Connecting Science to Practice
Investigational drug service (IDS) pharmacists play an important
role in clinical trials, in which they work with a multidisciplinary
research team to provide patient care. The IDS team has a valuable
part in helping coordinate investigational drugs for clinical trials.
The traditional IDS pharmacist’s responsibilities include medication
management, site initiation visits, and the development of
procedures that detail the IDS workflow. Nontraditional activities
include patient counseling, providing supportive-care management,
investigational drug and standard-of-care dose modifications,
medication adherence assessments, and other clinical tasks.
The objective of this study is to define the value of nontraditional
IDS pharmacy practice through the evaluation of traditional and
nontraditional IDS pharmacy metrics to expand the pharmacy IDS
practice. By defining nontraditional IDS pharmacy metrics, the
goal is to give ideas for supporting the IDS pharmacy staff’s responsibilities
to include more direct patient care.
The current models for conducting clinical trials in oncology involve a multidisciplinary research team working collectively to provide patient care. The research team is led by the site’s principal investigator who oversees operations conducted by clinical research coordinators, regulatory specialists, research assistants, clinical trial analysts, and investigational drug services (IDS).1-3 The IDS is comprised of a team of pharmacists and pharmacy technicians. These individuals play a central role in the successful delivery of clinical trials involving investigational drugs. The Hematology/Oncology Pharmacy Association (HOPA) has published its Investigational Drug Service Best Practice Standards to facilitate the delivery of quality oncology care for patients enrolled in clinical trials. It is recommended that all healthcare centers participating in clinical trials that include investigational drugs have an IDS-dedicated pharmacist as a member of the multidisciplinary research team.4 As oncology research programs continue to expand, it is necessary for IDS staffing to increase to meet the rising demands and increased complexity of clinical trials. Justification of these roles is complex, especially because daily tasks encompass traditional and nontraditional IDS pharmacy activities.
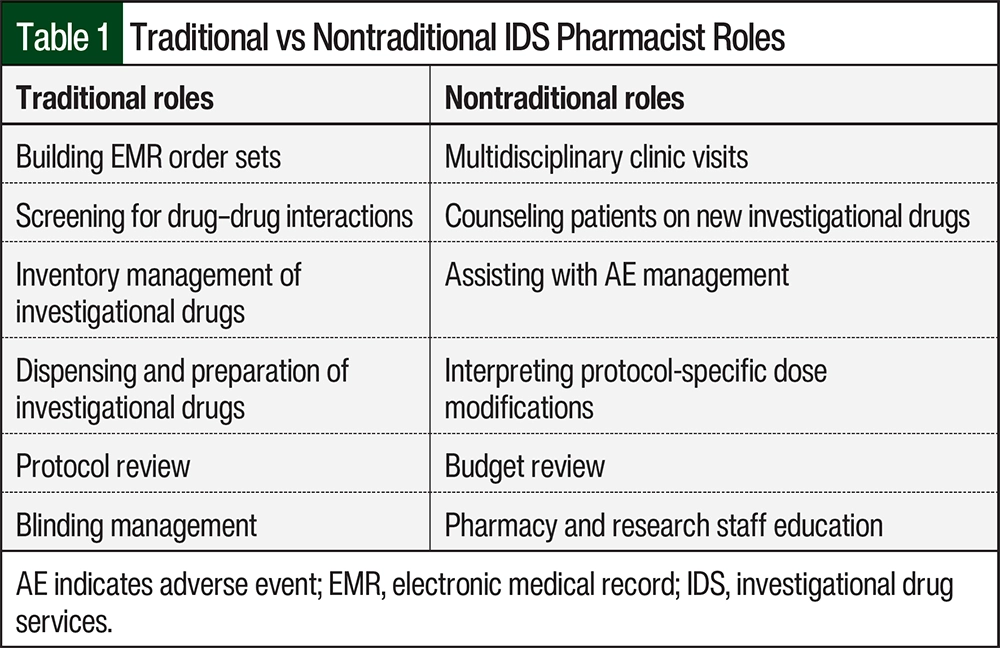
Traditional IDS pharmacists’ responsibilities include tasks such as inventory tracking, temperature management, protocol review, site initiation visits, blinding management, creation of protocol-specific medication order sets, and the development, implementation, and maintenance of policies and procedures that detail the IDS workflow.4-6 Of these activities, traditional IDS metrics have included the numbers of dispensations, investigational drug shipments received, and active protocols. Tracking additional study metrics is often difficult because of the various complexity levels associated with protocol initiation and maintenance. Complexity can range in studies for multiple reasons, including novel drugs, complex formulations, and specific protocol requirements.4-6 To define research staff workload and account for protocol-specific complexities, research departments often assign an internally developed complexity score to each protocol. To estimate the workload, Good and colleagues developed a complexity scoring system that uses a 4-point acuity score, with 1 being the least complex and 4 being the most complex. An acuity score of 1 was used for an observation or registry trial, whereas a score of 4 was assigned to protocols consisting of multidrug regimens, a high potential for adverse events (AEs), and multiple non–standard-of-care data collection points. These metrics were then used to estimate the workload and determine the number of nurses needed based on the acuity score.7
In a study conducted in Italy, Milani and colleagues evaluated the average time required for nursing to complete various clinical research tasks to develop a tool to estimate the nursing staff requirements.8 The study compared phase 1 trials with phase 2 and 3 trials to evaluate the differences in time requirements. The tasks that were measured included education, administration of the drug, reviewing protocols, and nursing documentation. The data collected were categorized into 30 different tasks that required completion for each study patient and the average time needed for each task was estimated. On average, a phase 1 trial required 1662 minutes per patient whereas phase 3 trials required 1124 minutes per patient.8
By assigning complexity levels, protocol-specific workload metrics can be combined with traditional metrics to better reflect the true staffing requirements. Although acuity scores have improved staff resource management for traditional study-related activities, there remains an unmet need for the determination of how to effectively track nontraditional activities through the use of metrics.
