Symptom Overview
Myelodysplastic syndromes (MDS) are a heterogenous group of acquired bone marrow disorders, with a median age of 70 years at diagnosis. The treatment options are often limited at this age, because of the intensity of therapy.1 Through clonal selection, MDS may progress to acute myeloid leukemia.1
The most common objective sequalae of MDS is anemia, which occurs in approximately 80% to 85% of patients.2 Approximately 30% to 45% of patients with MDS have thrombocytopenia, and approximately 40% of patients have neutropenia at diagnosis.2
Red blood cell transfusions are a cornerstone of supportive care for MDS-related anemia.1,3,4 However, such transfusions also lead to iron overload, which negatively affects the function of the liver, heart, and the endocrine system.5 Furthermore, observational studies have shown that iron overload can lead to poor clinical outcomes and can shorten the survival of patients with low-risk or intermediate-1-risk MDS.6,7
Symptomatically, patients at all levels of disease risk, including those with mild cytopenias, report that MDS negatively affects their quality of life, with fatigue being a particularly troublesome symptom.4 Often, fatigue is caused by symptomatic anemia, and treating anemia with transfusions does not completely restore a patient’s quality of life, as assessed by the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30.4
Etiology
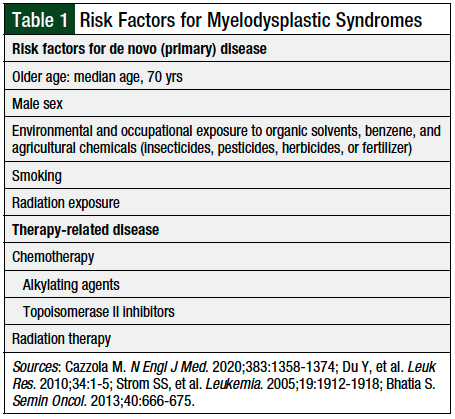
The risk factors for primary MDS are not well-established.8,9 However, exposure to chemotherapy, such as alkylating agents or topoisomerase II inhibitors, and radiation therapy increases the risk for therapy-related MDS.10 Table 1 lists the known risk factors for primary and therapy-related MDS.
The diagnosis of MDS and risk stratification are made according to different prognostic scoring systems, including the World Health Organization Prognostic Scoring System, International Prognostic Scoring System (IPSS), and the revised IPSS (IPSS-R).1,3
These risk-stratification systems were created to address the diverse nature of MDS, and each system has its own advantages and disadvantages.3 Each prognostic scoring system weighs cytogenetics in a different manner. MDS cytogenetics are predominantly expressed as genetic gains and losses versus genetic translocations.11
The most frequent among these are chromosome 7 deletion (del 7q) and del 5q, duplication 1q, del 20q, del 11q, del 12p/t(12p), del 17p/iso(17q), del 18q, +21q gains, del 13q, and +der(1;7)(q10;p10).11 Overall, the treatment choices for patients with MDS are directed by the risk categories from these different prognostic scoring systems.1,3
Treatment Options
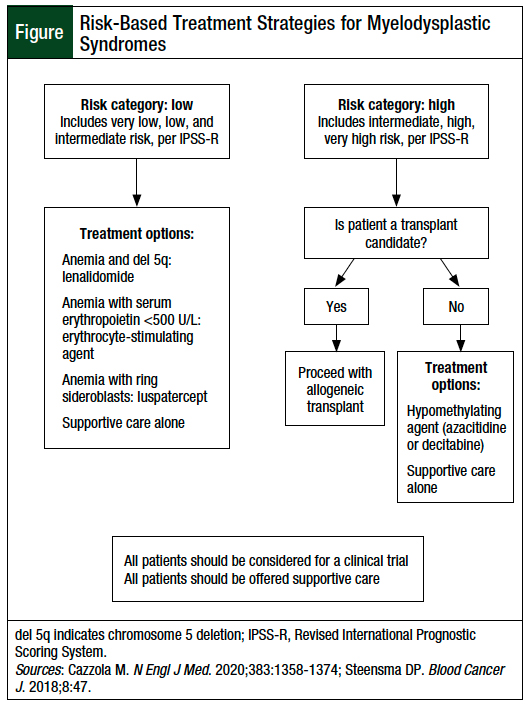
Currently, the only curative therapy for MDS is an allogeneic stem-cell transplant.1,3 However, because of the patients’ older age at MDS presentation, as well as the increased incidence of patients’ comorbidities, the treatment may focus on symptom management or low-intensity chemotherapy.1,3 The Figure shows the overall treatment of MDS, based on the patient’s risk level. The current treatment options for anemia associated with lower-risk MDS discussed include lenalidomide; erythropoiesis-stimulating agents (ESAs), with or without granulocyte colony-stimulating factors (G-CSFs); luspatercept; transfusion; and iron chelation for patients with iron overload.
