Katie O’Reilly, PharmD, BCOP, presented a useful roundup of the current and emerging treatment landscape in small cell lung cancer (SCLC), including an overview of immunotherapy in SCLC and the research supporting its use in a limited-stage setting. Her presentation, Updates in Small Cell Lung Cancer, was featured at the 2026 HOPA annual conference, March 25-27, in New Orleans, LA.
SCLC is strongly associated with exposure to tobacco and accounts for approximately 15% of lung cancer diagnoses. It is known to have a high proliferation rate, early metastases, and poor prognosis. Most patients will be diagnosed as having extensive-stage disease at initial evaluation, and it is an aggressive cancer, with a 5-year survival of <5% among patients with extensive-stage disease. SCLC is highly responsive to platinum-based chemotherapy; however, many patients will relapse within months to a year.
SCLC Treatment Landscape
The treatment landscape for SCLC in both initial and relapse settings had remained the same for many years. In patients newly diagnosed with limited-stage SCLC, surgery is typically followed by adjuvant chemotherapy with a platinum-based drug plus etoposide. Patients who could not undergo surgery commonly receive a platinum-based chemotherapy including etoposide with concurrent or sequential radiation treatment. Patients with extensive-stage SCLC typically receive platinum chemotherapy plus etoposide and immunotherapy. This regimen is subsequently followed by maintenance immunotherapy.
In patients with SCLC who have relapsed, treatment choice can depend on the chemotherapy-free interval after initial progression. Patients whose chemotherapy-free interval was <3 months commonly receive single-agent chemotherapy (options include topotecan, lurbinectedin). Patients whose chemotherapy-free interval was ≥3 months and who were receiving maintenance immunotherapy should be engaged in a discussion of whether it is better to retry platinum-based chemotherapy or initiate a single agent, such as topotecan or lurbinectedin. If the decision is made to retry the platinum-based chemotherapy, and the patient’s cancer subsequently progresses, then the single agent can be initiated next. Similarly, in patients whose chemotherapy-free interval is ≥3 months and who were not receiving maintenance immunotherapy, it may be appropriate to retry the platinum-based chemotherapy with or without immunotherapy, followed by maintenance immunotherapy. If the patient’s cancer progresses, then it may be appropriate to initiate single-agent therapy such as topotecan or lurbinectedin. In recent years, however, new options have emerged, which are very exciting for the patients who are diagnosed with this aggressive disease.
Emerging Therapies in SCLC
At the end of 2024, ASCO released updated guidance on the management of SCLC. The updates covered 2 main patient categories: patients with limited-stage SCLC who completed concurrent chemoradiation therapy and whose cancer had not progressed, and patients with relapsed SCLC whose chemotherapy-free interval was <90 days. According to the ASCO updates, patients in the former category should be offered consolidation immunotherapy with durvalumab, whereas patients in the latter category should be offered single-agent therapy with topotecan, lurbinectedin, or the bispecific T-cell engager tarlatamab, which was approved by the FDA in May 2024.
Immune Checkpoint Inhibitors
An increased understanding of the pathophysiology of SCLC shows how immunotherapy plays a leading role in its treatment. SCLC has a high mutation rate, which suggests that the tumors may be immunogenic and could respond to immune checkpoint inhibitors (ICIs). The IMpower133 and Caspian trials helped establish the clinical benefit of adding ICIs to first-line platinum-based chemotherapy plus etoposide. The IMpower133 trial led to the approval of atezolizumab based on an overall survival (OS) benefit of 10.3 months in the placebo group versus 12.3 months in the atezolizumab group. More recently, the EXTENTORCH trial showed that toripalimab improved progression-free survival (PFS) and OS, and the RATIONALE-312 trial showed similar benefits with tislelizumab.
