Oral tyrosine kinase inhibitors (TKIs) have demonstrated efficacy in the treatment of multiple malignant solid tumors, including renal-cell carcinoma, hepatocellular carcinoma, lung cancer, and thyroid cancer.1,2 TKIs inhibit specific molecular targets and signaling cascades within cancer cells that promote excessive tumor growth and proliferation.1 TKIs may be used as monotherapy or in combination with systemic therapy, depending on the specific cancer type and the phase of treatment.
The inclusion of a TKI in anticancer therapy improves outcomes for patients with early-stage cancer, as well as for patients with advanced or metastatic disease, leading to the increased use of this treatment in oncology.2 The adverse-event profile of TKIs varies, depending on the specific class and mechanism of action. TKIs that share the same physiologic and molecular targets often have a similar side-effect profile. For example, TKIs that target the vascular endothelial growth factor pathway often lead to hypertension.3
Although every TKI has specific recommendations for treatment adjustments within prescribing information files, it is a general recommendation in each drug’s prescribing information to stop these agents for all grade 3 or 4 adverse events, with the resumption of therapy at reduced doses if clinically appropriate.
More than 55% of patients with thyroid cancer required dose modifications of their TKI because of treatment intolerability and toxicity.1 The incidence and severity of TKI-related adverse events may depend on factors such as organ function, severity of disease, and concomitant drug or food interactions.1 In addition, the concentration of a TKI may vary from patient to patient, leading to unpredictable tolerability and clinical outcomes.1,3 Notably, TKIs are largely protein-bound, predominantly to albumin and to alpha-1-acid glycoprotein.4 The concentration of unbound, or “free,” drug is more indicative of efficacy and safety than total drug concentration.5 Hypoalbuminemia, which is clinically defined as serum albumin of <3.5 g/dL, is prevalent among patients with cancer.6
The Common Terminology Criteria for Adverse Events defines grade 1 hypoalbuminemia as serum albumin from less than the lower limit of normal to 3 g/dL; grade 2 as from <3 g/dL to 2 g/dL; and grade 3 as <2 g/dL.7 Although the incidence of hypoalbuminemia varies based on the type of cancer, its underlying mechanisms involve inflammatory processes and malnutrition.8
Cancer causes a state of high physiological stress through tumor hypoxia and necrosis, which drives the release of proinflammatory cytokines.9 In response to this stress, hepatocytes increase the production of acute-phase proteins and decrease the production of albumin.10 This inflammatory process, combined with nutritional deficiencies, drives the increased incidence of reduced serum albumin level in patients with cancer.9
The impact of hypoalbuminemia on highly protein-bound medications has been previously identified with chemotherapy, as well as with medications not related to cancer.9,11 The increased risk for encephalopathy with ifosfamide therapy in patients with hypoalbuminemia has been well-established.11
In their retrospective study, Drisaldi and colleagues analyzed the relationship between albumin and drug concentrations of other highly protein-bound medications, such as valproic acid; they evaluated a preestablished equation that provides a predicted value of unbound valproic acid concentrations with a correction for serum albumin compared with measured free drug levels.12 The results showed a significant discordance between the predicted and measured drug levels, with 97% of the predicted values underestimating the measured concentrations.12 These results further emphasize the impact that hypoalbuminemia has on free drug concentrations and the role that this may play in the tolerability of highly protein-bound medications.12
Minimal published data exist on the connection between hypoalbuminemia and unbound TKI drug concentrations. In their pharmacokinetic study, Yoo and colleagues found a correlation between decreased serum albumin and increased serum concentration of the TKI imatinib in patients with gastrointestinal stromal tumors.13 As a result of this link between albumin and serum concentrations of highly protein-bound TKIs established by Yoo and colleagues, low serum albumin may result in an increased incidence of adverse events caused by an increase in unbound drug concentration.13
In their retrospective review, Murdock and colleagues identified a shorter time to TKI therapy discontinuation because of adverse events in patients with hypoalbuminemia (defined as serum albumin <3.5 g/dL) than in patients without hypoalbuminemia.14 Although Murdock and colleagues’ study is the only published study that investigated the connection of low albumin to highly protein-bound TKI tolerability in patients with cancer,14 the collective results of these studies indicate the need for closer monitoring and dose adjustments in patients who present with hypoalbuminemia and are receiving drugs that are highly protein-bound.12-14
Considering that TKIs are used as first-line treatment for many malignant solid tumors, it is important to determine the relationship between low serum albumin and TKI tolerability. The objective of this study was to evaluate the effect of hypoalbuminemia on treatment tolerability and duration in patients who were receiving highly protein-bound TKIs for solid-tumor malignancies.