Lenalidomide
Lenalidomide is a first-line treatment option for patients with MDS associated with symptomatic anemia and chromosomal del 5q, with or without additional cytogenetic abnormalities.1,3,12,13 Lenalidomide can also be used in patients without del 5q; however, the rate of response is diminished, the adverse events increase, and no survival benefit is seen with this treatment.1,3,12
The phase 2 MDS-003 clinical trial examined the efficacy of lenalidomide 10 mg daily, or 10 mg daily for 21 days and 7 days off.12 The study participants were transfusion-dependent and had del 5q that was isolated or was accompanied by additional cytogenetic abnormalities.12 In all, 67% of the patients became transfusion-independent while receiving lenalidomide during this study.12 The median time to response was 4.6 weeks, and the duration of transfusion independence with lenalidomide use has been between 2 and 3 years, with assessment ongoing at last report.1,12
In that study, patients did not have increased adverse events with continuous dosing versus intermittent dosing, which established the current dosing of lenalidomide 10 mg orally daily for MDS.12,13 The primary adverse events reported with lenalidomide are moderate-to-severe neutropenia (55%) and thrombocytopenia (44%).12,13 Because of the risk for venous and arterial thromboembolism, patients should be prescribed antithrombotic prophylaxis to mitigate this risk.12,13 It is important to note that lenalidomide is only available through a Risk Evaluation and Mitigation Strategy program because of the risk for embryo-fetal toxicity.13
Erythropoiesis-Stimulating Agents
ESAs can be considered for the treatment of patients with lower-risk MDS and symptomatic anemia whose serum erythropoietin level is <500 U/L.1,3 Serum erythropoietin is strongly correlated with response to ESAs.3 Data suggest that patients with a baseline serum erythropoietin level of <200 U/L are most likely to respond to treatment1,3; those with levels of <100 U/L have a >70% chance of responding to ESAs, whereas levels of >500 U/L correlate to a response rate of <10%.3
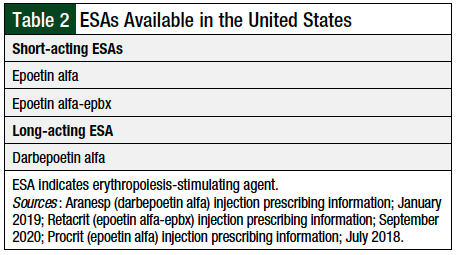
The ESAs currently available in the United States include short-acting epoetin alfa, the biosimilar epoetin alfa-epbx, and darbepoetin (Table 2).14-16 These agents act on the erythropoietin receptor located on red blood cells and help signal the proliferation and differentiation of red blood cell precursors. The goal of treatment with ESAs is to reduce the need for transfusions.17 Although epoetin alfa and darbepoetin transfusion requirements have not been assessed in head-to-head studies, several studies discussed by Park and colleagues show that these drugs reduce the need for transfusion.17
It is important that patients are not iron-deficient before the use of ESAs so that the ESA doses can be kept as low as possible.14-16 Supplemental iron should be administered when the serum ferritin level is <100 mcg/L, or when the transferrin saturation is <20%. Treatment with ESAs should not begin unless the patient’s hemoglobin level is <10 g/dL. In addition, the lowest possible dose of an ESA should be used to reduce red blood cell transfusions and to minimize adverse events.14-16
The standard dosage of epoetin alfa is 40,000 to 60,000 units, once weekly subcutaneously, and the dosage of darbepoetin alfa is 150 mcg to 300 mcg every other week, or 500 mcg every 2 to 3 weeks subcutaneously.15,16 Darbepoetin alfa may be more convenient than epoetin alfa for patients, because of its decreased frequency of administration.