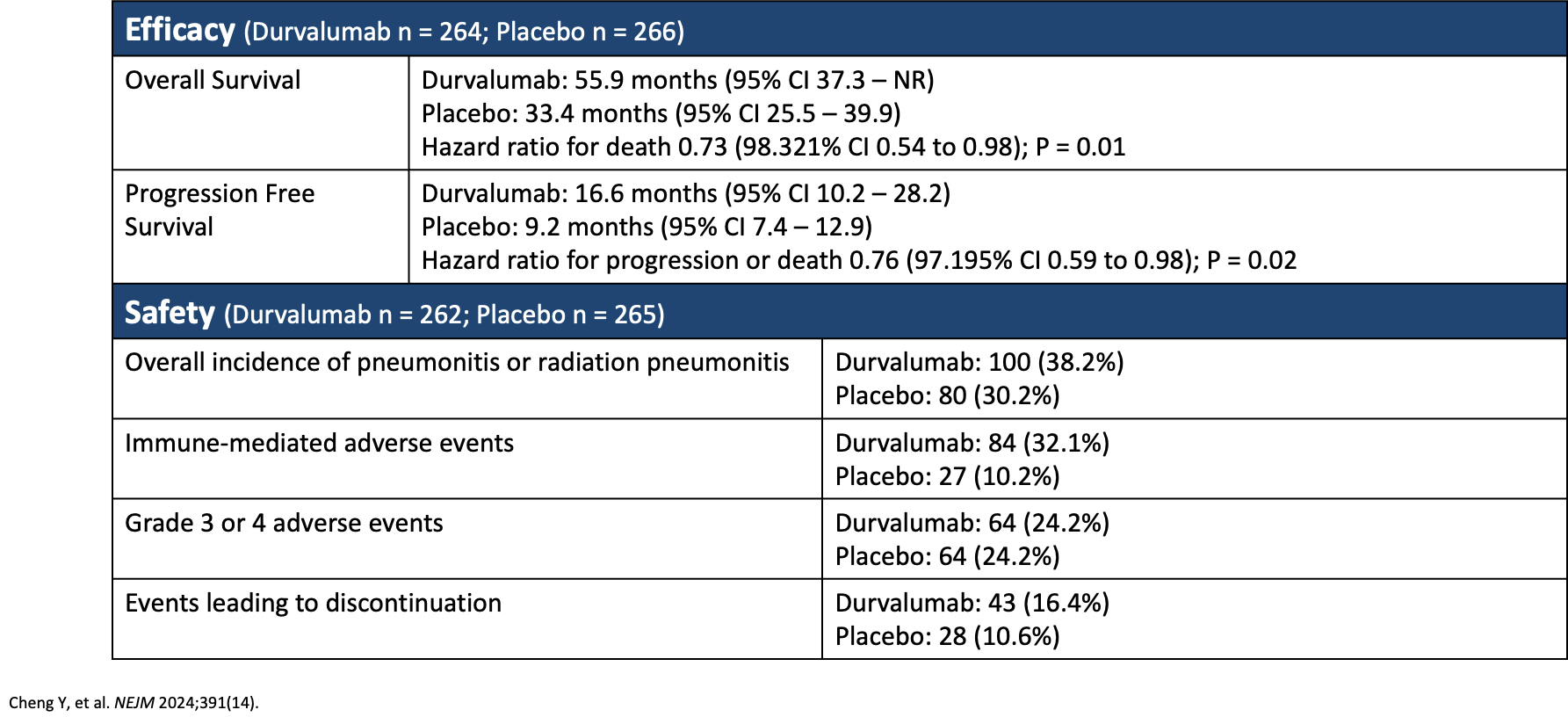
The ADRIATIC trial evaluated patients who had received concurrent chemoradiation therapy followed by the addition of durvalumab. The researchers’ findings are shown in Table 1, and they concluded that adjuvant therapy with durvalumab led to significantly longer OS and PFS compared with the group not receiving durvalumab. Based on these results, consolidation therapy with durvalumab is recommended in patients with SCLC who are able to receive immunotherapy.
