Immune checkpoint inhibitors are drugs that use the immune system to induce antitumor response. Currently, several checkpoint inhibitors are approved by the US Food and Drug Administration, including cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) inhibitors and programmed cell death 1 (PD-1) or PD ligand 1 (PD-L1) inhibitors. These agents enhance T-cell function against cancer cells by inhibiting mechanisms in the immune system that downregulate T-cell activity.1
Because immune checkpoint inhibitors affect the immune system, they can cause immune-related adverse events. These adverse events can affect most human organ systems and may occur any time after starting immune checkpoint inhibitor therapy.2 The incidence of immune-related adverse events varies across therapies and clinical trials. One systematic review of 35 randomized controlled clinical trials reported the incidence of all-grade immune-related adverse events to range from 74% to 90%, depending on the checkpoint inhibitor class.3
The real-world data outside of clinical trials on the incidence rates of immune-related adverse events are still emerging. There is a lack of data regarding immune-related adverse events in the US veteran population.
The goals of this study were to evaluate the characteristics of immune-related adverse events in a real-world veteran population to provide practical insight about the safety of increasingly used checkpoint inhibitor therapy, and to identify the factors that may predict the occurrence of these adverse events.
Methods
This retrospective study was based on chart review of US veterans who received at least 1 dose of an immune checkpoint inhibitor therapy between January 1, 2014, and October 31, 2019, at the Veterans Affairs (VA) Long Beach Healthcare System in Long Beach, CA.
A total of 146 patients received at least 1 dose of a checkpoint inhibitor during the study period. Of these, 6 patients were excluded from the study, because they had no documentation of clinical monitoring (eg, chart progress notes, laboratory values) after receiving a checkpoint inhibitor therapy. Therefore, a total of 140 patients were included in the study. The patients were divided into 2 groups: the immune-related adverse events group included patients who had at least 1 immune-related adverse event, and the no immune-related adverse events group included patients who did not have any such adverse event. The data on the checkpoint inhibitor therapies received by patients in the study were obtained from the VistA Chemotherapy Manager, a computerized physician order entry system for anticancer treatments used in the study institution.
The checkpoint inhibitor agents included in the study were CTLA-4 inhibitors, PD-1 inhibitors, PD-L1 inhibitors, and combination treatment with a CTLA-4 inhibitor (ipilimumab) and a PD-1 inhibitor (nivolumab). If patients switched between checkpoint inhibitors within the study period, only their first checkpoint inhibitor was included in the study, and subsequent therapies were not accounted for.
We conducted the chart reviews using the Computerized Patient Record System of VA Long Beach Healthcare System to assess if immune-related adverse events occurred after receiving 1 or more doses of a checkpoint inhibitor. The study focused on 5 specific immune-related adverse events, based on the National Comprehensive Cancer Network’s (NCCN) guidelines for the management of immunotherapy-related toxicities, version 1.2020, including diarrhea/colitis, kidney injury, pneumonitis, thyroid disorder, and transaminitis.4
These 5 adverse events are frequently reported in the literature5-7; it was possible to analyze these adverse events retrospectively through chart review, laboratory values, and imaging results. Diarrhea/colitis was captured based on physician documentation. Kidney injury was assessed using serum creatinine laboratory values. Pneumonitis was identified based on physician documentation and computerized tomography scans. Thyroid disorder was evaluated using laboratory values of thyroid-stimulating hormone and free T4, as well as physician documentation of symptoms. Transaminitis was determined based on laboratory values of aspartate aminotransferase, alanine aminotransferase, and total bilirubin. These 5 adverse events were graded according to definitions in the NCCN guidelines for the management of immunotherapy-related toxicities.4
In addition, we conducted a chart review for data on every patient’s baseline characteristics, including age, sex, race, and cancer type. The Eastern Cooperative Oncology Group (ECOG) performance status was not assessed by individual patients because of the retrospective nature of this study and the absence of consistent documentation.
However, the VA National Criteria for Use guidelines for checkpoint inhibitor initiation explicitly allow use only for ECOG performance status score of 0 or 1 for all immunotherapy agents, with some exceptions for ECOG performance status score of 0 to 2 for nivolumab and pembrolizumab. Therefore, patients had a qualifying ECOG performance status score to meet the VA guidelines before initiating checkpoint inhibitor therapy.
This study was approved by the VA Long Beach Healthcare System Research Office in accordance with VA Administration Program Guide 1200.21 as a nonresearch Veterans Health Administration operations activity.