The expected time to response to ESA treatment is 3 months.17 Therefore, if after 3 months of treatment no significant increase in hemoglobin levels or a significant decrease in transfusion requirements is observed, then G-CSFs can be added to ESA treatment potentially to increase the response to therapy.17 The dose of G-CSF is 300 mcg subcutaneously 2 times weekly,17 (although in real practice we sometimes titrate to 3 times weekly). The benefit of adding G-CSFs to lower doses of ESAs is an improvement in the response rate, ranging from 25% to 47%.17
As a class, ESAs have similar side-effect profiles.14-16 The serious side effects of ESAs include increased mortality, myocardial infarction, stroke, and thromboembolism.14-16 Using ESAs to target a hemoglobin level of >11 g/dL can increase the risk for serious cardiovascular-related adverse events. In addition, patients’ blood pressure should be monitored and controlled before the initiation of ESA therapy. Patients should be advised about the importance of compliance with antihypertensive therapy and incorporating lifestyle modifications to maintain blood pressure control, if applicable.14-16
Luspatercept
The recombinant fusion protein luspatercept-aamt binds to select endogenous transforming growth factor beta superfamily ligands, thereby reducing SMAD2 and SMAD3 signaling.18-20 Luspatercept promotes erythroid maturation via late-stage erythroblast differentiation, which can result in increases in hemoglobin and red blood cell concentrations.18-20
Luspatercept has been the only drug approved for low-risk MDS since lenalidomide was approved in 2005, and represents a novel treatment option to gain transfusion independence in very-low-risk, low-risk, or intermediate-risk MDS (per IPSS-R) with ring sideroblasts.13,19,20