As research departments continue to expand, so will the roles and responsibilities of IDS pharmacists. It is important that these individuals are willing to accept nontraditional clinical responsibilities in addition to the traditional roles described above. The nontraditional activities of an IDS pharmacist include activities that are recommended by HOPA’s Investigational Drug Service Best Practice Standards.4 These tasks include patient counseling, attending multidisciplinary appointments with the research team, providing supportive care recommendations, reporting AEs, and assessing medication adherence.4 In addition, clinical technology can be used in nontraditional responsibilities, such as the development of screening alerts in the electronic medical record (EMR).6 Historically in our experience, identifying and tracking nontraditional metrics has been a difficult task and therefore requires additional investigation to effectively define their value.
Although many institutions that conduct clinical trial research incorporate an IDS pharmacist into their workflow, few have practice models that integrate a pharmacist within their multidisciplinary team. At Avera Cancer Institute, pharmacists participate in traditional IDS services to operationalize clinical trials through building EMR order sets, screening for drug–drug interactions, and dispensing investigational drugs. In addition, IDS pharmacists also manage nontraditional responsibilities, such as attending multidisciplinary clinic visits, counseling patients on new investigational drugs, assisting with AE management, and interpreting protocol-specific dose modifications. Additional evaluation and research are needed to define nontraditional IDS responsibilities and metrics.
This pilot project aims to describe and review the interventions associated with a nontraditional IDS practice model. The study results will be used to define the value of measuring traditional and nontraditional IDS services through the use of metrics at the Avera Cancer Institute. This study’s results may be used at other institutions to provide full-time equivalent (FTE) justification or potential opportunities for IDS growth.
Methods
In this retrospective, multisite study, the investigators identified all patients receiving services from Avera Cancer Institute’s research department in October and November of every year from 2018 to 2021. A 2-month time period was selected for this pharmacy residency project because of the archiving limitations associated with paper research records. The data were collected from October and November of each year and were averaged to determine the metrics and workload. This study was deemed exempt from review by the local institutional review board (IRB).
All patients considered for, screened for, or enrolled in a clinical trial and those receiving services or investigational drugs from the Avera Cancer Institute’s research team during the time periods mentioned above were included. IRB-approved oncology treatment trials that were open at the Avera Cancer Institute and required pharmacy involvement within the time periods were included. Patients aged <18 years or patients who were assigned to observation and did not require IDS activities were excluded.
Data were collected from the EMR and research records over the specified time frame and included patients receiving active treatment during the defined study period, patients dispensed investigational drugs associated with a clinical trial, the location of the dispense, and adherence to the investigational drugs. The clinical interventions by IDS pharmacists were tracked by category, which included drug–drug interaction reviews, investigational drug counseling, dose modification/AE management, and miscellaneous interventions. Other data collected included the number of IRB-approved oncology treatment trials, the complexity level of the trials, the total number of investigational drug dispensations, the number of trials with start-up fees, the average start-up time per new study, the number of protocol amendments completed, the number of investigational drug shipments received, and the IDS pharmacy hours staffed monthly. The traditional and nontraditional metrics for pharmacists that were collected are shown in Table 1.
