With commentaries by

Vamsi Velcheti, MD, MBA, FASCO, FACP, FCCP
Professor of Medicine, NYU Grossman School of Medicine
Medical Director, Thoracic Oncology Program
Laura and Isaac Perlmutter Cancer Center
New York, NY
Dr. Velcheti was paid for his contribution to this interview. Please see end of article for additional industry-related disclosures.
Introduction
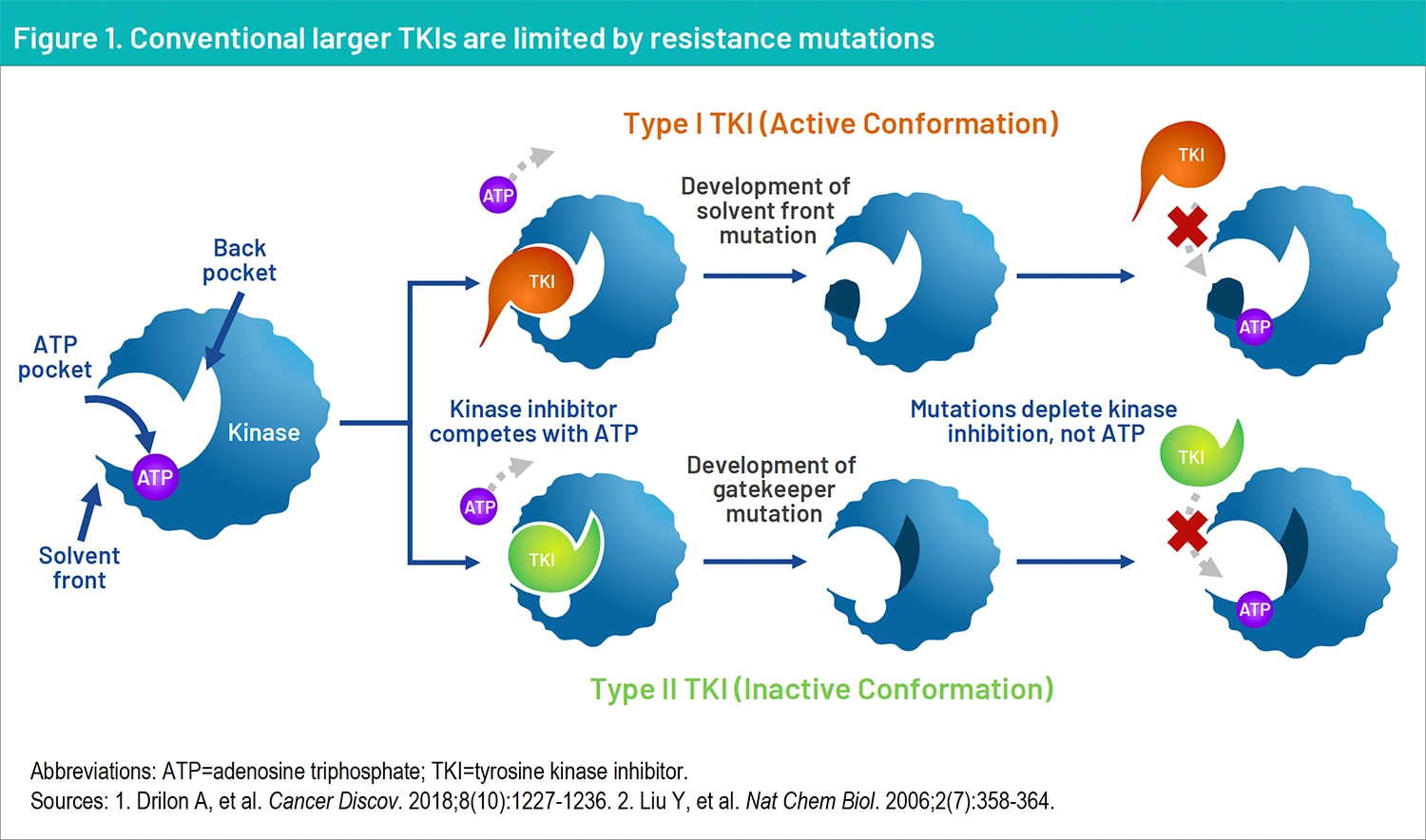
Lung cancer is the second most common cancer in the United States (excluding skin cancer) and approximately 80% to 85% of lung cancers are non-small cell lung cancer (NSCLC).1 ROS1 fusions, which are known to be oncogenic drivers, are observed in less than 2% of individuals diagnosed with NSCLC.2 Targeted therapy with tyrosine kinase inhibitors (TKIs) has transformed treatment for cancers with driver mutations. These small molecule inhibitors bind the ATP-binding site of the kinase, inhibiting its catalytic activity.3 Several TKIs are FDA approved for use in patients with driver mutations; however, tumors often develop resistance to TKI exposure. Resistance can occur by acquisition of “on-target” mutations at the kinase solvent front (SFMs) or in the gatekeeper position, which prevent TKIs from accessing the kinase ATP-binding pocket (Figure 1).4
Conventional larger TKIs can be limited by resistance mutations. Treatment for patients with NSCLC harboring TKI-resistant driver mutations represents a substantial unmet need. Next-generation TKIs, developed to combat resistance mutations, have become first-line treatments.5,6
Based on the phase 1 and phase 2 trial, TRIDENT-1, AUGTYRO was FDA-approved on November 15, 2023, as first-line therapy for locally advanced or metastatic ROS1-positive NSCLC.
