Hyperglycemia has been reported in approximately 10% to 15% of pediatric patients who are receiving chemotherapy for acute lymphoblastic leukemia (ALL).1-4 Such hyperglycemic episodes have been attributed to the use of asparaginase and glucocorticoids, with concurrent use further increasing the likelihood of hyperglycemia. Asparaginase therapy encompasses several agents, including l-asparaginase (Escherichia coli asparaginase, which is off now the market in the United States), Erwinia asparaginase, pegaspargase (peg-asparaginase), or calaspargase pegol.
The proposed mechanisms of action of asparaginase-induced hyperglycemia include decreasing insulin synthesis via the depletion of asparagine, decreasing insulin secretion from pancreatic B-cells, and impairing insulin receptor function.5-7 An alternative mechanism of action is acute pancreatitis leading to pancreatic beta-cell dysfunction.8
Although rare, hyperglycemic episodes may manifest as diabetic ketoacidosis in approximately 0.8% of pediatric patients receiving asparaginase therapy.9 The risk factors for diabetic ketoacidosis in pediatric patients with ALL include age older than 10 years, obesity, history of impaired glucose intolerance, and Down syndrome.9
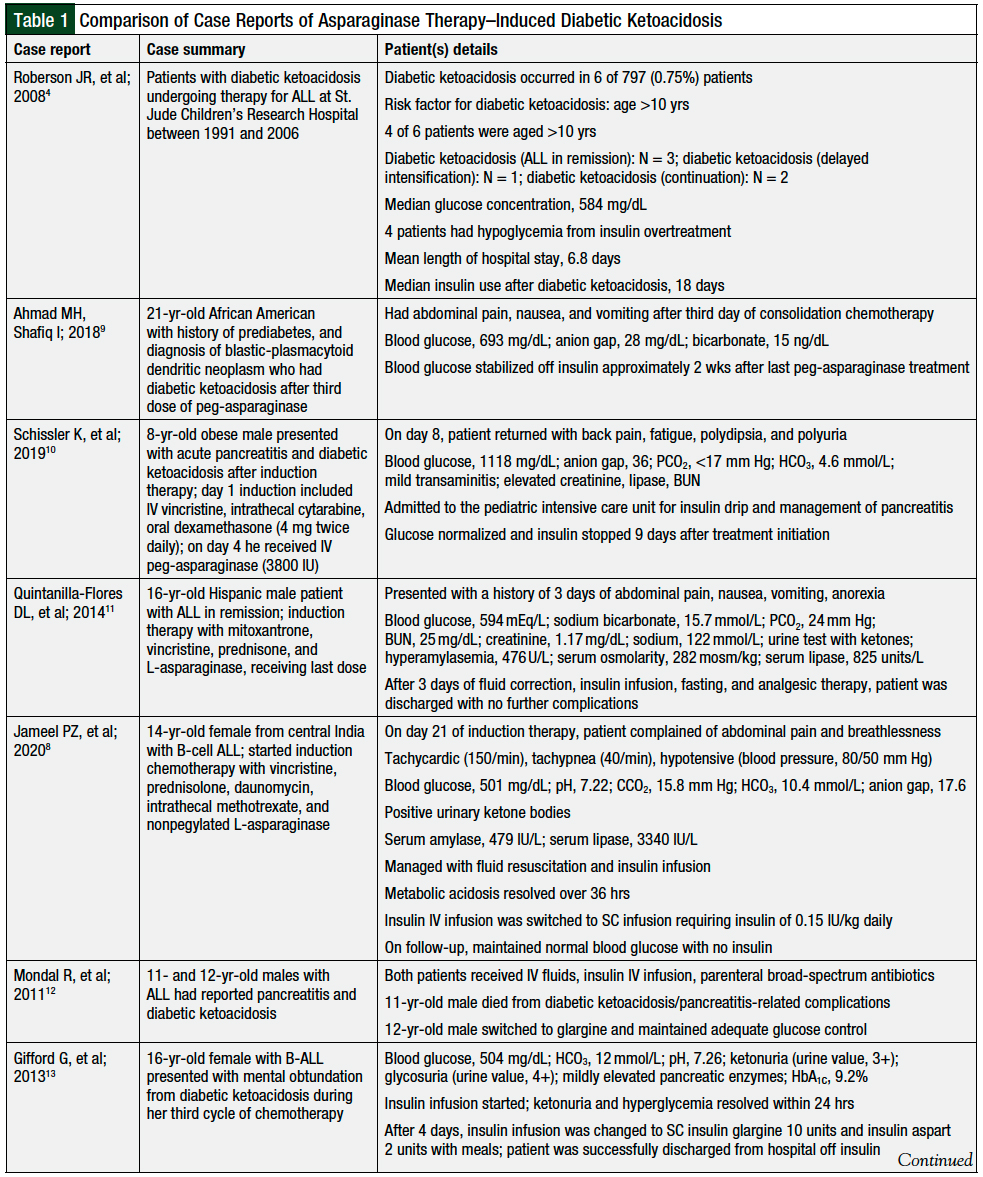
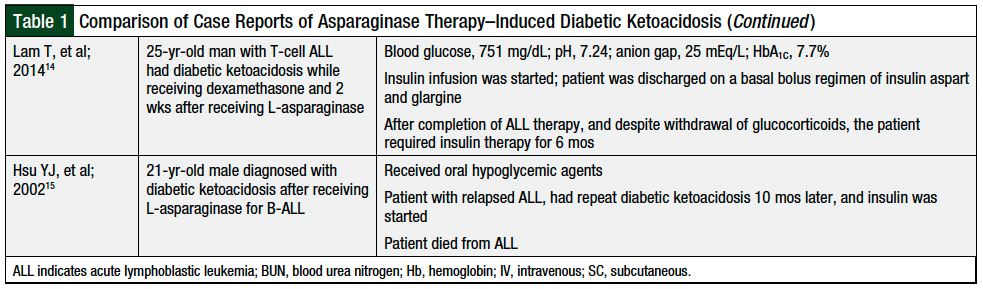
On presentation, diabetic ketoacidosis in the setting of chemotherapy for ALL is generally mild; however, it may result in short-term morbidity and hospital admission.8 Multiple case reports have been published describing patients with asparaginase therapy–induced diabetic ketoacidosis (Table 1).4,8-15
Adolescents and young adults with ALL typically receive treatment for ALL under the pediatric protocols, which includes asparaginase therapy. However, to our knowledge, in patients with ALL who were older than age 18 years, only 3 cases of diabetic ketoacidosis related to asparaginase therapy have been reported,9,14,15 and only 1 of those patients was receiving pegaspargase therapy.9
We describe the case of diabetic ketoacidosis in a 20-year-old woman with ALL who received pegaspargase therapy.
Case Report
