Approximately 12% of women in the United States will be diagnosed with breast cancer during their lifetime.1 In patients with metastatic hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative disease, early treatment is based on endocrine therapy and the addition of a cyclin-dependent kinase (CDK)4/6 inhibitor, such as palbociclib.2
The ability to initiate therapy in a timely manner after deciding on a treatment plan is important to patients and to providers. Obtaining oral oncology specialty medications, such as palbociclib, has become increasingly complex, given the interactions needed among providers, specialty pharmacies, insurance companies, pharmacy benefit managers (PBMs), and often drug manufacturers or independent foundations’ financial assistance programs.3-5
The barriers to obtaining oral oncology specialty medications may include issues with insurance coverage, direct copay cost, finding additional financial support for expensive medications, the identification of an insurance-required pharmacy, the completion of a prior authorization (PA), potential PA appeals by providers, and obtaining specialty medications.6-13 Previous reports show that the median or mean time to medication access for various oral cancer therapies ranges from 6 to 12 days.7,9-13
Increasingly, health systems work to address these barriers by creating and/or using their own health-system specialty pharmacies (HSSPs) or medically integrated dispensing pharmacies, with HSSPs reporting faster access to medications than external pharmacies.14 Optimally, insurance plans would allow these institutions to dispense oral oncolytic medications to their own patients. Instead, many health plans require patients to use the PBM-owned specialty pharmacy.15
In a previous study, Giridhar and colleagues identified various delays in patients receiving radiosensitizing capecitabine in combination with radiation for the treatment of rectal cancer.16 That study showed that significantly more patients had delays in receiving capecitabine when working with external specialty pharmacies compared with the Mayo Clinic HSSP (52.9% vs 11.4%, respectively) to receive the drug. The delay in the time to first fill resulted in patients initiating radiation therapy without concurrent radiosensitizing capecitabine treatment.16
Based on these data with capecitabine, we were interested in finding out if there were also significant delays in obtaining palbociclib via external pharmacies, and what the magnitude of these delays might be. The objective of this study was to evaluate whether a significant delay exists in the time to the first fill of palbociclib between our HSSP and external specialty pharmacies. This information could be further used to determine ways in which pharmacists and other members of the healthcare team could expedite the delivery of specialty medications to our patients.
Methods
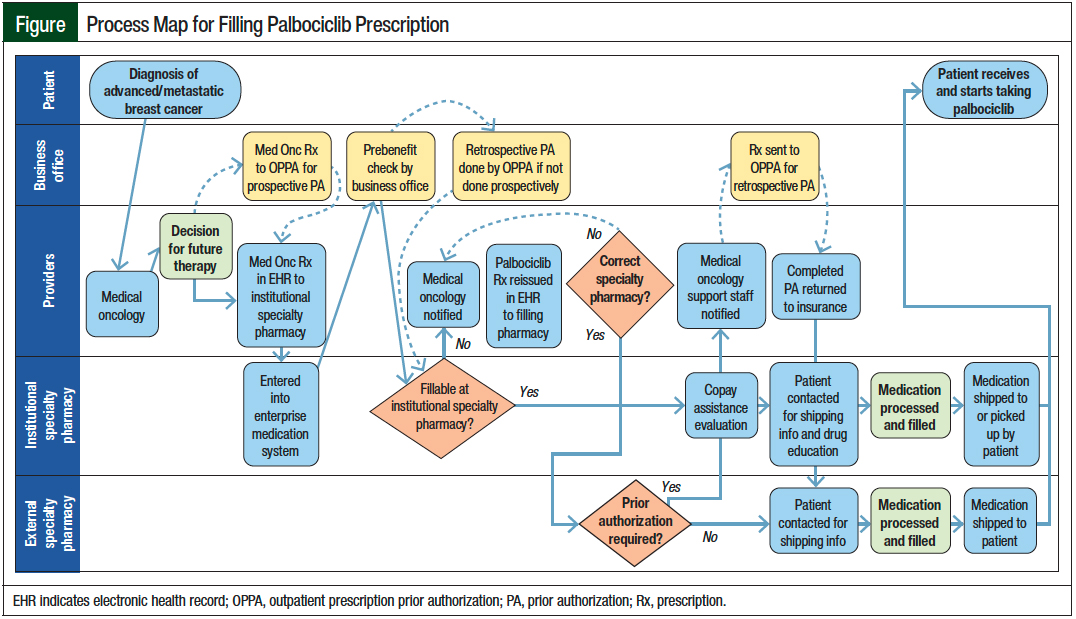
In this single-center study, we first created a process map to understand the individual steps needed to obtain oral oncolytic specialty agents. We then identified the potential barriers to efficient prescription filling. We conducted a single-center, retrospective review of prescription data for patients aged ≥18 years who were diagnosed with advanced or metastatic HR-positive, HER2-negative breast cancer and had an initial palbociclib prescription between May 5, 2018, and June 30, 2019, at the Mayo Clinic Cancer Center in Rochester, MN.
