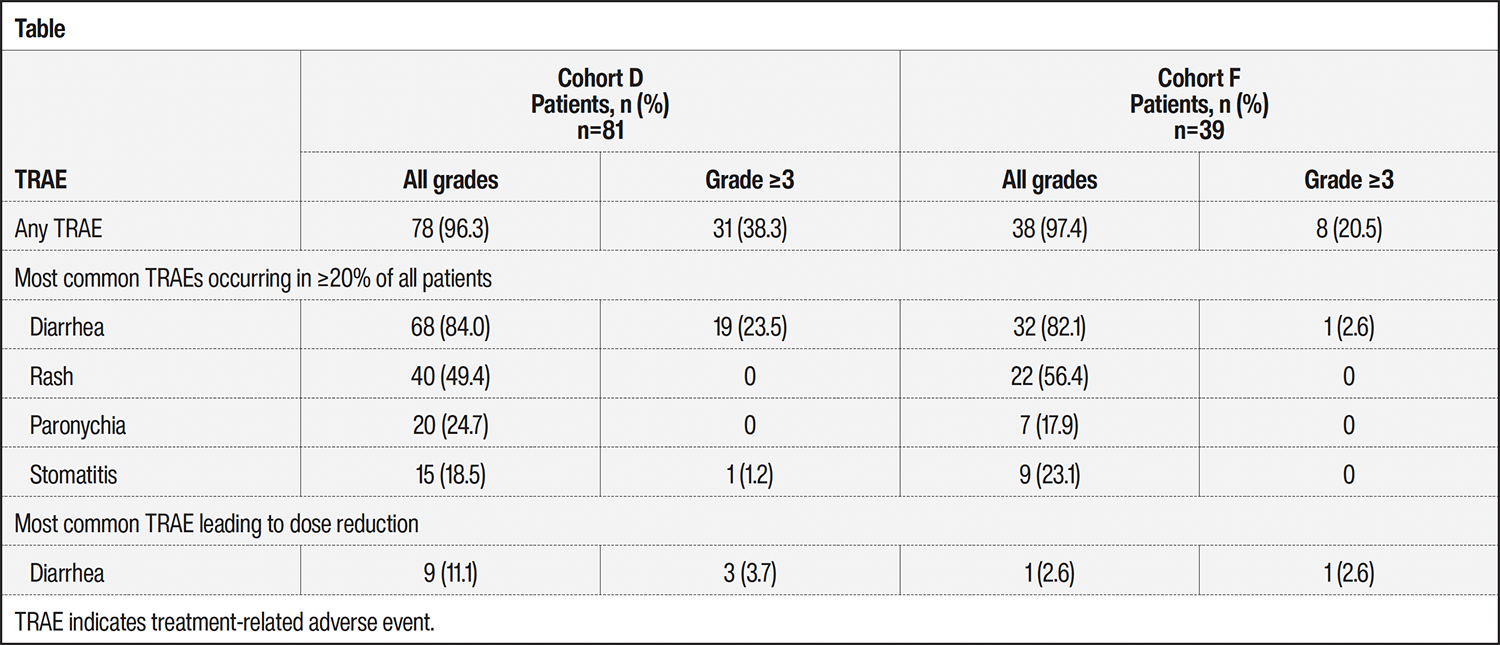
RESULTS: As of October 14, 2024, 81 patients in cohort D and 39 in cohort F received treatment. The median ages were 60 years in cohort D and 65 years in cohort F, 61.7% of patients in cohort D and 64.1% in cohort F were female, 61.7% in cohort D and 79.5% in cohort F had never smoked, and 43.2% of patients in cohort D had received ≥2 systemic therapies. All patients were analyzed for safety and efficacy; response was based on the full analysis set. Treatment-related adverse events (TRAEs) were observed in 96.7% of the patients; diarrhea was the most common TRAE leading to dose reduction in 8.3% of patients (Table). None of the patients discontinued sevabertinib treatment because of diarrhea, and there were no cases of interstitial lung disease. The investigator-assessed objective response rates were 59.3% (95% confidence interval [CI], 47.8-70.1) in cohort D and 59% (95% CI, 42.1-74.4) in cohort F. The disease control rates (confirmed response or stable disease for ≥12 weeks) were 84% (95% CI, 74.1-91.2) in cohort D and 84.6% (95% CI, 69.5-94.1) in cohort F. One patient in cohort D achieved a complete response.
CONCLUSION: Sevabertinib demonstrated manageable safety in both cohorts, which is consistent with previous reports. Diarrhea was the most common TRAE, but it was manageable and did not lead to treatment discontinuation. Similar response rates were observed in patients with advanced HER2 mutation–positive NSCLC who received pretreatment but were naïve to HER2-targeted therapy and in those who received treatment in the first-line setting.
- Siegel F, Karsli-Uzunbas G, Kotynkova K, et al. Preclinical activity of BAY 2927088 in HER2 mutant non-small cell lung cancer. Cancer Res. 2023;83(7 suppl):Abstract 4035.