With commentaries by

Joyce A. O’Shaughnessy, MD
Disease Chair, Breast Cancer Research Executive Committee
Sarah Cannon Research Institute
Texas Oncology - Baylor Charles A. Sammons Cancer Center
Dallas, TX
Introduction
Distant breast cancer recurrences, even following 5 years of adjuvant endocrine therapy (ET), remain a frequent and life-threatening complication for patients with early, hormone receptor (HR)-positive breast cancer with high-risk clinicopathological features.1 Patients with high-risk, HR-positive, HER2-negative early breast cancer (eBC) have approximately a 40% risk of developing metastatic breast cancer at 20 years postdiagnosis.1
A real-world study showed that patients with node-positive eBC who presented with features characteristic of high risk of disease recurrence had approximately a 30% risk of invasive disease recurrence or death by 5 years.2 In another real-world study, all node-positive subgroups (N1 high-risk, N2, N3) representative of monarchE cohort 1 had an increased recurrence risk ≥2.7-fold higher.3
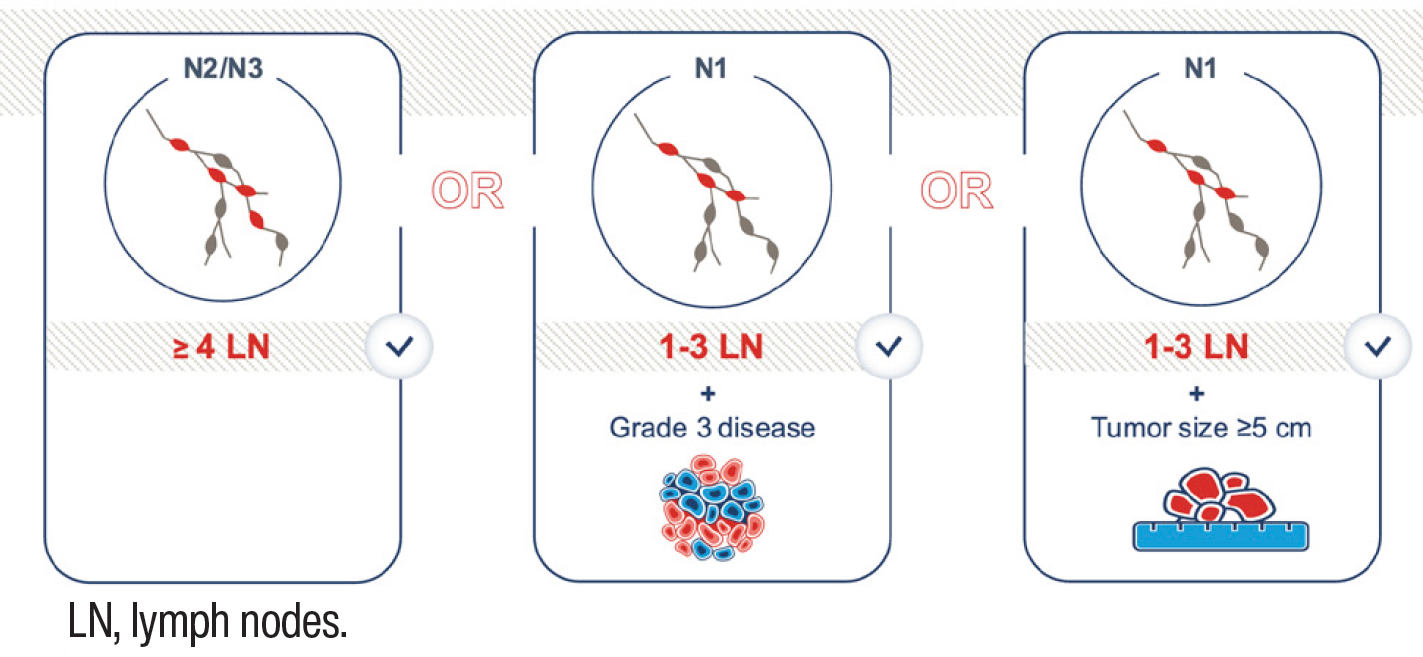
Features characteristic of high risk of disease recurrence include2:
- ≥4 positive lymph nodes (LNs) or
- 1-3 positive LNs and at least 1 of the following:
- Histological grade 3
- Tumor size ≥5 cm
The data highlights the need for a therapy that may potentially reduce the risk of breast cancer recurrence among patients with high-risk features.
