Kelly Fritz, PharmD, BCOP, and Sara deHoll, PharmD, BCOP, presented a comprehensive exploration of metastatic brain cancer in their presentation, Mind Over Metastases: Navigating Central Nervous System Involvement in Solid Tumors, which was featured at the 2026 HOPA annual conference, March 25-27, in New Orleans, LA.
Central nervous system (CNS) involvement in patients diagnosed with solid tumors is common. The most common solid tumor types implicated in CNS metastases include lung and breast cancers (HER2-positive or triple-negative breast cancer) and melanoma, but renal cell carcinoma and colon and testicular cancers are also seen. CNS metastases typically take one of 2 forms: parenchymal/intracranial metastases or leptomeningeal disease (LMD). Due to increasing success treating cancer and increased survival, 25% or more of patients with cancer will develop brain metastases, and these are 10 times more common than primary brain tumors.
Symptoms are dependent on the location of the brain mass, which can occur in the cerebrum, cerebellum, and brainstem, with global symptoms such as headache, nausea, seizures, and altered mental status occurring; more focal symptoms such as visual changes, hemiparesis, sensory loss, personality changes, and aphasia are also seen. Dr Fritz emphasized that any new neurologic symptom should trigger brain imaging. Brain magnetic resonance imaging is considered the gold standard for the diagnosis of brain metastases, but a biopsy may also be needed to study the tumor tissue.
It is believed that the pathophysiology of CNS metastatic disease involves cancerous cells spreading via blood, lymphatics, direct extension of tumor into the CNS, or tumor-cell migration from tumors that are adjacent to the CNS, such as head and neck cancers or melanoma or squamous cells on the face. Once the cancer has spread to the CNS, the “seed and soil” hypothesis describes how and why these cells go on to thrive: the microenvironment in the brain represents a ready source of blood, oxygen, and nutrients. In addition, the blood-brain barrier prevents cytotoxic chemicals from affecting the CNS masses. Of note, once the cancerous cells have invaded the CNS, additional mutations or discordant mutations can occur.
Treatment of Intracranial Brain Metastases
Intracranial metastases are commonly treated by some combination of surgery, radiation therapy, or systemic therapy. Decision-making surrounding these 3 methods includes consideration of the number of tumors present, the status of the extracranial disease, the patient’s symptoms and whether the treatment will improve them, and sequencing with other therapies.
Specific factors that make a patient a candidate for surgery include if the primary tumor is unknown, if the brain metastasis is a single large tumor, and if the systemic disease is controlled. Patients who would not be candidates for surgery include those who have multiple brain metastases, uncontrolled systemic disease, and involvement of regions of the brain responsible for speech, motor movement, sensory processing, and vision.
A consideration with radiation therapy includes the potential for cognitive decline following whole brain radiation; thus, focal treatment with stereotactic radiation therapy is more common. Patients who would be candidates for whole brain radiotherapy, however, include those with unresectable lesions, multiple lesions, or leptomeningeal disease with spinal involvement. Patients who would be candidates for stereotactic radiosurgery include those who have multiple lesions <4 cm each, those with unresectable lesions, and those who are medically inoperable.
Studies of systemic, or brain-penetrating, agents have been adding to the data that are available on the use of these agents in CNS metastases. These include the EGFR inhibitors osimertinib, amivantamab (Table 1), and lazertinib; the ALK inhibitors alectinib and brigatinib; the checkpoint inhibitors pembrolizumab, ipilimumab, and nivolumab (Table 2); the BRAF inhibitors dabrafenib and trametinib; and the HER2 inhibitors tucatinib, capecitabine, trastuzumab, and fam-trastuzumab deruxtecan-nxki (Table 3). It is interesting to note that the designs of studies of systemic therapies do not typically include patients with symptomatic brain or untreated metastases, which may affect the response rates in these trials.