The complexity level for each study was defined and standardized at the institutional level. A complexity level of 1 required the least amount of pharmacy involvement (eg, clinical trial involving an FDA-approved agent, but a new regimen), whereas a complexity level of 3 required the most pharmacy involvement (eg, first-in-human phase 1 clinical trial). In addition, an internal survey was conducted among research staff members to determine how other research staff members viewed pharmacy services. The survey was also used to identify potential growth areas for pharmacy involvement based on the feedback provided. The survey was adapted from an expert panel survey on pharmacists’ roles in IDS (Appendix Table).9 All responses from the survey were anonymous.
The primary objective of this study was to define the value of nontraditional IDS pharmacy practice through the evaluation of traditional and nontraditional IDS pharmacy metrics to expand the pharmacy IDS practice. The 2 secondary objectives of this study were to identify future traditional and nontraditional metrics to promote the expansion of a nontraditional IDS pharmacy practice based on historical site data, and to identify areas of future development for IDS pharmacists within the research team to more effectively deliver patient care through the analysis of a research department survey.
All categorical values were reported as a single count with frequencies. Data collection and analysis were performed in Microsoft Excel. No statistical tests were performed for this study.
Results
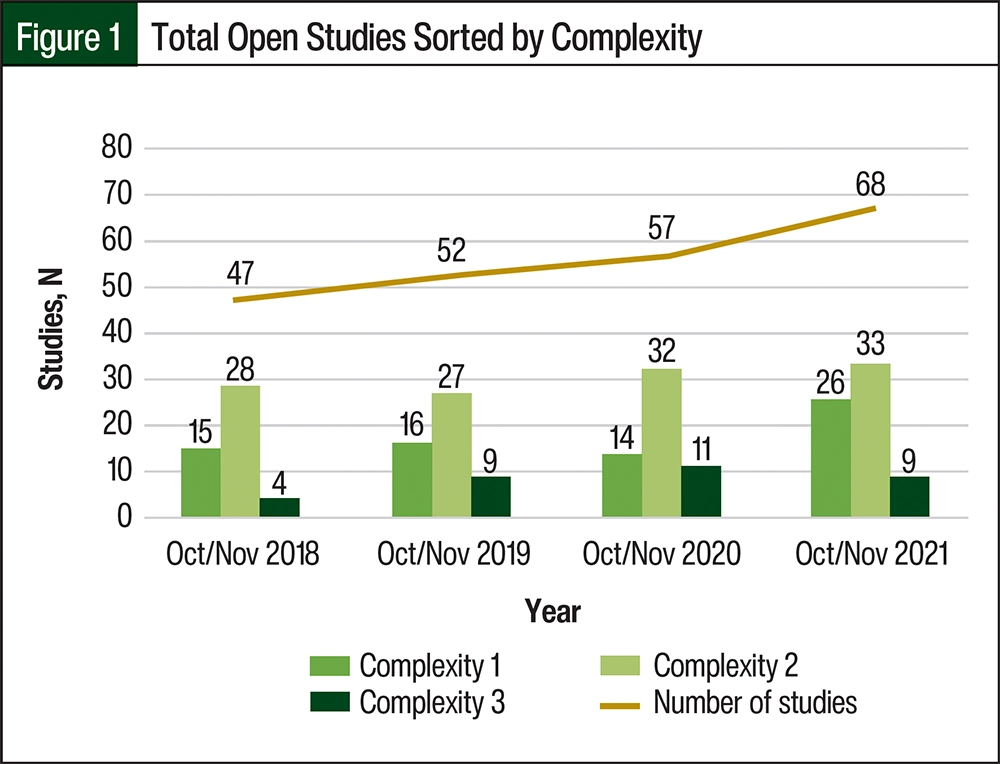
During the months of October and November from 2018 to 2021, there was an increase in the number of clinical trials open to enrollment. A total of 47 studies were open in 2018, 52 in 2019, 57 in 2020, and 68 in 2021 (Figure 1). Throughout the study time period, there was an approximately 45% increase in open studies at the site. In 2018, approximately 32% of open studies were classified as complexity level 1, 60% were complexity level 2, and 8% were complexity level 3. In 2021, the level 3 studies had increased to approximately 13%, which was almost double the number of level 3 trials in 2018.