A 20-year-old woman who was receiving treatment for B-cell ALL called our clinic complaining of polyuria, polydipsia, polyphagia, and acetone smell. She was receiving active therapy with the AALL1131 Interim Maintenance II regimen, which includes vincristine, Capizzi methotrexate, pegaspargase, and intrathecal methotrexate.16
During the AALL1131 Interim Maintenance II regimen, she received her sixth dose of pegaspargase. Her symptoms began 3 days after receiving the sixth dose of pegaspargase. The patient had not received steroids in more than 10 months after she had steroid-induced psychosis during the AALL1131 Consolidation I treatment.
The patient was instructed to go to the emergency department for further workup. In the emergency department, she reported weakness, dizziness, and difficulty walking. The initial workup revealed she had ketonuria, and her initial blood glucose was approximately 800 mg/dL. Computed tomography of the pancreas was unremarkable. The patient’s hemoglobin A1c level was 5%, with a body surface area of 2.04 m2 and a body mass index of 27.91 kg/m2.
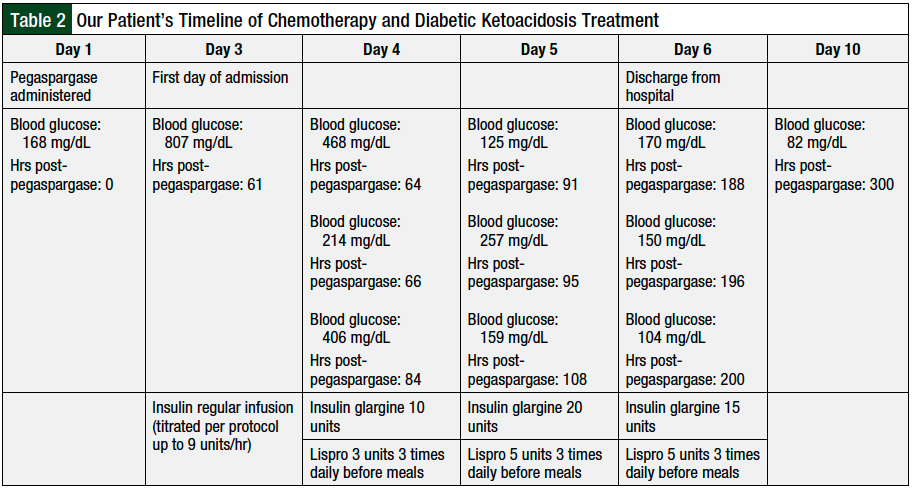
The patient was transferred to the intensive care unit and received intravenous dextrose with 1/2 normal saline hydration and an insulin continuous infusion. After 13 hours of receiving the insulin infusion, her blood glucose normalized and the anion gap closed. She was then transitioned to a basal-bolus insulin regimen. The patient was discharged home with this regimen after a 4-day hospitalization. Table 2 outlines her treatment details from the day of pegaspargase administration through day 10 post–pegaspargase administration.
Two weeks later, the patient followed up with the endocrinology department and her insulin regimen was discontinued. To date, her glucose levels have been normal. Our team made the decision to forego any further pegaspargase treatment (1 dose was omitted).
Discussion
Our case involves a patient with pegaspargase-induced diabetic ketoacidosis. Pegaspargase is an important component of chemotherapy used in the treatment of patients with ALL. In previously reported cases, diabetic ketoacidosis was seen days to weeks after asparaginase therapy.4,8-15 Our patient had diabetic ketoacidosis within 3 days of receiving her sixth dose of pegaspargase.