Methods
This retrospective, single-center study included adults with solid-tumor malignancies who were receiving highly protein-bound TKIs at the Medical University of South Carolina in Charleston. Patients were included if they were aged ≥18 years and received at least 1 highly protein-bound TKI between April 1, 2017, and March 31, 2020. Highly protein-bound TKIs were defined as ≥95% protein-bound, as listed in the pharmacokinetics section of the respective prescribing information of the TKIs included in this study. Patients were excluded from the study if the TKI was initiated for the treatment of hematologic malignancies or for any indication that was not approved by the US Food and Drug Administration.
The patients were divided into 2 groups based on baseline serum albumin: patients with hypoalbuminemia in one group and those without hypoalbuminemia in the other group. Hypoalbuminemia was defined as a baseline serum albumin level of <3.5 g/dL, based on our institutional definitions. The primary end point was all-cause discontinuation of a TKI. The secondary end points included TKI discontinuation because of intolerability (ie, adverse events), time to discontinuation, the incidence of adverse events, time to adverse events, and the incidence of hypoalbuminemia throughout the treatment with a TKI.
All-cause TKI discontinuation was chosen as the primary end point to establish the overall rate of discontinuation in patients with hypoalbuminemia. The primary end point was further delineated into the secondary end points, with the goal of potentially demonstrating a connection between low albumin levels and TKI intolerability in patients with solid tumors.
Data were collected from the electronic medical record for baseline characteristics, including age, sex, Eastern Cooperative Oncology Group performance status, weight, liver disease, proteinuria, type of cancer, the presence of metastases at baseline, baseline albumin at the time of TKI initiation, the oral TKI prescribed, the TKI starting dose, and the lines of therapy before the TKI was received.
Additional data collection for analysis included the incidence of and time to any-grade adverse events; the incidence of and time to TKI dose reductions and interruptions; the date of TKI discontinuation; the reason for dose reductions, interruptions, or discontinuation; and serum albumin levels at the time of each adverse event. In addition, specific reasons for TKI treatment discontinuation were collected, including disease progression, intolerability, adherence, and cost.
Statistical Analysis
This study was approved by the local Institutional Review Board. Continuous variables are reported as a median with interquartile range (IQR), and nominal variables are reported as a count with percentage. The outcomes were compared via Mann–Whitney U tests for continuous data and with chi-square or Fisher’s exact tests for categorical data. Cox regression was used to evaluate the association between hypoalbuminemia and the primary end point (ie, all-cause TKI discontinuation) after adjusting for covariates, including performance status, previous lines of therapy, baseline metastases, and liver disease.
The covariates were identified based on previous data indicating that the differences among these covariates may contribute to hypoalbuminemia and worse disease status and therefore may confound the end point of all-cause treatment discontinuation.14,15 P values of <.05 were considered to represent a significant difference. Statistical analysis was completed via IBM SPSS version 25.0 (IBM Corp; Armonk, NY).