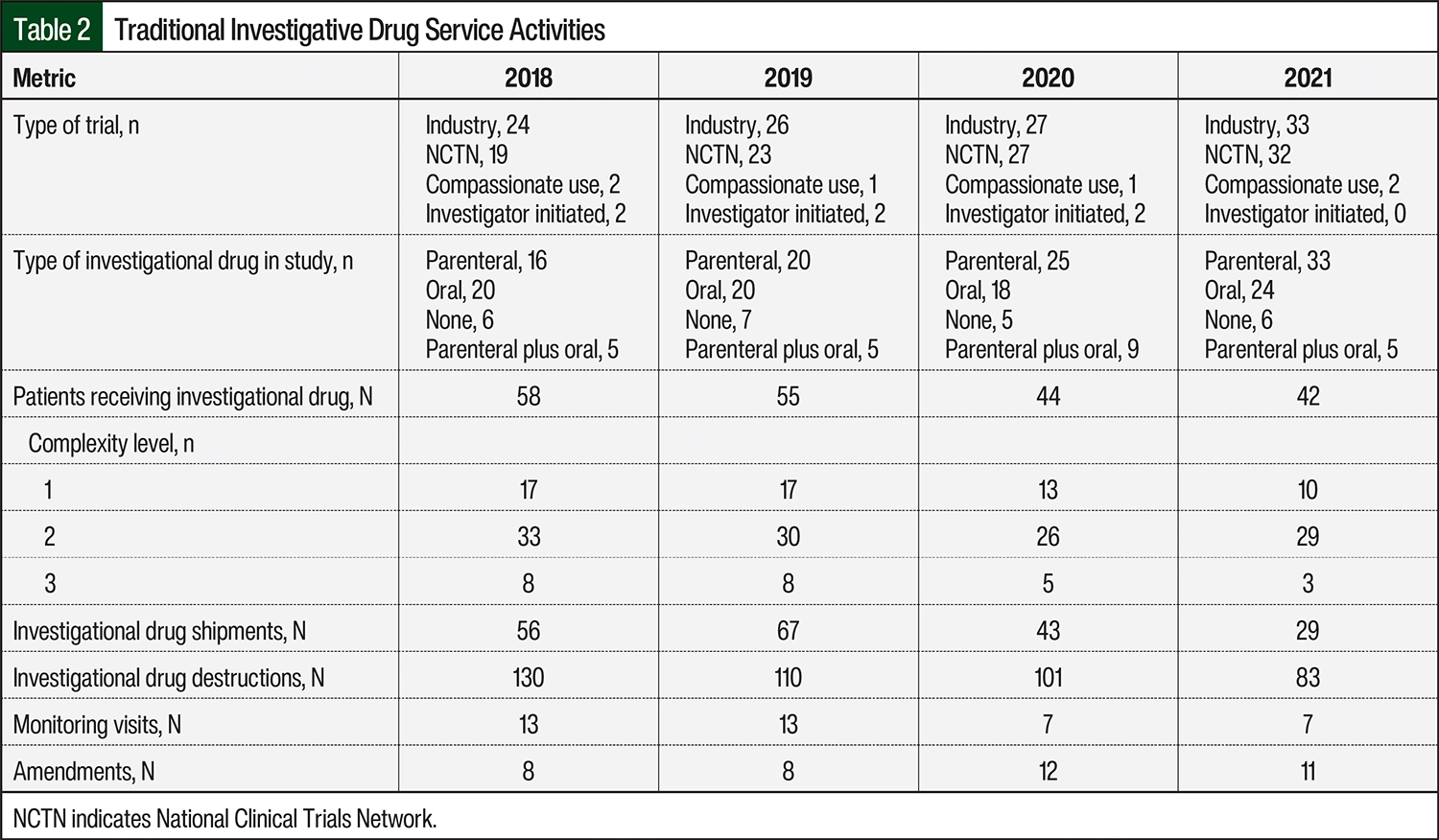
The trial types (eg, compassionate use, investigator-initiated, industry, and National Clinical Trials Network [NCTN]) across the duration of the study were consistent (Table 2). During the study’s time period, the approximate average number of open studies by trial type included 28 for industry, 26 for NCTN, 2 for compassionate use, and 2 for investigator-initiated studies. Over the 4-year time period, the number of open NCTN trials increased by 68% whereas the number of open industry trials increased by 38%. Compassionate use and investigator-initiated studies comprised a small percentage of the open studies throughout the study duration.
The studies were further classified into investigational drug route of administration categories, including parenteral, oral, parenteral plus oral, or none (Table 2). From 2018 to 2021, the number of studies with parenteral investigational drugs doubled from 16 to 33 studies. The number of trials with oral investigational drugs also increased, from 20 studies in 2018 to 24 studies in 2021. The number of studies that had no investigational drugs or parenteral investigational drugs plus oral investigational drugs were consistent throughout the study duration.