The start date coincided with the local going live of a new institution-wide electronic health record (EHR), which included new workflow for oral cancer therapies. The time-to-first-fill of palbociclib data were calculated as the difference in calendar days between the date the prescription was electronically ordered and the sold date, defined as the date the prescription was picked up by or shipped to the patient. The sold date was obtained through our HSSP reporting functions. External specialty pharmacies used by our patients to fill their palbociclib prescription were contacted by phone to obtain the palbociclib prescription’s sold and/or shipping date information.
To delineate our institutional time spent in obtaining approval for the prescriptions and redirecting them outside of the institution, the modified time to fill was defined as the difference between the time the prescription was sent electronically to the external insurance-preferred pharmacy and the sold date. We also evaluated the differences between the insurance coverage types, the external specialty pharmacies, and the impact of sending new prescriptions once the insurance-preferred specialty pharmacy was identified. The time to first fill was compared between our HSSP and the external specialty pharmacies. Additional comparisons were completed using a Wilcoxon rank-sum test.
Because the majority of our institution’s PA and appeal work is done before a prescription is sent to an external pharmacy, we further evaluated the impact of PA approval and pharmacy network determination processes on the time-to-first-fill comparisons. The PA data and appeal information were gathered by the outpatient prescription PA team using our EHR, the CoverMyMeds network, and other internal revenue cycle documentation.
To assess the impact of the time required for PAs, the study patients were sorted into 3 subgroups based on their initial status: PA approved, PA denied, and PA data not found or PA not needed. The PA approval and the appeal approval dates used in this study were based on the documented decision date on which the insurance company approved the PA or the appeal (eg, the fax header date on an approval letter, the approval date if electronically received). Based on the PA approval date, we were also able to compare the HSSP and the external specialty pharmacies in terms of the time to the PA approval, as well as the time from the PA approval to the drug being shipped or sold.
The Figure outlines the process for filling a specialty medication used to identify the steps that may be contributing to delayed filling time. Once a patient is diagnosed with cancer and is seen by the medical oncology team, the decision to pursue treatment is made and a prescription is sent electronically to our HSSP.
Our institutional PA process is designed to be managed by a centralized outpatient prescription PA team on behalf of the provider. PAs are triggered either prospectively (ie, direct referral to the outpatient prescription PA team to obtain the PA approval before the release of the prescription to the pharmacy) or retrospectively (ie, after the pharmacy has received the prescription, processed a claim, and determined that a PA is needed).
A prebenefit check is initiated to determine whether our HSSP can fill the prescription. When our HSSP is not in the insurance company network, identifying the required external pharmacy may happen before or after the PA is approved; regardless, a new prescription is then reissued to the correct pharmacy. The completion of the insurance benefits investigation and the reissuing of the prescription add further steps to the overall process.
Once the prescription reaches the insurance-preferred filling pharmacy, an evaluation for copay assistance is completed. Then, the patient is contacted to gather the billing and shipping information, and to provide patient education. Finally, the prescription is processed, filled, and shipped or picked up by the patient.
