CAR T cells have induced dramatic responses in pediatric patients with B-lineage hematologic malignancies,1 yet there are numerous obstacles in translating this success to children with central nervous system (CNS) tumors.2,3 A central clinical obstacle is that a systemic delivery significantly limits on-tumor, on-target efficiency because of the blood–brain barrier.4 The results of an early clinical study of the delivery of intracavitary CAR T cells in adults showed that doses of CAR T cells were generally well-tolerated, resulted in no dose-limiting adverse events (AEs), and had evidence of antitumor activity.5 In addition, intracerebroventricular CAR T cells demonstrated tumor control in the setting of recurrent multifocal glioblastoma, suggesting that this method of delivery helps control disseminated disease.6
Building on this initial translational success in adults, Seattle Children’s Hospital opened 3 trials of patients with CNS that employ repeated locoregional delivery of CAR T cells (NCT03500991, NCT03638167, NCT04185038).7-9 As of October 2022, these trials had cumulatively provided 307 intracranial CAR T-cell doses to 65 enrolled children with recurrent or refractory CNS tumors or with newly diagnosed diffuse intrinsic pontine glioma (DIPG) or diffuse midline glioma (DMG).7-9 This approach requires a CAR T-cell production platform that can make sufficient CAR T cells for multiple doses, as well as a collaborative partnership with an investigational drug pharmacy that can prepare doses for rapid clinical delivery.
Although we have published preliminary tolerability and correlative study data from children with recurrent CNS tumors who received HER2-specific CAR T cells10 and children with DIPG who received B7-H3–specific CAR T cells,11 here we expand on those reports to summarize the largest clinical experience of investigational drug service (IDS) pharmacy activities for providing CAR T cells for repeated locoregional delivery to children with CNS tumors.
Methods
The DNA constructs, lentivirus, and preclinical studies have been previously published.10-12 Clinical data from July 2018 to October 2022 were included in this manuscript. BrainChild-01, BrainChild-02, and BrainChild-03 (NCT03500991, NCT03638167, and NCT04185038, respectively) are phase 1 studies of CNS locoregional adoptive therapy with autologous CD4+ and CD8+ T cells lentivirally transduced to express HER2-, EGFR-, or B7-H3-specific CAR and EGFRt (truncated form of EGFR) delivered intracranially to children and young adults aged 1 year to 26 years with recurrent or refractory CNS tumors or with DIPG or DMG.7-9 The clinical methods for BrainChild-01 and BrainChild-03 have been described, while BrainChild-02 mirrors BrainChild-01 but delivers EGFR-specific CAR T cells rather than HER2-specific CAR T cells.7-11
The dose-limiting–monitoring period included repeated intracranial CAR T-cell doses. Disease restaging occurred at the end of treatment course 2. These studies are conducted in accordance with the FDA and the International Conference on Harmonization Good Clinical Practice guideline, the Declaration of Helsinki, and applicable institutional review board requirements; the study protocol was approved by the Seattle Children’s Institutional Review Board. All patients or their guardians provided written informed consent in accordance with local regulatory review.
The enrolled patients underwent leukapheresis. CD4+ and CD8+ T cells from apheresis products were bioengineered to express the second-generation CAR T cells. The requirements to receive CAR T-cell infusions included having a CNS catheter in place; being ≥5 days after having surgery; evidence of persistent, evaluable disease; not breastfeeding nor pregnant; meeting the defined washout periods from any bridging therapy; having adequate organ function as defined by specified laboratory values used for eligibility; not having encephalopathy or uncontrolled seizure activity; complying with the administration of the prescribed antiepileptic drug; and having no evidence of active severe infection. To receive subsequent infusions, patients were also required to have had no dose-limiting AEs. Beyond treatment course 2, the patients were eligible to receive additional infusions at the previous maximum tolerated dose level if the above criteria were met and if sufficient CAR T cells were available. Response was assessed after treatment course 2 and at subsequent even-numbered treatment courses using magnetic resonance imaging of the brain and spine and cerebrospinal fluid cytology. The correlative studies collections varied by biospecimen and by assigned treatment arm.