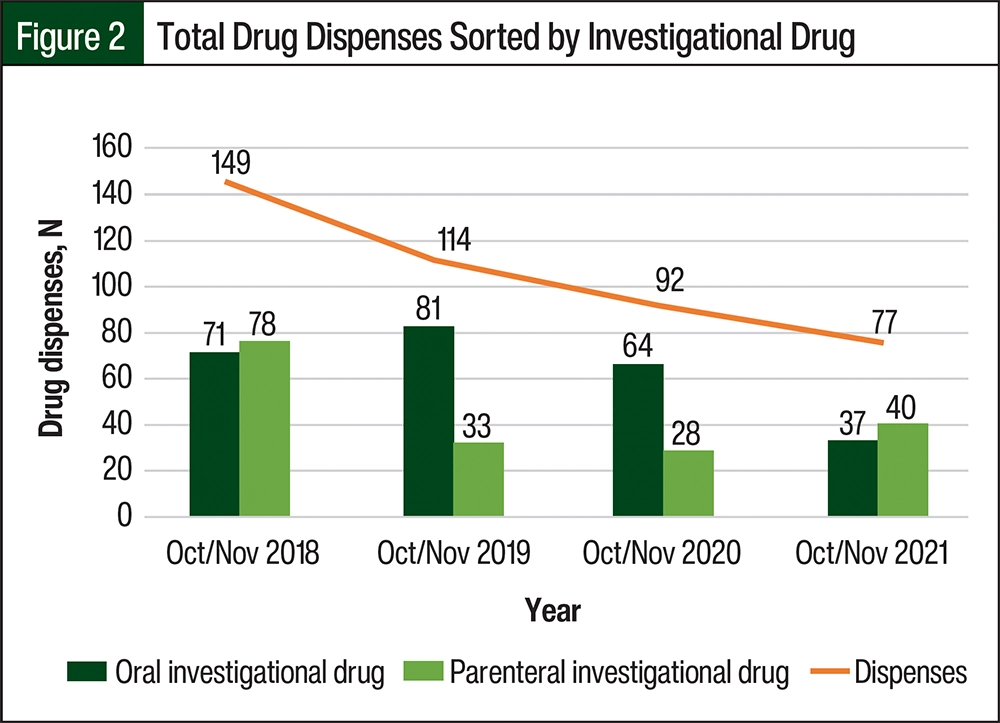
The total number of patients who received investigational drug dispensations was 58 in 2018, 55 in 2019, 44 in 2020, and 42 in 2021 (Table 2). The total number of investigational drug shipments also decreased when comparing 2018 to 2021 (Figure 2).
The number of patients in complexity level 2 studies remained constant throughout the study’s duration (Table 2). The number of patients in complexity level 3 studies decreased from 8 patients in 2018 to 3 patients in 2021.
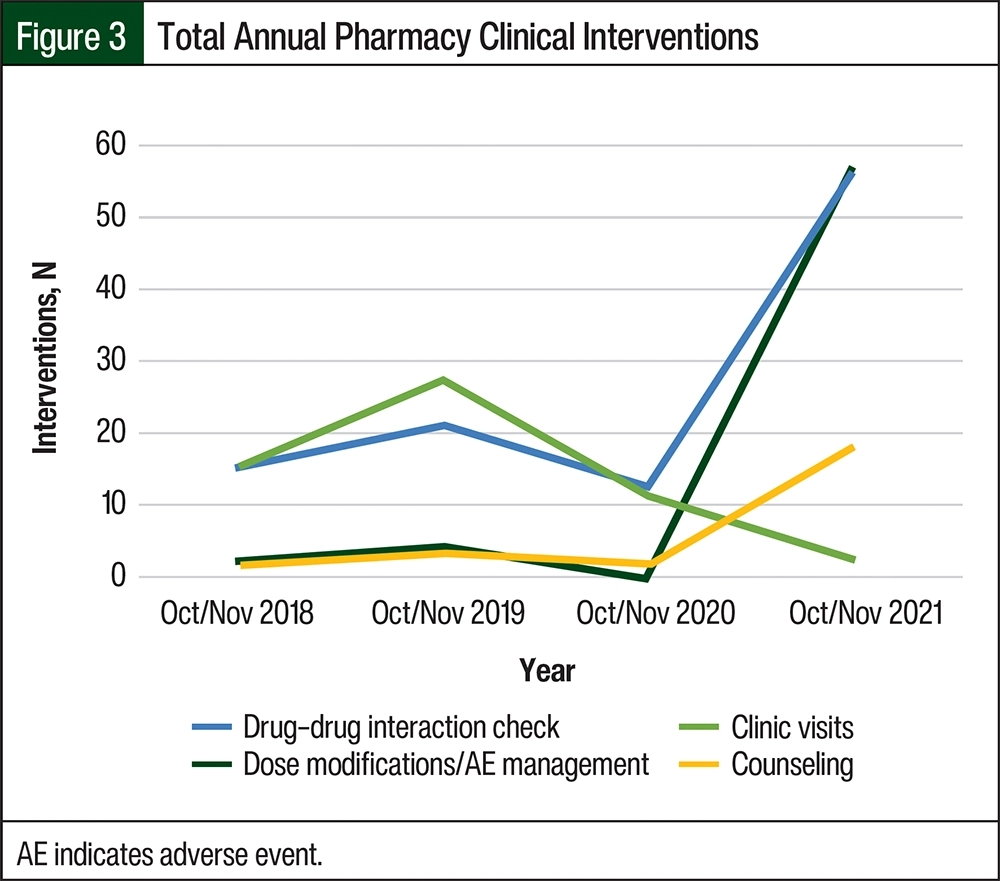
Over the duration of the study, the cumulative number of pharmacist interventions increased, including counseling, dose modifications/AE management, and drug–drug interaction checks (Figure 3). Phase 1 trial clinic visits over the study duration decreased because of the closure of 2 high-enrolling studies. From 2018 to 2020, clinical intervention data were collected from retrospective research chart reviews and internal correspondences. A formal intervention tracking process was implemented in 2021 and was used for data collection. The average time per pharmacy intervention in 2021 was 0.6 hours.