Results
External Specialty Pharmacies
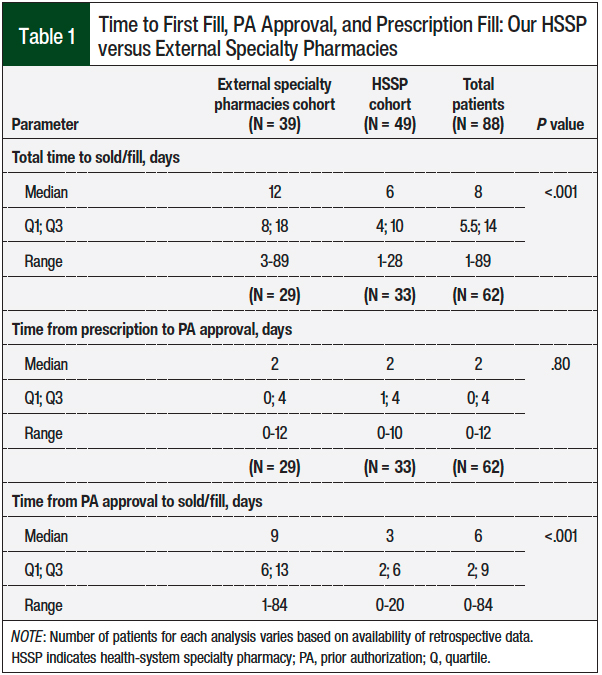
A total of 88 patients met the study inclusion criteria and had prescription fill data for palbociclib available for analysis. Overall, 39 patients had their first fill at an external specialty pharmacy, and 49 patients had their first fill at our HSSP. A median time to first fill for our HSSP cohort was 6 days (interquartile range [IQR], 4-10); it was 12 days (IQR, 8-18; P <.001) for the external specialty pharmacies cohort (Table 1).
When excluding institutional time spent on insurance approval, benefits investigations, and redirecting prescriptions externally, the external specialty pharmacy–modified time to fill was a median of 7 days (IQR, 3-10). This modified time to first fill for the external pharmacies was not significantly different from the HSSP total time to first fill (P = .60); however, the steps included in each group in this comparison were vastly different, because the majority of the external pharmacies received the new prescription with the PA and insurance approval already in place (after being processed by our HSSP). This will require further evaluation of the impact of the insurance approval process to streamline this process better.
No significant difference in the time to first fill was observed in the various external specialty pharmacies included in this study (P = .26). Subsequently, we assessed the factors that might have influenced the time to first fill compared with those who did not have a PA denial, including the number of prescription redirects, PA process, type of insurance, and individuals with prolonged delays to initiating therapy.
Overall, 2 patients in our study required a free drug, which took 18 days to apply for and 20 days to obtain the medication through the patient assistance program.
Prior Authorization Process
Of the 70 patients with PA information available for evaluation, 62 prescriptions were initially approved (internal, N = 33; external N = 29) and 8 were initially denied and then approved (internal, N = 3; external, N = 5). As can be expected, the patients with an initial PA denial had a significantly longer median time to their first prescription fill compared with those who did not have a PA denial: 14 days (IQR, 5.25-13) versus 7.5 days (IQR, 9.75-19.5), respectively (P <.001).
The median time from when the prescription was written to the PA approval was 2 days for the patients who filled the prescription at our HSSP and at external pharmacies. Once the PA was approved, the remaining steps to the prescription fill took a median of an additional 3 days at our internal HSSP versus 9 days for patients who filled at an external pharmacy (P = <.001).
In general, external pharmacies received prescriptions with the PA already approved, because insurance approval was obtained before or on the day of the prescription reissue to the external pharmacy in 23 of 29 (79.3%) patients. The median PA time included in the modified time to fill was 0 days, and only 2 patients exceeded 48 hours to have the PA completed. The time required to obtain insurance approval did not add to the median modified time to fill once the prescription was reissued to the external pharmacy.