Table 1: Treating Intracranial Metastases With EGFR Inhibitors in NSCLC: Amivantimab Plus Lazertinib
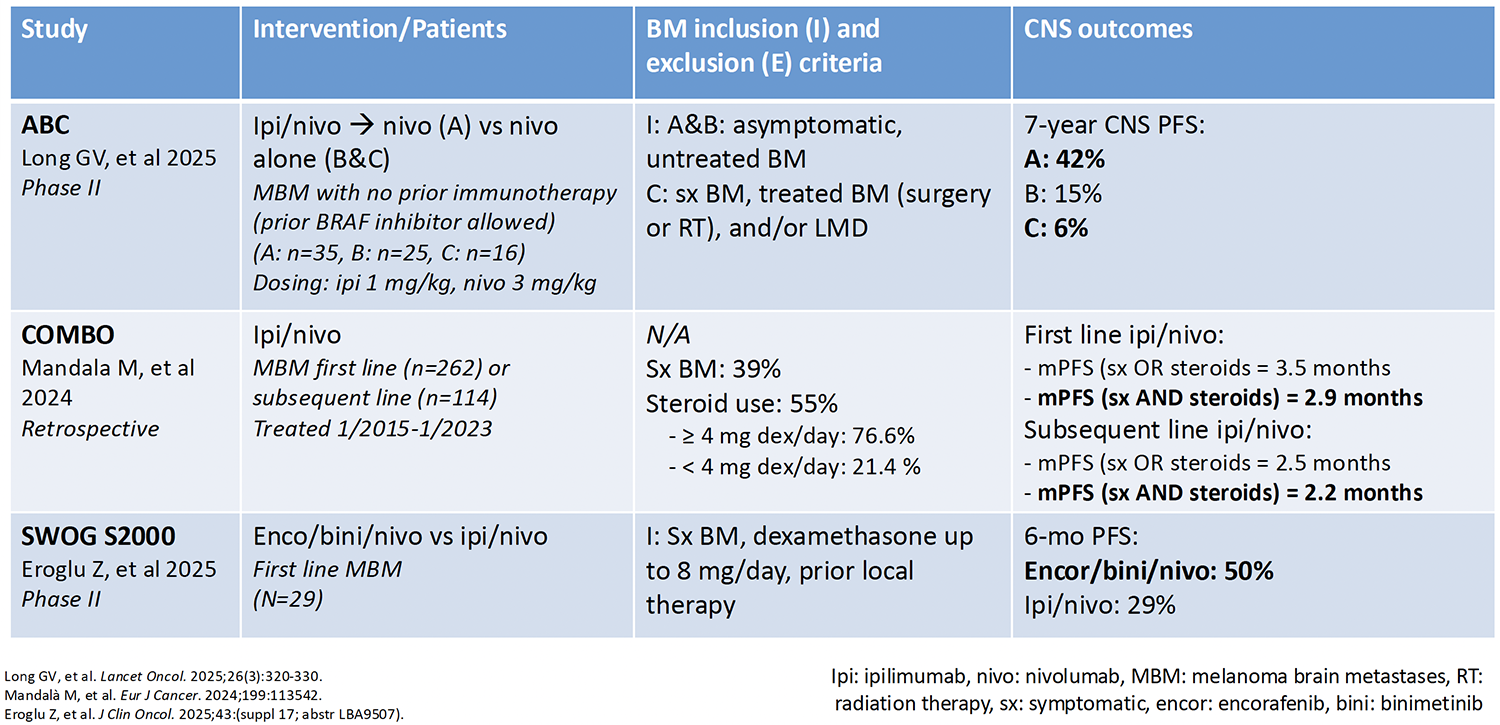
Table 2: Treating Intracranial Metastases With Checkpoint Inhibitors in Melanoma: Ipilimumab Plus Nivolumab
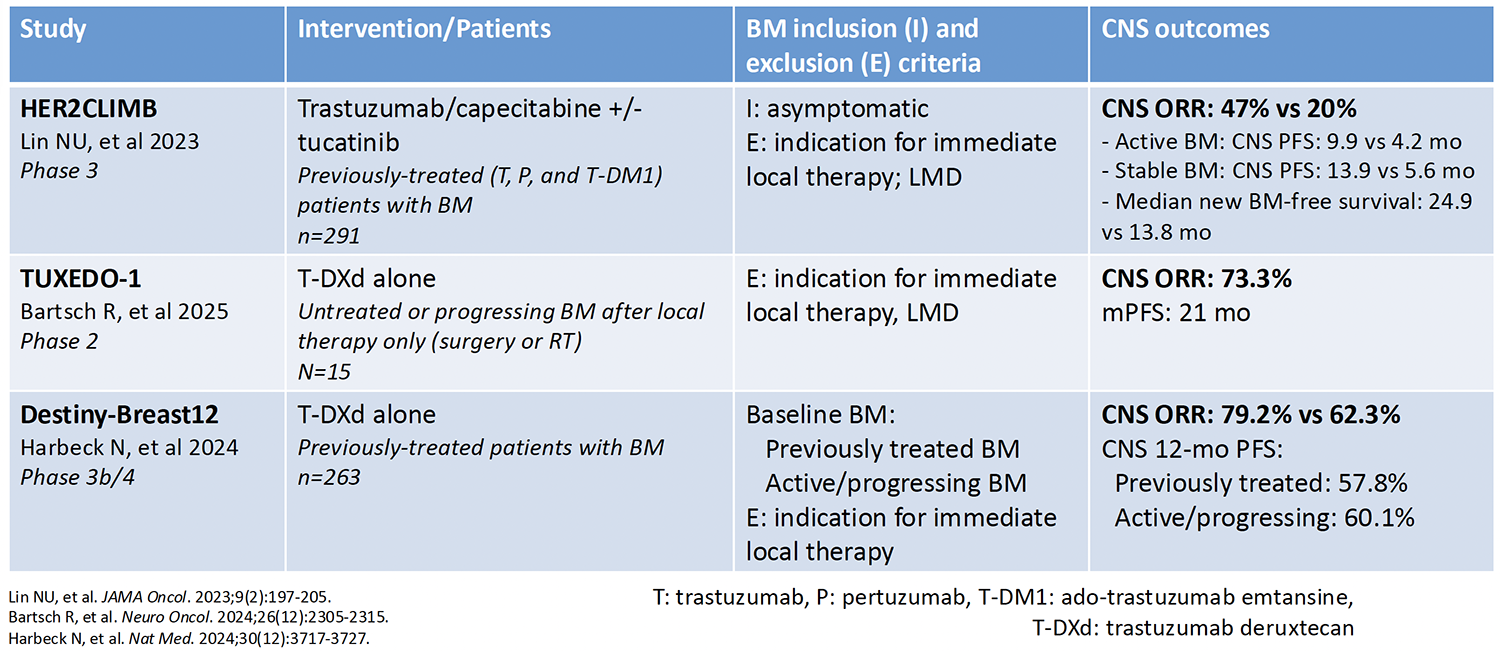
Table 3: Treating Intracranial Metastases With HER2 Inhibitors in Breast Cancer: Tucatinib Plus Capecitabine Plus Trastuzumab vs Trastuzumab Deruxtecan
Sequencing and Timing of Therapies
The sequencing and timing of these options for the treatment of de novo brain metastases varies depending on the extracranial disease. For non–small cell lung cancer (NSCLC), melanoma, and HER2-positive breast cancer, the decision-tree initially involves local therapy with focal radiation or surgery, or systemic therapy. Although no head-to-head studies are available to guide a decision between radiation therapy and surgery, Dr Fritz reported that most patients prefer 1 of these 2 options. Some patients may be candidates for systemic treatment. These agents include EGFR or ALK inhibitors for patients with NSCLC, ipilimumab/nivolumab for melanoma, and tucatinib, trastuzumab, and capecitabine for HER2-positive breast cancer. Second-line therapies for those who initially received surgery include focal radiation or systemic therapy. For those initially started on focal radiation, second-line therapy can include systemic treatment or observation. Third-line treatment for those receiving second-line focal radiation includes systemic treatment or observation.
