Treatment for breast cancer has become especially patient-specific, with genetic factors, tumor size, and the stage of cancer influencing the initial and subsequent treatment regimens. Many patients who receive chemotherapy for breast cancer are treated with a taxane, either sequentially or in combination with an anthracycline and an alkylating agent.1 The majority of patients tolerate taxane-containing chemotherapy regimens well; however, some patients have hypersensitivity reactions that require a change in therapy.2 The symptoms of taxane hypersensitivity reactions include acute changes in blood pressure (hypertension or hypotension), syncope, dyspnea, throat tightness, chest pain, back pain, and abdominal pain.2
The etiology of taxane hypersensitivity reactions remains poorly understood.2 Three mechanisms have been proposed to account for taxane hypersensitivity reactions, including complement activation induced by Cremophor EL (formulated with paclitaxel) and polysorbate 80 (formulated with docetaxel), which triggers basophil and/or mast-cell activation3,4; histamine release through an effect of paclitaxel on basophils5; and an immunoglobulin (Ig)E- or IgG-mediated mechanism directed against the taxane or the solvent.6
Premedication with corticosteroids, antihistamines, and H2 receptor antagonists reduces the incidence of hypersensitivity reactions in patients who are receiving taxanes,2 yet severe reactions to paclitaxel and docetaxel still occur in approximately 1% to 5% of patients who received appropriate premedications.2,7
In severe cases of taxane-related hypersensitivity reactions, one strategy to prevent the recurrence of these reactions is to use nab-paclitaxel in place of other taxanes.2 Nab-paclitaxel does not contain Cremophor EL or polysorbate 80,8 the likely causes of hypersensitivity reactions to a taxane.3,4 Instead, nab-paclitaxel is an albumin-bound form of paclitaxel, formulated as a powder for suspension reconstituted with 0.9% sodium chloride.9
In the treatment of cancer, nab-paclitaxel that is routinely given without premedications has a much lower rate of hypersensitivity reactions than paclitaxel that is given with premedications.10,11 However, there is a paucity of data describing how to substitute appropriately nab-paclitaxel for other taxanes.12,13
The National Comprehensive Cancer Network’s breast cancer guidelines recommend substituting nab-paclitaxel for paclitaxel or for docetaxel in cases of hypersensitivity reactions1; however, most nab-paclitaxel substitutions are not supported by evidence from randomized controlled trials.
Case reports and small clinical trials have described substituting nab-paclitaxel for other taxanes in the treatment of gynecologic cancers12,13 and breast cancers,14,15 and have shown that nab-paclitaxel is safe and well-tolerated as a substitute to other taxanes. Here we report 2 cases of using nab-paclitaxel in patients with breast cancer who had an adverse reaction to a taxane, 1 patient with a hypersensitivity reaction to paclitaxel and 1 patient with a hypersensitivity reaction to docetaxel.
Patient 1
A 38-year-old Hispanic woman presented to our clinic and was diagnosed with unilateral grade 3 estrogen receptor (ER)-negative, progesterone receptor (PR)-negative, human epidermal growth factor receptor 2 (ERBB2/HER2)-negative (1+, negative), triple-negative invasive ductal adenocarcinoma with ductal carcinoma in situ measuring 1.1 × 0.7 × 1.1 cm, with no lymph node involvement. Staging showed clinical stage IIB (T2cN0M0G3) premenopausal triple-negative invasive ductal carcinoma of 1 breast.
Genetic testing showed that the patient was not a carrier of any actionable mutations, including the ATM, BARD1, BRCA1, BRCA2, CHEK2, PALB2, and TP53 genes. Her medical history included obesity, with a body mass index (BMI) of 35 kg/m2, and hypertension.
The patient had no known drug allergies before starting chemotherapy. She had no pertinent surgical history before the cancer diagnosis, and no history of tobacco use.