monarchE: Primary analysis
The monarchE study was a global, open-label, randomized, phase 3 trial composed of 5637 adults with high-risk, HR-positive, HER2-negative eBC who received prior treatment with surgery and radiotherapy and/or chemotherapy (Figure 1).4 Some of the approved patient population for the monarchE trial included patients with T1 tumors and micrometastases if their tumors were grade 3, patients with stage II disease, and patients with stage III disease. High-risk disease was defined as either ≥4 positive axillary lymph nodes (ALN), or between 1 and 3 positive ALN and either grade 3 disease or tumor size of ≥5 cm (cohort 1, n=5120).5 A smaller group of patients were enrolled with 1 to 3 ALN and Ki-67 of at least 20% as an additional risk factor (cohort 2, n=517).5
Commentary by Joyce O’Shaughnessy, MD: In terms of who should get adjuvant abemaciclib, ASCO has endorsed the broader intent-to-treat population in monarchE (patients from cohorts 1 and 2).
All patients underwent 1:1 randomization of abemaciclib 150 mg orally twice daily plus current standard-of-care adjuvant ET or current standard-of-care adjuvant ET alone for 2 years (treatment duration).4 The primary endpoint of the study was invasive disease-free survival (IDFS), defined as time from randomization to the first occurrence of local or regional recurrence, contralateral recurrence, second primary non-breast invasive cancer, distant recurrence, or death attributable to any cause. Secondary endpoints included distant relapse-free survival (DRFS), overall survival (OS), safety, and patient-reported outcomes.4 A prespecified OS interim analysis is planned to occur 2 years after the primary outcome analysis for IDFS.5
Commentary by Joyce O’Shaughnessy, MD: I’ve had a lot of experience now using adjuvant abemaciclib both in the monarchE trial and then after its approval. Patients are generally very open to taking abemaciclib because they know they’re at high risk.
monarchE: Outcomes at 2 years of treatment and 42-month median follow-up
Compared with adjuvant ET alone, adjuvant abemaciclib plus ET persistently showed significant improvement in IDFS (hazard ratio: 0.664; 95% confidence interval [CI], 0.578-0.762; nominal P<.0001) and DRFS (hazard ratio: 0.659; 95% CI, 0.567-0.767; nominal P<.0001).5 OS data were immature, and significance between the groups was not reached at the time of the prespecified OS analysis.5 Efficacy of adjuvant abemaciclib was generally consistent across relative dose intensities and uncompromised by dose reductions.6 A majority (72.2%) of patients completed the full 2 years of treatment. Manageable, acceptable, and predictable safety profile and tolerability were consistent with the well-established safety profile of abemaciclib.7 The most common adverse events (incidence ≥20%) were diarrhea, neutropenia, nausea, abdominal pain, infections, fatigue, anemia, leukopenia, decreased appetite, vomiting, headache, alopecia, and thrombocytopenia.8 Only 5.3% of patients discontinued abemaciclib because of diarrhea. A positive benefit-risk balance was demonstrated in patient-reported outcomes.7,9 Patient quality of life was not impacted by diarrhea. From ≥3 months, most patients who experienced diarrhea in the abemaciclib arm reported having diarrhea “a little bit” or “somewhat.” These 2-year data led to regulatory approval in the United States and European Union of adjuvant abemaciclib in combination with ET in selected patients with eBC and high risk of recurrence.8,10 Following the positive interim analysis, patients continued to be followed for IDFS, distant recurrence, and OS.