Indication
AUGTYRO® (repotrectinib) is indicated for the treatment of adult patients with locally advanced or metastatic ROS1-positive NSCLC. Please see Important Safety Information below.
The safety of AUGTYRO was evaluated in 426 patients in TRIDENT-1.
AUGTYRO SUMMARY OF WARNINGS AND PRECAUTIONS
AUGTYRO is associated with the following warnings and precautions: central nervous system (CNS) adverse reactions, interstitial lung disease (ILD)/pneumonitis, hepatotoxicity, myalgia with creatine phosphokinase (CPK) elevation, hyperuricemia, skeletal fractures, and embryo-fetal toxicity.
Key results from TRIDENT-1 will be reviewed in this publication.
AUGTYRO, a structurally novel ROS1 TKI
AUGTYRO is a next-generation ROS1 TKI with a compact structure that is smaller than first generation ROS1 TKIs.8 Its structure potentially enables AUGTYRO to circumvent known ROS1 resistance mutations*, and provides its physiochemical parameters for enhanced intercranial activity.8
*TRIDENT-1 enrolled patients with resistance mutations following TKI therapy, and responders included patients with solvent front (G2032R), gatekeeper (L2026M), and other mutations (S1986F/Y). In addition, repotrectinib exhibited anti-tumor activity in cultured cells expressing ROS1fusions and mutations including SDC4-ROS1, SDC4 ROS1G2032R, CD74 ROS1, CD74-ROS1G2032R, CD74-ROS1D2033N, and CD74 ROS1L2026M.
TRIDENT-1: Design of the AUGTYRO first in-human study
The primary endpoint of this study was objective response rate (ORR) as assessed by blinded independent central review (BICR) per RECIST version 1.1.2,9 Key secondary endpoints consisted of duration of response (DOR), progression-free survival (PFS), and intracranial objective response rate (icORR) according to modified RECIST version 1.1, as assessed by BICR.2,9 To be included in the study, patients had to have ROS1 plus locally advanced or metastatic NSCLC, and ECOG performance status ≤1, measurable disease per RECIST version 1.1, and ≥8 months from the first dose.9 Patients were excluded from the study if they had symptomatic brain metastases.9
This study was divided into 2 phases: Phase 1 was a dose-escalation phase that determined the recommended phase 2 dose, and phase 2 was the dose-expansion phase that consisted of the efficacy cohorts.2 During phase 1, patients received AUGTYRO 160 mg once a day for 14 days; day 15 and onward, patients received AUGTYRO 160 mg twice a day.2,9 The phase 2 efficacy population included patients who received at least 1 dose of AUGTYRO.2 The primary efficacy population consisted of the TKI-naïve cohort and the 1 prior TKI and no prior chemotherapy cohort as depicted in the study design (Figure 2).2,9 The safety analysis population included all patients who received treatment with the phase 2 dose, regardless of the tumor or fusion type.2 This publication will focus on the results from the TKI-naïve cohort.
