Rigors are involuntary episodes of severe shaking or shivering that can be accompanied by a rise in temperature resulting from altered thermoregulation in the hypothalamus.1 Although the precise etiology of rigors is unknown, rituximab and antithymocyte globulin (ATG) induce cell lysis, which results in the release of various inflammatory cytokines that cause rigors.2,3 Treatment with rituximab has been shown to induce rigors in 13% to 33% of patients, and the incidence rate of rigors related to treatment with ATG is approximately 55%.4,5 These reactions can be unpleasant for patients and may cause early discontinuation of therapy or delays in treatment.
Treatment for rigors associated with chemotherapy is not well studied, and the current treatments are extrapolated from research in postanesthetic shivering.6 In a systematic review of randomized, controlled trials, Kranke and colleagues evaluated the use of various medications for the treatment of postoperative shivering.6 Meperidine stopped shivering in 87%, 91%, and 88% of patients after 5 minutes, 10 minutes, and 15 minutes, respectively.6 Historically, at Atrium Health Wake Forest Baptist in Winston-Salem, NC, meperidine was used to stop rigors associated with rituximab or ATG treatments, although the use of meperidine has several disadvantages. Meperidine’s active metabolite, normeperidine, is renally cleared and is associated with risks for anxiety, myoclonus, seizures, and mood changes within 24 hours of use.7 Meperidine’s benefit in the treatment of rigors or shivering is related to decreasing the shivering threshold by binding to μ and κ opioid receptors, which can be targeted by various agents within the opioid drug class.1 In its 2007 bulletin, the Institute for Safe Medication Practices called for the avoidance of treatment with meperidine, particularly in older patients and in those with renal insufficiency.8
At Atrium Health Wake Forest Baptist, morphine has been used since 2003 as a treatment for rigors associated with rituximab and ATG in lieu of meperidine in an effort to minimize the use of meperidine. Treatment with morphine is effective in the treatment of rigors associated with amphotericin administration and is extrapolated to be effective for other drug-induced rigors through pathways similar to those where meperidine has shown benefit.9
As a supportive care measure at Atrium Health Wake Forest Baptist, all treatment plans with rituximab and ATG have morphine prebuilt to be administered for rigors. Per the treatment plan, 2 mg of intravenous (IV) morphine is administered at the onset of rigors, followed by another 2 mg of IV morphine if no response is obtained within 15 minutes. The dose of morphine is commercially available in prefilled syringes and can be stored on the floor in automated drug cabinets that are easily accessible by nursing staff to quickly obtain and administer when advised by a provider that a patient has had a reaction to treatment with rituximab or ATG. It is at the provider’s discretion whether to administer subsequent doses of IV morphine or to escalate treatment to 12.5 mg or 25 mg of IV meperidine.
The purpose of this study was to evaluate the effectiveness of morphine as an alternative therapy to meperidine in stopping rigors after receiving treatment with rituximab or ATG.
Methods
This study was a pharmacist-led, observational, single-center, retrospective chart review conducted at Atrium Health Wake Forest Baptist. The institutional review board and oncology protocol review committee at Atrium Health Wake Forest Baptist approved the study protocol. The patients were identified from a report generated through the electronic medical records (EMRs) to include patients whose treatment plans contained rituximab or ATG. The data were collected from a review of the medication records and nursing and providers’ notes. All end points were analyzed using descriptive statistics.
Patients were included in the review if they were aged ≥18 years, received rituximab or ATG at Atrium Health Wake Forest Baptist in either an infusion clinic or the inpatient setting for a hematology or oncology indication, and received ≥1 doses of 2-mg IV morphine for the treatment of rigors. The exclusion criteria included receiving a medication other than morphine for the treatment of the initial rigor event, rigors occurring after receiving a medication other than rituximab or ATG, patients already meeting the inclusion criteria for a previous episode of rigors, and the use of morphine for an indication other than rigors within the past 12 hours.