Results
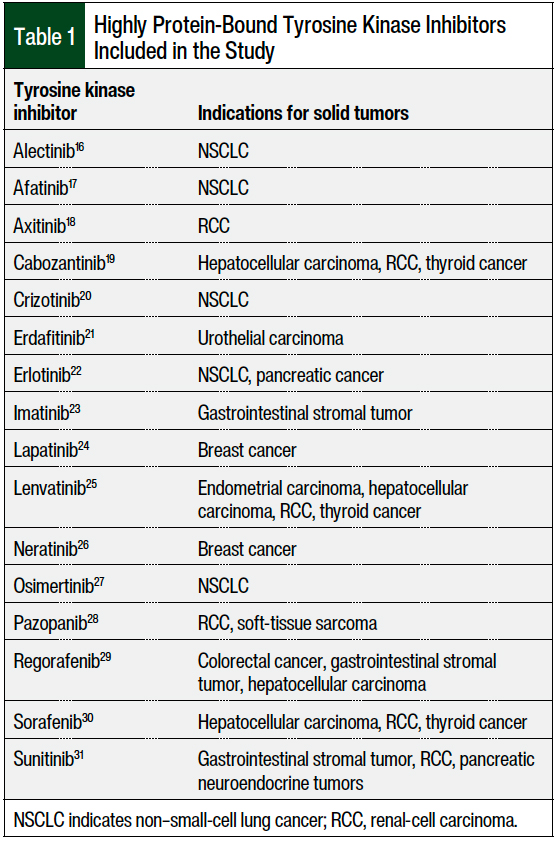
A total of 16 TKIs were included for analysis in the study based on our institution’s formulary and their protein-binding properties being ≥95% (Table 1).16-31 After screening patient charts for exclusion criteria, a total of 220 patients were eligible for analysis, including 101 (45.9%) patients with baseline hypoalbuminemia and 119 (54.1%) patients without hypoalbuminemia at baseline.
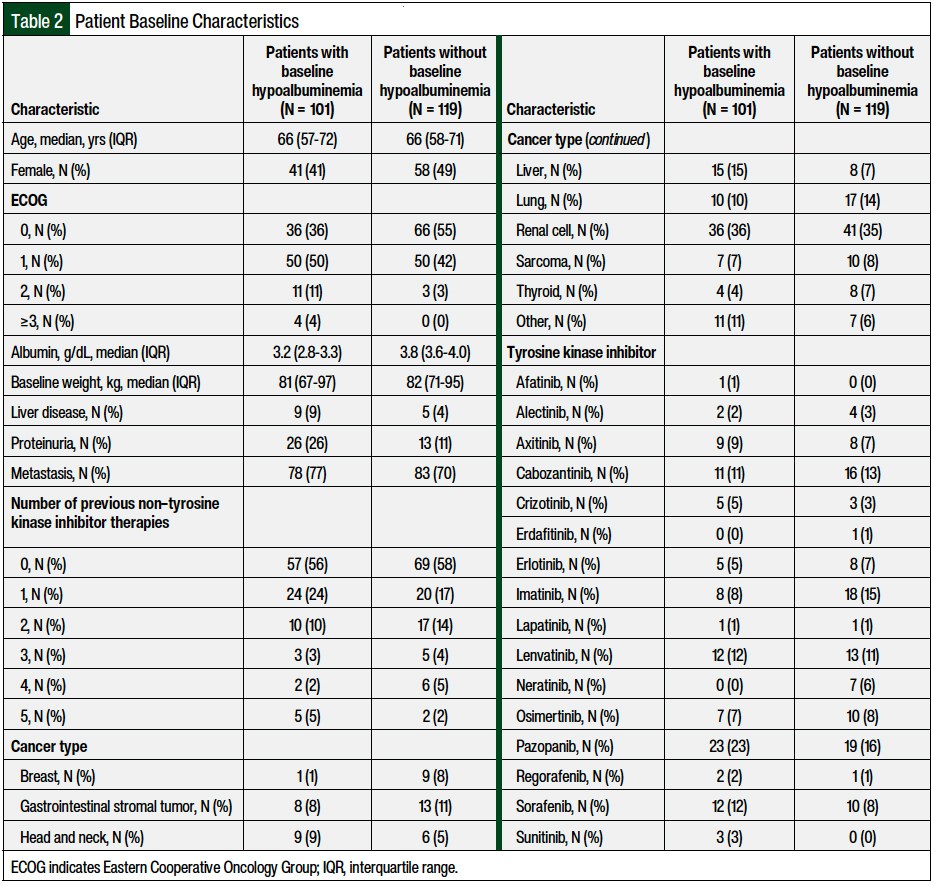
Table 2 shows the patients’ baseline characteristics. The median age of the overall study population was 66 years, and approximately 55% of the patients were male. The 3 most common types of cancer were renal-cell carcinoma (35%), lung cancer (12%), and liver cancer (11%). The median serum albumin for the hypoalbuminemia group was 3.2 g/dL (IQR, 2.8-3.3 g/dL). The patients with hypoalbuminemia had worse performance status than those without the condition (64% vs 45%, respectively), more liver disease (9% vs 4%, respectively), and proteinuria (26% vs 11%, respectively). The use of previous lines of therapy was similar between the 2 groups.