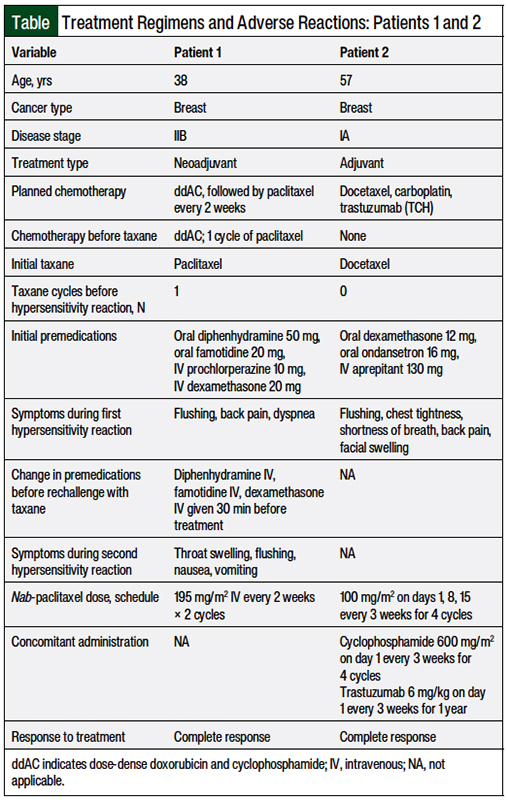
The initial treatment plan consisted of neoadjuvant chemotherapy with intravenous (IV) dose-dense doxorubicin 60 mg/m2 and IV cyclophosphamide 600 mg/m2 (ddAC) every 2 weeks for 4 cycles, followed by paclitaxel (175 mg/m2 every 2 weeks for 4 cycles). All the chemotherapy doses were based on her actual body weight rather than on adjusted body weight. After completing chemotherapy, the patient had a partial mastectomy and received adjuvant radiation therapy.
The patient tolerated 4 cycles of ddAC without incident, and she received appropriate antiemesis prophylaxis with dexamethasone, ondansetron, lorazepam, and IV aprepitant emulsion. She also received pegfilgrastim therapy appropriately to prevent neutropenia.
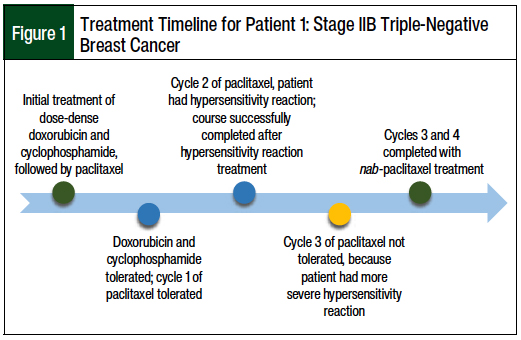
The patient tolerated cycle 1 of paclitaxel without incident, and she received appropriate premedications with oral diphenhydramine 50 mg, oral famotidine 20 mg, IV prochlorperazine 10 mg, and IV dexamethasone 20 mg 30 minutes before starting treatment with paclitaxel. She received pegfilgrastim 6-mg subcutaneous autoinjector activated 24 hours after her infusion.
During cycle 2 of paclitaxel, the patient again received appropriate premedications with oral diphenhydramine 50 mg, oral famotidine 20 mg, and IV dexamethasone 20 mg 30 minutes before starting paclitaxel. However, 22 minutes (and 37 mg) into the 300-mg paclitaxel infusion, she had a hypersensitivity reaction that included the symptoms of flushing, back pain, and dyspnea, which is defined as grade 2 Common Terminology Criteria for Adverse Events (CTCAE).16
On noting the patient’s symptoms, the nurse stopped the infusion and the provider was notified. The symptoms resolved with the administration of IV methylprednisolone 125 mg, IV diphenhydramine 25 mg, and IV famotidine 20 mg, as indicated by the health system’s standard operating procedure for the treatment of moderate chemotherapy hypersensitivity or infusion reactions. The infusion was resumed at the initial rate, and the patient finished the treatment cycle without further incidence.
