5-fluorouracil (5-FU) is a fluoropyrimidine chemotherapeutic agent that forms the backbone of various regimens that are frequently used in the treatment of malignancy.1,2 In the treatment of solid tumors, 5-FU has been used for more than 40 years and is the third most often used agent internationally, and fluoropyrimidine-based therapies are the most frequently administered systemic agents at National Comprehensive Cancer Network (NCCN) Member Institutions.1-3 As a frequently used chemotherapy, the mechanism of action and adverse effect profile of 5-FU has been well-described in the literature.
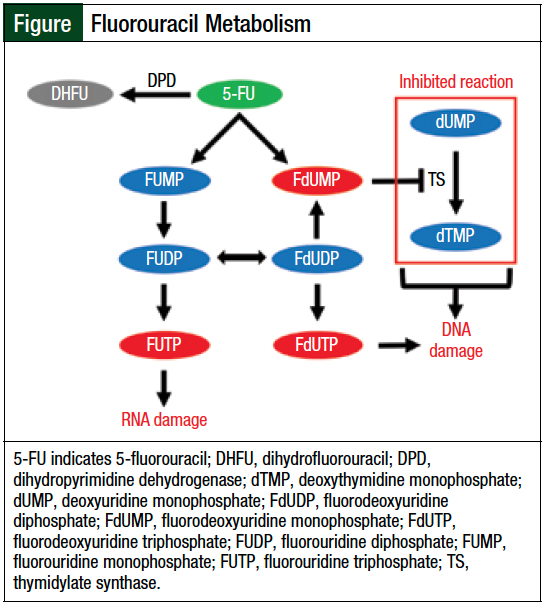
All DNA antimetabolite agents exhibit similar mechanisms characteristic to this drug class. 5-FU is a pyrimidine analog that inhibits RNA and DNA synthesis through several mechanisms.4 5-FU is converted into 1 of 3 active metabolites that have pharmacodynamic effects within the cell, including fluorouridine triphosphate, fluorodeoxyuridine monophosphate, and fluorodeoxyuridine triphosphate (Figure).4
Dihydropyrimidine dehydrogenase is an enzyme that is responsible for converting approximately 85% of 5-FU into inactive 5-fluorodihydrouracil.5 Dihydropyrimidine dehydrogenase deficiency occurs in 4% to 7% of the population, and can be achieved through several distinct mutations in the DPYD gene.5,6 Some DPYD polymorphisms will lead to decreased metabolism and/or decreased or absent enzyme activity, resulting in increased 5-FU side effects.5,6 In addition, these patients with dihydropyrimidine dehydrogenase deficiency will be at increased risk for rare fluoropyrimidine-induced cardiotoxicity and neurotoxicity.5,6
The common adverse effects of 5-FU that have been observed with the modified FOLFOX-6 (folinic acid, fluorouracil, and oxaliplatin) regimen include nausea (29.1%), mucositis (33.3%), diarrhea (23.6%), and vomiting (17.0%).7 The mechanisms of these adverse events are relatively well-understood, but the rarer cardiac adverse events are not well-elucidated.4
Cardiac adverse events have been documented to be manifesting as benign chest pain or acute coronary syndrome.8 Of note, all chest pain should warrant emergent medical attention. Benign chest pain in this context refers to angina without any troponin leak or any other cardiac abnormalities requiring intervention; such chest pain resolves with the cessation of fluoropyrimidine administration. Other reports have described even less common cardiac adverse events, such as arrhythmias, myocarditis, pericarditis, and heart failure.9-11 Such cases of acute-onset heart failure most often involve patients who have no history of cardiac issues and describe hypoactive left-ventricular function on echocardiogram without structural changes.11
For patients who have 5-FU or capecitabine (an orally administered fluoropyrimidine) overdoses or severe life-threatening adverse reactions, especially involving the cardiovascular or central nervous systems, rescue with uridine triacetate should be attempted.12 Not all patients who have cardiac adverse events, however, require rescue with uridine triacetate; some fluoropyrimidine-related chest pain is self-limiting and likely has no long-term consequences. The repletion of uridine competes with the fluorouridine triphosphate metabolite and helps to restore normal RNA synthesis and processing to mitigate adverse events. Uridine triacetate has been the most extensively studied, and is the most effective, when initiated within 96 hours of the end of a 5-FU infusion or capecitabine administration.12
Uridine triacetate is dosed at 10 g orally every 6 hours for 20 doses total.12 When used in this manner, 96% of patients who overdosed may survive, with reversal of cardiotoxicity and neurotoxicity and prevention of mucositis and leukopenia.12 More recent reports provide limited data for rescue to be initiated after 96 hours; therefore, late rescue may be considered on a case-by-case basis.13,14 Once uridine triacetate rescue therapy is initiated, the patient should complete all 20 doses, regardless of a change in the signs or symptoms of adverse reactions.