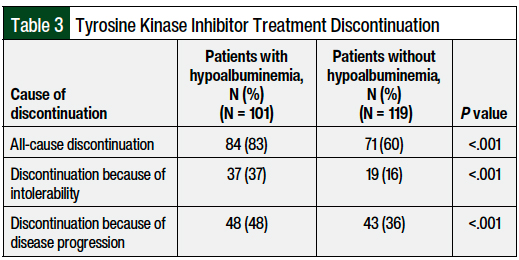
Table 3 outlines the incidence of TKI discontinuation among all the patients. All-cause TKI discontinuation was more common in the group with baseline hypoalbuminemia than in the patients without hypoalbuminemia at baseline (83% vs 60%, respectively; P <.001). After adjusting for covariates, the presence of baseline hypoalbuminemia was associated with a higher incidence of TKI discontinuation than seen in patients without hypoalbuminemia (hazard ratio, 2.68; 95% confidence interval, 1.97-3.76).
TKI discontinuation because of intolerability was significantly higher in patients with hypoalbuminemia (37% vs 16%, respectively; P <.001). None of the patients discontinued TKI treatment because of cost barriers, and only 1 patient discontinued TKI therapy because of adherence issues. Of patients who discontinued TKI treatment because of intolerability, 28 did so after the first adverse event, 23 after the second adverse event, and 5 after the third adverse event.
The median time to all-cause TKI discontinuation was shorter in the patients with hypoalbuminemia than in those without hypoalbuminemia (96 days vs 288 days, respectively; P <.001). There was also a significantly shorter time to TKI discontinuation because of intolerability in patients with hypoalbuminemia at baseline (55 days vs 147 days, respectively; P = .03). Dose adjustments or interruptions because of intolerability was 64% in the patients with hypoalbuminemia and 62% in those without hypoalbuminemia (P = .74).
The proportion of patients who had an adverse event of any grade was similar between the 2 groups, with 81% of patients with hypoalbuminemia and 75% of patients without hypoalbuminemia having an adverse event (P = .26). However, there was a significant difference in the time to adverse events between the groups (25 days vs 31 days, respectively; P = .03).
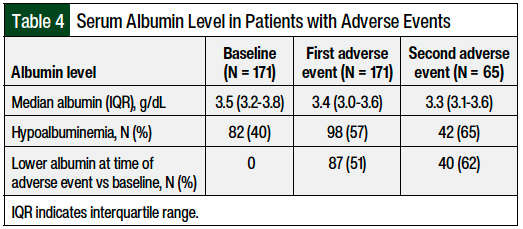
Among the 171 patients who had an adverse event, 40% had baseline hypoalbuminemia and 57% had hypoalbuminemia at the time of the first adverse event (Table 4). Furthermore, among the 65 patients who had a second adverse event, 65% had hypoalbuminemia at the time of the adverse event. The median serum albumin level decreased from 3.5 g/dL at baseline to 3.3 g/dL in patients with more than 1 adverse event.