The approval of luspatercept was based on the results of the MEDALIST trial, which showed that transfusion independence for ≥8 weeks was observed in 38% of patients during the first 24 weeks of treatment.19 Luspatercept is approved by the US Food and Drug Administration for the treatment of anemia associated with very-low-risk to intermediate-risk MDS with ring sideroblasts or with myelodysplastic or myeloproliferative neoplasm with ring sideroblasts and thrombocytosis in adults who have not responded to treatment with an ESA and who require ≥2 red blood cell units over 8 weeks.20
Luspatercept treatment is started at 1 mg/kg every 3 weeks and is administered as a subcutaneous injection.20 If the patient is still receiving red blood cell transfusions after at least 2 consecutive doses (ie, 6 weeks) subsequent to the starting dose, luspatercept dosing should be increased to 1.33 mg/kg. If the patient is still receiving transfusions after at least 2 consecutive 1.33-mg/kg doses of luspatercept, the patient can be dose-escalated to the highest dose of 1.75 mg/kg. If the patient does not have a reduction in transfusion burden after at least 3 consecutive doses of luspatercept at the maximum dose, then treatment with luspatercept should be discontinued.20
Of the patients who were transfusion-independent for at least 8 weeks in the MEDALIST trial, 90% responded at the starting dose of 1 mg/kg of luspatercept, and 7% had their first response after dose increases.19 Treatment with luspatercept is generally well-tolerated; the most common side effects include diarrhea (22%), asthenia (20%), and back pain (19%).19 These side effects may decrease with continued use of luspatercept and are not associated with dose increases.19
Transfusion and Iron Chelation
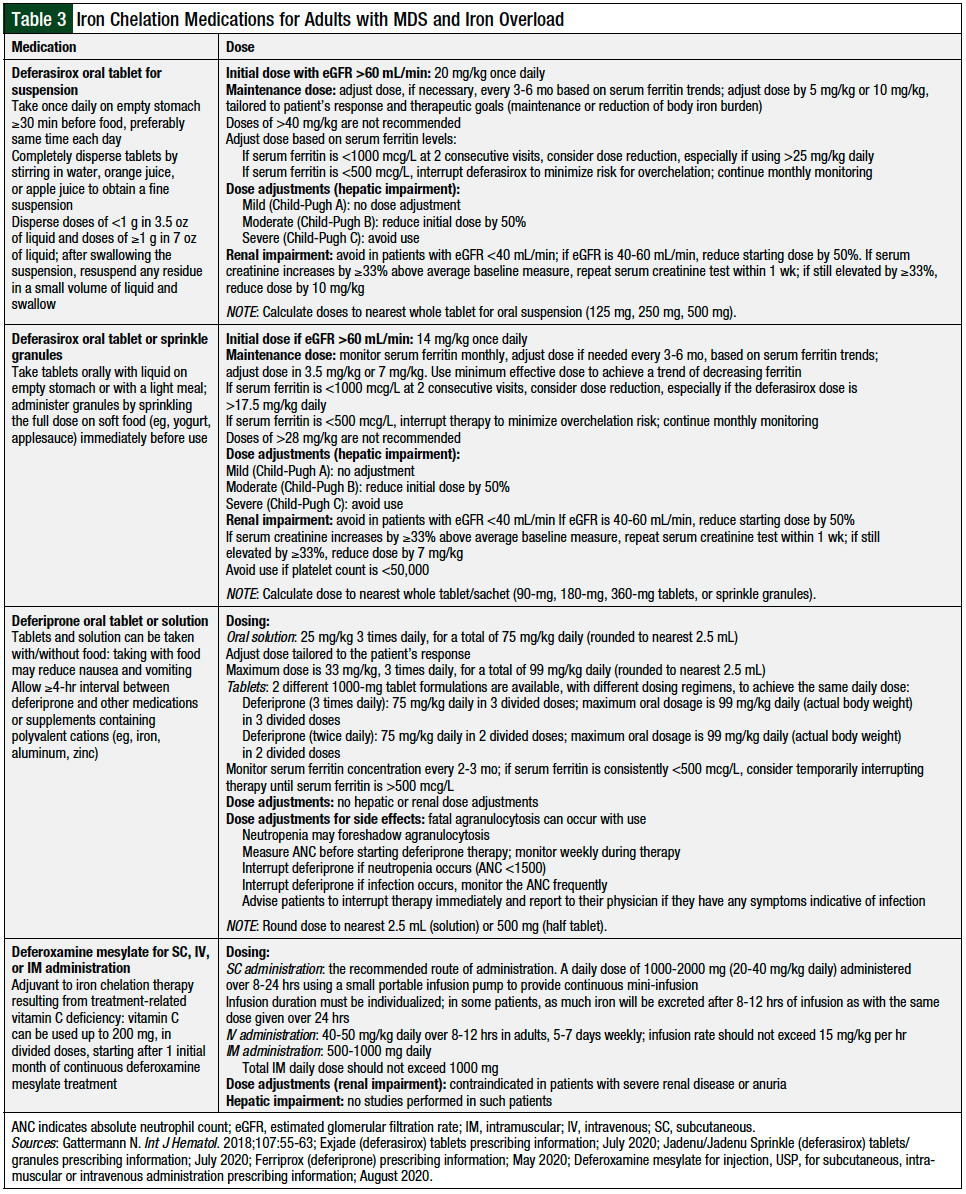
Red blood cell transfusions are recommended for patients with MDS and symptomatic anemia (ie, shortness of breath, weakness, fatigue, chest pain).5,21 Frequent blood cell transfusions can lead to iron overload,21 because 1 unit of blood contains between 200 mg and 250 mg of iron. Significant morbidity and mortality are associated with iron overload.1,5 Table 3 lists the iron chelation medications and monitoring.