The primary study objective was to assess the efficacy of IV morphine in stopping rigors associated with rituximab and ATG treatments. For morphine, efficacy was defined as the stopping of rigors after the administration of IV morphine, without escalation to treatment with meperidine and within 60 minutes of the first dose. Morphine’s therapy benefit in ≥88% of the patients receiving treatment for rigors was considered comparable with the benefit of treatment with meperidine. The secondary study objectives were to determine the dose of morphine required to stop an episode of rigors, and to evaluate the time to the resolution of rigors after the administration of IV morphine or IV meperidine.
Results
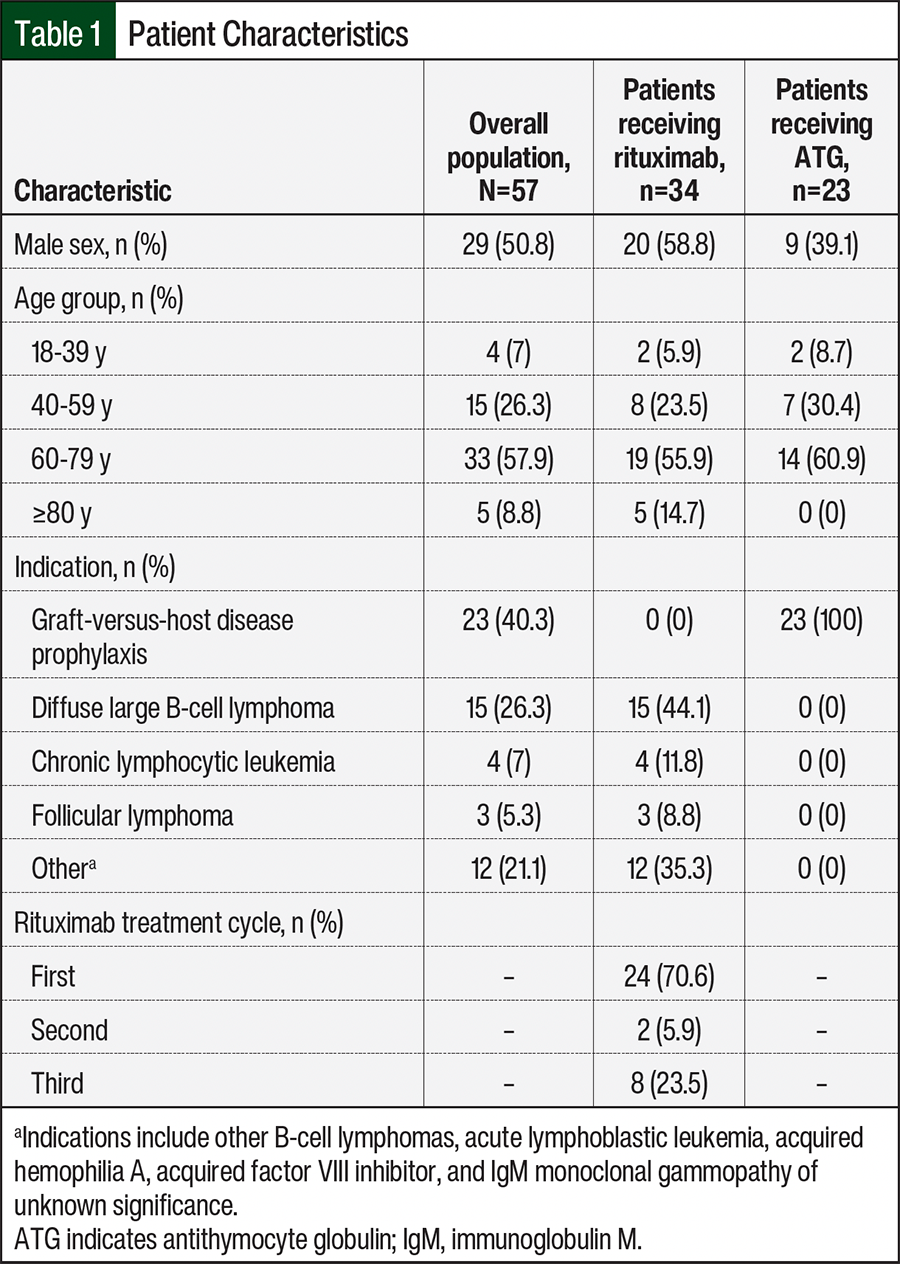
From July 2016 through July 2019, 620 individual patient charts were collected that included treatment with rituximab (n=574) or with ATG (n=46). Of the 620 patients who received either rituximab or ATG, 57 met the study’s inclusion criteria (rituximab, n=34; ATG, n=23). Patients were primarily excluded from the study because they did not have an episode of rigors or did not require intervention for rigors. The incidence rates for rigors in this 3-year period were 6% and 50% for rituximab and ATG, respectively. Most of the patients were aged between 60 years and 79 years and received rituximab for the treatment of diffuse large B-cell lymphoma or ATG for the prevention of graft-versus-host disease (Table 1). The patients who received rituximab were more likely to have an episode of rigors during their first treatment cycle (Table 1).