Commentary by Joyce O’Shaughnessy, MD: We need to see the patients every 2 weeks for 2 months as was done per protocol because the toxicity management takes place in the first 2 months. We do tell the women that diarrhea occurs the most frequently in the first 4 to 6 weeks and prophylatic diarrhea treatment is provided at the initiation of abemaciclib. It can get better after that but may still persist at a generally low level in some women.
At a median follow-up of 42 months, the steady separation of IDFS and DRFS curves along with a notable increased absolute benefit at 4 years, indicated continuous improvement beyond treatment. The effects of abemaciclib were generally consistent across relative dose intensity (RDI) subgroups with no clinically meaningful differences in the estimated 4-year IDFS rates between the RDI subgroups (4-year rates: 87.1, 86.4, and 83.7).6 At a median follow-up of 42 months, dose reductions were essential to managing adverse events for maximal adherence to treatment and achievement of optimal response; however, this did not adversely affect the efficacy of abemaciclib (Figure 2).6 Of patients treated with adjuvant abemaciclib plus ET, 1221 (43.7%) patients had dose reductions, including 29.8% with 1 and 13.9% with 2 dose reductions.6
In addition, dose reductions did not have an impact on IDFS or DRFS during the 2-year treatment period. The abemaciclib dose of 150 mg BID plus ET had a similar IDFS compared with doses of 100 mg BID or 50 mg BID with a hazard ratio of 0.899 (95% CI, 0.718-1.1125). Similarly, the abemaciclib dose of 150 mg BID plus ET had a similar DRFS compared with doses of 100 mg BID or 50 mg BID with a hazard ratio of 0.958 (95% CI, 0.750-1.223).