Decision-making regarding sequencing and timing for brain metastases occurring during therapy in these patient groups depends largely on whether the patient was receiving systemic therapy when the metastasis occurred. If the patient was receiving systemic therapy, then the second-line treatment may include local radiation therapy or surgery. Third-line treatment for these patients may include continuing the previous therapy in patients who have stable extracranial disease or a therapy switch in patients with progressing extracranial disease. If the patient was not receiving systemic therapy when the metastasis occurred, then the options include local radiation therapy or surgery, or the patient can be switched to a different systemic treatment. A third-line option in such patients would be switching to a different systemic therapy.
Symptom Management
Common symptoms in patients with intracranial metastases include edema, seizures, thrombosis, and cognitive decline. Radiation therapy and surgery can result in edema in these patients, and dexamethasone can be used in managing this adverse event (AE). Historically, dexamethasone was given as a bolus of 10 mg to 40 mg followed by 4 mg intramuscularly every 4 hours; however, more recent reports suggest once or twice daily dosing is preferred. Patients with brain metastases that are near the cerebral cortex are at increased risk for seizures, which include temporal and parietal lobe involvement. Nonenzyme-inducing antiepileptic drugs such as levetiracetam and lacosamide can manage seizures once present. In addition, carbamazepine, phenytoin, and valproic acid should be avoided. Hemorrhagic brain metastases are more common in patients with melanoma, renal cell carcinoma, and lung cancer. Nonhemorrhagic brain metastases with thrombus can be managed with direct oral anticoagulants and low-molecular-weight heparin, although these agents should not be used if the patient has had a recent intracranial hemorrhage, an urgent need for surgery, or a platelet count <50,000. It is currently unknown whether hemorrhagic brain metastases with a thrombus can be treated without causing increased risk for intracranial hemorrhage. Cognitive decline, a known adverse event (AE) associated with whole brain radiotherapy, was shown to have a nonsignificant response to memantine in a study by Brown and colleagues. Although neuropsychiatric testing to monitor cognitive decline is not performed consistently in clinical practice, it remains important to always counsel patients with brain metastases with a caregiver present because cognitive decline in these patients is likely.
Leptomeningeal Disease
LMD, characterized by tumor cells in the cerebrospinal fluid (CSF) or in the lining of the tissue or the spinal cord in the brain, occurs in approximately 10% of patients with cancer. The type of symptoms present will depend on the location of the brain metastases, but headache, vision changes, gait changes, and nausea are commonly seen. The work-up of a patient suspected to have LMD should include magnetic resonance imaging, lumbar puncture, and, rarely, leptomeningeal biopsy. A firm diagnosis can be challenging and would include CSF or a biopsy positive for tumor cells; a diagnosis of probable LMD is based on neurologic testing and imaging results suggestive of LMD.
The prognosis of patients with LMD is generally poor. In patients with lung cancer, overall survival is 4.2 to 8.1 months; in patients with breast cancer, it is 3.8 to 5.4 months; and in melanoma, it is 4.8 months. Treatments for LMD include intrathecal therapies, systemic therapies, and radiation. Response to treatment can be measured via the Response Assessment in Neuro-Oncology–Leptomeningeal Metastasis criteria, which includes a neurologic assessment, CSF evaluation, and imaging studies. In addition, the Neurological Assessment in Neuro-Oncology–Leptomeningeal Metastasis scale is an assessment tool comprised of 14 neurologic domains that can provide a more comprehensive picture on neurologic function.
Treatment of LMD: Intrathecal Therapies
Important issues to consider when treating patients with LMD with intrathecal therapies include whether the therapy can provide the desired increased drug concentration in the CSF while decreasing systemic AEs. The administration of intrathecal therapies is invasive, involving either a lumbar puncture or Ommaya reservoir, an indwelling, dome-shaped silicone device placed under the scalp with a catheter extending into one of the brain's lateral ventricles. Lumbar puncture may be preferred because it is a less invasive procedure compared with the Ommaya reservoir; in addition, it is the preferred option if the patient is expected to have a small number of intrathecal therapy administrations. Factors that make lumbar puncture less desirable include the need to discontinue anticoagulation, the potential for discomfort/paresthesia, the need to lie flat for 30 to 60 minutes after the procedure, and orthostatic headache following administration. An Ommaya reservoir may be preferred because it allows the patient to continue with anticoagulant therapy, is associated with less discomfort than a lumbar puncture, and allows for uniform drug distribution. Factors that may make an Ommaya reservoir less desirable include that it is an invasive procedure and can result in hematoma and infection. Additional issues surrounding the use of intrathecal therapy concern whether the patient should receive concurrent radiation and/or systemic therapy and the optimal duration of therapy.