Commentary by Vamsidhar Velcheti, MD, MBA, FASCO, FACP, FCCP: Patients with ROS1-positive non-small cell lung cancers is a rare subgroup. It’s about 1% to 2% of all non-small cell lung cancer. They often present at an advanced stage, they have extensive metastatic disease burden, unfortunately, they often do very poorly with standard treatments. There are some targeted therapy treatment options for patients with ROS1-positive tumors, including repotrectinib, a highly selective ROS1 inhibitor with its data from the TRIDENT-1 study. And it’s welcomed treatment option for patients with metastatic ROS1-positive non-small cell lung cancer. This highlights the need for these next- generation tyrosine kinase inhibitors and especially in patients with ROS1-positive tumors
Phase 1-2 results reported from the TRIDENT-1 study
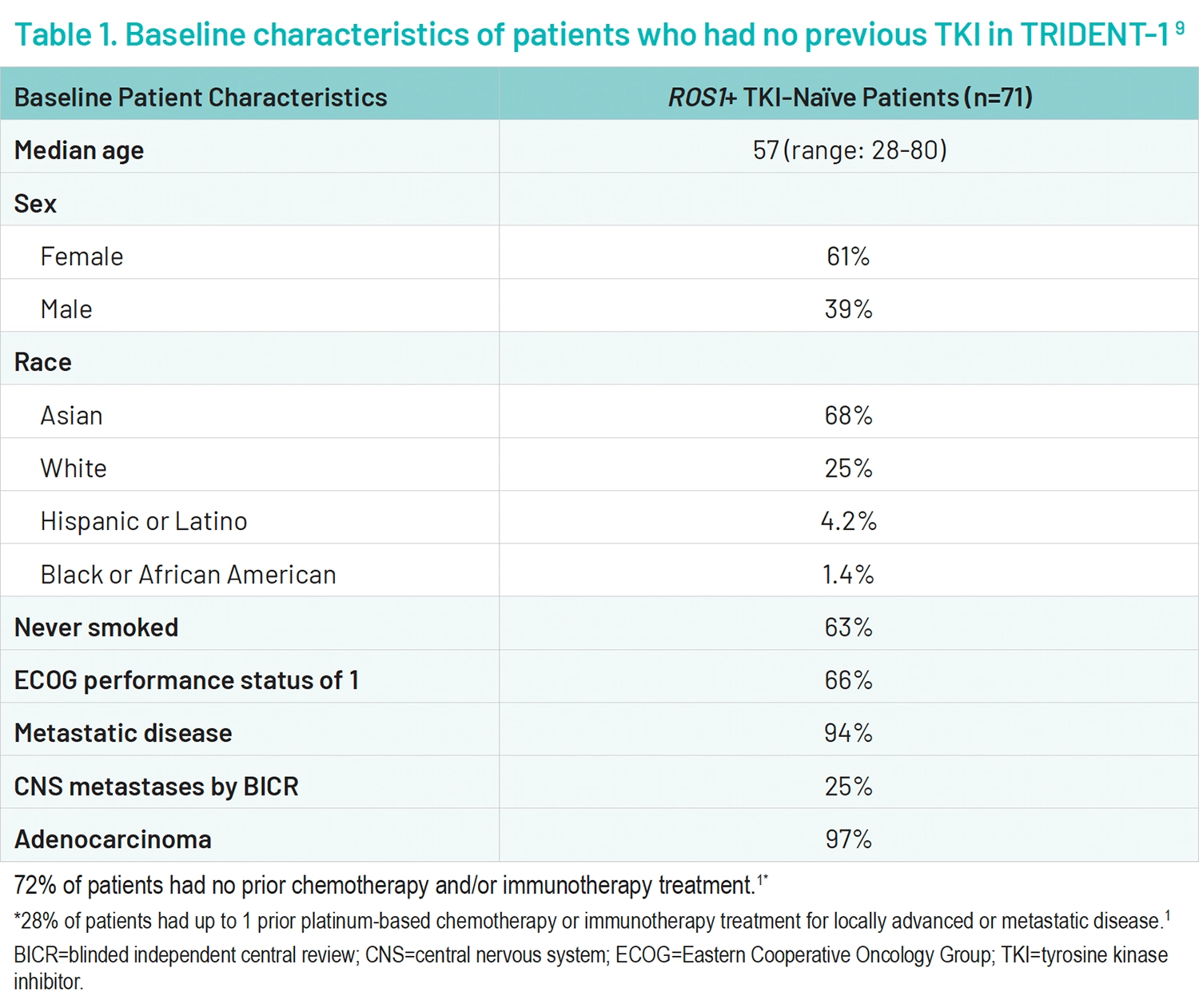
A total of 519 patients received on ≥1 doses of AUGTYRO; 103 patients were treated in phase 1 and 416 patients were treated in phase 2.2 Within the primary efficacy population, there were 71 patients who had no previous TKI treatment (Table 1).2,9
For the primary endpoint of confirmed objective response, among TKI-naïve patients (n=71) a confirmed overall response rate occurred in 79% (n=56/71) of patients (95% confidence interval [CI], 68-88)2,9; 6% (n=4/71) of patients had a complete response, and 73% (n=52/71) had a partial response.9 The median time to response was 1.8 months (range, 0.9-5.6).2 The median follow-up for DOR was 24.0 months (range, 14.2-66.6), the median follow-up for ORR and icORR was 18.1 months, and the median duration of response was 34.1 months (95% CI, 25.6, not evaluable; range, 1.4+- 42.4+ months).2,9 70% of patients were still responding at 12 months of treatment (DOR landmark analysis is based on the observed DOR). In the phase 2 trial, an intracranial response occurred in 7 of 8 patients with measurable CNS metastasis at baseline in those who had no previous TKI.9