Table 1. ADRIATIC Trial: Safety and Efficacy of Consolidation Therapy With Durvalumab in Patients With Limited Stage SCLC
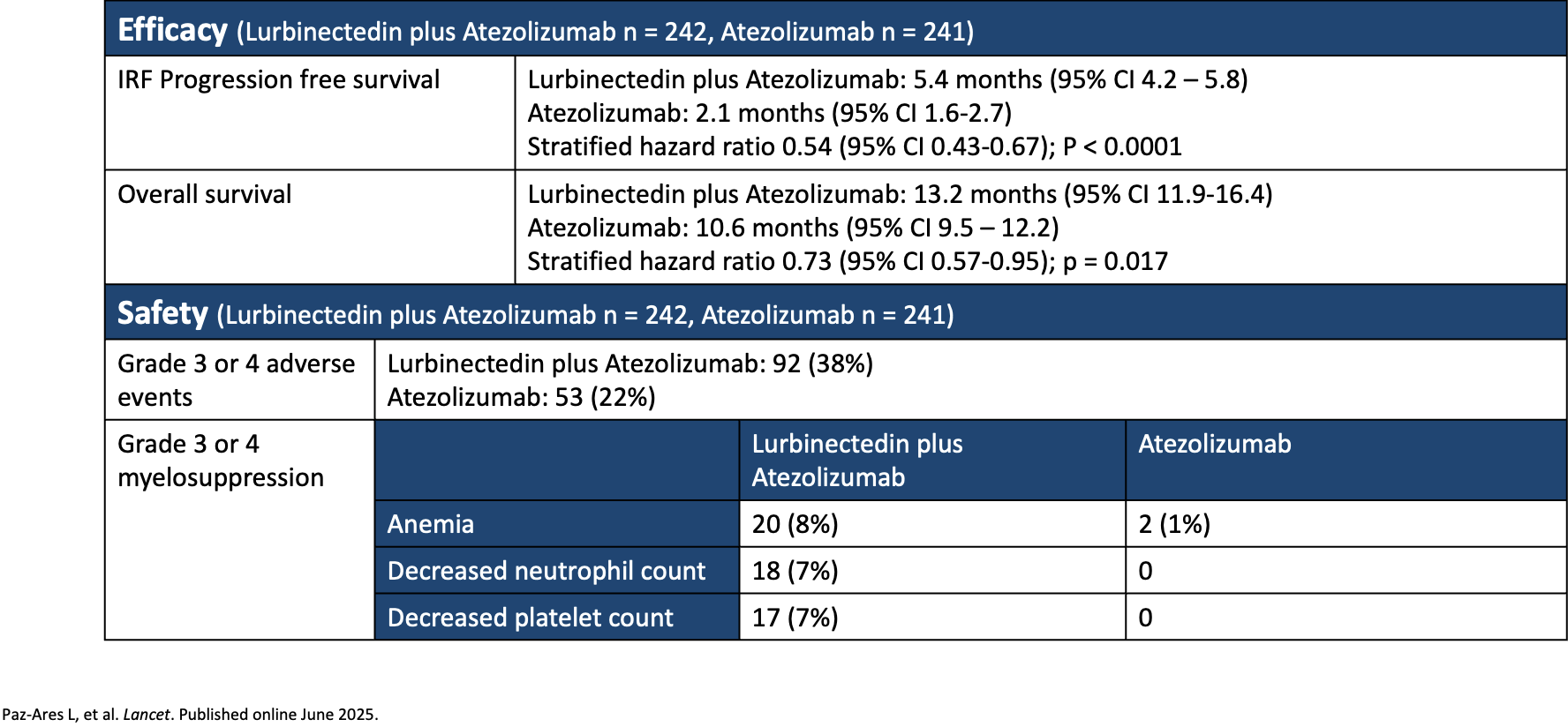
Researchers for the IMforte trial compared the efficacy and safety of first-line lurbinectedin plus atezolizumab versus atezolizumab monotherapy as maintenance treatment in patients with extensive-stage SCLC whose disease had not progressed after first-line induction treatment with atezolizumab, carboplatin, and etoposide. The primary end point included investigator-assessed PFS and OS. OS was 13.2 months with lurbinectedin plus atezolizumab versus 10.6 months with atezolizumab monotherapy. The researchers concluded that lurbinectedin plus atezolizumab had longer PFS and OS compared with atezolizumab, but the combination also had a higher incidence of adverse events (Table 2).
