Symptom Overview
Damage to motor, sensory, or autonomic nerves secondary to antineoplastic chemotherapy can result in varying degrees of dysfunction and/or pain, which can lead to significant morbidity and diminished quality of life for affected patients, with up to 70% of patients having some element of chemotherapy-induced peripheral neuropathy (CIPN) after chemotherapy.1,2
A variety of chemotherapy classes, with varying effects by agent within a class, are known neurotoxins with a different affinity for nerves (Table 1).
The effects of CIPN are generally symmetrical, distal, in a stocking-glove distribution, and primarily affect the upper and lower extremities. These are usually sensory effects (burning, freezing, electroshock-like); however, there are more agent-specific neuropathies, such as constipation seen with vincristine; orthostasis with bortezomib; or temperature-related effects with oxaliplatin.3,4 In general, these effects are cumulative, dose-dependent, variable by agent, and attenuate to different levels once therapy is discontinued.
Etiology
Although not fully understood, the mechanisms of nerve damage vary by agents, with symptoms seen on nerve-cell microtubules or their axonal transport systems, mitochondria, or dorsal root ganglia, all of which can ultimately result in neuronal degradation. Additional neurologic dysfunction can occur, depending on the underlying disease, especially diseases that may result in mechanical compression of nerves or nerve roots. Several patient attributes and conditions are also known to increase the risk for CIPN (Table 2).
The symptoms of CIPN frequently attenuate after therapy, but they can be dose-limiting and result in suboptimal chemotherapy dosing.5 Treatment goals include the prevention or attenuation of neuropathies associated with therapy, the relief of symptoms, improving patient quality of life, and maintaining optimal chemotherapy dosing.
Treatment Options
A recent review of CIPN showed that no medications have enough evidence to suggest that they should be used to prevent CIPN, and there was adequate evidence to suggest that a variety of agents should not be used for that purpose, because of lack of efficacy, risk for harm, or both.7 The medications that should not be used for the prevention of CIPN include2,7:
- Acetyl-L-carnitine
- Amifostine
- Amitriptyline
- Calcium and magnesium for oxaliplatin-based chemotherapies
- Diethyldithiocarbamate
- Glutamine for taxanes
- Glutathione for paclitaxel/carboplatin chemotherapies
- Nimodipine
- Org 2766
- Recombinant human leukemia inhibitor factor
- All-transretinoic acid
- Venlafaxine ±, may affect antitumor effects of platinums
- Vitamin E.
Once neuropathy is present, the best approach to management is chemotherapy dose interruption. If chemotherapy interruption is not possible, then the addition of pharmacologic and/or nonpharmacologic therapies is common. Antidepressants, anticonvulsants, analgesics, antioxidants, nutritional supplements, and other agents have been used in attempts to prevent or ameliorate the symptoms of CIPN.
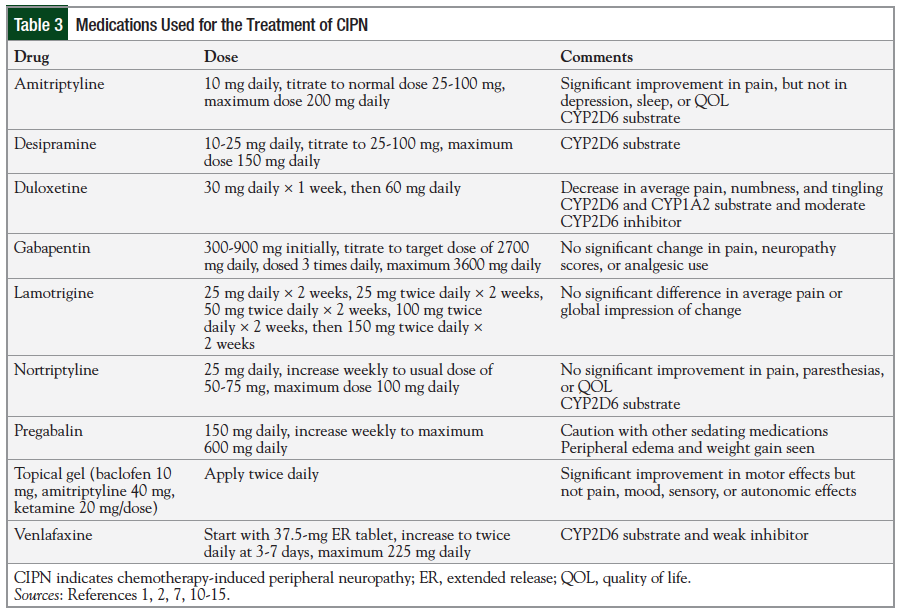
Several medications may be helpful for the treatment of CIPN, as has been shown in placebo-controlled trials (Table 3)1,2,7,10-15; however, because of the risk for adverse events, the potential benefit from each agent should be matched to the individual patient’s neuropathy profile.