Before cycle 3 of paclitaxel, the patient received premedication with IV diphenhydramine 50 mg and IV famotidine 20 mg (as opposed to oral formulations of these medications, which were given with cycle 2) to ensure faster and adequate premedication in case the patient had any absorption difficulties with oral medications and IV dexamethasone 20 mg. Although a more aggressive steroid premedication regimen could have been used, we believed that changing from oral to IV premedications would be sufficient.
After infusing paclitaxel for 8 minutes (14.6 mg), the patient began to have throat swelling, flushing, and nausea. The infusion was stopped, and the protocol for severe reaction was initiated; the patient received a normal saline continuous infusion, IV diphenhydramine 25 mg, and IV methylprednisolone 125 mg. The patient continued to report throat tightness, and intramuscular epinephrine 0.3 mg was administered.
The patient then had nausea and 1 episode of emesis, for which IV prochlorperazine 10 mg and IV famotidine 20 mg were administered. The patient’s symptoms resolved 33 minutes after they began. The paclitaxel infusion was not restarted, and paclitaxel treatment was discontinued.
Because the patient was unable to complete cycle 3 of paclitaxel, she resumed cycle 3 with nab-paclitaxel 5 days after her hypersensitivity reaction to paclitaxel. The new treatment plan for cycles 3 and 4 consisted of pretreatment with IV prochlorperazine 10 mg, followed by dose-dense IV nab-paclitaxel 195 mg/m2 every 2 weeks, with pegfilgrastim 6-mg autoinjector placed after the completion of the nab-paclitaxel infusion.
After discussion with colleagues at our parent hospital, the dose of nab-paclitaxel of 195 mg/m2 every 2 weeks was initiated to maintain a dose-dense strategy. Nab-paclitaxel 260 mg/m2, as previously used successfully for adjuvant treatment,17,18 has proved too toxic for patients at our institutions, thus an empiric 25% dose reduction has been instituted and used for patients who receive nab-paclitaxel on a dose-dense schedule.
The treatment course for this patient case is shown in Figure 1. The patient tolerated treatment well and had no adverse reactions.
After completing neoadjuvant treatment, she underwent a localized right lumpectomy and right sentinel lymph node biopsy and had no residual disease in the breast. She recovered well from surgery and reported some residual neuropathy in her fingertips 2 months after the completion of neoadjuvant nab-paclitaxel treatment.
Because the patient underwent a lumpectomy, she required radiation therapy with 40 Gy to the whole breast, followed by a 10-Gy tumor bed boost, for a total dose of 50 Gy over 4 weeks. All the regimens she received and her hypersensitivity reactions are listed in the Table.
As of this writing, 18 months after completing radiation therapy and 24 months after completing chemotherapy, the patient has no evidence of disease, and requires no further treatment. She had a follow-up mammogram 16 months after completing radiation therapy that showed no evidence of recurrent malignancy.
Patient 2
A 57-year-old Hispanic woman presented to our clinic and was diagnosed with stage IA (T1cN0M0G3) ER-positive (66%), PR-negative, ERBB2/HER2-positive (3+) invasive ductal carcinoma in situ measuring 1.6 cm, with no lymph node involvement. Her medical history included arthritis; obesity, with a BMI of 46 kg/m2; depression; prediabetes controlled with diet; and hypothyroidism. She had no pertinent surgical history before the cancer diagnosis, consumed 2 beers per week, and had a history of less than a 1 pack-year cigarette use.
After cancer staging, the patient underwent a lumpectomy and sentinel lymph node biopsy, followed by re-excision because of a retained clip 48 days later. After re-excision, the tumor margins were negative.