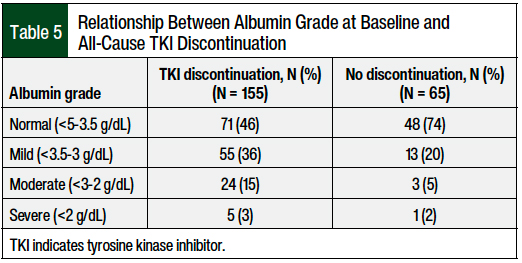
The majority of patients had a lower albumin level at the time of the adverse event than at baseline; although the majority of patients had baseline normal or mild hypoalbuminemia, many patients’ albumin status worsened at the time of the adverse event and TKI discontinuation. As hypoalbuminemia increased in severity, patients were more likely to discontinue TKI therapy (Table 5). Of the patients with baseline moderate-to-severe hypoalbuminemia, 88% discontinued TKI therapy compared with 80% of patients with baseline mild hypoalbuminemia (a nonsignificant difference).
Discussion
The patients with baseline hypoalbuminemia had a significantly higher rate of all-cause TKI discontinuation occurring at a significantly shorter time after TKI initiation compared with patients with normal baseline albumin. TKI discontinuation resulting from intolerability was also more common among the patients with hypoalbuminemia than among those without it.
The incidence of hypoalbuminemia in our study (45.9%) was similar to that seen in clinical practice (49.3%) in relation to advanced cancers.15 Major differences in baseline characteristics of patients in our study with baseline hypoalbuminemia versus those without hypoalbuminemia included a worse performance status and higher incidences of liver disease and proteinuria.
Given the multifactorial mechanisms of decreased serum albumin levels in patients with cancer, these differences may contribute to the incidence of hypoalbuminemia and are to be expected. Although we found a slightly higher incidence of metastases at baseline in the population with hypoalbuminemia than in those without hypoalbuminemia, the proportion of patients having received any previous lines of therapy was similar between the 2 groups.
Although the majority of all patients in our study had an adverse event of any grade, the incidence of adverse events was similar between both groups. There was, however, a significantly shorter time to a documented adverse event in the patients with hypoalbuminemia than in those without. In addition, in patients who had multiple adverse events related to TKI therapy, the proportion of patients with hypoalbuminemia increased. We were not able to determine the severity of adverse events and identify the differences between the 2 groups, because of the retrospective nature of the study design.
Currently only minimal data are available regarding the link between hypoalbuminemia and TKI tolerability in patients with cancer. Murdock and colleagues conducted a retrospective evaluation of the tolerability of highly protein-bound oral oncolytic drugs in solid tumors,14 with similar results to the results of our study. Their analysis showed a similar incidence of adverse events between the groups (81% with hypoalbuminemia vs 75% without hypoalbuminemia), as well as an increased incidence of all-cause TKI discontinuation (83% vs 57%, respectively) with a significantly shorter time to TKI discontinuation (4.2 months vs 13.1 months, respectively) for the group with hypoalbuminemia.14
The incidence (83% vs 60%, respectively) and time to all-cause TKI discontinuation (3.2 months vs 9.6 months, respectively) in patients with hypoalbuminemia versus patients without hypoalbuminemia in our analysis were very similar to those reported by Murdock and colleagues.14 There were, however, a few significant differences in the results, including the analysis of TKI discontinuation because of intolerability. Murdock and colleagues identified a median time to TKI discontinuation because of intolerability of 22 months for the hypoalbuminemia population and median time was not reached in the population without hypoalbuminemia14 compared with a median time of 1.8 months in our analysis for patients with hypoalbuminemia. Possible reasons for this difference were the small sample size in the study by Murdock and colleagues, as well as the disparities between their study and ours in the incidence of TKI discontinuation as a result of intolerability.
The analysis by Murdock and colleagues included 143 patients, of which 21 (15%) discontinued treatment as a result of intolerability.14 Our analysis included 220 patients, of which 56 (25%) discontinued treatment as a result of intolerability. These relatively small numbers could have led to increased variability regarding the findings.