The primary objective of the BrainChild studies was to assess the feasibility, safety, and tolerability of CAR T cells. Feasibility was defined as the generation of sufficient therapeutic product to receive all scheduled doses in courses 1 and 2 at the intended dose level per the assigned dose regimen after 2 attempts using a single apheresis product for the starting material. Safety and tolerability are determined by data that include history and physical examinations, laboratory and radiographic evaluations, and Common Terminology Criteria for Adverse Events v5.0. Further details on clinical evaluations have been published.10,11
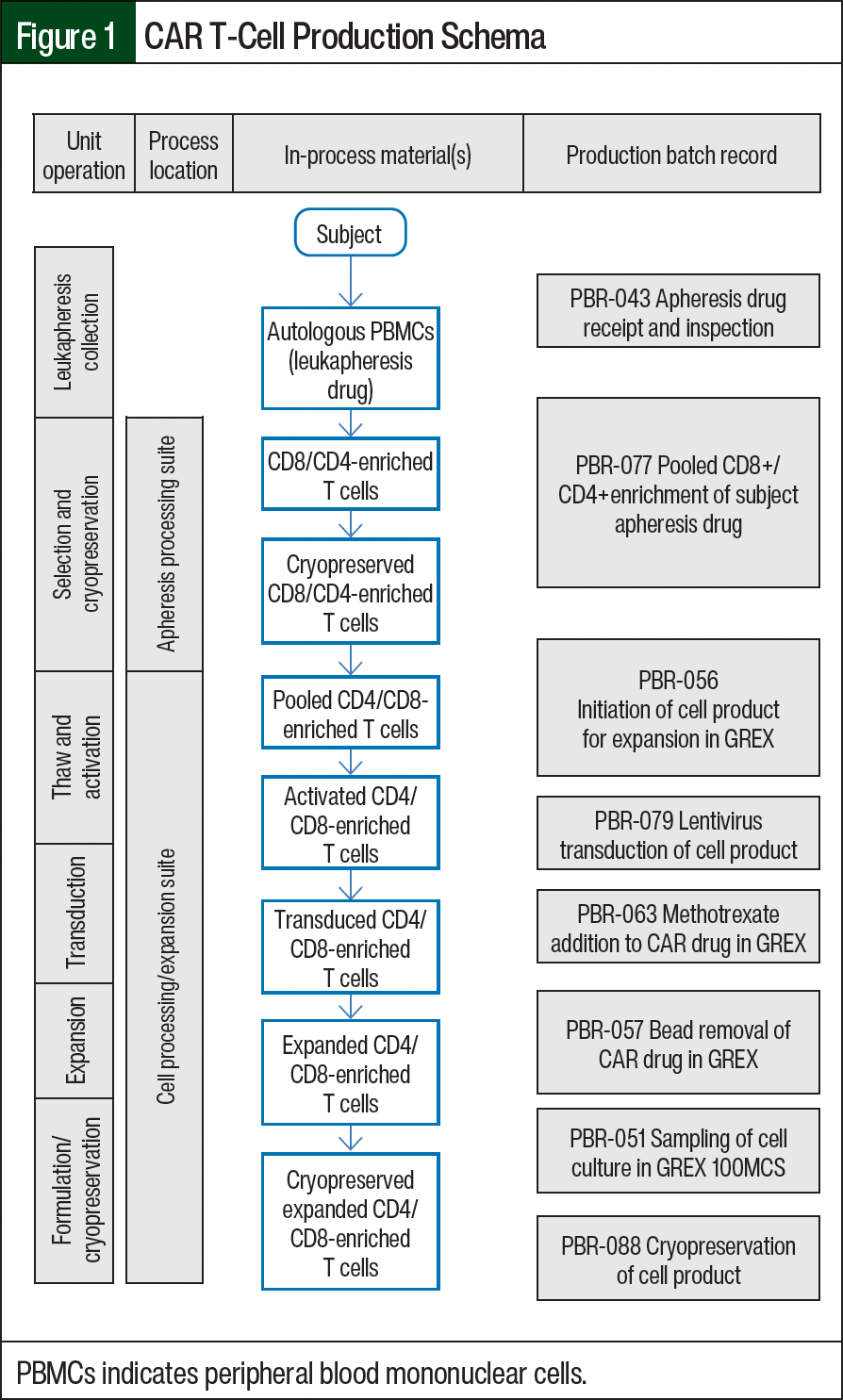
Our most recent cell product manufacturing procedures have been published.10,11 Cells are manufactured in a Good Manufacturing Practice (GMP) facility. In summary, CD4- and CD8-positive T cells are isolated from the patient’s apheresis products. A 1:1 mixture of CD4- and CD8-enriched cell fractions are pooled and stimulated. Cells are then transduced using a GMP-grade self-inactivating lentivirus that encodes CAR and cell-surface markers. The cell product is enriched on day 7 of culture. After 14 to 18 days in culture, the cells are harvested, washed, and resuspended for cryopreservation in CellSeal closed-system cryogenic vials. The full CAR T-cell production pipeline is shown in Figure 1. Before the CAR T-cell administration, a thaw-and-wash process was performed between April 2019 and December 2021 for 13 patients and 123 doses. Starting April 2021, a thaw-and-dilute process was implemented, although any cell product cryopreserved before April 2021 continued to be prepared using the thaw-and-wash process.
Results
The patients who are enrolled in the BrainChild clinical trials undergo standard outpatient leukapheresis at Seattle Children’s Hospital (Figure 1). The leukapheresis sample is next delivered to Seattle Children’s Hospital’s Therapeutic Cell Production Core (TCPC), which is housed within Seattle Children’s Research Institute’s free-standing Building Cure research facility. Primary CD8+/CD4+ T cells are isolated through a pooled positive enrichment. Then, cells are cryopreserved for future initiation or are cultured fresh in X-VIVO 15 media with supplemented 2% KnockOut Serum Replacement. The cells are stimulated on day 0 with Gibco Cell Therapy Systems (CTS) Dynabeads CD3/CD28 and are supplemented with recombinant human interleukin (rhIL)-7, rhIL-15, and rhIL-21 cytokines.