The use of iron chelation therapy in patients with iron overload caused by frequent transfusions was recently supported in the TELESTO clinical trial of patients with low-risk or intermediate-1-risk MDS.5 To be included in this study, patients had to have serum ferritin levels of >2247 pmol/L, have received 15 to 75 packed red blood cell units, and were anticipated to be transfused with at least 8 units of red blood cells annually during the study.5
The primary end point was event-free survival with deferasirox versus placebo.5 The median event-free survival was prolonged by almost 1 year with deferasirox. The median overall survival was 5.2 years (confidence interval [CI], 3.9 years-not evaluable upper bound) in the deferasirox group versus 4.1 years (CI, 3.0-4.9 years) in the placebo group, with overlapping 95% CIs (hazard ratio, 0.83 [CI, 0.54-1.28]).5
The most common side effects of deferasirox treatment in the TELESTO study were diarrhea (24.7%), pyrexia (21.8%), and increased serum creatinine (15.9%).5 Supportive care for diarrhea related to deferasirox treatment includes discontinuing laxatives, dosing the medication at night, and the use of antidiarrheal medications.21 Increases in creatinine concentrations is a well-known side effect of deferasirox.5,21 In all, 20% of patients have an initial increase in creatinine levels after starting deferasirox therapy.21 With proper dose adjustment, most patients will not have a continued rise in serum creatinine levels thereafter; however, some older patients with MDS must discontinue deferasirox treatment because of further increases in serum creatinine.21 The use of deferasirox is contraindicated with a creatinine clearance of <40 mL/min/1.73 m2.22,23
Less frequently used iron chelators include deferiprone and deferoxamine. Deferiprone is an oral iron chelator that is available in tablet and oral solution forms, and is dosed either 2 or 3 times daily.24 In a comparative study of deferasirox and deferiprone used for the treatment of iron overload in 113 patients with MDS, iron levels were reduced by 50% in 18 of the 65 (27.7%) patients who received deferasirox, but not in any of the 48 patients who received deferiprone.25
An additional, unique side effect of deferiprone treatment is agranulocytosis, which is the reason why deferiprone historically has rarely been used, particularly in patients with MDS.24,25 One benefit to the use of deferiprone over other iron chelators is that it requires no renal or hepatic dose adjustments, which makes deferiprone an option for patients who are renally or hepatically compromised.24,25
Deferoxamine is an iron chelator that is administered subcutaneously via a small portable infusion pump that provides continuous mini-infusion, intravenously or intramuscularly.26 The use of deferoxamine in patients with MDS who have received transfusions is low because of the way deferoxamine is administered, its side effects, and the concerns about patient compliance.27 Notable warnings for deferoxamine use include ocular and auditory disturbances and increases in serum creatinine, as well as flushing of the skin, urticaria, hypotension, and shock if deferoxamine is given via rapid intravenous injection.26 However, if a patient is unable to take oral medications, deferoxamine is the only iron chelator available for that person.
Conclusion
The management of lower-risk MDS and associated anemia is largely focused on supportive care. Treatment options have recently expanded with the approval of luspatercept, which can reduce transfusions in select MDS morphologies. In addition, the completion of the TELESTO trial has helped to answer questions regarding survival outcomes in patients receiving iron chelation drugs for low-risk and intermediate-1-risk MDS; however, the significant side effects of iron chelators should be considered as well. Despite these advances, there is still much work to be done regarding disease modulation and therapy development, particularly for transplant-ineligible patients with MDS.
Author Disclosure Statement
Dr Rowen has no conflicts of interest to report.
References
- Cazzola M. Myelodysplastic syndromes. N Engl J Med. 2020;383:1358-1374.
- Steensma DP, Bennett JM. The myelodysplastic syndromes: diagnosis and treatment. Mayo Clin Proc. 2006;81:104-130.
- Steensma DP. Myelodysplastic syndromes current treatment algorithm 2018. Blood Cancer J. 2018;8:47.
- Efficace F, Gaidano G, Breccia M, et al. Prevalence, severity and correlates of fatigue in newly diagnosed patients with myelodysplastic syndromes. Br J Haematol. 2015;168:361-370.