When we removed the barriers of PA and external prescription redirection, by comparing the time to first fill after the PA approval and prescription reissue, the processing or assistance time was 3 days (IQR, 2-6) for our HSSP compared with 7 days (IQR, 3-10) for the modified time to fill for external pharmacies. Overall, patients could start therapy more quickly when they were able to use the HSSP instead of an insurance-preferred or insurance-mandated external specialty pharmacy.
Type of Insurance
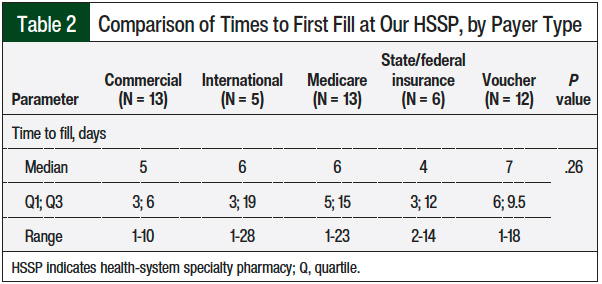
We reviewed the cohort of 49 patients who filled their prescription at our HSSP to determine any potential difference between the various types of insurance coverage used. As seen in Table 2, no significant difference was observed in payers’ first fill (P = .26). However, vouchers were used by some patients with Medicare to avoid further delays when additional financial assistance was sought.
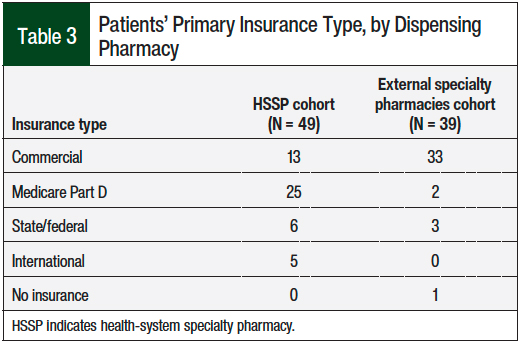
By contrast, the type of insurance for the HSSP cohort and the external pharmacy cohort differed dramatically (Table 3). Only 13 of the 49 (26.5%) patients with HSSP first fill were commercially insured compared with 33 of the 39 (85%) patients who were required to fill their prescription at an external specialty pharmacy. Furthermore, only 2 of the 27 (7.5%) total patients with Medicare Part D coverage filled their prescription at an external pharmacy (Table 3), making a comparison of that aspect between the groups difficult.
Prescriptions Issued
The need for multiple electronic prescription redirections when the patient’s insurance company required a specific filling pharmacy was a potential cause for the delay in the time to first fill. Of the 39 patients who required their prescription(s) to be redirected to an external pharmacy, 8 (20.5%) had prescriptions redirected at least twice.
Particularly with medications that have a limited distribution network, payers may require a prescription to be filled through a pharmacy that does not have access to dispense the medication.10 This leads to significant frustration and time wasted before the payer ultimately allows an override for a pharmacy that can dispense the medication. This occurred in only 1 of these 8 patients in our cohort, however, because most major PBM-owned specialty pharmacies have access to palbociclib. The 7 remaining patients had prescriptions issued to each patient’s local pharmacy before being redirected.
After manual electronic chart review, these prescriptions were either sent to the local pharmacy based on a patient’s desire to obtain the drug locally, or they were possibly inadvertently sent locally based on the EHR default settings. Of these 8 patients with more than 2 prescriptions issued, it took a median of 1 day (range, 0-5 days) from the identification of the correct pharmacy until the prescription was reissued. The insurance requirement to use an external pharmacy results in additional work and time spent by our institutional staff to resend these prescriptions, which could delay the time to the initiation of therapy for patients.
Delays to Therapy Initiation
A manual chart review of the prolonged time (>6 days) to first fill at our HSSP revealed several reasons for the prolonged delays in therapy initiation beyond pharmacy processing, which can be classified into categories such as financial, personal, clinical, insurance, and communication. At least 5 patients had some degree of delaying the start of treatment as a result of the initiation of a patient assistance program application process, and 1 patient had a delay in responding with insurance information, leading to a prolonged time of 18 days to first fill.