Outcomes in TKI-naïve patients treated with AUGTYRO
ORR – 79% (n=56/71)
DOR – 34.1 months
icORR – 7/8 patients with measurable CNS metastasis at baseline
Commentary by Vamsidhar Velcheti, MD, MBA, FASCO, FACP, FCCP: The remarkable improvements in overall response rate (ORR) observed in treatment-naïve patients, , make a compelling case for the early use of repotrectinib. Leveraging repotrectinib in the frontline setting could potentially improve overall response outcomes for patients with ROS1-positive lung cancer.
I think the major challenge with ROS1-positive tumors, and just like with other targeted therapies as well, is most of these patients, even though they respond to targeted therapy, they often develop resistance. And the next-generation ROS1 inhibitor, repotrectinib, has a compact structure, and it has an ability to target some of the solvent-front mutations, and may provide durable responses, as supported by objective response rate and median duration of response and also may provide intracranial responses, which is actually a critical gap for treatment with these ROS1-positive tumors. The first-generation ROS1 inhibitors are very limited in terms of efficacy in the brain and these tumors tend to develop resistance quickly with solvent-front mutations. So using a more tailored drug like repotrectinib, could potentially increase the durability of response, and intracranial responses.
Safety of AUGTYRO
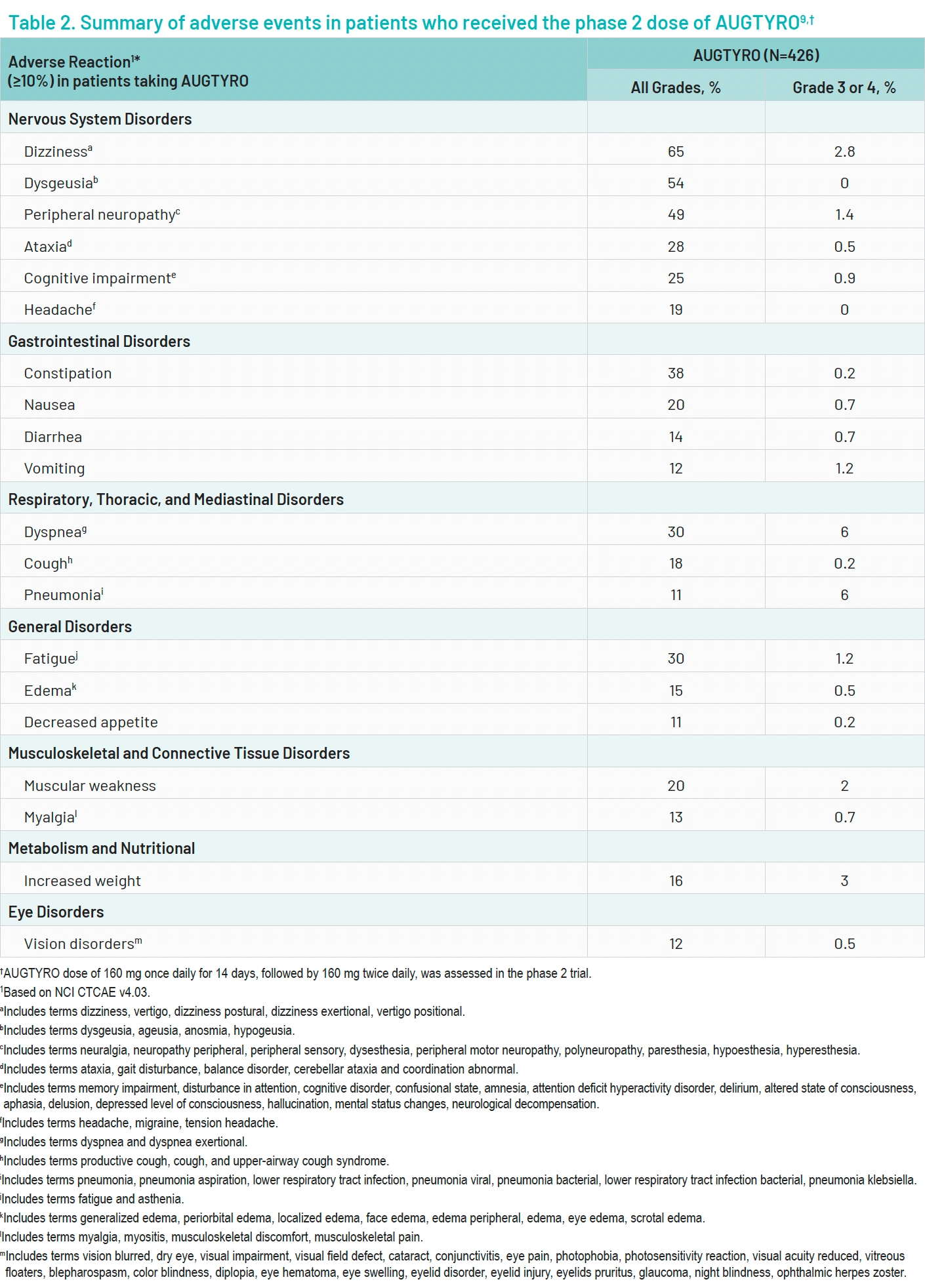