At the end of the study period, 1 patient in each treatment group required escalation from IV morphine to IV meperidine. The patient receiving rituximab who required escalation to IV meperidine did so after not responding to 2 doses of IV morphine (4 mg total) in a 30-minute period and had cessation of rigors after receiving 12.5 mg of IV meperidine. Meperidine was used in one other patient who received ATG subsequent to treatment with 1 dose of IV morphine (2 mg total); after chart review, it was unclear why treatment escalation to meperidine was required or why a second dose of morphine was not administered instead.
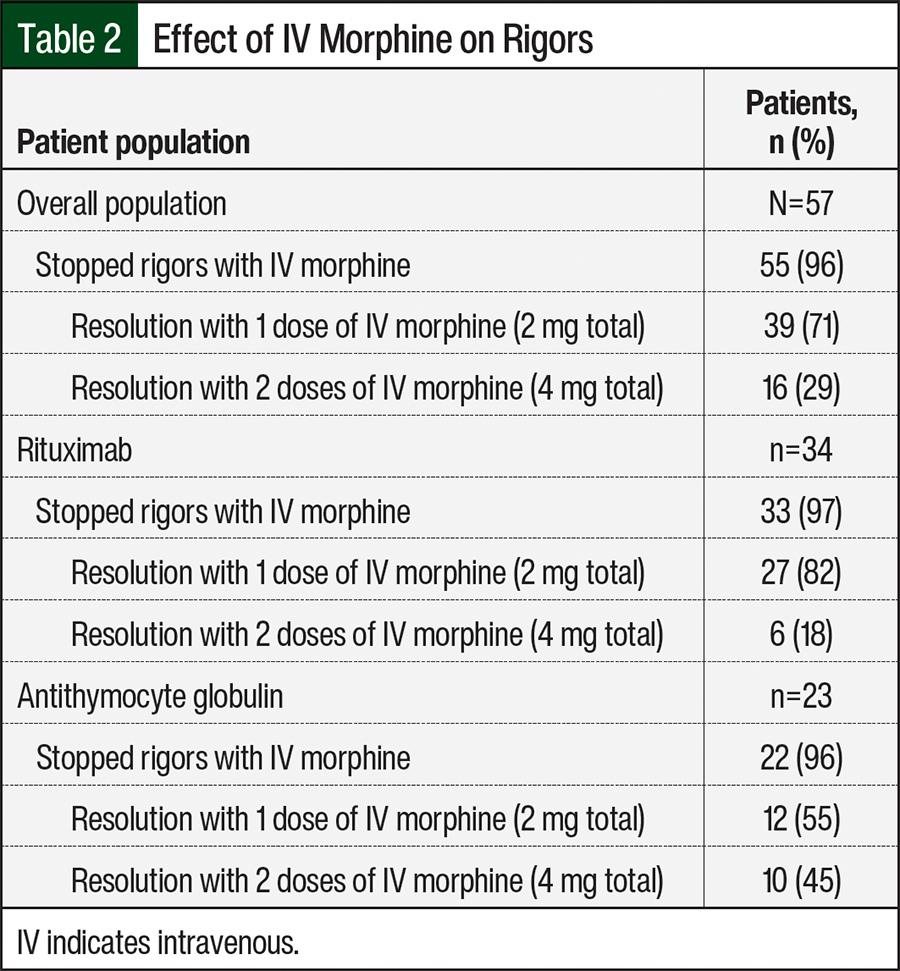
As shown in Table 2, most of the patients who had rigors after receiving treatment with rituximab had a response to 1 dose of IV morphine, whereas more patients who received ATG required a second dose of morphine compared with the patients in the rituximab group. The only adverse events (AEs) were in 2 patients who had fatigue after they received IV morphine. Neither of the 2 patients who received treatment with meperidine had any AEs. Because of a lack of standardized documentation, the time to the resolution of rigors after receiving morphine or meperidine could not be accurately assessed.