In addition, 3 patients chose to delay the initiation of therapy for personal reasons. Of these, 2 patients were admitted to the hospital and 1 patient was awaiting biopsy results, which led to an increased time to first fill ranging from 11 to 23 days.
Insurance type was another major driver of prolonged time to first fill. A total of 3 of 44 patients with noninternational insurance had a PA denial, which prolonged the time to first fill by 4 days in 2 patients and by 6 days in 1 patient. Unclear communication between the patient, the provider, and/or the pharmacy was responsible for delays for 4 patients (range, 5-7 days).
Discussion
Our results show a significant difference in the time to first fill of palbociclib between patients using our HSSP and those using external specialty pharmacies. It took at least twice as long for patients at Mayo Clinic Cancer Center to fill a new palbociclib prescription when they used an external pharmacy than when they used our HSSP, and nearly 3 weeks if the patients had to obtain the medication from the manufacturer’s free drug program.
Our data illustrate that the basic time-to-first-fill metrics may not be directly comparable between pharmacies without a deep understanding of what steps are completed before the receipt of the prescription at an insurance-mandated external pharmacy. This became clear after assessing additional data points showing that all the time and work that went into the PA approval and the pre–benefit investigation had been done before the external pharmacy–modified time-to-fill clock started.
The reasons for a delayed time to first fill in our HSSP cohort were multifactorial, including financial, insurance, clinical, personal, and communication barriers. These wide-ranging variables indicate that patient circumstances can play a large role in the time to first fill, and no single solution will eliminate all major delays. For example, improvements in internal workflows (potentially requiring additional staffing) could trim time from several steps, including the time for prescriptions to be redirected to an external pharmacy.
However, the biggest gains in the time to first fill could be made by PBMs by allowing patients to fill palbociclib at their HSSP instead of at a plan-mandated external pharmacy. This is best illustrated by comparing the time to first fill after the PA was approved between the groups: it took 3 times longer from this point for patients to fill their prescription using an external pharmacy than using an HSSP (median time, 9 days vs 3 days, respectively). This has the potential to decrease the time to first fill, and it streamlines the workflow process and reduces staff burden in re-work and external pharmacy coordination. By removing the external barriers that free up staff time, more focus could be placed on improving therapeutic and quality-of-life outcomes.
The use of manufacturer hub services for benefits investigations could be considered and may be helpful in some practices. However, in an HSSP with internal resources for PAs, benefits investigations, and patient assistance, the provider is only required to submit an e-prescription to the HSSP instead of manually filling out multiple pages of enrollment forms, which fragments the process outside of the institution.
The hubs may help identify, but usually do not obtain, the PAs, appeals, or independent foundation grants on the provider’s behalf, so additional communication and paperwork still come back to the provider to complete when using these services. Although we did not use hub services upfront for benefits investigations, these services were required to pursue free drugs.
The manual paperwork is outside of the EHR and can be difficult to track. Ideally, hub services must incorporate technologic improvements that reduce the burden of the insurance authorization process with electronic PAs and real-time prescription benefit tool requirements. Integrating these tools directly into the EHR is imperative for maximum benefit and practice efficiency.
Our results add to the growing evidence that HSSPs are able to provide therapy more quickly to patients than when the use of an external pharmacy is mandated. The study by Wyatt and colleagues showed a faster time, 4.7 days, to medication access when the prescription was filled internally compared with 7.3 days for limited distribution medications that the pharmacy did not have manufacturer access to dispense.9
The time to insurance approval of 1.9 days in the study by Wyatt and colleagues was similar to our study at 2 days.9 They also evaluated the addition of dedicated specialty pharmacy staff to the clinic. These dedicated resources improved the time to medication access for the 2 limited distribution medications obtained externally compared with the previous institutional process, but the time was still slower than when internal dispensing occurred.9
Wang and colleagues evaluated 29 different oral oncology medications and concluded that insurance-required transfers to external pharmacies took longer to receive than those filled internally at the hospital.10 Much different from our data, they noted that the proportion of transfers did not differ significantly between Medicare patients and patients with commercial insurance. Their study also showed that it took Medicare patients more than double the time (4.7 days longer) to receive therapy than commercially insured patients.10 This difference is not unexpected given the additional cost and assistance barriers faced by patients with Medicare Part D coverage compared with commercially insured patients.