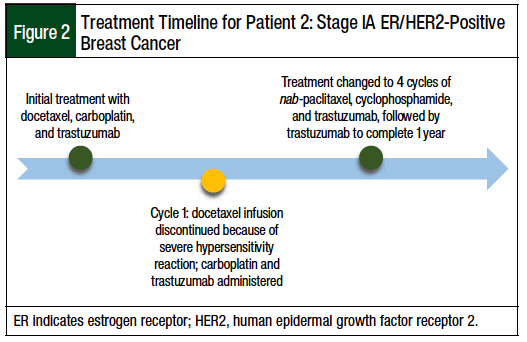
Because of the patient’s ERBB2/HER2-positive status, adjuvant chemotherapy was initiated. Her baseline left-ventricular ejection fraction was >55%. Initially, she started treatment with IV docetaxel 75 mg/m2, IV carboplatin area under the curve 6, and IV trastuzumab 8 mg/kg (TCH) once every 3 weeks for 6 cycles, with pegfilgrastim planned for each cycle, followed by IV trastuzumab 6 mg/kg every 3 weeks to complete 1 year of therapy. All the chemotherapy doses were based on her actual body weight rather than on adjusted body weight. The patient was also scheduled to receive radiation therapy after completion of 6 cycles of the TCH regimen.
During cycle 1 of TCH, the patient received oral dexamethasone 12 mg, oral ondansetron 16 mg, and IV aprepitant emulsion 130 mg before the docetaxel infusion. Docetaxel was stopped 5 minutes (9 mg) into the 150-mg infusion after the patient reported symptoms consisting of flushing, chest tightness, shortness of breath, back pain, and facial swelling, which were classified as grade 2 CTCAE hypersensitivity reaction.16
We initiated our institution’s severe reaction protocol, and the patient received a normal saline continuous infusion, IV diphenhydramine 25 mg, IV methylprednisolone 125 mg, IV famotidine 20 mg, and intramuscular epinephrine 0.3 mg. Her symptoms resolved completely after 30 minutes. The team decided not to rechallenge docetaxel, because the reaction was severe. After resolution of the reaction, the patient received carboplatin and trastuzumab, followed by pegfilgrastim as previously planned.
After the hypersensitivity reaction, the treatment regimen was changed to nab-paclitaxel 100 mg/m2 on days 1, 8, and 15, cyclophosphamide 600 mg/m2 on day 1, and trastuzumab 6 mg/kg on day 1 every 3 weeks for 4 cycles, followed by trastuzumab 6 mg/kg every 3 weeks to complete a year of treatment, as described in a study of patients with early-stage breast cancer.19 The course of this patient’s treatment is shown in Figure 2.
The patient tolerated cycle 1 day 1 of the new treatment regimen well, with no hypersensitivity reactions; she received premedication with oral acetaminophen 650 mg, oral ondansetron 16 mg, and oral dexamethasone 12 mg. After cycle 4 of adjuvant chemotherapy, she received radiation therapy consisting of 40 Gy to the whole breast, followed by a 10-Gy tumor bed boost, for a total dose of 50 Gy over 4 weeks.
After the completion of radiation therapy, the patient started endocrine therapy with oral letrozole 2.5 mg daily for her ER-positive status. All the regimens that the patient received and her hypersensitivity reaction are shown in the Table. The patient remains in remission 12 months after completing 1 year of trastuzumab therapy.
Discussion
Taxanes serve as the backbone regimen of many breast cancer treatments,1 yet some patients do not tolerate paclitaxel or docetaxel because of hypersensitivity reactions.2 Nab-paclitaxel is associated with a lower incidence of hypersensitivity reactions than other taxanes, likely because it is not formulated with Cremophor EL or polysorbate 80.2,8 However, nab-paclitaxel has not been extensively studied in breast cancer and is not considered the standard of care for this disease.1
Multiple strategies to continue taxane chemotherapy in patients with a hypersensitivity reaction to a taxane exist in addition to changing the treatment to nab-paclitaxel.20-22 One such strategy includes premedication with dexamethasone 20 mg 12 hours and 6 hours before the infusion of the taxane.23,24
Another strategy includes a rapid desensitization protocol to the taxane, whereby the patient is given premedications with diphenhydramine (or hydroxyzine), famotidine (or ranitidine), and lorazepam immediately before treatment; in addition, dexamethasone is administered on the night before and on the day of the protocol.25 After appropriate premedications, higher doses of the taxane are administered consecutively under close clinic supervision until the target dose is achieved.24
Other desensitization regimens can also be used.22 Our institution does not have a desensitization protocol, because we do not have sufficient clinic space for a multi-hour protocol.