In previous reports (Table 1),4,8-15 patients had risk factors for pegaspargase-induced diabetic ketoacidosis that included obesity, age older than 10 years, and prediabetes. Our patient’s only risk factor was age >10 years. Of note, our patient was not receiving concurrent steroids, as was the case in most of the previously reported case reports.8,10,11,14
Hyperglycemia is a common side effect of asparaginase therapy; however, progression to diabetic ketoacidosis is rare. Diabetic ketoacidosis in patients receiving asparaginase therapy resolves quickly with appropriate management. As was reported in several cases cited in Table 1,9-11,13 our patient, too, had resolution of the hyperglycemia within 2 weeks of receiving pegaspargase.
Conclusion
Pegaspargase is an important component of chemotherapy in young patients with ALL. Hyperglycemia is a common side effect of asparaginase therapy; however, progression to diabetic ketoacidosis is rare. Our patient had resolution of hyperglycemia within 2 weeks of receiving pegaspargase. Patients undergoing asparaginase therapy should be monitored closely for hyperglycemia. The feasibility and safety of resuming treatment with pegaspargase after diabetic ketoacidosis could be an area for future review.
Author Disclosure Statement
Dr Madarang and Dr Gallardo have no conflicts of interest to report. Dr Bradley is on the Speaker’s Bureau and the Advisory Board of AbbVie, and is a Consultant to and on the Speaker’s Bureau of Novartis.
References
- Iyer RS, Rao SR, Pai S, et al. L-asparaginase related hyperglycemia. Indian J Cancer. 1993;30:72-76.
- Jaffe N, Traggis D, Das L, et al. Comparison of daily and twice-weekly schedule of L-asparaginase in childhood leukemia. Pediatrics. 1972;49:590-595.
- Pui CH, Burghen GA, Bowman WP, Aur RJA. Risk factors for hyperglycemia in children with leukemia receiving L-asparaginase and prednisone. J Pediatr. 1981;99:46-50.
- Roberson JR, Raju S, Shelso J, et al. Diabetic ketoacidosis during therapy for pediatric acute lymphoblastic leukemia. Pediatr Blood Cancer. 2008;50:1207-1212.
- Lavine RL, Brodsky I, Garofano CD, Rose LI. The effect of E. coli L-asparaginase on oral glucose tolerance and insulin release in man. Diabetologia. 1978;15:113-116.
- Rao SP, Castells S. Hyperglucagonemia in L-asparaginase induced diabetes mellitus. Am J Pediatr Hematol Oncol. 1986;8:83-85.
- Turner GR, Marks JF, Buchanan GR. Relative hyperglucagonemia in L-asparaginase-and prednisone-induced glucose intolerance in management of acute lymphocytic leukemia. Clin Pediatr (Phila). 1983;22:363-367.
- Jameel PZ, Lohiya S, Dongre A, et al. Concurrent diabetic ketoacidosis and pancreatitis in Paediatric acute lymphoblastic leukemia receiving L-asparaginase. BMC Pediatr. 2020;20:228.
- Ahmad MH, Shafiq I. Diabetic ketoacidosis following PEG-asparaginase therapy. Endocrinol Diabetes Metab Case Rep. 2018;2018:18-0064.
- Schissler K, Diaz N, Sarmiento A, et al. Acute pancreatitis and diabetic ketoacidosis in an 8-year-old following induction therapy for pre-B acute lymphocytic leukemia. J Endocr Soc. 2019;3(suppl_1):Abstract SUN–276.
- Quintanilla-Flores DL, Flores-Caballero MÁ, Rodríguez-Gutiérrez R, et al. Acute pancreatitis and diabetic ketoacidosis following L-asparaginase/prednisone therapy in acute lymphoblastic leukemia. Case Rep Oncol Med. 2014;2014:139169.
- Mondal R, Nandi M, Tiwari A, Chakravorti S. Diabetic ketoacidosis with L-asparaginase therapy. Indian Pediatr. 2011;48:735-736.
- Gifford G, Milliken S, Greenfield J. Diabetic ketoacidosis secondary to L-asparaginase in acute lymphoblastic leukaemia. Intern Med J. 2013;43:946-948.
- Lam T, Chipps D, Gunton JE. Diabetic ketoacidosis secondary to L-asparaginase and dexamethasone during treatment for acute lymphoblastic leukaemia. J Diabetes Metab. 2014;5:422.
- Hsu YJ, Chen YC, Ho CL, et al. Diabetic ketoacidosis and persistent hyperglycemia as long-term complications of L-asparaginase-induced pancreatitis. Zhonghua Yi Xue Za Zhi (Taipei). 2002;65:441-445.
- Burke MJ, Salzer WL, Devidas M, et al. Replacing cyclophosphamide/cytarabine/mercaptopurine with cyclophosphamide/etoposide during consolidation/delayed intensification does not improve outcome for pediatric B-cell acute lymphoblastic leukemia: a report from the COG. Haematologica. 2019;104:986-992.