Concomitant opioid use was recorded for any opioid dose given within 12 hours of the administration of rituximab or ATG to evaluate if there was any impact on rigors or on the response to morphine. There was no distinguishable difference in the incidence of rigors between the patients who were already receiving opioids continuously and the patients who were not already receiving an opioid. However, of the patients who had rigors, most did not receive treatment with opioids within the past 12 hours (75.4% vs 24.6% of those who did).
Of the 2 patients who required treatment escalation to meperidine, the patient who received rituximab received 4 mg of oral hydromorphone total within the past 12 hours, whereas the patient who received ATG had no recent opioid exposure. Of the 14 patients who received opioids, 9 received oxycodone (5-60 mg orally), 2 received hydromorphone (0.6 mg IV in 1 patient who was receiving oxycodone and 4 mg orally in the other patient), 2 received fentanyl alone (50-100 µg), and 3 received tramadol (50-100 mg orally, 1 concurrently with oxycodone). None of the patients received hydrocodone, methadone, or morphine.
The receipt of morphine in the 12 hours before the start of rigors was an exclusion criterion for this evaluation; however, none of the patients were excluded from this study for recent morphine use. Most of the patients who received rituximab were premedicated with a 3-drug regimen containing diphenhydramine, acetaminophen, and a corticosteroid. The patients who received ATG were premedicated with a 2-drug regimen containing diphenhydramine and a corticosteroid. Both of the patients who required escalation to treatment with meperidine had received the standard institutional premedications for their respective agents. The corticosteroid premedication was omitted for only 4 of the 34 patients who received rituximab. Of the 23 patients who received ATG and had rigors, 1 patient did not receive premedication with a corticosteroid. In general, when comparing the patients who received all of the standard premedications per our institution’s recommendations with the 5 patients who did not receive the standard premedications, there was no discernible influence on morphine’s efficacy in stopping rigors.
Discussion
There is a paucity of information regarding the treatment of rigors associated with rituximab and ATG, with most data being extrapolated from patients who had postanesthesia rigors. This retrospective analysis provides supporting evidence that treatment with morphine is an effective alternative to meperidine, and the use of morphine may avoid the potential AEs associated with meperidine. However, this study was not designed to prove the noninferiority of morphine versus meperidine because there is a low rate of meperidine use at our institution. Although there remains a concern for the accumulation of morphine and meperidine in the setting of renal dysfunction, the primary concern for morphine accumulation is respiratory depression, which treatment with meperidine in lieu of morphine would not mitigate, and respiratory depression was not observed in this review.
Of the 57 patients who received morphine, fatigue was the only AE documented, which occurred in 2 patients. Although no AEs were reported with the use of meperidine in this study, utilization was too low to draw any definitive conclusions. The inability to determine the time to response for the treatment of rigors after morphine administration also limits the assessment of how quick a resolution can be achieved or expected. However, most of the patients only required 1 dose of morphine, and 8 of the 16 patients who received 2 doses received the second dose of morphine at least 1 hour after the first dose, which potentially results from the resumption of treatment with rituximab or ATG and the recurrence of rigors.
A strength of this study was the collection of concomitant opioid use, as it was unknown if actively receiving opioids could impact whether patients had rigors or if they would be less responsive to treatment with morphine. However, a lack of response to morphine was not observed in patients receiving other opioid medications; only 1 of the 14 patients who received treatment with opioids required treatment escalation to meperidine.