We used nab-paclitaxel in both cases presented here to prevent further hypersensitivity reactions. In the first case, we deemed that a transition to IV premedications would provide sufficient premedication to prevent a repeated hypersensitivity reaction; however, the patient still had a second, much more severe, hypersensitivity reaction. In the second case, we deemed that the initial hypersensitivity reaction was too severe to attempt additional premedication.
As mentioned previously, several studies and case reports have demonstrated that nab-paclitaxel can be used safely and effectively in patients who have a hypersensitivity reaction to another taxane.26,27 Nab-paclitaxel has been successfully used in the neoadjuvant setting for the treatment of ERBB2/HER2-negative nonmetastatic breast cancer.26,27 Nab-paclitaxel was not statistically different from paclitaxel when both agents were dosed weekly for 3 weeks, followed by 1 week off, for a total of 4 cycles, followed by 4 cycles of an anthracycline.27 Because the patient in our first case had already received 2 cycles of dose-dense paclitaxel before having a hypersensitivity reaction, the treatment team decided to continue with treatment in a dose-dense manner, administering nab-paclitaxel every 2 weeks.
Patients with triple-negative breast cancer (TNBC) have worse overall survival than patients with other types of breast cancer.28 Localized TNBC has a 5-year survival rate of 91%,29 whereas the 5-year survival rate for all localized breast cancer (triple-negative; ER/PR-positive, ERBB2/HER2-negative) is 99%.30 Because TNBC is associated with worse outcomes, a more aggressive treatment strategy is typically used, such as early dose-dense chemotherapy.28
In a meta-analysis comparing standard versus dose-dense chemotherapy for breast cancer, the 10-year overall breast cancer mortality rate was 21.3% with standard chemotherapy versus 18.9% with dose-dense chemotherapy.31 Our first patient with TNBC has had in-clinic follow-up and remains alive and recurrence-free 24 months after completing nab-paclitaxel therapy.
The dosing regimen for the second patient was based on a phase 2 pilot study consisting of 63 patients.19 The study evaluated nab-paclitaxel and cyclophosphamide (with trastuzumab added in patients with ERBB2/HER2-positive disease) for the treatment of early breast cancer.19 The regimen was safe and effective; at the time of the study’s publication, the median treatment follow-up was only 17.8 months, with all 63 patients alive and in remission.19 In that study, only 8 patients had ERBB2/HER2-positive disease19; thus, the information from our second case report adds further evidence that nab-paclitaxel is safe and effective as adjuvant treatment in early-stage ERBB2/HER2-positive breast cancer.
The 5-year overall survival in patients with ER/PR-negative, ERBB2/HER2-positive breast cancer is 78.8%, whereas the 5-year progression-free survival rate for these patients is 66%.32 Our patient in case 2 remains alive and recurrence-free 12 months after completing 1 year of trastuzumab therapy.
Conclusion
Our 2 case reports demonstrate the successful use of nab-paclitaxel in patients with breast cancer, indicating possible nab-paclitaxel dosing strategies for patients with breast cancer who had a hypersensitivity reaction to paclitaxel or to docetaxel treatment. Standard chemotherapy treatment for breast cancer often includes the use of a taxane; however, there is currently limited evidence regarding the appropriate dosing of nab-paclitaxel in patients with breast cancer who have a hypersensitivity reaction to another taxane.