The primary outcome variable was the incidence of immune-related adverse events in veterans receiving checkpoint inhibitor therapy at the VA Long Beach Healthcare System. The secondary outcome variables were the time to immune-related adverse event onset after starting checkpoint inhibitor therapy and the predictors of adverse events occurrence.
Statistical Analysis
The categorical variables (sex, race, cancer type, and checkpoint inhibitor received) were compared between the 2 groups using Fisher’s exact tests. The continuous variables (age and checkpoint inhibitor doses received) were not normally distributed as indicated by Shapiro-Wilk tests; therefore, nonparametric statistics (ie, Mann-Whitney U test) were used to compare these variables between the groups.
Logistic regression analysis was used to evaluate whether a patient’s age, sex, race, cancer type, checkpoint inhibitor received, and checkpoint inhibitor doses received predicted the occurrence of immune-related adverse events. All statistical analysis was conducted using IBM SPSS Statistics 26 software (IBM Corporation; Armonk, NY). P value of <.05 was considered statistically significant.
Results
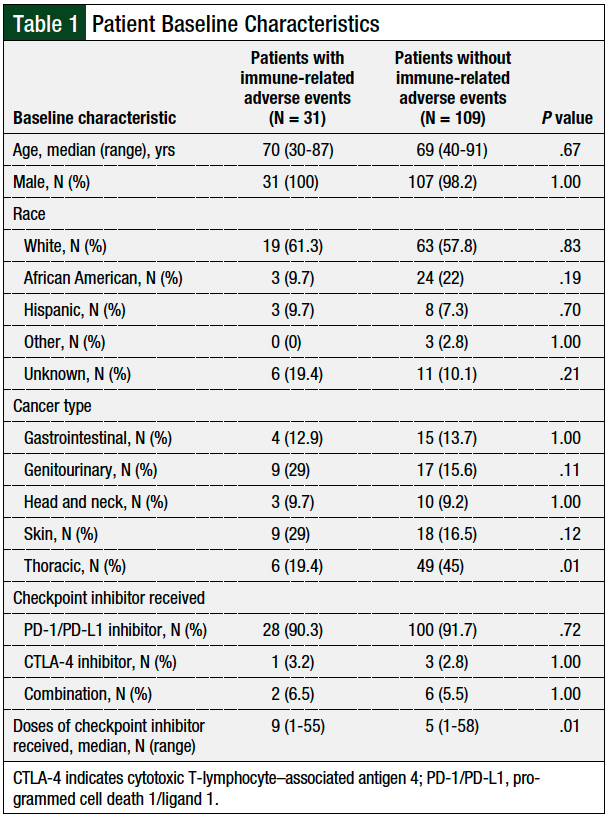
Table 1 summarizes the patients’ baseline characteristics. There were 31 patients who had at least 1 immune-related adverse event (immune-related adverse events group), whereas the remaining 109 patients had no immune-related adverse events (no immune-related adverse events group). No significant difference was detected in demographic characteristics (ie, age, sex, race) between patients in the 2 groups. No significant difference was detected between the groups in the prevalence of cancer types, except for thoracic cancer (19.4% in the immune-related adverse events group vs 45% in the no immune-related adverse events group; P = .01).
In addition, there was no significant difference between the groups for the type of immune checkpoint inhibitor therapy received. In both groups, a large majority of patients received a PD-1 or PD-L1 inhibitor, and only a few patients received a CTLA-4 inhibitor or immune checkpoint inhibitor combination therapy. Patients with immune-related adverse events received a significantly greater median number of immune checkpoint inhibitor doses than the patients without immune-related adverse events.