- ngelucci E, Li J, Greenberg P, et al; for the TELESTO study investigators. Iron chelation in transfusion-dependent patients with low- to intermediate-1–risk myelodysplastic syndromes: a randomized trial. Ann Intern Med. 2020;172:513-522.
- de Swart L, Smith A, Fenaux P, et al. Transfusion-dependency is the most important prognostic factor for survival in 1000 newly diagnosed MDS patients with low- and intermediate-1 risk MDS in the European LeukemiaNet MDS Registry. Blood. 2011;118:Abstract 2775.
- Malcovati L, Della Porta MG, Strupp C, et al. Impact of the degree of anemia on the outcome of patients with myelodysplastic syndrome and its integration into the WHO classification-based Prognostic Scoring System (WPSS). Haematologica. 2011;96:1433-1440.
- Du Y, Fryzek J, Sekeres MA, Taioli E. Smoking and alcohol intake as risk factors for myelodysplastic syndromes (MDS). Leuk Res. 2010;34:1-5.
- Strom SS, Gu Y, Gruschkus SK, et al. Risk factors of myelodysplastic syndromes: a case–control study. Leukemia. 2005;19:1912-1918.
- Bhatia S. Therapy-related myelodysplasia and acute myeloid leukemia. Semin Oncol. 2013;40:666-675.
- Ogawa S. Genetics of MDS. Blood. 2019;133:1049-1059.
- Talati C, Sallman D, List A. Lenalidomide: myelodysplastic syndromes with del(5q) and beyond. Semin Hematol. 2017;54:159-166.
- Revlimid (lenalidomide) capsules, for oral use [prescribing information]. Celgene Corporation; October 2019.
- Retacrit (epoetin alfa-epbx) injection, for intravenous or subcutaneous use [prescribing information]. Pfizer; September 2020.
- Procrit (epoetin alfa) injection, for intravenous or subcutaneous use [prescribing information]. Janssen; July 2018.
- Aranesp (darbepoetin alfa) injection, for intravenous or subcutaneous use [prescribing information]. Amgen; January 2019.
- Park S, Greenberg P, Yucel A, et al. Clinical effectiveness and safety of erythropoietin-stimulating agents for the treatment of low- and intermediate-1–risk myelodysplastic syndrome: a systematic literature review. Br J Haematol. 2019;184:134-160.
- Platzbecker U, Germing U, Götze KS, et al. Luspatercept for the treatment of anaemia in patients with lower-risk myelodysplastic syndromes (PACE-MDS): a multicentre, open-label phase 2 dose-finding study with long-term extension study. Lancet Oncol. 2017;18:1338-1347. Erratum in: Lancet Oncol. 2017;18:e562.
- Fenaux P, Platzbecker U, Mufti GJ, et al. Luspatercept in patients with lower-risk myelodysplastic syndromes. N Engl J Med. 2020;382:140-151.
- Reblozyl (luspatercept-aamt) for injection, for subcutaneous use [prescribing information]. Celgene Corporation; April 2020.
- Gattermann N. Iron overload in myelodysplastic syndromes (MDS). Int J Hematol. 2018;107:55-63.
- Exjade (deferasirox) tablets, for oral suspension [prescribing information]. Novartis Pharmaceuticals Corporation; July 2020.
- Jadenu/Jadenu Sprinkle (deferasirox) tablets/granules, for oral use [prescribing information]. Novartis Pharmaceuticals Corporation; July 2020.
- Ferriprox (deferiprone) [prescribing information]. Chiesi; May 2020.
- Cermak J, Jonasova A, Vondrakova J, et al. A comparative study of deferasirox and deferiprone in the treatment of iron overload in patients with myelodysplastic syndromes. Leuk Res. 2013;37:1612-1615.
- Deferoxamine mesylate for injection, USP, for subcutaneous, intramuscular or intravenous administration [prescribing information]. Hospira; August 2020.
- Mitchell M, Gore SD, Zeidan AM. Iron chelation therapy in myelodysplastic syndromes: where do we stand? Expert Rev Hematol. 2013;6:397-410.