These 2 cases provide additional information regarding nab-paclitaxel dosing for adjuvant and neoadjuvant breast cancer treatment. Further randomized controlled clinical trials are needed to demonstrate the long-term efficacy of nab-paclitaxel in early breast cancer. In addition, developing a tool to risk-stratify patients based on the probability of taxane hypersensitivity reactions could help clinicians decide which patients may benefit from avoiding paclitaxel and docetaxel and receiving nab-paclitaxel treatment instead.
Author Disclosure Statement
Dr Straughn, Dr Hardie-Hood, and Dr Carrera have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Breast Cancer. Version 5.2021. September 13, 2021. www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed October 22, 2021.
- Picard M, Castells MC. Re-visiting hypersensitivity reactions to taxanes: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:177-191.
- Szebeni J, Muggia FM, Alving CR. Complement activation by Cremophor EL as a possible contributor to hypersensitivity to paclitaxel: an in vitro study. J Natl Cancer Inst. 1998;90:300-306.
- Weiszhár Z, Czúcz J, Révész C, et al. Complement activation by polyethoxylated pharmaceutical surfactants: Cremophor-EL, Tween-80 and Tween-20. Eur J Pharm Sci. 2012;45:492-498.
- Essayan DM, Kagey-Sobotka A, Colarusso PJ, et al. Successful parenteral desensitization to paclitaxel. J Allergy Clin Immunol. 1996;97(pt 1):42-46.
- Shimozuma K, Ohashi Y, Takeuchi A, et al. Taxane-induced peripheral neuropathy and health-related quality of life in postoperative breast cancer patients undergoing adjuvant chemotherapy: N-SAS BC 02, a randomized clinical trial. Support Care Cancer. 2012;20:3355-3364.
- Weiss RB, Donehower RC, Wiernik PH, et al. Hypersensitivity reactions from Taxol. J Clin Oncol. 1990;8:1263-1268.
- Stinchcombe TE. Nanoparticle albumin-bound paclitaxel: a novel Cremphor-EL-free formulation of paclitaxel. Nanomedicine (Lond). 2007;2:415-423.
- Abraxane for injectable suspension (paclitaxel protein-bound particles for injectable suspension) (albumin-bound) for intravenous use [prescribing information]. Celgene Corporation; August 2020. https://packageinserts.bms.com/pi/pi_abraxane.pdf. Accessed November 1, 2021.
- Ibrahim NK, Samuels B, Page R, et al. Multicenter phase II trial of ABI-007, an albumin-bound paclitaxel, in women with metastatic breast cancer. J Clin Oncol. 2005;23:6019-6026.
- Rowinsky EK, Donehower RC. Paclitaxel (Taxol). N Engl J Med. 1995;332:1004-1014. Erratum in: N Engl J Med. 1995;333:75.
- de Leon MCB, Bolla S, Greene B, et al. Successful treatment with nab-paclitaxel after hypersensitivity reaction to paclitaxel and docetaxel. Gynecol Oncol Case Rep. 2013;5:70-71.
- Fader AN, Rose PG. Abraxane for the treatment of gynecologic cancer patients with severe hypersensitivity reactions to paclitaxel. Int J Gynecol Cancer. 2009;19:1281-1283.
- Robert N, Krekow L, Stokoe C, et al. Adjuvant dose-dense doxorubicin plus cyclophosphamide followed by dose-dense nab-paclitaxel is safe in women with early-stage breast cancer: a pilot study. Breast Cancer Res Treat. 2011;125:115-120.
- Kimura K, Tanaka S, Iwamoto M, et al. Safety of nanoparticle albumin-bound paclitaxel administered to breast cancer patients with clinical contraindications to paclitaxel or docetaxel: four case reports. Oncol Lett. 2013;6:881-884.
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5x11.pdf. Accessed June 21, 2021.