Then, 18 to 24 hours after stimulation, the cells are transduced using lentiviral vector at a defined volume. After transduction, the culture volume is increased to 1000 mL (the nominal volume in the bioreactor) and the cells are incubated for 6 days until day 7. In some trials, the cell product includes a methotrexate selection step. In these trials, the lentivirus contains a human DHFR variant, which confers resistance to antifolates and provides a dominant selection marker.13 Cells are then harvested for stimulation bead removal using the Gibco CTS DynaCellect Magnetic Separation System and are counted. Depending on the number of cells required to achieve testing and dosing needs, the cells are expanded or cryopreserved. The cells that are intended for testing, infusion, and retention are transferred to vapor-phase cryogenic storage. The contents from 1 vial are set aside for US Pharmacopeia microbiology testing. The representative final product testing vials are then complete the release testing assays and must meet all predetermined release testing specifications before the product release and/or infusion.
Once the CAR T-cell manufacture is complete and the patient is scheduled to return to our outpatient pediatric cancer clinic for infusion, Seattle Children’s Hospital’s IDS pharmacy submits a request for the delivery of the prepared CAR T cells. Once delivered, the IDS pharmacy oversees all pharmacy-related processes for the cell product at the hospital, including storage, thawing, and delivery to the bedside. The IDS pharmacy is staffed by dedicated pharmacists and pharmacy technicians with specialized training in handling and documentation requirements for investigational products. The IDS workspace is physically located within Seattle Children’s Hospital’s inpatient pharmacy and shares key resources, such as the clean room, with other pharmacy services.