Primary Outcome
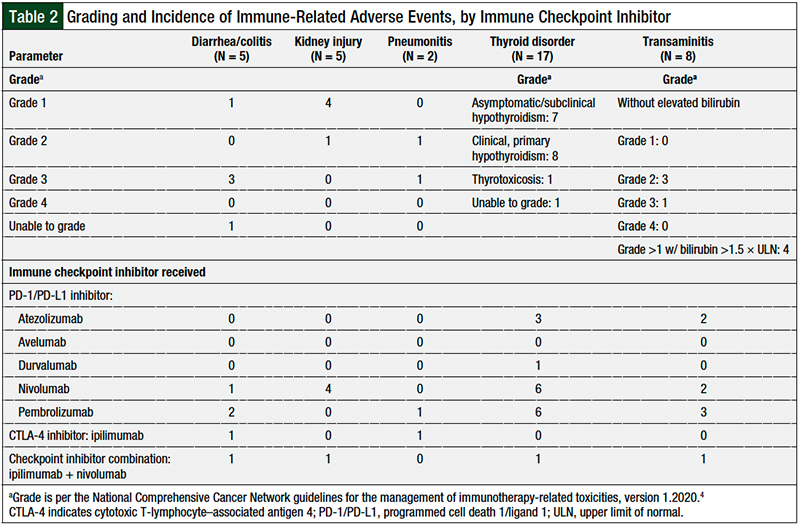
Of 140 patients, 31 (22.1%) had at least 1 immune-related adverse event. Of these 31 patients, 26 patients had 1 immune-related adverse event, 4 patients had 2 immune-related adverse events, and 1 patient had 3 immune-related adverse events, for a total of 37 immune-related adverse events. There were 5 incidences of diarrhea/colitis, 5 of kidney injury, 2 of pneumonitis, 17 of thyroid disorder, and 8 cases of transaminitis.
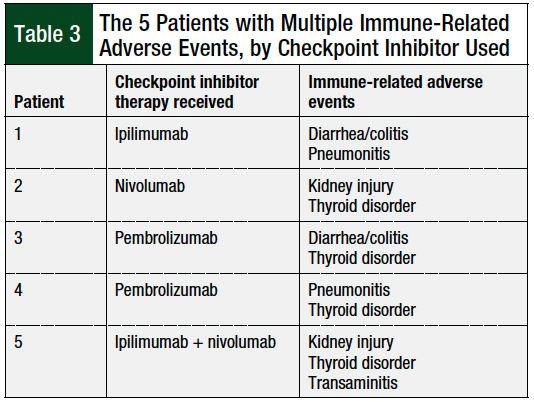
Table 2 describes the grading of the 37 cases of immune-related adverse events and details their incidence per the specific checkpoint inhibitor used. The severity of adverse events ranged from grade 1 to 3, with no incidences of grade 4 or death attributed to immune-related adverse events. Table 3 lists the 5 patients with multiple adverse events and their checkpoint inhibitor regimen and the specific immune-related adverse events they had.
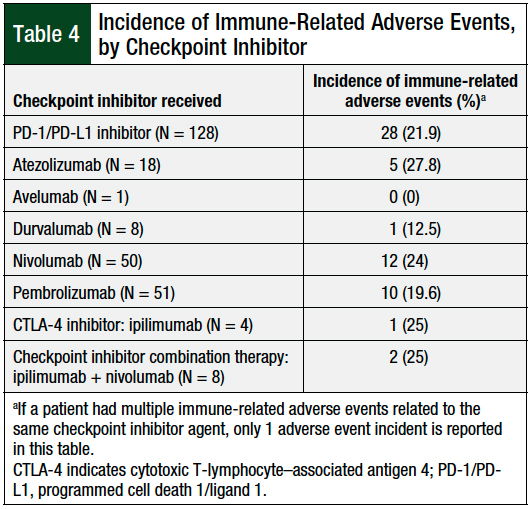
Table 4 describes the overall incidence of immune-related adverse events by the type of checkpoint inhibitor. Of the 128 patients who received a PD-1 or PD-L1 inhibitor, 28 (21.9%) had an immune-related adverse event; of these, 3 patients had 2 immune-related adverse events. Of the 4 patients who received a CTLA-4 inhibitor, 1 (25%) patient had 2 immune-related adverse events. Of the 8 patients who received combination therapy with a PD-1 inhibitor and a CTLA-4 inhibitor, 2 (25%) had an immune-related adverse event, with 1 of these patients having 3 immune-related adverse events.