Commentary by Joyce O’Shaughnessy, MD: I think one thing that’s so important is that we have these very important data now from monarchE showing that women who had 1 or even 2 dose reductions, didn’t have a worse outcome than patients who had no dose reduction.
Commentary by Joyce O’Shaughnessy, MD: When patients start taking abemaciclib at the full dose, 150 mg twice daily, there is about 8% of patients who have very substantial diarrhea. And it’s very important to tell them to stop it if that happens. And then we’ll reduce the dose. Now it turns out that when you decrease the dose of 150 mg to 100 mg, it generally makes a really big difference for patients in terms of being able to easily manage the diarrhea, if they have any at all. Also, when we reduce the dose to help patients stay on abemaciclib, we know we will see fewer recurrences because of the abemaciclib. And so, getting that dose reduction really helps people stay on it without loss of efficacy.
Here, we present updated efficacy outcomes of the 5-year landmark analyses.
monarchE: 5-year data
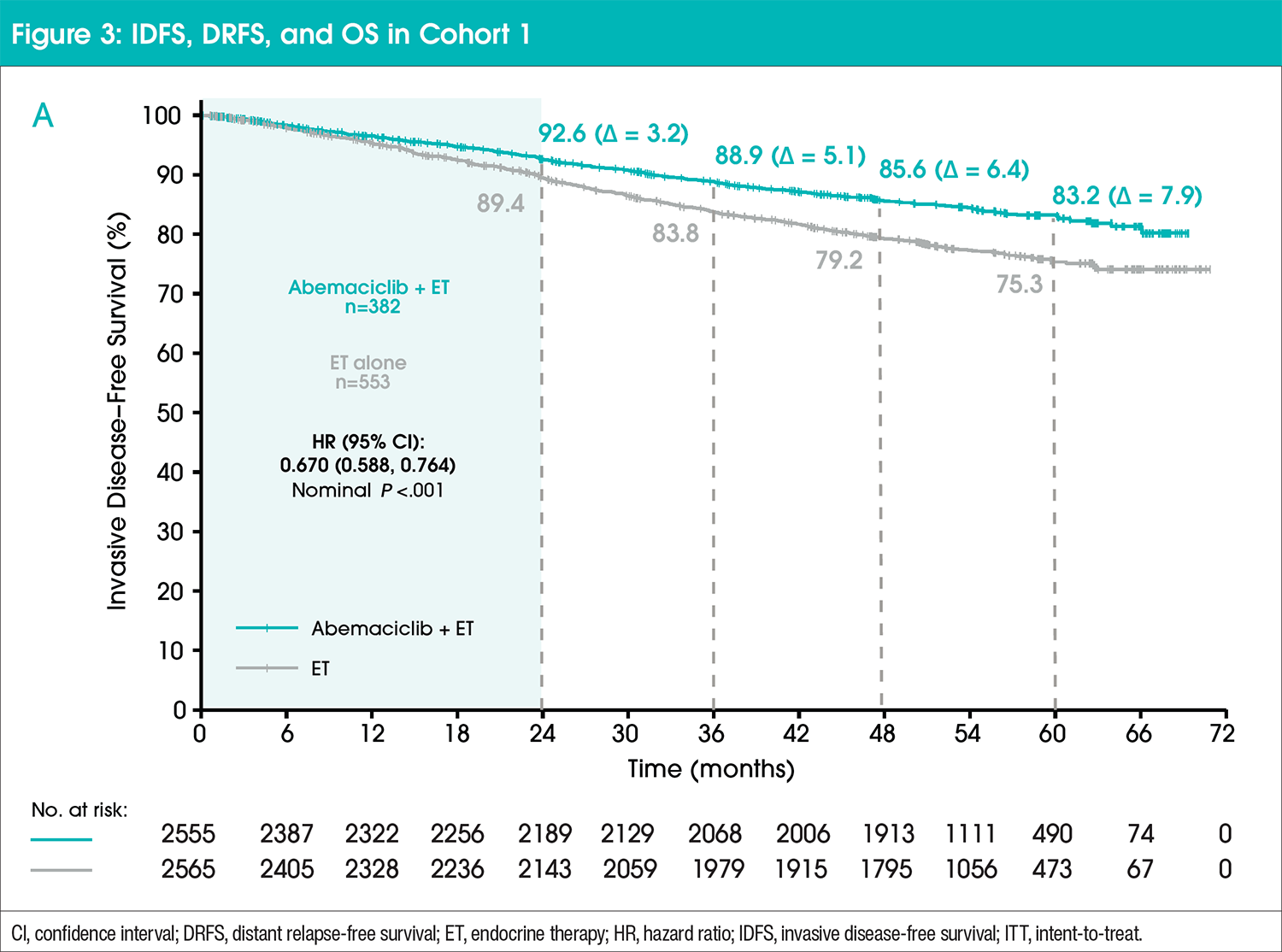
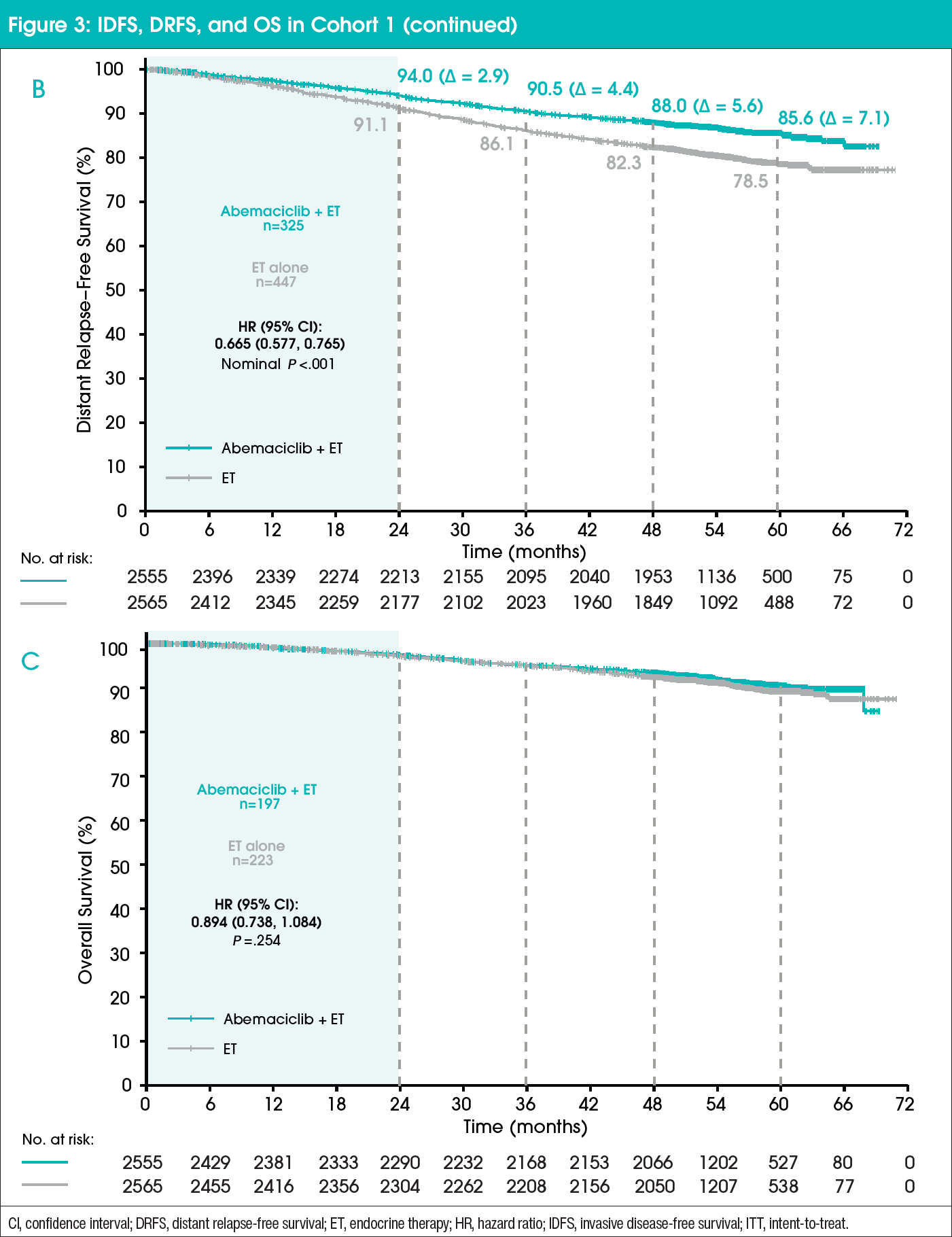
In cohort 1, IDFS, DRFS, and OS were consistent with the intent-to-treat population (Figure 3).11 Absolute improvement in IDFS rates further deepened at 5 years (7.6%) compared with 6%, 4.8%, and 2.8% at 4, 3, and 2 years, respectively.11 The addition of abemaciclib to ET also resulted in an improvement in DRFS compared with ET alone.10 At 5 years, the absolute