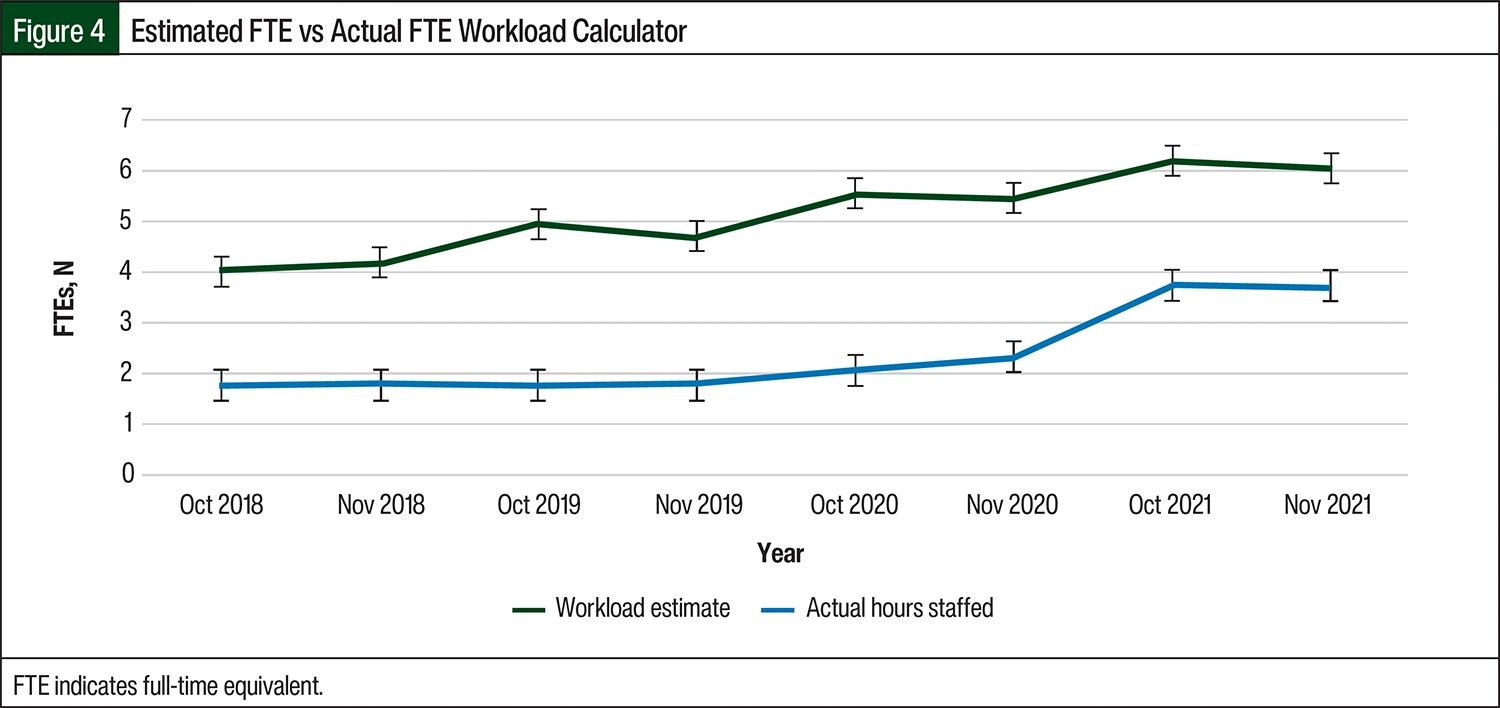
A pharmacy workload estimate calculator was developed by incorporating multiple factors. The first factor was study start-up time based on study complexity. The average pharmacy start-up time based on complexity was 13.68 hours for complexity level 1 studies (n=13), 15.29 hours for level 2 studies (n=12), and 33.12 hours for level 3 studies (n=7). The second factor was related to time requirements for study maintenance. To find the average study maintenance time based on the study complexity level, pharmacists tracked their daily work for 2 separate weeks, 1 week in October 2021 and 1 week in November 2021. The study maintenance activities included protocol amendments, monitoring visits, and protocol-specific email correspondence.