Among the 426 patients who were treated at the phase 2 dose, the most common treatment-related adverse events of any grade included dizziness (65%), dysgeusia (54%), and peripheral neuropathy (49%).9 The most common grade 3 or 4 adverse events were dyspnea, pneumonia, and increased weight occurring at 6%, 6%, and 3%, respectively.9 Adverse events led to treatment discontinuation in 7% of patients. Adverse events led to dose reduction in 38% of patients and dose interruption in 50% of patients.9 Fatal adverse events occurred in 3.5% of patients (Table 2).9
Commentary by Vamsidhar Velcheti, MD, MBA, FASCO, FACP, FCCP: There are some unique toxicities like dizziness and dysgeusia. Most of these side effects are grade 1 and 2. The discontinuation rate, because of the side effects, is fairly low.
Adverse events related to treatment with AUGTYRO were primarily grade 1 or 2.
Summary and conclusions
Results from the phase 1-2 TRIDENT-1 study demonstrated that AUGTYRO showed activity in patients with ROS1-positive NSCLC. AUGTYRO had durable antitumor activity based on the objective response rate and median duration of response.2 AUGTYRO was active against intracranial disease.2 The selectivity and intracranial activity of AUGTYRO underscores its unique therapeutic value.
Commentary by Vamsidhar Velcheti, MD, MBA, FASCO, FACP, FCCP: I think every eligible patient with ROS1-positive locally advanced or metastatic non-small cell lung cancer, should be considered for treatment with repotrectinib up front. I really think given what we are seeing with the ORR in the frontline setting in treatment-naïve patients, eligible patients should receive repotrectinib in the frontline setting.
Based on its safety and efficacy profiles, as well as its intracranial activity, AUGTYRO proves to be a suitable option for the initial treatment of patients diagnosed with ROS1-positive NSCLC.