The current report presents a case of acute-onset, decompensated congestive heart failure in a patient who received 5-FU–based chemotherapy for adjuvant treatment of stage III adenocarcinoma of the colon and rescue treatment with uridine triacetate.
Case Report
A 59-year-old Caucasian woman was admitted to the cardiovascular intensive care unit (ICU) at the University of Kansas Hospital with tachycardia and hypotension. Her medical history was significant for stage IIIC (TNM stage T4b N2b M0) adenocarcinoma of the colon, complex regional pain syndrome, and osteoarthritis. Her surgical history included an unspecified back surgery and recent appendectomy, cholecystectomy, and colectomy. She reported rare alcohol use and has been smoking 3 cigarettes daily for “many years.”
The patient had no known drug or food allergies, and her medication list before her ICU admission included alprazolam (0.25 mg at bedtime, as needed), cholecalciferol (1000 units once daily), dexamethasone (8 mg daily on days 2 and 3 of each chemotherapy cycle), mexiletine (150 mg twice daily), mometasone (100 mcg in each nostril daily), ondansetron (8 mg daily on days 2 and 3 of each chemotherapy cycle), oxycodone sustained release (40 mg every 12 hours), and prochlorperazine (10 mg every 6 hours, as needed).
Mexiletine was being used off label by her primary care physician for the management of chronic pain associated with complex regional pain syndrome. Her oncologist had prescribed 6 months of adjuvant chemotherapy with 5-FU and oxaliplatin, as administered in the modified FOLFOX-6 regimen (the complete dosing and schedule of the regimen are shown in Table 1), after complete resection of her tumor, which is the standard of care per the NCCN guidelines.2
The patient had received modified FOLFOX-6 infusions without incident on day 1 of her treatment while at the cancer center and was sent home with a 5-FU ambulatory pump. She returned on day 3 to be unhooked from the now-empty 5-FU pump, and she reported dizziness to her nurse. An examination revealed a heart rate of 120 beats per minute, blood pressure of 82/43 mm Hg, breathing of 25 respirations per minute, and her body temperature was 36.6°C. A provider at the cancer center examined the patient and recommended that her caregiver take her directly to the emergency department.
In the emergency department, a full workup was performed. Her complete blood count with differential was unremarkable. The patient’s blood chemistry was significant for serum creatinine of 1.98 mg/dL (baseline, 0.6-0.7 mg/dL), lactic acid of 4.5 mg/dL, a troponin level of 0.47 ng/mL, and a brain naturetic peptide level of 1672 ng/L. An electrocardiogram showed normal sinus rhythm, with no ST-segment or T-wave abnormalities, and an echocardiogram showed left-ventricle ejection fraction (LVEF) of 25%.
The patient had not had a previous echocardiogram to compare against the latest echocardiogram but it was presumed that she did not have existing structural nor functional cardiac abnormalities before this event. A review of symptoms was only positive for dizziness (grade 1), fatigue (grade 1), nausea (grade 2), and vomiting (grade 2). She declared that she had chest pain (grade 2) that was sharp, substernal, nonradiating, and nonexertional, and that waxed and waned over several hours daily since starting the 5-FU infusion.
The patient was transferred to the cardiovascular ICU for the management of acute, decompensated congestive heart failure, where she stayed for 7 days. Over the next 24 hours, her troponin level rose to a peak of 1.53 ng/mL. Her serum creatinine level continued to rise for 4 days after hospital admission, to 4.53 mg/dL, before returning to baseline over the next several days. During her stay in the cardiovascular ICU, she required treatment with dobutamine for 5 days, norepinephrine and vasopressin for 2 days each, and a furosemide infusion for 4 days.