Before the initiation of work, the IDS pharmacy team met with the pharmacy leadership to achieve consensus on several key clinical aspects. First, this protocol required substantial additional storage space for protocol-specific equipment. Within the hospital’s pharmacy, the installation of a liquid nitrogen freezer and tank with alarm monitoring was required to store cryogenically frozen cell vials. Second, the cell product had to be prepared in a biosafety cabinet, which requires approximately 2 hours to prepare a single dose. During this time, the biosafety cabinet is unavailable to the inpatient pharmacy, which required significant coordination to avoid disruption to the inpatient pharmacy workflow. Third, a minimum of 2 IDS pharmacy staff members would be required to prepare and dispense each CAR T-cell dose; therefore, the IDS pharmacy had to ensure adequate staffing to provide uninterrupted workflow for other research studies.
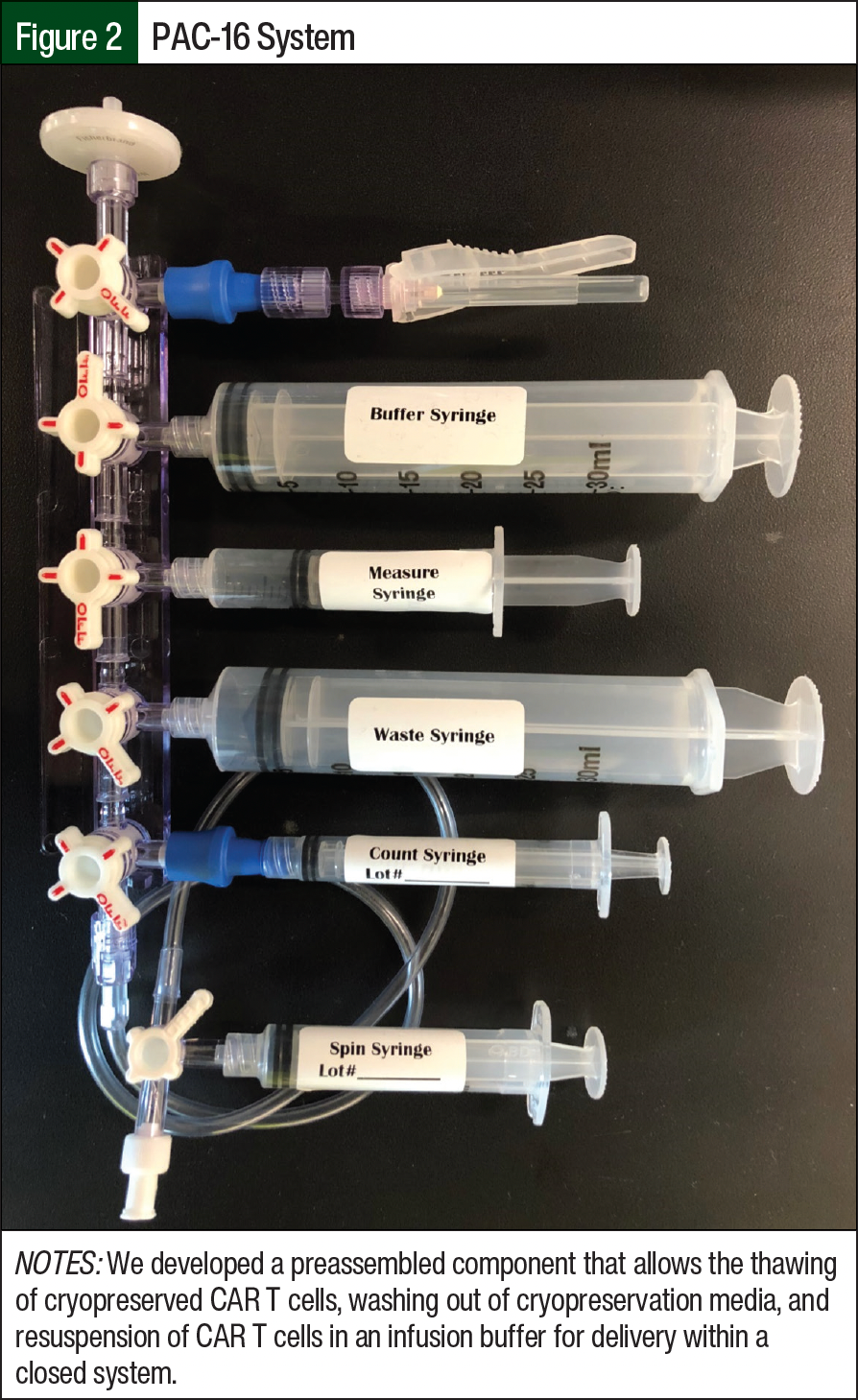
Before opening BrainChild-01, the IDS pharmacy staff underwent an intensive training program, which was led by our TCPC team, to train and certify IDS pharmacy team members on procedures related to T-cell products. Training was broadly divided into the 3 categories of equipment operation, the manipulation of CAR T cells (dose preparation), and dose calculations. One of the first steps was to adapt GMP-based standard operating procedures to the pharmacy standards. The IDS pharmacy staff were trained in the operation of equipment, including the preassembled closed system syringe (PAC-16), welder, thawer, centrifuge, and Beckman Coulter Vi-CELL XR Cell Viability Analyzer. Two individuals were required to carry out the thaw-and-wash/dilute procedure, including a verifier and an operator. The verifier was responsible for reading the standard operating procedures and verifying that each step was performed as described, whereas the operator was responsible for performing the procedure.
Because the CAR T cells are cryopreserved in a medium containing 5% dimethyl sulfoxide (DMSO), the first goal was to reduce this concentration for infusion to minimize the risk for DMSO-related AEs.14 We developed a preassembled component (PAC), the PAC-16, that would allow us to thaw the cryopreserved CAR T cells, wash out the cryopreservation media, and resuspend