IMPORTANT SAFETY INFORMATION
Warnings and precautions
Central Nervous System Adverse Reactions
- Among the 426 patients who received AUGTYRO in Study TRIDENT-1, a broad spectrum of central nervous system (CNS) adverse reactions including dizziness, ataxia, and cognitive disorders occurred in 77% of patients with Grade 3 or 4 events occurring in 4.5%.
- Dizziness, including vertigo, occurred in 65%; Grade 3 dizziness occurred in 2.8% of patients.
- Ataxia, including gait disturbance and balance disorder, occurred in 28% of patients; Grade 3 ataxia occurred in 0.5%.
- Cognitive impairment, including memory impairment and disturbance in attention, occurred in 25% of patients. Cognitive impairment included memory impairment (15%), disturbance in attention (12%), and confusional state (2%); Grade 3 cognitive impairment occurred in 0.9% of patients.
- Mood disorders occurred in 6% of patients. Mood disorders occurring in >1% of patients included anxiety (2.6%); Grade 4 mood disorders (mania) occurred in 0.2% of patients.
- Sleep disorders including insomnia and hypersomnia occurred in 18% of patients. Sleep disorders observed in >1% of patients were somnolence (9%), insomnia (6%) and hypersomnia (1.6%).
- The incidences of CNS adverse reactions reported were similar in patients with and without CNS metastases.
- Advise patients not to drive or use machines if they are experiencing CNS adverse reactions. Withhold and then resume at same or reduced dose upon improvement, or permanently discontinue AUGTYRO based on severity.
Interstitial Lung Disease (ILD)/Pneumonitis
- ILD/pneumonitis (pneumonitis [2.8%] and ILD [0.2%]) occurred in 3.1%; Grade 3 ILD/pneumonitis occurred in 1.2%.
- Monitor patients for new or worsening pulmonary symptoms indicative of ILD/pneumonitis. Immediately withhold AUGTYRO in patients with suspected ILD/pneumonitis and permanently discontinue AUGTYRO if ILD/pneumonitis is confirmed.
Hepatotoxicity
- Increased alanine transaminase (ALT) occurred in 38%, increased aspartate aminotransferase (AST) occurred in 41%, including Grade 3 or 4 increased ALT in 3.3% and increased AST in 2.9%.
- Monitor liver function tests, including ALT, AST and bilirubin, every 2 weeks during the first month of treatment, then monthly thereafter and then as clinically indicated. Withhold and then resume at same or reduced dose upon improvement or permanently discontinue AUGTYRO based on the severity.
Myalgia with Creatine Phosphokinase (CPK) Elevation
- AUGTYRO can cause myalgia with or without creatine phosphokinase (CPK) elevation. Myalgia occurred in 13% of patients, with Grade 3 in 0.7%. Concurrent increased CPK within a 7-day window was observed in 3.7% of patients.
- Advise patients to report any unexplained muscle pain, tenderness, or weakness. Monitor serum CPK levels during AUGTYRO treatment and monitor CPK levels every 2 weeks during the first month of treatment and as needed in patients reporting unexplained muscle pain, tenderness, or weakness. Initiate supportive care as clinically indicated. Based on severity, withhold and then resume AUGTYRO at same or reduced dose upon improvement.
Hyperuricemia
- 21 patients (5%) experienced hyperuricemia reported as an adverse reaction, 0.7% experienced Grade 3 or 4 hyperuricemia. One patient without pre-existing gout required urate-lowering medication.
- Monitor serum uric acid levels prior to initiating AUGTYRO and periodically during treatment. Initiate treatment with urate-lowering medications as clinically indicated. Withhold and then resume at same or reduced dose upon improvement, or permanently discontinue AUGTYRO based on severity.
Skeletal Fractures
- Fractures occurred in 2.3% of patients and involved the ribs (0.5%), feet (0.5%), spine (0.2%), acetabulum (0.2%), sternum (0.2%), and ankles (0.2%). Some fractures occurred at sites of disease and prior radiation therapy.
- 26 evaluable patients in an ongoing open-label study in pediatric patients, fractures occurred in one 12-year-old patient (ankle/foot) and one 10-year-old patient (stress fracture). AUGTYRO is not approved for use in pediatric patients less than 12 years of age.