A heparin infusion was administered for 48 hours in accordance with guidelines for the management of type II non–ST-segment elevation myocardial infarction.15 The patient was taken to the cardiac catheterization laboratory for a diagnostic right heart catheterization. The results were consistent with severe left-ventricle systolic dysfunction, but no atherosclerotic disease or cardiomyopathy was found.
Perhaps the most important intervention during the patient’s hospitalization was the initiation of uridine triacetate. Because of the unique presentation and rarity of cardiotoxicity associated with fluoropyrimidines, the etiology of the cardiogenic shock was not recognized until 2 or 3 days after admission. Uridine triacetate should be initiated within 96 hours of the end of the 5-FU infusion to be most effective.
Uridine triacetate was urgently acquired; it is not kept in stock because of cost and low frequency of use, as well as the availability of rapid ordering and delivery. Treatment with uridine triacetate was started 82.5 hours from the end of the 5-FU infusion. The patient received 10 g every 6 hours for a total of 20 doses. The average wholesale price for a 5-FU single 10-g dose is $4815.60, totaling $96,312 for a full 20-dose course of rescue therapy.16
The patient was tested for 3 genotypes associated with dihydropyrimidine dehydrogenase deficiency: 2846A>T, DYPD*13 (also known as 1679T>G), and DYPD*2A (also known as 1905+1G>A). None of these gene mutations, which are known alleles that express decreased or absent enzyme activity, were detected in this patient, which does not necessarily preclude her from having other untested or unknown dihydropyrimidine dehydrogenase mutations, which may cause the same effect. However, it was discovered that she harbored 2 distinct thymidylate synthase alterations, both of which are associated with decreased thymidylate synthase expression of enzyme and therefore are linked to increased risk for fluoropyrimidine-related adverse reactions. The mutation that was detected was 6 bp deletion allele (also known as rs34489327), and the patient had a 2R/2R thymidylate synthase genotype.
Once the patient was tapered off of all vasopressors and other infusions, her LVEF improved to 50% 6 days after the initial echocardiogram; on recheck 6 months later, her LVEF remained within normal limits, at 55% to 60%. She was discharged with new prescriptions recommended by the cardiology consult service, including lisinopril (2.5 mg orally once daily), metoprolol succinate (12.5 mg orally once daily), atorvastatin (80 mg orally once daily), aspirin (81 mg orally once daily), spironolactone (25 mg orally once daily), and furosemide (20 mg orally every other day).
At the outpatient oncology follow-up, it was decided that the patient should switch to single-agent pembrolizumab to complete her adjuvant treatment, because she had a microsatellite instability-high tumor. This is not a standard-of-care adjuvant treatment for stage III colon cancer,2 but because a rechallenge with fluoropyrimidines was not an option, the pembrolizumab approach was approved by the University of Kansas Health System Pharmacy & Therapeutics Committee.
The patient completed 24 months of adjuvant treatment with pembrolizumab (200 mg intravenously every 3 weeks). She has been under surveillance for 12 months since completing her adjuvant pembrolizumab treatment and so far has no evidence of cancer recurrence.
Discussion
Modified FOLFOX-6 is frequently used for the treatment of various gastrointestinal malignancies; most pertinent to this case, the regimen includes the administration of 5-FU over 46 hours (Table 1).2 In the outpatient setting, this regimen requires the patient to be connected to an ambulatory infusion pump, and the patient returns to the clinic on day 3 of the cycle, to be unhooked from the pump.
Although overdoses may be physically apparent on review of compounds or pumps, it is more difficult to identify patients who have unusually severe adverse reactions that warrant rescue therapy. Pharmacogenetic testing is one modality by which some patients may be identified as being susceptible to fluoropyrimidine side effects. However, these genetic tests often take weeks to get the results, which requires decisions about side-effect management without this key information.