the T cells in an infusion buffer for delivery (Figure 2). This PAC allowed the process to be performed in a closed-system manner, thereby increasing safety and reducing sterility risks before delivery. The cells were thawed and processed through the PAC and washed twice before final resuspension and count (ie, the thaw-and-wash process). This recovery count was used to determine the volume for infusion. During the development and qualification of this PAC, samples were removed at various points in the process and were sent to an independent laboratory for analysis of the DMSO content. From these analyses, the final infusion concentration of DMSO was reduced to approximately 1%.
The IDS pharmacy team was responsible for the storage, preparation, handling, and delivery of the final cell doses to the patient’s bedside. The manufactured patient-specific cell vials were transported from TCPC to the IDS pharmacy and were stored in the pharmacy’s liquid nitrogen freezer until the time of administration. On notification of patient eligibility on the day of infusion, the IDS pharmacy would be informed to begin to thaw and prepare the CAR T-cell product.
For the thaw-and-wash process, the IDS pharmacy would first calibrate the cell viability analyzer. Then, the CellSeal cryogenic vial would be retrieved from the liquid nitrogen freezer and the PAC-16 would be prepared with the Normosol-R electrolyte buffer solution in a buffer syringe. Next, the cell vial would be thawed using the CellSeal automated thawing system. The first wash is then performed by drawing up the cells into the PAC-16 and mixing them with Normosol-R. The cell solution is withdrawn into a spin syringe that is detached from the PAC-16 and used for centrifugation. The cells are centrifuged at 1400 rpm for 10 minutes to ensure that a cell pellet is formed in the spin syringe. The spin syringe is reattached to the PAC-16 and the supernatant is expelled.
The second wash is performed as well as a second centrifugation, which resuspends the cells in Normosol-R. A small cell sample is withdrawn to assess the viability cell count, which is performed using a cell analyzer to ensure that the product viability meets the specification. The cell dose and volume are calculated based on cell count. Last, the final dose is prepared for delivery in a labeled syringe.