- Promptly evaluate patients with signs or symptoms (e.g., pain, changes in mobility, deformity) of fractures. There are no data on the effects of AUGTYRO on healing of known fractures and risk of future fractures.
Embryo-Fetal Toxicity
- Based on literature reports in humans with congenital mutations leading to changes in tropomyosin receptor tyrosine kinase (TRK) signaling, findings from animal studies, and its mechanism of action, AUGTYRO can cause fetal harm when administered to a pregnant woman.
- Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective non-hormonal contraception during treatment with AUGTYRO and for 2 months following the last dose, since AUGTYRO can render some hormonal contraceptives ineffective.
- Advise male patients with female partners of reproductive potential to use effective contraception during treatment with AUGTYRO and for 4 months after the last dose.
Adverse Reactions
- The safety of AUGTYRO was evaluated in 426 patients in TRIDENT-1. The most common adverse reactions (≥20%) were dizziness, dysgeusia, peripheral neuropathy, constipation, dyspnea, fatigue, ataxia, cognitive impairment, muscular weakness, and nausea.
Drug Interactions
Effects of Other Drugs on AUGTYRO
Strong and Moderate CYP3A Inhibitors
- Avoid concomitant use with P-gp inhibitors, strong or moderate CYP3A inducers, and strong or moderate CYP3A inhibitors. Discontinue CYP3A inhibitors for 3 to 5 elimination half-lives of the CYP3A inhibitor prior to initiating AUGTYRO.
Effects of AUGTYRO on other Drugs
Certain CYP3A4 Substrates
- Avoid concomitant use unless otherwise recommended in the Prescribing Information for CYP3A substrates, where minimal concentration changes can cause reduced efficacy. If concomitant use is unavoidable, increase the CYP3A4 substrate dosage in accordance with approved product labeling.
- Repotrectinib is a CYP3A4 inducer. Concomitant use of repotrectinib decreases the concentration of CYP3A4 substrates, which can reduce the efficacy of these substrates.
Contraceptives
- Repotrectinib can decrease progestin or estrogen exposure to an extent that could reduce the effectiveness of hormonal contraceptives.
- Avoid concomitant use of AUGTYRO with hormonal contraceptives. Advise females of childbearing potential to use an effective nonhormonal contraceptive.
Please see U.S. Full Prescribing Information for AUGTYRO.
References
- American Cancer Society. What is lung cancer? 2024. Accessed May 17, 2024. https://www.cancer.org/cancer/types/lung-cancer/about/what-is.html
- Drilon A, Camidge DR, Lin JJ, et al. Repotrectinib in ROS1 fusion-positive non-small-cell lung cancer. N Engl J Med. 2024;390:118-131.
- Chen Y-F, Fu L-W. Mechanisms of acquired resistance to tyrosine kinase inhibitors. Acta Pharmaceutica Sinica B. 2011;1:197-207.
- Fancelli S, Caliman E, Mazzoni F, et al. Chasing the target: new phenomena of resistance to novel selective ret inhibitors in lung cancer. Updated evidence and future perspectives. Cancers. 2021;13:1091.
- Shaw AT, Bauer TM, De Marinis F, et al. First-line lorlatinib or crizotinib in advanced ALK-positive lung cancer. N Engl J Med. 2021;383:2018-2029.
- Ramalingam SS, Vansteenkiste J, Planchard D, et al. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N Engl J Med. 2020;382:41-50.
- FDA. FDA approves repotrectinib for ROS1-positive non-small cell lung cancer. 2023. Accessed May 17, 2024. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-repotrectinib-ros1-positive-non-small-cell-lung-cancer
- Drilon A, Ou SI, Cho BC, et al. Repotrectinib (TPX-0005) is a next-generation ROS1/TRK/ALK inhibitor that potently inhibits ROS1/TRK/ALK solvent-front mutations. Cancer Discov. 2018;8:1227-1236.
- AUGTYRO prescribing information. Bristol Myers Squibb. Princeton, NJ.
- Cho BC, Camidge DR, Lin JJ, et al. Repotrectinib in patients with ROS1 fusion-positive non-small cell lung cancer: update from the pivotalphase 1/2 TRIIDENT-1 trial. Presented at: 2023 World Conference on Lung Cancer; September 9-12, 2023; Singapore.
3600-US-2500020 05/25