In our study, nearly all the Medicare Part D patients were in the HSSP group, whereas commercially insured patients represented a high proportion of the external specialty pharmacy group. The data from Wang and colleagues10 suggest that, at a minimum, the insurance demographic differences would not be expected to be the reason for the longer time to fill for external pharmacies in our cohort and possibly could have been a disadvantage with regard to the HSSP group’s time to first fill results in our study.
Limitations
This study had several limitations, including its retrospective and observational design. Our results may not extrapolate to institutions without dedicated resources for PAs and patient assistance programs. Although we did not find a difference in the time to first fill between the payer types using our HSSP, vouchers were used by some Medicare Part D patients to reduce the time to first fill, whereas free drug and/or independent grants for ongoing therapy continued to be pursued. If not for the availability and use of the vouchers, it is possible that the Medicare Part D cohort might have had a significantly longer time to first fill compared with commercially insured patients. Also, the mix of insurance types varied dramatically between our HSSP group and the external pharmacies group.
Nearly all of the external pharmacy patients were commercially insured, which typically allows for patients’ out-of-pocket (OOP) copays to be eligible for a manufacturer’s copay card. Our HSSP group had a high percentage of Medicare Part D patients, who have high OOP expenses and are ineligible for copay assistance from the drug manufacturer. To enable most eligible patients to afford their Medicare Part D copays, grant funding must be sought from independent foundations. When not available, assistance is then sought from the free drug program. These applications frequently require income and/or tax documentation and can substantially increase the time spent working on financial assistance options relative to a copay card.
For 5 patients, grants were not available initially but became available after the free drug process was started, but before approval. This resulted in additional time and effort by all involved and subsequently delayed the time to first fill. The actual time spent pursuing patient assistance, however, was not directly measurable in this study. Given the high OOP expenses and no cap associated with oral cancer therapies covered through Medicare Part D plans, a redesign of the structure of Medicare Part D itself would have a dramatic impact on patient financial barriers, as well as minimize the untold hours of time devoted to seeking copay assistance.
We also did not have PA data available for all patients, and the method of notification for payer decisions was inconsistent among the payers (ie, electronic, mail, fax, verbal notification). Appeals of a PA denial can be even more problematic, because they typically cannot be done in the same electronic manner as many PAs are processed.
In at least 1 case, an appeal approval notification did not reach the provider until 6 days after the approval determination, thus delaying the rest of the fill process. Real-time prescription benefit and electronic PAs have the potential to streamline the approval process, but significant improvements in the quality, consistency, usability, and availability of these tools must be made at the time of prescribing to reduce these barriers to therapy initiation. For example, some of the electronic PAs in our cohort were approved in a matter of minutes, yet others were delayed by requirements of multiple submissions of additional forms that were not available up front, the use of portals outside of our EHR, and/or manual faxed forms.
Conclusion
This study demonstrates a doubling in the total time to first fill when patients used an external specialty pharmacy compared with our integrated HSSP. Delays in the time to first fill can be multifactorial and can vary widely, from financial toxicity and insurance barriers to hospitalization or a personal decision to delay the start of therapy. Further exploration of this impact and the identification of common barriers leading to delays in the time to first fill are being addressed in an effort to decrease the time to first fill and to expedite the treatment of patients with advanced or metastatic breast cancer who are prescribed CDK4/6 inhibitors.
Broad, dramatic improvements in electronic PAs, real-time prescription benefit tools, and insurance plan copay design also have the potential to streamline the entire prescription medication delivery process. Given the current system, additional data showing improved outcomes for patients using an HSSP or medically integrated dispensing practice may be beneficial in encouraging insurance plans to allow institutions to dispense these specialty medications for their own patients.