- McArthur HL, Rugo H, Nulsen B, et al. A feasibility study of bevacizumab plus dose-dense doxorubicin–cyclophosphamide (AC) followed by nanoparticle albumin–bound paclitaxel in early-stage breast cancer. Clin Cancer Res. 2011;17:3398-3407.
- Pippen J, Paul D, Vukelja S, et al. Dose-dense doxorubicin and cyclophosphamide followed by dose-dense albumin-bound paclitaxel plus bevacizumab is safe as adjuvant therapy in patients with early stage breast cancer. Breast Cancer Res Treat. 2011;130:825-831.
- Yardley D, Burris H III, Peacock N, et al. A pilot study of adjuvant nanoparticle albumin-bound (nab) paclitaxel and cyclophosphamide, with trastuzumab in HER2-positive patients, in the treatment of early-stage breast cancer. Breast Cancer Res Treat. 2010;123:471-475.
- Boulanger J, Boursiquot JN, Cournoyer G, et al. Management of hypersensitivity to platinum- and taxane-based chemotherapy: CEPO review and clinical recommendations. Curr Oncol. 2014;21:e630-e641.
- Feldweg AM, Lee CW, Matulonis UA, Castells M. Rapid desensitization for hypersensitivity reactions to paclitaxel and docetaxel: a new standard protocol used in 77 successful treatments. Gynecol Oncol. 2005;96:824-829.
- Robinson JB, Singh D, Bodurka-Bevers DC, et al. Hypersensitivity reactions and the utility of oral and intravenous desensitization in patients with gynecologic malignancies. Gynecol Oncol. 2001;82:550-558.
- Kwon JS, Elit L, Finn M, et al. A comparison of two prophylactic regimens for hypersensitivity reactions to paclitaxel. Gynecol Oncol. 2002;84:420-425.
- Trudeau ME, Eisenhauer EA, Higgins BP, et al. Docetaxel in patients with metastatic breast cancer: a phase II study of the National Cancer Institute of Canada-Clinical Trials Group. J Clin Oncol. 1996;14:422-428.
- Castells MC, Tennant NM, Sloane DE, et al. Hypersensitivity reactions to chemotherapy: outcomes and safety of rapid desensitization in 413 cases. J Allergy Clin Immunol. 2008;122:574-580.
- Zong Y, Wu J, Shen K. Nanoparticle albumin-bound paclitaxel as neoadjuvant chemotherapy of breast cancer: a systematic review and meta-analysis. Oncotarget. 2017;8:17360-17372.
- Gianni L, Mansutti M, Anton A, et al. Comparing neoadjuvant nab-paclitaxel vs paclitaxel both followed by anthracycline regimens in women with ERBB2/HER2-negative breast cancer—the Evaluating Treatment with Neoadjuvant Abraxane (ETNA) trial: a randomized phase 3 clinical trial. JAMA Oncol. 2018;4:302-308.
- Li X, Yang J, Peng L, et al. Triple-negative breast cancer has worse overall survival and cause-specific survival than non-triple-negative breast cancer. Breast Cancer Res Treat. 2017;161:279-287.
- American Cancer Society. Triple-negative breast cancer. Updated January 27, 2021. www.cancer.org/cancer/breast-cancer/about/types-of-breast-cancer/triple-negative.html. Accessed June 21, 2021.
- American Cancer Society. Cancer Facts & Figures 2020. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2020/cancer-facts-and-figures-2020.pdf. Accessed June 21, 2021.
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Increasing the dose intensity of chemotherapy by more frequent administration or sequential scheduling: a patient-level meta-analysis of 37,298 women with early breast cancer in 26 randomised trials. Lancet. 2019;393:1440-1452.
- Onitilo AA, Engel JM, Greenlee RT, Mukesh BN. Breast cancer subtypes based on ER/PR and Her2 expression: comparison of clinicopathologic features and survival. Clin Med Res. 2009;7:4-13.