Table 2. IMforte Trial: Efficacy and Safety of First-Line Maintenance Therapy With Lurbinectedin Plus Atezolizumab in Extensive-Stage SCLC
Not all trials with ICIs in patients with SCLC have resulted in positive results. Examples of trials in SCLC with less robust findings include KEYNOTE-604, which looked at pembrolizumab plus chemotherapy with platinum and etoposide in patients with extensive-stage SCLC. A dual primary end point of OS and PFS was assessed, but the researchers did not find a significant improvement in the OS group. Similarly, researchers for the CheckMate 451 trial evaluated patients with extensive-stage SCLC who received nivolumab plus ipilimumab versus nivolumab monotherapy following first-line chemotherapy. The researchers did not find a significant OS benefit in the group receiving nivolumab plus ipilimumab.
Updated Approach to Initial Treatment: Immunotherapy
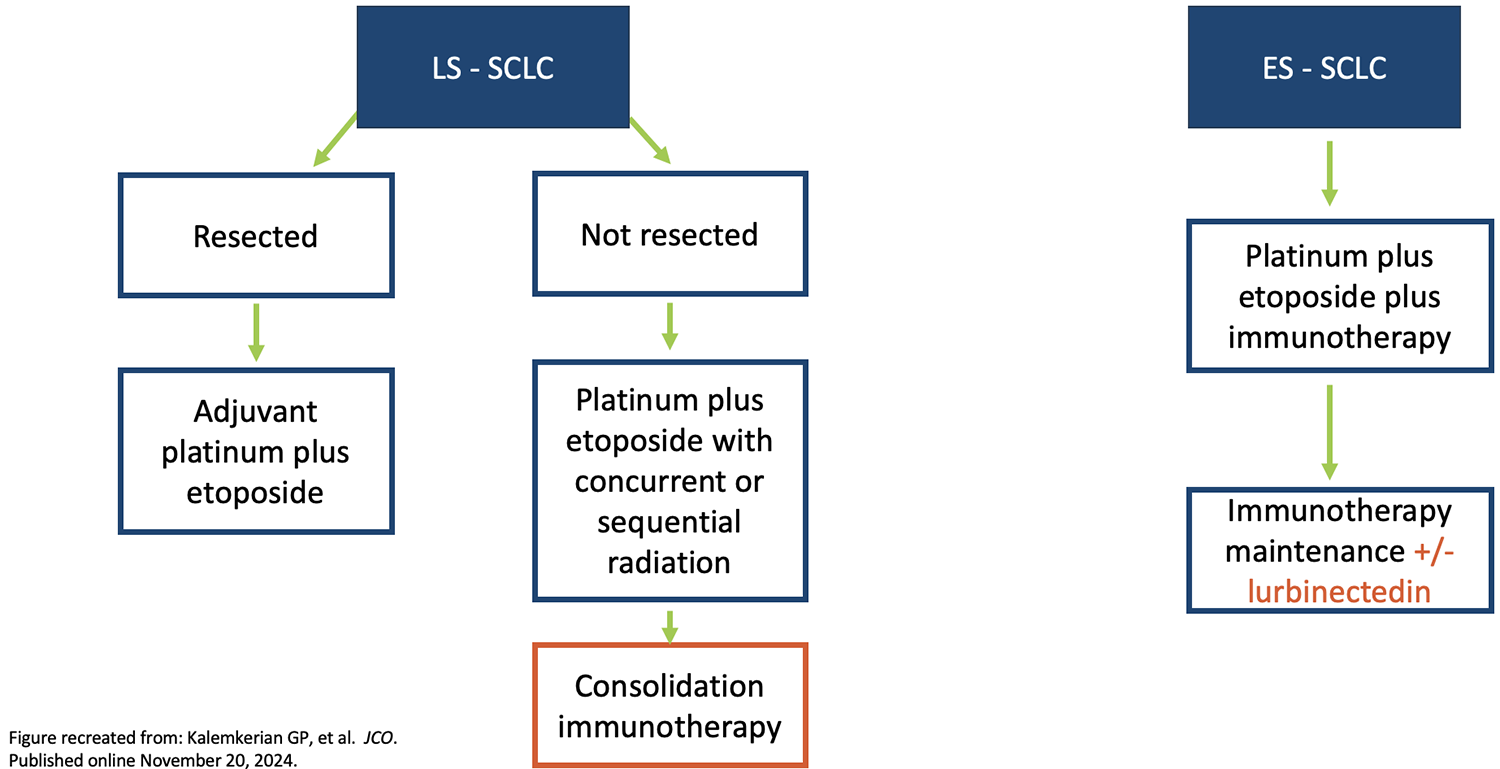
The updated approach to initial treatment of patients with newly diagnosed SCLC now includes consolidation immunotherapy for those with limited-stage SCLC, as well as the option to add lurbinectedin to maintenance immunotherapy in those with extensive-stage disease (Figure). Updates are also needed in the setting of relapsed disease because previously, second-line treatment options have been limited, with durations of response that have ranged from 3.6 to 5.3 months and an OS that has rarely exceeded 8 months.