Titration to target dose, as tolerated by the patient, is required for most medications. For compounded pharmaceutical agents, the concern is regarding nonstandardization of agents and the minimal long-term safety data.
The duration of use of each agent is unclear, with most studies evaluating the treatment of CIPN over periods ranging from ‹2 weeks to as much as 12 weeks.1,4 Effects of therapy are often seen within days of initiating treatment and may take days to weeks for the full benefit to be seen.
Although these therapies can lessen the effects of CIPN, they rarely cure it, and the patient’s need is frequently longer than most study periods. As a result, the clinician is often left to evaluate the safety of the extended use of specific agents based on experiences from managing other disease states with the same agent. Therefore, longer-term use is common, with each patient’s duration of therapy customized based on the individual response. Therapy is frequently continued until the neuropathy wanes, until the patient has serious therapy-related side effects, or until there is an indication to stop the medication.
At the time of medication discontinuation, accepted guidelines for the discontinuation of antidepressants or other medications should be followed to reduce the risk for a discontinuation syndrome. Many potentially beneficial agents are currently in early-phase clinical trials or are reported by anecdotal experience. In addition, work is ongoing to identify genetic markers for patients with increased risk for developing CIPN to identify agents that would not be likely to cause neuropathy in individual patients.16
It is hoped that useful agents for the prevention and management of neuropathies can be found, and that patients with increased risk for neuropathies could be offered alternative therapies.
References
- Aziz MT, Good BL, Lowe DK. Serotonin-norepinephrine reuptake inhibitors for the management of chemotherapy-induced peripheral neuropathy. Ann Pharmacother. 2014;48:626-632.
- Piccolo J, Kolesar JM. Prevention and treatment of chemotherapy-induced peripheral neuropathy. Am J Health Syst Pharm. 2014;71:19-25.
- Delforge M, Bladé J, Dimopoulos MA, et al. Treatment-related peripheral neuropathy in multiple myeloma: the challenge continues. Lancet Oncol. 2010;11:1086-1095.
- Durand JP, Deplanque G, Montheil V, et al. Efficacy of venlafaxine for the prevention and relief of oxaliplatin-induced acute neurotoxicity: results of EFFOX, a randomized, double-blind, placebo-controlled phase III trial. Ann Oncol. 2012;23:200-205.
- Speck RM, Sammel MD, Farrar JT, et al. Impact of chemotherapy-induced peripheral neuropathy on treatment delivery in nonmetastatic breast cancer. J Oncol Pract. 2013;9:e234-e240.
- Okada N, Hanafusa T, Sakurada T, et al. Risk factors for early-onset peripheral neuropathy caused by vincristine in patients with a first administration of R-CHOP or R-CHOP-like chemotherapy. J Clin Med Res. 2014;6:252-260.
- Hershman DL, Lacchetti C, Dworkin RH, et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2014;32:1941-1967.
- Quasthoff S, Hartung HP. Chemotherapy-induced peripheral neuropathy. J Neurol. 2002;249:9-17.
- Harnicar S, Adel N, Jurcic J. Modification of vincristine dosing during concomitant azole therapy in adult acute lymphoblastic leukemia patients. J Oncol Pharm Pract. 2009;15:175-182.
- Barton DL, Wos EJ, Qin R, et al. A double-blind, placebo-controlled trial of a topical treatment for chemotherapy-induced peripheral neuropathy: NCCTG trial N06CA. Support Care Cancer. 2011;19:833-841.
- Hammack JE, Michalak JC, Loprinzi CL, et al. Phase III evaluation of nortriptyline for alleviation of symptoms of cis-platinum-induced peripheral neuropathy. Pain. 2002;98:195-203.
- Kautio A-L, Haanpää M, Saarto T, Kalso E. Amitriptyline in the treatment of chemotherapy-induced neuropathic symptoms. J Pain Symptom Manage. 2008;35:31-39 .
- Rao RD, Michalak JC, Sloan JA, et al; for the North Central Cancer Treatment Group. Efficacy of gabapentin in the management of chemotherapy- induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo- controlled, crossover trial (N00C3). Cancer. 2007;110:2110-2118.
- Rao RD, Flynn PJ, Sloan JA, et al. Efficacy of lamotrigine in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo-controlled trial, N01C3. Cancer. 2008;112:2802-2808.
- Lavoie Smith EM, Pang H, Cirrincione C, et al; for the Alliance for Clinical Trials in Oncology. Effect of duloxetine on pain, function, and quality of life among patients with chemotherapy-induced painful peripheral neuropathy: a randomized clinical trial. JAMA. 2013;309:1359-1367.
- de Graan A-J, Elens L, Sprowl JA, et al. CYP3A4*22 genotype and systemic exposure affect paclitaxel-induced neurotoxicity. Clin Cancer Res. 2013;19:3316-3324.