In addition, there were significant differences between the populations of each study. Murdock and colleagues included abiraterone and enzalutamide in the list of TKIs, and a large proportion (36.4%) of patients with prostate cancer made up their total population,14 whereas our analysis had no patients with prostate cancer. These hormonal therapies have a very different safety profile from the TKIs included in our study. Furthermore, patients with prostate cancer have low incidence of liver metastases, which can be a main factor in having liver dysfunction and subsequent hypoalbuminemia.
At baseline, the median albumin for patients with hypoalbuminemia in our analysis was slightly lower than that reported by Murdock and colleagues (3.2 g/dL vs 3.4 g/dL, respectively).14
Although both analyses identified a higher incidence of liver dysfunction in patients with hypoalbuminemia than in those without hypoalbuminemia, Murdock and colleagues did not report on the proportion of patients with metastatic disease,14 and they conducted the assessment of performance status as well as previous lines of therapy in a different way from our study, making it difficult to compare directly the severity of illness between the patient populations in the 2 studies. It is also difficult to identify the primary reason for the discrepancy in the time to TKI discontinuation caused by intolerability between the 2 analyses; however, the differences in the patient populations might have been a contributing factor.
Of note, the study by Murdock and colleagues14 and our study were single-center, retrospective chart reviews, and thus relied on provider documentation to determine if and when TKI discontinuation because of intolerability occurred. These data are only available in chart notes and are not consistently documented, and treatment discontinuation of TKIs for other reasons (mainly disease progression) is common; thus, it is possible that there were differences in the documentation regarding the reason for TKI discontinuation between our center and the center in the study by Murdock and colleagues that affected the findings.
Nevertheless, our results support the observation of the increased incidence of TKI discontinuation, as well as shorter time to TKI discontinuation for patients with hypoalbuminemia and the need to further classify the relationship between serum albumin and TKI tolerability. Therefore, patients with hypoalbuminemia should be identified at baseline and throughout TKI therapy. As serum albumin decreased throughout the course of TKI treatment, patients continued to be at an increased risk for adverse events and discontinuation of therapy.
Further studies are warranted to identify the relationship between hypoalbuminemia, TKI tolerability, and clinical outcomes. With earlier discontinuation of therapy, patients must receive subsequent lines of therapy earlier in the course of their disease. Alternative empiric dosing strategies cannot be recommended at this time, because of a lack of data on the clinical impact of decreased dosing intensities on survival outcomes. Close monitoring of albumin and adverse events may help to identify patients who are at increased risk for early TKI discontinuation.
An interdisciplinary team that incorporates a dedicated oncology pharmacist can benefit patient care significantly in the supportive care management of adverse events and can make early interventions to adjust the dosing for intolerability to avoid the all-cause discontinuation of TKI therapy.
Limitations
This study has several limitations. The retrospective design of the study meant that the data collection relied on provider documentation for the assessment of adverse events and the reason for TKI discontinuation. Adherence and cost were options for reasons for discontinuation in an attempt to capture nonadherence or financial toxicity. However, no patients had documentation of TKI discontinuation because of cost barriers, and only 1 patient discontinued TKI therapy because of adherence issues.
Reliance on provider documentation rather than prescription fill history might have led to an underestimation of the rates of nonadherence; however, prescription fill history was not readily available during the data collection.
It was also not possible to assess the severity of the adverse events, and therefore grading could not be applied to the incidence of adverse events. We also could not identify the differences between the 2 groups in the severity of the adverse events because of the retrospective design of the study. In addition, it is possible that not all adverse events were documented. Another limitation of this study’s retrospective design is the impact on specific dates for the occurrence of adverse events.
Additional limitations based on the selection of data collection and on end points include a lack of an assessment of concomitant medications to evaluate possible drug interactions. Because drug displacement via drug interactions is a potential mechanism for increased free drug concentration, drug interactions might also have played a role in increased adverse events and might have affected the rates of TKI discontinuation.