Although there are data indicating that the universal testing of all patients for dihydropyrimidine dehydrogenase deficiency before treatment with fluoropyrimidines may be cost-effective, the NCCN does not recommend this practice at this time.2
Genetic variations in the thymidylate synthase genes can also cause patients to have altered responses to fluoropyrimidines, which may manifest as a decreased response to treatment or increased side effects.6,17 Mutations resulting in the increased expression of thymidylate synthase or increased activity of the enzyme will compete with fluoropyrimidines, and patients with these mutations may have a decreased response to therapy but also fewer side effects.17 Conversely, mutations resulting in the decreased expression of thymidylate synthase or the decreased activity of the enzyme will potentiate the effects of fluoropyrimidines, and patients with these mutations may have an increased response to therapy, as well as more side effects.17
Factors that may identify patients who are at increased risk for fluoropyrimidine side effects other than assessing for genetic mutations and overdoses have been studied.18 Early and severe side effects are increased with decreased creatinine clearance, increased body surface area, and advanced age. Finally, pretreatment uracil levels, although frequently checked, may be an additional risk factor for severe side effects.18
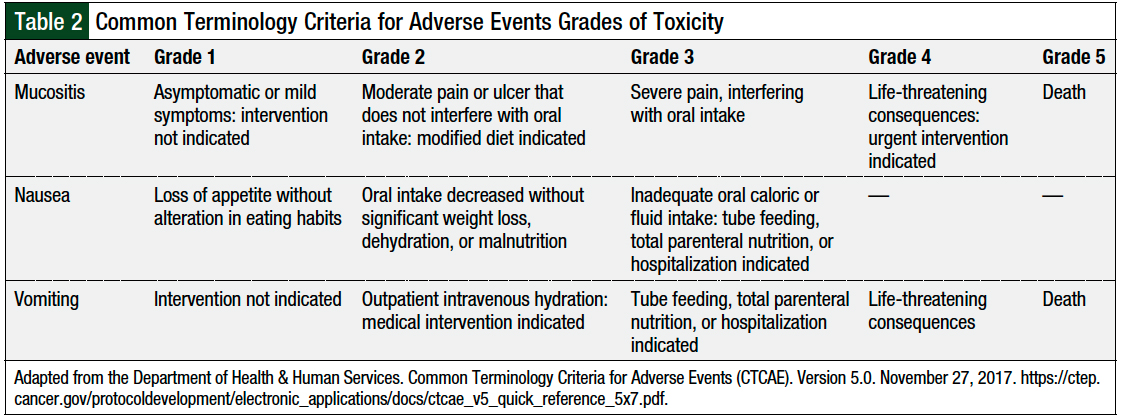
Prompt evaluation of the presenting signs and symptoms of side effects in all patients with cancer is crucial to proper disease management. Certain warning signs may help distinguish the patients who receive 5-FU and will have severe and escalating adverse reactions from those who have expected and stable reactions. Some of the expected grade 1 or 2 adverse events after the administration of the modified FOLFOX-6 regimen include diarrhea, nausea, vomiting, mucositis, and peripheral neuropathy.7 The Common Terminology Criteria for Adverse Events grading of the adverse events attributed to 5-FU are listed in Table 2.19
The rates of grade 3 or 4 adverse events for the modified FOLFOX-6 regimen are approximately 33.3% for diarrhea, 2.1% for nausea, 14.3% for vomiting, and 11.4% for mucositis.7 Although these percentages are approximations, they highlight which adverse reactions are uncommon to present as severe reactions (ie, grade 3 or 4). Low grades of nausea and vomiting likely result from improved antiemetic agents and regimens, but notably, mucositis infrequently presents as grade 3 or 4.
Vigilance regarding new or worsening cardiac conditions (eg, heart failure or atrial fibrillation) and possible altered mental status help to clarify the evaluation of these conditions, because these are not expected side effects of fluoropyrimidine therapy. Assessing the dose appropriateness and timing of the modified FOLFOX-6 regimen, namely, whether doses and infusion rates were suitable, and if this was the first or subsequent cycle of treatment, should be the first step in evaluating severe adverse events.
Altogether, through the use of objective measures of known risk factors, assessing the grades of adverse events, noting the presence of rare side effects, and the timing of the presentation, the clinician may be able to identify patients who overdosed or who have polymorphisms that may cause life-threatening fluoropyrimidine side effects and who would greatly benefit from rescue treatment with uridine triacetate.
The patient in this case report might not have been identified by any of the risk factors previously described, but her presenting signs and symptoms of chest pain, tachycardia, and hypotension in the setting of no cardiac history after her first dose of 5-FU served as a crucial signal of the etiology of her heart failure.