benefit in DRFS rates increased to 6.7% compared with 5.3%, 4.1%, and 2.5% at 4, 3, and 2 years, respectively.11 OS data remain immature.11 No new safety findings were found in the long-term follow-up with no cumulative or persistent symptoms observed after treatment completion.11 Furthermore, these results demonstrate a 3-year carryover effect, with treatment benefits persisting beyond the 2-year treatment period.11
The benefit of adjuvant abemaciclib plus ET was also seen in subpopulations including premenopausal patients and patients who received neoadjuvant chemotherapy. In premenopausal patients, the 4-year IDFS rate was 88.1 (95% CI, 86.1-89.9) in the abemaciclib plus ET arm versus 80.7 (95% CI, 78.3-82.9) in the ET arm with a hazard ratio of 0.597. In patients with prior neoadjuvant chemotherapy, the 4-year IDFS rate in the abemaciclib plus ET arm was 81.0 (95% CI, 78.4-83.4) versus 71.9 (95% CI, 68.9-74.6) in the ET arm with a hazard ratio of 0.649.11
Commentary by Joyce O’Shaughnessy, MD: What was so important about the 5-year data is to see the women at 5 years, when everybody’s been off abemaciclib for at least 2 to 3 years. The amount of recurrences is smaller in the abemaciclib arm than in the ET arm leading to further curve separation. The IDFS benefit continues to get larger over time; it hasn’t plateaued. So that was a big deal. The benefit is improving. There continues to be proportionally more recurrences in those who didn’t get abemaciclib than in those women who did get abemaciclib. I’m looking forward to seeing longer follow-up to see if the IDFS improvement gets even larger.