Conclusion
The results of our study show significant differences in all-cause TKI discontinuation, as well as discontinuation because of intolerability between the 2 groups. The Cox regression analysis was conducted to adjust for the differences in the patients’ baseline characteristics, and showed an independent, significant association between the presence of hypoalbuminemia and the rates of TKI discontinuation.
Our findings show that patients with baseline hypoalbuminemia had adverse events earlier, leading to a shorter duration of therapy as a result of intolerability and disease progression. Baseline hypoalbuminemia, therefore, may be an important clinical measure when initiating TKI treatment. Because of lack of data supporting clinical outcomes, preemptive dose reductions or alternate dosing strategies cannot be recommended at this time. However, patients with hypoalbuminemia at baseline or throughout the duration of their TKI therapy warrant closer monitoring to identify and manage adverse events and adjust the therapy, without discontinuing TKI therapy.
Oncology pharmacists can play a prominent role in the follow-up of these patients who are at increased risks for TKI intolerability and discontinuation.
Author Disclosure Statement
Dr Sion is on the Advisory Board of Merck, Exelixis, and Oncology Management, and became an employee of Merck after the completion of this study; Dr Marini, Dr Bass Hammond, Dr Rennebaum, Dr Weeda, and Dr Misniakiewicz have no conflicts of interest to report.
References
- Liu JW, Chen C, Loh EW, et al. Tyrosine kinase inhibitors for advanced or metastatic thyroid cancer: a meta-analysis of randomized controlled trials. Curr Med Res Opin. 2018;34:795-803.
- Huang L, Jiang S, Shi Y. Tyrosine kinase inhibitors for solid tumors in the past 20 years (2001–2020). J Hematol Oncol. 2020;13:143. doi: 10.1186/s13045-020-00977-0.
- Schmidinger M. Understanding and managing toxicities of vascular endothelial growth factor (VEGF) inhibitors. EJC Suppl. 2013;11:172-191.
- Jackson PR, Tucker GT, Woods HF. Altered plasma drug binding in cancer: role of α1-acid glycoprotein and albumin. Clin Pharmacol Ther. 1982;32:295-302.
- Villarroel MC, Pratz KW, Xu L, et al. Plasma protein binding of sorafenib, a multi kinase inhibitor: in vitro and in cancer patients. Invest New Drugs. 2012;30:2096-2102.
- Gatta A, Verardo A, Bolognesi M. Hypoalbuminemia. Intern Emerg Med. 2012;7(suppl 3):S193-S199.
- Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf. Accessed August 24, 2021.
- McMillan DC. Systemic inflammation, nutritional status and survival in patients with cancer. Curr Opin Clin Nutr Metab Care. 2009;12:223-226.
- Nazha B, Moussaly E, Zaarour M, et al. Hypoalbuminemia in colorectal cancer prognosis: nutritional marker or inflammatory surrogate? World J Gastrointest Surg. 2015;7:370-377.
- Kowalski-Saunders PW, Winwood PJ, Arthur MJ, Wright R. Reversible inhibition of albumin production by rat hepatocytes maintained on a laminin-rich gel (Engelbreth-Holm-Swarm) in response to secretory products of Kupffer cells and cytokines. Hepatology. 1992;16:733-741.
- Brink AL, Bowe C, Dains JE. Risk factors for ifosfamide-related encephalopathy in adult cancer patients: an integrative review. J Adv Pract Oncol. 2020;11:368-380.
- Drisaldi A, Weeda E, Neyens R, et al. Accuracy of valproic acid concentration correction based on serum albumin. Neurocrit Care. 2019;30:301-306.
- Yoo C, Ryu MH, Ryoo BY, et al. Changes in imatinib plasma trough level during long-term treatment of patients with advanced gastrointestinal stromal tumors: correlation between changes in covariates and imatinib exposure. Invest New Drugs. 2012;30:1703-1708.