The incidence of overall immune-related adverse events in the study was compared with the data reported in 2 previously published studies (Table 5).3,8
Secondary Outcomes
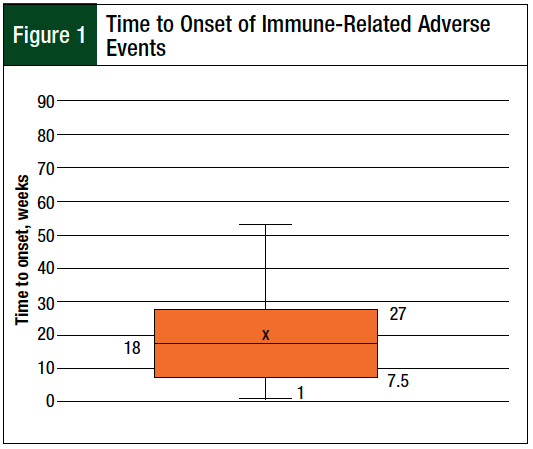
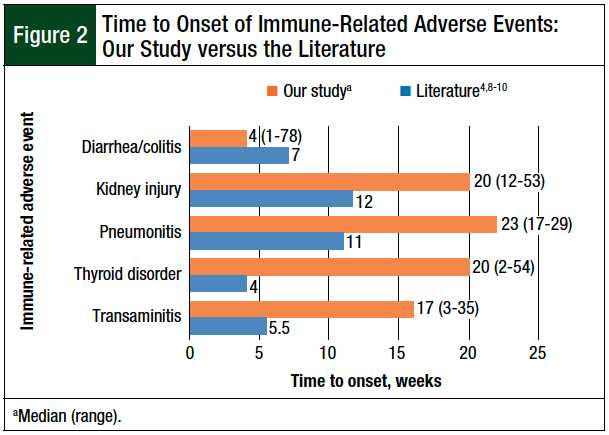
For the 37 immune-related adverse events, the median time to adverse events onset was 18 weeks, and the onset time ranged widely, from 1 week to 78 weeks (Figure 1). We also compared the immune-related adverse event onset times in our study with data published in the literature (Figure 2).4,8-10 For all immune-related adverse events, except diarrhea/colitis, the study patients had a longer time to adverse event onset compared with the data in the literature.4,8-10
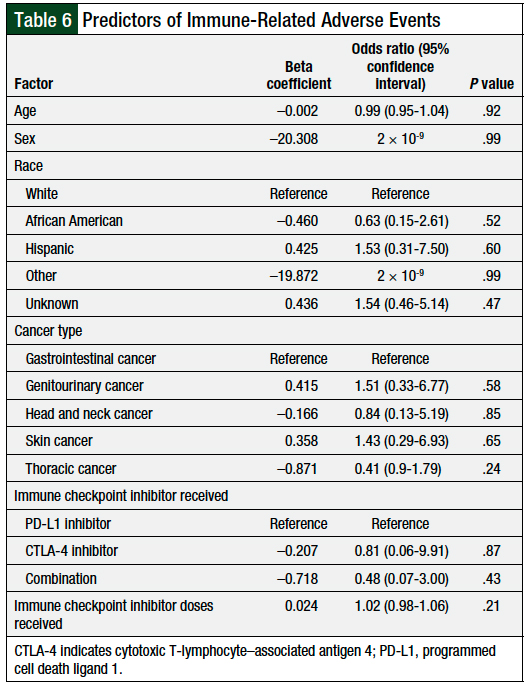
The logistic regression model for this study included predictor variables such as age, sex, race, cancer type, immune checkpoint inhibitor therapy received, and immune checkpoint inhibitor doses received. The outcomes from the logistic regression analysis indicated that the model was not a significant predictor (P = .32) for the occurrence of immune-related adverse events (Table 6).
Discussion
Of the 5 immune-related adverse events evaluated in our study, the incidence rates are largely consistent with the literature.3,8 Of the 37 immune-related adverse events in the study, the majority were related to thyroid disorder (N = 17), whereas pneumonitis had the lowest frequency (N = 2). We found no incidences of life-threatening (grade 4) adverse events.
When comparing the incidence of immune-related adverse events among immune checkpoint inhibitors, previous data suggest that treatment with CTLA-4 inhibitors is generally associated with more frequent and more severe adverse events than PD-1/PD-L1 inhibitors.6,11 In terms of specific immune-related adverse events, CTLA-4 inhibitors are often associated with gastrointestinal adverse events, whereas PD-1/PD-L1 inhibitors are more frequently associated with thyroid-related adverse events and pneumonitis.3
Within the PD-1/PD-L1 inhibitor classes, an increased rate of any-grade immune-related adverse events has been observed with PD-1 inhibitors compared with PD-L1 inhibitors.11 In addition, Medina and colleagues found higher frequency and severity of immune-related adverse events associated with the combination of checkpoint inhibitors compared with single-agent checkpoint inhibitor therapy.11
Given the smaller sample sizes in our study of patients who received checkpoint inhibitors, an accurate comparison of the incidence of immune-related adverse events among the different checkpoint inhibitors is limited. As a percentage, we found a higher incidence of adverse events in patients who received a CTLA-4 inhibitor alone or a combination of 2 checkpoint inhibitors compared with patients who received a PD-1/PD-L1 inhibitor. Patients who received a PD-1 inhibitor generally had a greater percentage of adverse events than those who received a PD-L1 inhibitor.