Conclusion
The nature of new or worsening chest pain and other cardiac symptoms universally warrants seeking emergent medical attention, and all patients should be educated on this. Although severe cardiotoxicity resulting from fluoropyrimidines is rare, it requires timely and decisive management. Early identification of severe adverse events and rescue therapy with uridine triacetate are highly effective in reducing morbidity and mortality. Molecular testing for dihydropyrimidine dehydrogenase deficiency and thymidylate synthase mutations do not result in a timely manner, and this information is not consistently available before treatment initiation with fluoropyrimidines. Therefore, clinical vigilance is currently the primary method for the identification and optimal management of these uncommon, life-threatening adverse events associated with 5-FU therapy.
Author Disclosure Statement
Dr Cascone has no conflicts of interest to report.
References
- Sara JD, Kaur J, Khodadadi R, et al. 5-fluorouracil and cardiotoxicity: a review. Ther Adv Med Oncol. 2018;10:1-18.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Colon Cancer. Version 2.2021. January 21, 2021. www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed June 2, 2021.
- Grem JL. 5-fluorouracil: forty-plus and still ticking. A review of its preclinical and clinical development. Invest New Drugs. 2000;18:299-313.
- Miura K, Kinouchi M, Ishida K, et al. 5-FU metabolism in cancer and orally-administrable 5-FU drugs. Cancers (Basel). 2010;2:1717-1730.
- Ezzeldin H, Diasio R. Dihydropyrimidine dehydrogenase deficiency, a pharmacogenetic syndrome associated with potentially life-threatening toxicity following 5-fluorouracil administration. Clin Colorectal Cancer. 2004;4:181-189.
- Mercier C, Ciccolini J. Severe or lethal toxicities upon capecitabine intake: is DPYD genetic polymorphism the ideal culprit? Trends Pharmacol Sci. 2007;28:597-598.
- Negarandeh R, Salehifar E, Saghafi F, et al. Evaluation of adverse effects of chemotherapy regimens of 5-fluoropyrimidines derivatives and their association with DPYD polymorphisms in colorectal cancer patients. BMC Cancer. 2020;20:560.
- Anand AJ. Fluorouracil cardiotoxicity. Ann Pharmacother. 1994;28:374-378.
- Stewart T, Pavlakis N, Ward M. Cardiotoxicity with 5-fluorouracil and capecitabine: more than just vasospastic angina. Intern Med J. 2010;40:303-307.
- Çalik AN, Çeliker E, Velibey Y, et al. Initial dose effect of 5-fluorouracil: rapidly improving severe, acute toxic myopericarditis. Am J Emerg Med. 2012;30:257.e1-257.e3.
- Robben NC, Pippas AW, Moore JO. The syndrome of 5-fluorouracil cardiotoxicity: an elusive cardiopathy. Cancer. 1993;71:493-509.
- Ma WW, Saif MW, El-Rayes BF, et al. Emergency use of uridine triacetate for the prevention and treatment of life-threatening 5-fluorouracil and capecitabine toxicity. Cancer. 2017;123:345-356.
- Baldeo C, Vishnu P, Mody K, Kasi PM. Uridine triacetate for severe 5-fluorouracil toxicity in a patient with thymidylate synthase gene variation: potential pharmacogenomic implications. SAGE Open Med Case Rep. 2018;6:1-4.
- Zurayk M, Keung YK, Yu D, Hu EHL. Successful use of uridine triacetate (Vistogard) three weeks after capecitabine in a patient with homozygous dihydropyrimidine dehydrogenase mutation: a case report and review of the literature. J Oncol Pharm Pract. 2019;25:234-238.
- Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the Management of Patients with Non–ST-Elevation Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;64:e139-e228. Erratum in: J Am Coll Cardiol. 2014;64:2713-2714.
- Uridine triacetate. Lexi-Drugs. Lexicomp. Hudson, OH: Wolters Kluwer. Updated December 13, 2020. http://online.lexi.com. Accessed June 2, 2021. [Subscription required.]
- Peters GJ, van Groeningen CJ, van der Wilt CL, et al. Time course of inhibition of thymidylate synthase in patients treated with fluorouracil and leucovorin. Semin Oncol. 1992;19(suppl 3):26-35.
- Meulendijks D, van Hasselt JGC, Huitema ADR, et al. Renal function, body surface area, and age are associated with risk of early-onset fluoropyrimidine-associated toxicity in patients treated with capecitabine-based anticancer regimens in daily clinical care. Eur J Cancer. 2016;54:120-130.
- Department of Health & Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf. Accessed February 26, 2021.