- Murdock JL, Duco MR, Reeves DJ. Tolerability of highly protein bound targeted oral oncolytic drugs in patients with hypoalbuminemia: a retrospective analysis. Ann Pharmacother. 2021;55:165-173.
- Viganó A, Bruera E, Jhangri GS, et al. Clinical survival predictors in patients with advanced cancer. Arch Intern Med. 2000;160:861-868.
- Alecensa (alectinib) capsules, for oral use [prescribing information]. Genentech; September 2021. www.gene.com/download/pdf/alecensa_prescribing.pdf. Accessed June 27, 2022.
- Gilotrif (afatinib tablets), for oral use [prescribing information]. Boehringer Ingelheim International; April 2022. https://docs.boehringer-ingelheim.com/Prescribing%20Information/PIs/Gilotrif/Gilotrif.pdf?DMW_FORMAT=pdf. Accessed June 27, 2022.
- Inlyta (axitinib) tablets, for oral administration [prescribing information]. Pfizer; June 2020. https://labeling.pfizer.com/ShowLabeling.aspx?id=759. Accessed June 27, 2022.
- Cabometyx (cabozantinib) tablets, for oral use [prescribing information]. Exelixis; September 2021. www.cabometyxhcp.com/sites/default/files/2021-03/prescribing-information.pdf. Accessed June 27, 2022.
- Xalkori (crizotinib) capsules, for oral use [prescribing information]. Pfizer; October 2021. https://labeling.pfizer.com/ShowLabeling.aspx?id=676. Accessed June 27, 2022.
- Balversa (erdafitinib) tablets, for oral use [prescribing information]. Janssen Pharmaceutical Companies; April 2022. www.janssenlabels.com/package-insert/product-monograph/prescribing-information/BALVERSA-pi.pdf. Accessed June 27, 2022.
- Tarceva (erlotinib) tablets, for oral use [prescribing information]. Astellas Pharma; Genentech; October 2016. www.gene.com/download/pdf/tarceva_prescribing.pdf. Accessed June 27, 2022.
- Gleevec (imatinib mesylate) tablets, for oral use [prescribing information]. Novartis; March 2022. www.novartis.us/sites/www.novartis.us/files/gleevec_tabs.pdf. Accessed June 27, 2022.
- Tykerb (lapatinib) tablets, for oral use [prescribing information]. Novartis; March 2022. www.novartis.us/sites/www.novartis.us/files/tykerb.pdf. Accessed June 27, 2022.
- Lenvima (lenvatinib) capsules, for oral use [prescribing information]. Eisai; December 2021. www.lenvima.com/-/media/Project/EISAI/Lenvima/PDF/prescribing-information.pdf. Accessed June 27, 2022.
- Nerlynx (neratinib) tablets, for oral use [prescribing information]. Puma Biotechnology; March 2022. https://nerlynxhcp.com/pdf/full-prescribing-information.pdf. Accessed June 27, 2022.
- Tagrisso (osimertinib) tablets, for oral use [prescribing information]. AstraZeneca; January 2022. www.azpicentral.com/pi.html?product=tagrisso&country=us&popup=no. Accessed June 27, 2022.
- Votrient (pazopanib) tablets, for oral use [prescribing information]. Novartis; December 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2021/022465Orig1s033lbl.pdf. Accessed June 27, 2022.
- Stivarga (regorafenib) tablets, for oral use [prescribing information]. Bayer HealthCare Pharmaceuticals; December 2020. https://labeling.bayerhealthcare.com/html/products/pi/Stivarga_PI.pdf. Accessed June 27, 2022.
- Nexavar (sorafenib) tablets, for oral use [prescribing information]. Bayer HealthCare Pharmaceuticals; July 2020. https://labeling.bayerhealthcare.com/html/products/pi/Nexavar_PI.pdf. Accessed June 27, 2022.
- Sutent (sunitinib malate) capsules, for oral use [prescribing information]. Pfizer; September 2021. https://labeling.pfizer.com/showlabeling.aspx?id=607. Accessed June 27, 2022.