Limitations
A limitation of this study was the low number of patients who were able to be evaluated because of a lower incidence of rigors after receiving rituximab in this study population compared with historical rates in clinical trials.4 The low incidence of rigors after rituximab treatment could be related to the cycle of treatment during the time frame of the study; this study aligns with other studies of rituximab in that there are higher rates of AEs with earlier cycles compared with later cycles.4 Also, differences in supportive-care medications (eg, premedications and pain medications) could have resulted in a lower number of events. However, although this study has a small population, having only 1 patient in each arm who did not achieve a response with morphine was promising for the continued use of this protocol.
One of the key secondary end points for this study was to evaluate the dose of morphine that is required to stop an episode of rigors. The standard protocol at this institution is to administer 2 mg of IV morphine, followed by another 2 mg of IV morphine if a response has not been achieved. Comparatively, fewer patients in the rituximab group required the additional 2 mg of IV morphine than patients in the ATG group. Because there was a low incidence of patients not responding to a second dose of IV morphine, we currently feel that the dosing schematic is appropriate for these patients. However, this study was not able to assess the severity of the episode of rigors, which could theoretically have required repeated doses of morphine or potentially a higher dose of morphine to achieve an adequate response.
The other secondary end point of time to the resolution of rigors could also impact dosing decisions. One limitation of this study was the inability to accurately evaluate the patients’ time to response because there was no standardized documentation in the EMR. Future prospective studies could be performed with a short monitoring form entered into the EMR to more accurately take into account the severity of rigors and to document the timing of the resolution of rigors to better elucidate when a subsequent dose of morphine should be administered.
Conclusion
This retrospective chart review provides supporting evidence to continue the use of IV morphine as an alternative to treatment with meperidine for rituximab- or ATG-related rigors at our institution. The doses of morphine that were used were well tolerated and are easily accessible to nursing staff if stocked via automated drug cabinets. Further studies could be performed to assess the benefit of using morphine for the treatment of rigors related to other chemotherapies or immunotherapies in oncology.
Author Disclosure Statement
Dr Lucas, Dr Edenfield, Dr Kennedy, and Dr Anders have no conflicts of interest to report.
References
- Lopez MB. Postanaesthetic shivering–from pathophysiology to prevention. Rom J Anaesth Intensive Care. 2018;25:73-81.
- Boye J, Elter T, Engert A. An overview of the current clinical use of the anti-CD20 monoclonal antibody rituximab. Ann Oncol. 2003;14:520-535. Erratum in: Ann Oncol. 2003;14:967.
- Pihusch R, Holler E, Mühlbayer D, et al. The impact of anti-thymocyte globulin on short-term toxicity after allogeneic stem cell transplantation. Bone Marrow Transplant. 2002;30:347-354.
- Rituxan (rituximab) injection, for intravenous use [prescribing information]. Genentech; December 2021. Accessed June 19, 2024. www.gene.com/download/pdf/rituxan_prescribing.pdf
- Thymoglobulin (anti-thymocyte globulin [rabbit]) injection, for intravenous use [prescribing information]. Genzyme Corporation; March 2023. Accessed June 19, 2024. https://products.sanofi.us/thymoglobulin/thymoglobulin.pdf
- Kranke P, Eberhart LH, Roewer N, Tramèr MR. Pharmacological treatment of postoperative shivering: a quantitative systematic review of randomized controlled trials. Anesth Analg. 2002;94:453-460.
- Latta KS, Ginsberg B, Barkin RL. Meperidine: a critical review. Am J Ther. 2002;9:53-68.
- Institute for Safe Medication Practices. High alert medication feature: reducing patient harm from opiates. February 22, 2007. Accessed March 28, 2022. (Requires subscription to access.) www.ismp.org/resources/high-alert-medication-feature-reducing-patient-harm-opiates
- Timmers GJ, Zweegman S, Simoons-Smit AM, et al. Amphotericin B colloidal dispersion (Amphocil) vs fluconazole for the prevention of fungal infections in neutropenic patients: data of a prematurely stopped clinical trial. Bone Marrow Transplant. 2000;25:879-884.