Figure. Updated Approach to Treating Newly Diagnosed SCLS
Bispecific T-Cell Engagers in SCLC
Bispecific T-cell engagers are immunotherapeutic molecules designed to harness the body’s immune system against tumors or infected cells. They bind to tumor-associated antigens such as delta-like ligand 3 (DLL3), which is expressed on the surface of cancer cells in 85% to 94% of cases of SCLC; they also bind to the CD3 receptors on T cells. It is important to monitor for the adverse events associated with these agents, including cytokine release syndrome and immune effector cell–associated neurotoxicity syndrome.
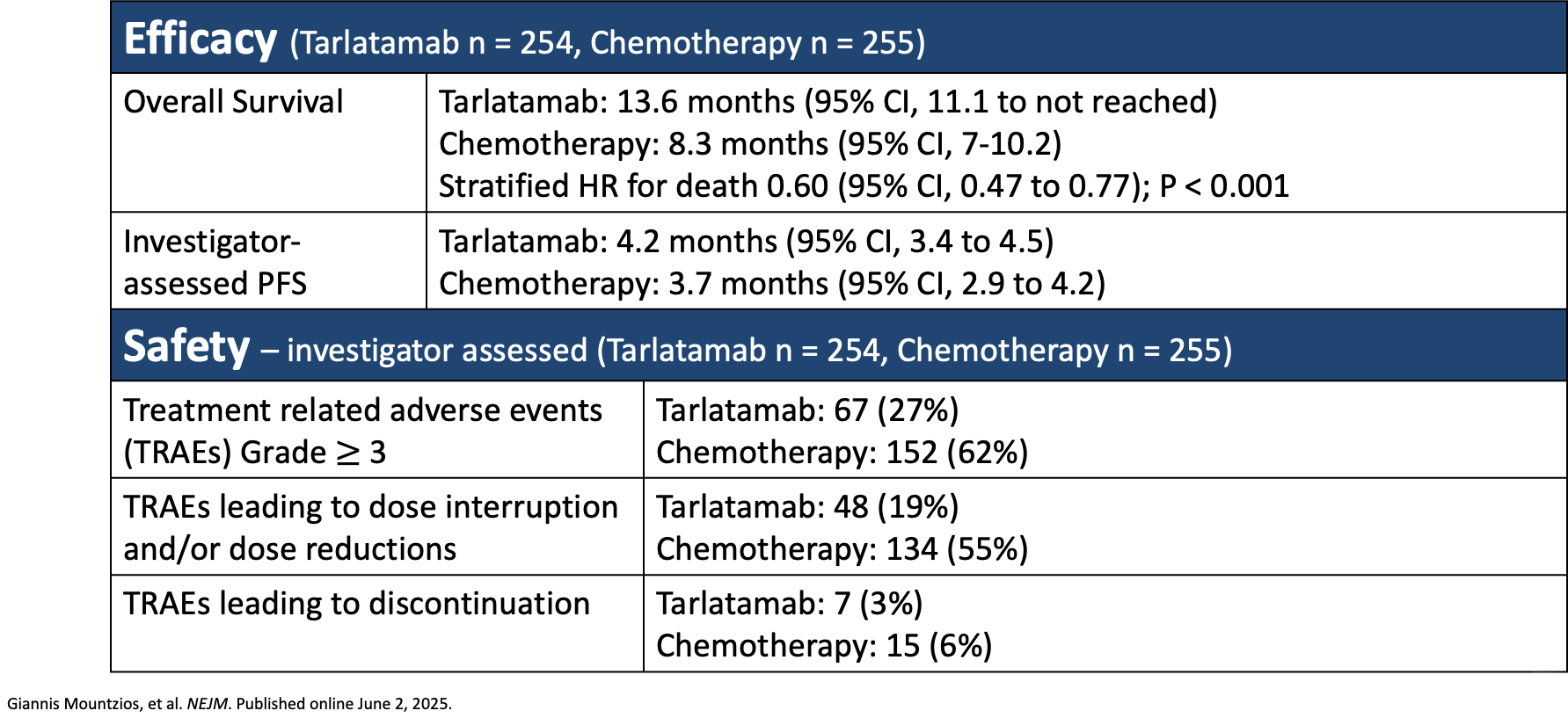
In the DELLphi-304 trial, researchers compared tarlatamab, a bispecific DLL3-directed T-cell engager, with chemotherapy (topotecan, lurbinectedin, or amrubicin) in patients with SCLC whose disease had progressed after receiving platinum-based chemotherapy. The primary end point was OS, and secondary end points included investigator-assessed PFS and patient-reported outcomes. The researchers found that OS was significantly longer in patients who had received tarlatamab (13.6 months) compared with the alternative chemotherapy regimen (8.3 months), and investigator-assessed PFS was also improved in the tarlatamab group (4.2 months) versus the chemotherapy group (3.7 months). In addition, tarlatamab showed a favorable safety profile. The incidence of investigator-assessed treatment-related adverse events grade ≥3 was 27% in the tarlatamab group and 62% in the chemotherapy group; in addition, treatment-related adverse events leading to discontinuation were 3% in the tarlatamab group versus 6% in the chemotherapy group (Table 3).
Table 3. DELLphi-304: Tarlatamab in Small Cell Lung Cancer After Platinum-Based Chemotherapy
Updated Approach to Treatment of the Patient Who Has Relapsed
The DELLphi-304 trial has resulted in updates to the treatment approach in the patient whose disease has relapsed. The usefulness of repeating a platinum-based chemotherapy regimen is currently being debated, but tarlatamab has been added as an option following disease progression in patients with a chemotherapy-free interval of <3 months, those with a chemotherapy-free interval of ≥3 months who are receiving maintenance immunotherapy, and those with a chemotherapy-free interval of ≥3 months who are not receiving maintenance immunotherapy.
Future Directions in SCLC
Bispecific/trispecific T-cell engagers in development include alveltamig, a trispecific T-cell engager that is currently in a phase 1 trial (NCT05978284). Gocatamig, another trispecific T-cell engager, is currently being evaluated in combination with ifinatamab deruxtecan (a B7-H3–targeting antibody-drug conjugate) and durvalumab (a PD-L1 inhibitor) to enhance antitumor activity. Gocatamig has 3 functional domains: CD3 binding for T-cell engagement, albumin binding for half-life extension, and DLL3 binding for tumor targeting.