In terms of specific immune-related adverse events, the incidence of diarrhea/colitis and pneumonitis was not associated with a particular checkpoint inhibitor class in contrast to data in the literature.3 Conversely, the incidence of thyroid disorder was associated more with the PD-1/PD-L1 inhibitors than with CTLA-4 inhibitors, which is consistent with data presented in a systematic review of the literature.3
Although the previous data report higher immune-related adverse event severity associated with CTLA-4 inhibitors and combination therapy compared with PD-1/PD-L1 inhibitors,6,11 this was not consistently seen in our study. Notably, however, despite the small sample sizes in our study, 25% of patients who received a CTLA-4 inhibitor and 25% of patients who received combination therapy in our study had multiple immune-related adverse events; in contrast, a lower percentage (21.9%) of patients who received PD-1/PD-L1 inhibitors had multiple immune-related adverse events. This finding related to combination therapy concurs with the study by Shankar and colleagues, which showed that patients who received combination therapy with a CTLA-4 inhibitor and PD-1/PD-L1 inhibitor had an increased risk for multisystem immune-related adverse events.12
Using logistic regression analysis, we also sought to identify patient risk factors that predicted the occurrence of immune-related adverse events. The independent variables of age, sex, race, cancer type, and type and dose of checkpoint inhibitor received did not significantly predict the occurrence of immune-related adverse events.
Other retrospective studies that examined the risk factors for immune-related adverse events have had variable findings.13,14 Owen and colleagues conducted a retrospective study of 91 patients with non–small-cell lung cancer who received a PD-1/PD-L1 inhibitor,13 and Eun and colleagues conducted a retrospective study of 391 patients who received the PD-1 inhibitor pembrolizumab.14 Both studies showed that the incidence of immune-related adverse events was not significantly affected by age, which agrees with our findings. Both studies also noted that patients who had immune-related adverse events received checkpoint inhibitors longer than those who did not have such adverse events.13,14
In contrast, although our study showed that patients who had immune-related adverse events received a significantly higher median number of checkpoint inhibitor doses compared with patients without such adverse events, the number of checkpoint inhibitor doses received did not significantly predict the occurrence of immune-related adverse events.
Our finding is consistent with a study by Sukari and colleagues of 182 patients who received a single-agent PD-1 inhibitor, which showed that the duration of checkpoint inhibitor treatment was not significantly associated with the overall number of grade ≥2 immune-related adverse events.7
As for differences in the incidence of immune-related adverse events between the disease states, Eun and colleagues identified a higher risk for adverse events in patients with melanoma than in patients with lung cancer.14 This outcome differs from our finding, which showed that the cancer type did not predict the occurrence of immune-related adverse events (Table 6), although we found a significant difference in thoracic cancer prevalence between 2 groups of patients with immune-related adverse events and those without adverse events.
In addition, Owen and colleagues and Cho and colleagues observed that ECOG performance status was not associated with immune-related adverse events.13,15
Although we did not specifically evaluate patients for ECOG performance status, patients were generally similar in performance status to meet the VA criteria to receive checkpoint inhibitors; therefore, an association with the occurrence of immune-related adverse events would not be expected. Overall, in contrast to other retrospective studies,7,12-15 our study was more heterogeneous, with different disease states, and included a variety of checkpoint inhibitors, making a direct comparison with other studies more limited.
Our patients generally had a longer time to the onset of immune-related adverse events than patients in other studies.4,8-10 The wide range of onset time of immune-related adverse events from 1 to 78 weeks in our study highlights the large patient variability and inconsistent time course of immune-related adverse events.
Currently, there is limited information on what factors may influence the onset time of immune-related adverse events. A study by Naidoo and colleagues showed that patients who received checkpoint inhibitor combination therapy tended to have a shorter onset time to adverse events than those who received checkpoint inhibitor monotherapy.9
In our study, 2 patients who received checkpoint inhibitor combination therapy had immune-related adverse events. One of these patients had a longer onset time for thyroid disorder compared with the median, whereas the other patient had an onset time for diarrhea/colitis that was consistent with the median. However, because of the small sample size of patients receiving checkpoint inhibitor combination therapy, these findings are inconclusive.