Conclusion
There is an unmet medical need for patients with eBC with high-risk features including ≥4 positive ALN and 1 to 3 positive ALN with at least 1 of the following: histological grade 3, or tumor size ≥5 cm. The addition of abemaciclib to standard-of-care ET in the adjuvant treatment of HR-positive, HER2-negative, high-risk eBC provided clinical benefit, even with dose reductions, with deepening of the absolute benefit in IDFS and DRFS rates at 5 years compared with that of previous years, suggesting a sustained benefit beyond the treatment period.
Commentary by Joyce O’Shaughnessy, MD: This is a major advance in the adjuvant setting. We haven’t had a big advance in HR-positive, HER2-negative, high-risk eBC since the 1990s. So this is a really clinically significant improvement in disease-free survival that we really need to discuss and recommend to every woman who fits the monarchE eligibility criteria. My personal approach is to offer the abemaciclib to all patients with HR-positive, HER2-negative, high-risk, node-positive eBC.
References
- Pan H, Gray R, Braybrooke J, et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377:1836-1846.
- Sheffield KM, Peachey JR, Method M, et al. A real-world US study of recurrence risks using combined clinicopathological features in HR-positive, HER2-negative early breast cancer. Future Oncol. 2022;21:2667-2682.
- Tolaney SM, Sammons S, Cortes J, et al. Real-world risk of recurrence by nodal status in patients with HR+, HER2-, node-positive, high-risk early breast cancer. Presented at: San Antonio Breast Cancer Symposium 47th Annual Meeting; San Antonio, TX; December 10-14, 2024.
- Johnston SRD, Harbeck N, Hegg R, et al. Abemaciclib combined with endocrine therapy for the adjuvant treatment of HR+, HER2-, node-positive, high-risk, early breast cancer (monarchE). J Clin Oncol. 2020;34:3987-3998.
- Johnston SRD, Toi M, O’Shaughnessy J, et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. 2023;1:77-90.
- Goetz M, Cicin I, Testa L, et al. Impact of dose reductions on adjuvant abemaciclib efficacy for patients with high-risk early breast cancer: analyses from the monarchE study. npj Breast Cancer. 2024;10:34.
- Rugo HS, O’Shaughnessy J, Boyle F, et al. Adjuvant abemaciclib combined with endocrine therapy for high-risk early breast cancer: safety and patient-reported outcomes from the monarchE study. Ann Oncol. 2022;6:616-627.
- Verzenios (abemaciclib) [prescribing information]. Netherlands: Eli Lilly Nederland; 2022.
- Tolaney SM, Guarneri V, Seo JH, et al. Long-term patient-reported outcomes from monarchE: abemaciclib plus endocrine therapy as adjuvant therapy for HR+, HER2-, node-positive, high-risk, early breast cancer. Eur J Cancer. 2024;199:113555.
- Verzenio (abemaciclib) [prescribing information]. Indianapolis, IN: Eli Lilly; 2021.
- Rastogi P, O’Shaughnessy J, Martin M, et al. Adjuvant abemaciclib plus endocrine therapy for hormone receptor–positive, human epidermal growth factor receptor 2–negative, high-risk early breast cancer: results from a preplanned monarchE overall survival interim analysis, including 5-year efficacy outcomes. J Clin Oncol. 2024;9:987-993.