Intrathecal therapy options include chemotherapies: cytarabine and methotrexate, thiotepa, topotecan, and pemetrexed. In addition, intrathecal targeted and immunotherapies include trastuzumab and nivolumab. Two studies of intrathecal thiotepa in patients with LMD and breast or lung cancer are summarized in Table 4. Two studies of intrathecal topotecan in patients with LMD and breast, lung, or CNS cancer are described in Table 5. Intrathecal pemetrexed is used in patients with LMD and lung cancer (Table 6). Intrathecal trastuzumab is used in patients with LMD and breast cancer (Table 7). Finally, Table 8 describes the use of nivolumab in patients with LMD and melanoma.
Table 4: Treating LMD: Intrathecal Thiotepa for Breast or Lung Cancer
Table 5: Treatment of LMD: Intrathecal Topotecan for Breast, Lung, CNS Malignancies
Table 6: Treatment of LMD: Intrathecal Pemetrexed for Lung Cancer
Table 7: Treatment of LMD: Intrathecal Trastuzumab for Breast Cancer
Table 8: Treatment of LMD: Intrathecal Nivolumab for Melanoma
Preventing AEs From Intrathecal Therapies
Guidelines and the evidence in the literature in this area are lacking. However, institution and provider guidance indicates that antiemetics (5-HT3 antagonist, dexamethasone, or olanzapine) and analgesics (acetaminophen) are beneficial for nausea and pain/headache, and chemical meningitis can be prevented with dexamethasone, prednisolone, or intrathecal hydrocortisone.
Treating LMD With Systemic Therapy
Ideally, the systemic therapy used to treat LMD will also help treat the systemic disease. Systemic therapies used to treat LMD include osimertinib, fam-trastuzumab deruxtecan-nxki, pembrolizumab, nivolumab plus ipilimumab, tucatinib plus capecitabine plus trastuzumab, BRAF/MEK inhibitors, and high-dose methotrexate. In a phase 2 single-arm, single-center trial of pembrolizumab, researchers studied 13 patients: high-grade gliomas and breast, lung, head and neck, and cutaneous squamous cell cancers with LMD. The patients received 200 mg intravenously every 3 weeks. The patients’ CNS response was 38%, median CNS progression-free survival was 2.9 months, and median overall survival was 4.9 months. Nivolumab plus ipilimumab was studied in a phase 2, single-arm, multicenter trial including 18 patients with breast, CNS, esophageal, lung, and ovarian cancers. Patients received the combination intravenously every 3 weeks for 4 cycles followed by nivolumab monotherapy. Patients’ median overall survival was 2.9 months, and the progression-free intracranial response was 1.93 months.
Treating LMD With Radiation Therapy
Focal radiation may be used to treat the symptoms of patients with cauda equina syndrome stemming from spinal tumors. It may also be used to treat nodular or linear nodular disease, and it is helpful in improving the delivery of intrathecal chemotherapy in patients with CNS flow obstruction. As previously mentioned, whole brain radiation therapy can lead to cognitive decline; however, it may be helpful as a palliative monotherapy, as data with this option in combination with intrathecal therapies are lacking. Cerebrospinal irradiation is effective in patients with diffuse LMD in the spine and brain, although it can cause myelosuppression, nausea, and vomiting.
Future Directions: CNS Metastases
Improving the diagnostic process for brain metastases and LMD is seen as a critical step in improving the management of these patients. Currently, research is exploring circulating tumor cells and cell-free tumor DNA in the CSF to better detect LMD and brain metastases. Research into treatments for brain metastases is focusing on combination intrathecal and systemic therapies and novel intrathecal therapies. In addition, research into the optimal sequencing of therapies, eg, combining radiation, systemic, and intrathecal therapy, is needed to further guide the treatments of these patients.