These integrated programs can mitigate many of the barriers seen with fragmented care delivery and get medication to the patient quickly.
Acknowledgments
We thank Erin Dop and Nicole Lorde-Matthews for collecting prior authorization data.
Funding Source
This study was supported by the Pfizer Global Medical Grants Quality Improvement Grant: Treatment Patterns of CDK4/6 Inhibitor Utilization in HR+/HER2- Metastatic Breast Cancer.
Author Disclosure Statement
Dr Golbach has received research support from Pfizer; Dr Taraba has received research support from Pfizer, is on the Advisory Board of Daiichi Sankyo, Novartis, and Eli Lilly, and is a consultant to Change Healthcare; Dr Smith has received research support from Pfizer; Dr Giridhar has received research support from Pfizer and is on the Advisory Board of Novartis; Ms Mara has no conflicts of interest to report.
References
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Breast Cancer. Version 4.2022. June 21, 2022. www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed September 22, 2022.
- American Medical Association. 2021 AMA prior authorization (PA) physician survey. 2022. www.ama-assn.org/system/files/2021-04/prior-authorization-survey.pdf. Accessed June 21, 2022.
- Surescripts. Pharmacist perspectives on the specialty fulfillment process. April 2020. https://surescripts.com/docs/default-source/default-document-library/surescripts-specialty-pharmacy-data-brief.pdf. Accessed June 21, 2022.
- Tompkinson M, Fine K, Gruber D, et al. Perceptions of health-care workers of the cost and safety of oral oncolytic agents for patients: a survey. J Adv Pract Oncol. 2020;11:354-365.
- Toman A. Specialty pharmacy: why does it take so long? Pharmacy Times. December 18, 2017. www.pharmacytimes.com/view/specialty-pharmacy-why-does-it-take-so-long. Accessed September 26, 2022.
- Anders B, Shillingburg A, Newton M. Oral antineoplastic agents: assessing the delay in care. Chemother Res Pract. 2015;2015:512016. doi: 10.1155/2015/512016. Accessed June 21, 2022.
- Agarwal A, Freedman RA, Goicuria F, et al. Prior authorization for medications in a breast oncology practice: navigation of a complex process. J Oncol Pract. 2017;13:e273-e282.
- Wyatt H, Peter M, Zuckerman A, et al. Assessing the impact of limited distribution drug networks based on time to accessing oral oncolytic agents at an integrated specialty pharmacy. J Hematol Oncol Pharm. 2020;10(4):198-205.
- Wang AA, Tapia C, Bhanji Y, et al. Barriers to receipt of novel oral oncolytics: a single-institution quality improvement investigation. J Oncol Pharm Pract. 2020;26:279-285.
- Geynisman DM, Meeker CR, Doyle JL, et al. Provider and patient burdens of obtaining oral anticancer medications. Am J Manag Care. 2018;24:e128-e133.
- Niccolai JL, Roman DL, Julius JM, Nadour RW. Potential obstacles in the acquisition of oral anticancer medications. J Oncol Pract. 2017;13:e29-e36.
- O’Neil D, Accordino MK, Wright JD, et al. Delay in receipt of newly prescribed oral anticancer drugs. J Clin Oncol. 2019;37(15 suppl):Abstract 6541.
- Bagwell A, Kelley T, Carver A, et al. Advancing patient care through specialty pharmacy services in an academic health system. J Manag Care Spec Pharm. 2017;23:815-820.
- Advisory Board. Pharmacy benefit managers, explained. Daily Briefing. November 13, 2019. www.advisory.com/daily-briefing/2019/11/13/pbms#:~:text=PBMs%20often%20restrict%20where%20health%20plan%20members%20can,PBMs%2C%20a%20trend%20which%20is%20expected%20to%20grow. Accessed June 21, 2022.
- Giridhar K, Pophali P, Sridharan M, et al. Identifying and overcoming barriers in delivering concurrent chemotherapy with radiation in the neoadjuvant treatment of rectal cancer. J Natl Compr Canc Netw. 2018;16:Abstract AB2018-12.