Analysis of these data provided an average weekly study maintenance time that was extrapolated for the month. Pharmacists documented the amount of time required to perform each protocol-specific task to determine the maintenance hours per study complexity level. The total number of studies opened for each month based on complexity was multiplied by the average start-up/maintenance hours to calculate an estimated workload. Based on the estimated workload calculator versus actual hours staffed, IDS pharmacy was understaffed by an average of 2 FTE staff members (Figure 4).
A total of 19 nonpharmacy research staff members completed the pharmacy involvement survey. The survey consisted of multiple statements with rating options from 1 (very important) to 5 (not important) pertaining to IDS pharmacy–related activities. The top responses that indicated pharmacy involvement as very important included reviewing and monitoring patients for drug interactions, evaluating patient profiles for medication therapy problems, preparing and dispensing investigational drugs, and answering drug information questions. The statements that were rated as the least important included publications and abstract editing and assisting with patient screening for potential clinical trial eligibility.
Discussion
To our knowledge, this is the first study that outlines IDS metrics to promote the growth and development of nontraditional IDS pharmacy practice. Our results show that there is important value in nontraditional IDS pharmacy practice when an IDS pharmacist is involved in direct patient care with the multidisciplinary team based on internal survey results conducted within the research department.
Capturing nontraditional IDS metrics has allowed our site to quantify the volume of pharmacist activities in the form of interventions despite a decline in traditional IDS metrics, including investigational drug dispenses and shipments. The decline in traditional IDS activities may have resulted from multiple factors, including the effects of the COVID-19 pandemic. As a result of the pandemic, patients were unable to leave their homes, and therefore screening and protocol enrollment declined.10
Another potential reason for the decline in total dispensations was related to sponsor allowance of larger quantities of oral investigational drugs per dispense to limit patient exposure risk. Before the COVID-19 pandemic, most study sponsors would only allow for the dispensation of 1 cycle of an oral investigational drug at each clinic visit. As a result of COVID-19 and the desire to reduce the number of in-clinic appointments, the study sponsors allowed the IDS pharmacy to dispense up to 3 months of oral investigational drugs, which contributed to the decline in annual overall dispensations.
As a result of a concentrated effort to increase the number of studies available, the total number of studies increased during the study duration. In addition to an increase in the total number of studies, the number of complexity level 3 studies doubled. As a result of the increased study volume, additional time and resources were spent on the study start-up process, with an average time of 18.5 hours per study.
In addition to the increase in study volume, the total number of pharmacy interventions also increased during the study period as a result of IDS pharmacist involvement with clinical tasks, such as counseling, dose modifications, AE management, and drug–drug interaction checks. A goal of this project was to establish a clinical intervention tracking system specific to IDS because there was no official method for doing so before 2021. This goal was achieved through the development of an IDS-specific tracking tool. Before the implementation of the tracking system, interventions were documented and archived via email, research records, and EMR documentation, which made it difficult to efficiently extract the metrics.
Overall, the estimated workload calculator demonstrated an understaffing of the IDS pharmacy department by 2 FTE staff members during the study period. The workload calculator has the benefit of accounting for the start-up time plus maintenance time of each study. One downfall of this tool is that it is an estimation based on the average start-up time, in which case the tool may underestimate or overestimate time requirements. The investigators attempted to account for this limitation by tracking study-specific start-up times plus the maintenance times to obtain the average time requirements based on complexity level. In addition, the workload calculator does not consider project time or time for pharmacy clinical interventions. Based on the workload calculator and pharmacy interventions, 1 FTE was added after this project was completed.