DeLLphi-305 (NCT06211036) is a phase 3 trial exploring tarlatamab plus durvalumab versus durvalumab alone in patients with extensive-stage SCLC after first-line chemoimmunotherapy. DeLLphi-306 (NCT06117774) is a phase 3 trial assessing tarlatamab in patients with limited-stage SCLC without progression after concurrent chemoradiation therapy.
Peluntamig is a bispecific T-cell engager that targets DLL3 and CD47. RO7616789 is a trispecific T-cell engager being studied in advanced SCLC and other neuroendocrine carcinomas, and QLS31904 is a DLL3 and CD3 bispecific antibody in phase 1 testing (NCT05461287). Obrixtamig is a bispecific T-cell engager that binds to DLL3 on tumor cells and to CD3 on T cells. In the DAREON-9 trial, researchers are assessing obrixtamig plus topotecan in relapsed/refractory SCLC, and the DAREON-8 trial assesses obrixtamig plus standard of care for patients with extensive-stage SCLC.
Antibody-drug conjugates in clinical development that target trophoblast cell surface antigen 2 include sacituzumab govitecan and ESG-401. Agents targeting B7-H3 include ifinatamab deruxtecan. ZL-1310 is an antibody-drug conjugate that targets DLL3, and ABBV-706 targets SEZ6.
Summary
Immunotherapy has been shown to have added benefit in enhancing survival outcomes in patients with limited- or extensive-stage disease. The treatment landscape for patients with extensive-stage disease is changing as novel chemotherapy regimens and targeted approaches with T-cell engagers for maintenance therapy are explored in clinical trials. The development of T-cell engagers and antibody-drug conjugates will continue to change treatment approaches for patients with SCLC.
Sources
Surveillance Research Program. SEER*Explorer. NCI, November 5, 2024.
Rudin CM, et al. Nat Rev Dis Primers. 2021;7(1):1-20.
Kim SY, et al. JAMA. 2025;333:1906-1917.
Zhai X, et al. J Hematol Oncol. 2025;18:37.
Khurshid H, et al. JCO Oncol Pract. 2025;21:754-758.
Kalemkerian GP, et al. J Clin Oncol. 2025;43:101-105.
Horn L, et al. N Engl J Med. 2018;379:2220-2229.
Dutta R, et al. Scientific Reports. 2023;13:3739.
Lim SM, et al. JTO Clin Res Reports. 2025;6:100871.
Paz-Ares L, et al. Lancet. 2019;394:1929-1939.
Cheng Y, et al. N Engl J Med. 2024;391:1313-1327.
Paz-Ares L, et al. Lancet. 2025;405:2129-2143.
Albayrak G, et al. Brit J Cancer. 2025;133:1241-1249.
Mountzios G, et al. N Engl J Med. 2025;393:349-361.
Wermke M, et al. J Clin Oncol. 2025;43(suppl 16):8094-8094.
Peters S, et al. J Clin Oncol. 2024;42(suppl 16):TPS8127-TPS8127.
Lim SM, et al. JTO Clin Res Reports. 2025;6:100871.
Dowlati A, et al. J Thorac Oncol. 2025;S1556-0864(24)025498.
Wang J, et al. J Thorac Oncol. 2024;19:S17.
Rudin C, et al. J Thorac Oncol. 2024;19(10).
Rosa K. Onclive.com. Accessed April 21, 2026. www.onclive.com/view/ifinatamab-deruxtecan-elicits-responses-in-previously-treated-extensive-stage-small-cell-lung-cancer
Ahn M-J, et al. Presented at: International Association for the Study of Lung Cancer (IASLC) 2025 World Conference on Lung Cancer; September 6-9, 2025; Barcelona, Spain. Abstract OA06.03.
Byers LA, et al. Presented at: International Association for the Study of Lung Cancer (IASLC) 2025 World Conference on Lung Cancer; September 6-9, 2025; Barcelona, Spain. Abstract OA06.04.