Sources
Le Rhun E, et al. ESMO Open. 2023;8:101624.
Le Rhun E, et al. Ann Oncol. 2021;32:1332-1347.
Vogelbaum MA, et al. J Clin Oncol. 2022;40:492-516.
Fares J, et al. Lancet Oncol. 2025;26:e111-121.
Lamba N, et al. Neuro Oncol. 2021;23:1447-1456.
Yang JCH, et al. N Engl J Med. 2025;393:1681-1693.
Cho BC, et al. N Engl J Med. 2024;391:1486-1498.
Besse B, et al. J Thorac Oncol. 2025;20:651-664.
Chen MF, et al. J Thorac Oncol. 2026;21:103505.
Long GV, et al. Lancet Oncol. 2025;26:320-330.
Mandalà M, et al. Eur J Cancer. 2024;199:113542.
Eroglu Z, et al. J Clin Oncol. 2025;43:(suppl 17): Abstract LBA9507.
Lin NU, et al. JAMA Oncol. 2023;9:197-205.
Bartsch R, et al. Neuro Oncol. 2024;26:2305-2315.
Harbeck N, et al. Nat Med. 2024;30:3717-3727.
Roth P, et al. Ann Oncol. 2021;32:171-182.
Sepúlveda-Sánchez JM, et al. Ann Oncol. 2021;32:139-141.
Lim-Fat MJ, et al. Neurosurgery. 2019;85:E607-E608.
Dietrich J, et al. Expert Rev Clin Pharmacol. 2011;4:233-242.
Kroon LL, et al. Neuro Oncol Prac. 2025;12:747-762.
Chang SM, et al. J Clin Oncol. 2019;37:1130-1135.
Achal AS, et al. Nat Rev Dis Primers. 2019;5:5.
Ranjan S, et al. Cancer. 2024;130:1577-1589.
Lee A, et al. Thromb Res. 2021;208:148-155.
Carney BJ, et al. Blood. 2018;132(suppl 1):2521.
Brown PD, et al. Neuro Oncol. 2013;15:1429-1437.
Schiff D, et al. J Clin Oncol. 2022;40:2271-2276.
Wilcox JA, et al. Neuro Oncol. 2024; 26:1781-1804.
Frechette KM, et al. Biomedicines. 2024;7:1792.
Chamberlain M, et al. Neuro Oncol. 2017;19:484-492.
Le Ruhn E, et al. Neuro Oncol. 2025;27;455-465.
Franklin A, et al. Neuro-Oncol. 2025;27:iii110.
Olmos-Jimenez R, et al. Farmacia Hospitalaria. 2017;41:105-129.
Grossman SA, et al. J Clin Oncol. 1993;11:561-569.
Cho KM, et al. Anticancer Research. 2015;35:5631-5638.
Groves MD, et al. Neuro Oncol. 2008;10:208-215.
Jaeckle KA, et al. Cancer Med. 2020;9:7935-7942.
Fan C, et al. ESMO Open. 2024;9:102384.
Glitza Oliva IC, et al. Nat Med. 2023;29:898-905.
Oberkampf F, et al. Neuro Oncol. 2023;25:365-374.
Chamberlain MC, et.al. J Neurosurg. 1997;87;694-699.
Frank RL. J Emerg Med. 2008;149-157.
Park S, et al. J Clin Oncol. 2024;42:2747-2756.
Alder L, et al. NPJ Breast Cancer. 2025;9:19.
Naidoo J, et.al. J Immunother Cancer. 2021;9:e002473.
Brastianos PK, et al. Nat Commun. 2021;12:5954.
Sener U, et.al. Curr Neurol Neurosci Rep. 2025;25:25.
Wilcox J, et.al. J Clin Oncol. 2025;43(suppl 16): Abstract 2032.
Le Rhun E, et al. J Clin Oncol. 2025;43:Abstract 2029.
ClinicalTrials.gov. Radiation therapy followed by intrathecal trastuzumab/pertuzumab in HER2+ breast leptomeningeal disease. Accessed April 15, 2026. https://clinicaltrials.gov/study/NCT04588545?intr=intrathecal%20pertuzumab&rank=2