The survey results reaffirmed the current IDS nontraditional pharmacy model. The highest “very important” statements ranked by research staff were considered nontraditional IDS activities. Of the top 4 “very important” statements, 3 included the direct patient care activities of review and monitor patients for drug interactions, evaluate patient profiles for medication therapy problems, and answer drug information questions. Only 1 of the top 4 “very important” pharmacy statements was considered a traditional activity, which was to prepare and dispense an investigational drug. These results support the proposition to have an IDS pharmacist more actively involved in the multidisciplinary team and to take an active role in direct patient care.
Based on this study’s results, investigators can continue to track pharmacy interventions to assist with workload justification in a nontraditional IDS pharmacy practice. In the future, investigators can develop a workload dashboard that will allow IDS pharmacy staff to efficiently track metrics over the course of each month.
Limitations
There were several limitations to this study. First, the data estimated for each year were only collected for the months of October and November. It is possible that the full year was not accurately portrayed because only 2 months from each year were examined.
Another limitation was that there was no formal process for collecting data on pharmacy interventions until 2021. Before 2021, all interventions were saved in the electronic or paper research chart. Because of variations in the intervention documentation, it is possible that some interventions were not adequately accounted for from 2018 to 2020. In addition, this study was a retrospective review and was not intended to measure interventions that impact patient care outcomes, such as adherence rates.
The final limitation of this study was related to the COVID-19 pandemic. In general, COVID-19 had a major effect on oncology care. As a result, the number of patients who were screened for clinical trials at our institution declined. COVID-19 was an unexpected confounder of its effect on enrollment, clinic visits, and dispensations.
Conclusion
Based on this study’s results, the investigators found benefit in tracking nontraditional metrics to support direct patient care activities for patients enrolled in clinical treatment trials through the promotion of a multidisciplinary approach. Overall, traditional IDS metrics decreased during the study duration whereas clinical activities and the total number of open studies increased. At the institution where this study was conducted, the IDS pharmacy’s activities have become more complex with a request for additional nontraditional IDS activities.
Although the workload calculator only accounted for traditional IDS activities, this study allowed the investigators to identify and quantify nontraditional IDS activities to improve the delivery of patient care and staff satisfaction. These improvements were demonstrated by an increase in the number of pharmacy clinical interventions with the survey’s results. Tracking and quantifying clinical IDS pharmacy interventions with traditional metrics may provide a unique opportunity for the expansion of the IDS pharmacy staff’s responsibilities outside of their traditional roles. The results of this study demonstrate the value of incorporating IDS pharmacists into a multidisciplinary clinical research team and expanding their clinical role in research.
Author Disclosure Statement
Dr Lockhorst has received honoraria from Pharmacy Times Continuing Education. Dr Schmidt, Dr Middendorff, and Dr Kirkpatrick have no conflicts of interest to report.
References
- Kleppinger C. FDA’s Clinical Investigator Training Course. Division of Good Clinical Practice Compliance. FDA. November 2013. Accessed October 14, 2021. www.fda.gov/files/science%20%26%20research/published/9-45---11-00-Investigator-Responsibilities-–-Regulation-and-Clinical-Trials.pdf
- Baer AR, Zon R, Devine S, Lyss AP. The clinical research team. J Oncol Pract. 2011;7:188-192.
- National Cancer Institute. How do clinical trials work? Research team members. 2020. Accessed September 15, 2024. www.cancer.gov/research/participate/clinical-trials/how-trials-work#research-team-members
- Hematology/Oncology Pharmacy Association. HOPA Investigational Drug Service Best Practice Standards. 2014. Accessed September 4, 2024. www.hoparx.org/documents/109/HOPA16_IDS_Guidelines.reviewed_2018.pdf
- Kay SC, Luke DG, Tamer HR. ASHP guidelines for the management of investigational drug products. Am J Health Syst Pharm. 2018;75:561-573.
- Brown JN, Britnell SR, Stivers AP, Cruz JL. Medication safety in clinical trials: role of the pharmacist in optimizing practice, collaboration, and education to reduce errors. Yale J Biol Med. 2017;90:125-133.
- Good MJ, Lubejko B, Humphries K, Medders A. Measuring clinical trial-associated workload in a community clinical oncology program. J Oncol Pract. 2013;9:211-215.
- Milani A, Mazzocco K, Stucchi S, et al. How many research nurses for how many clinical trials in an oncology setting? Definition of the Nursing Time Required by Clinical Trial—Assessment Tool (NTRCT-AT). Int J Nurs Pract. 2017;23:e12497.
- Wascher M, Mighty J, Brown V, et al. Establishing an investigational drugs and research residency at an academic medical center. Am J Health Syst Pharm. 2019;76:1862-1867.
- Chen RC, Haynes K, Du S, et al. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7:878-884.