Limitations
This study has several limitations. This study is retrospective in nature and only details a single-center experience. Because immune-related adverse events were identified through chart review, the evaluation might have been limited by inconsistent documentation of chart notes, such as the underreporting of patient symptoms.
The small sample size in our study is a major limitation. The small number of patients who received a CTLA-4 inhibitor, a PD-1/PD-L1 inhibitor, or a combination of immune checkpoint inhibitors limits the comparison between our findings and the findings in other studies related to these checkpoint inhibitor classes.
Finally, the heterogeneity of the different disease states and checkpoint inhibitors included in our study hampers our ability to compare our results with other studies that focus on a specific disease state or on a specific checkpoint inhibitor agent. Our data may only be generalizable to populations similar to those included in our study.
Conclusion
The incidence of immune-related adverse events in the veteran population at our institution is generally consistent with the incidence reported in the literature. The longer time to onset of immune-related adverse events in our study underscores the importance of ongoing monitoring of patients during checkpoint inhibitor therapy, because such adverse events can occur at any time, and we found no patient factors that can help to predict their occurrence.
Author Disclosure Statement
Dr Tran, Dr Wu, Dr Sevak, and Dr Gupta have no conflicts of interest to report.
References
- Lee L, Gupta M, Sahasranaman S. Immune checkpoint inhibitors: an introduction to the next-generation cancer immunotherapy. J Clin Pharmacol. 2016;56:157-169.
- Liu YH, Zang XY, Wang JC, et al. Diagnosis and management of immune related adverse events (irAEs) in cancer immunotherapy. Biomed Pharmacother. 2019;120:109437.
- Arnaud-Coffin P, Maillet D, Gan HK, et al. A systematic review of adverse events in randomized trials assessing immune checkpoint inhibitors. Int J Cancer. 2019;145:639-648.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Management of Immunotherapy-Related Toxicities. Version 1.2020. December 16, 2019. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed February 9, 2020.
- Xu C, Chen YP, Du XJ, et al. Comparative safety of immune checkpoint inhibitors in cancer: systematic review and network meta-analysis. BMJ. 2018;363:k4226.
- Marin-Acevedo JA, Chirila RM, Dronca RS. Immune checkpoint inhibitor toxicities. Mayo Clin Proc. 2019;94:1321-1329.
- Sukari A, Nagasaka M, Alhasan R, et al. Cancer site and adverse events induced by immune checkpoint inhibitors: a retrospective analysis of real-life experience at a single institution. Anticancer Res. 2019;39:781-790.
- Cortazar FB, Marrone KA, Troxell ML, et al. Clinicopathological features of acute kidney injury associated with immune checkpoint inhibitors. Kidney Int. 2016;90:638-647.
- Naidoo J, Wang X, Woo KM, et al. Pneumonitis in patients treated with anti–programmed death-1/programmed death ligand 1 therapy. J Clin Oncol. 2017;35:709-717. Erratum in: J Clin Oncol. 2017;35:2590.
- Morganstein DL, Lai Z, Spain L, et al. Thyroid abnormalities following the use of cytotoxic T-lymphocyte antigen-4 and programmed death receptor protein-1 inhibitors in the treatment of melanoma. Clin Endocrinol. 2017;86:614-620.
- Medina P, Jeffers KD, Trinh VA, Harvey RD. The role of pharmacists in managing adverse events related to immune checkpoint inhibitor therapy. J Pharm Pract. 2020;33:338-349.
- Shankar B, Zhang J, Naqash AR, et al. Multisystem immune-related adverse events associated with immune checkpoint inhibitors for treatment of non-small cell lung cancer. JAMA Oncol. 2020;6:1952-1956.
- Owen DH, Wei L, Bertino EM, et al. Incidence, risk factors, and effect on survival of immune-related adverse events in patients with non–small-cell lung cancer. Clin Lung Cancer. 2018;19:e893-e900.
- Eun Y, Kim IY, Sun JM, et al. Risk factors for immune-related adverse events associated with anti-PD-1 pembrolizumab. Sci Rep. 2019;9:14039.
- Cho JY, Kim J, Lee JS, et al. Characteristics, incidence, and risk factors of immune checkpoint inhibitor-related pneumonitis in patients with non-small cell lung cancer. Lung Cancer. 2018;125:150-156.