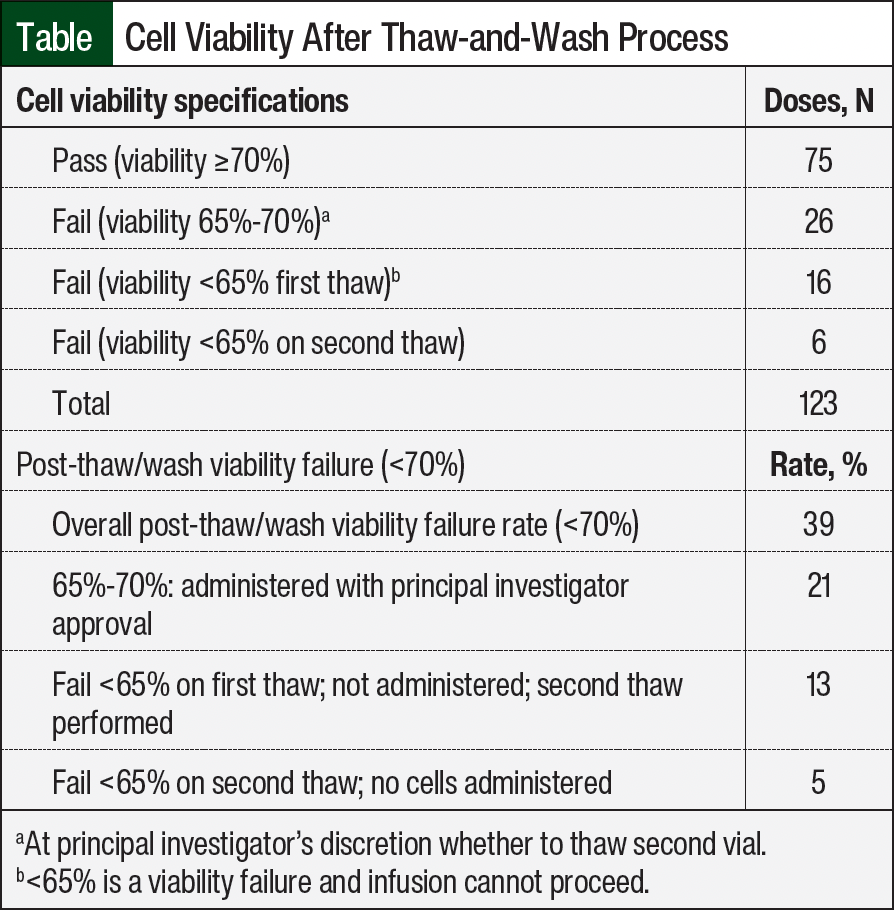
A major challenge of the thaw-and-wash process was the procedure’s negative effect on cell viability. If cell viability did not meet the specification of ≥70%, the principal investigator was contacted to determine whether to move forward with the infusion or to perform a second thaw-and-wash procedure using a second vial. Under this system, 123 doses were prepared for 13 subjects (Table). In all, 75 (61%) doses met the viability specification of ≥70%. Within the failed 39% of doses, 26 (21%) doses had 65% to 70% viability but were administered with the principal investigator’s approval, 16 (13%) doses had a viability of <65% and required the principal investigator’s approval for a second thaw, and 6 (5%) doses did not meet the viability of >65% on the second thaw and no dose was administered.
Another challenge was ensuring that the cell analyzer instrument was operational. If the cell analyzer failed calibration on the morning of infusion, the IDS pharmacy staff had to coordinate with the laboratory staff to perform a just-in-time manual cell count. In addition, the length of time it took to complete the thaw-and-wash process (approximately 2-2.5 hours) was arduous and labor intensive.
Because of the intensive labor investment and strain on cell viability required for a wash process, we investigated a thaw-and-dilute process that aimed to preserve cell viability and limit demands on personnel as clinical enrollments increased. The thaw-and-dilute procedure steps included retrieving the CellSeal cryogenic vial from the liquid nitrogen freezer, thawing the vial using the CellSeal automated thawing system, and drawing up the dose volume needed from the cell vial into a single luer-lock syringe. The cells were then diluted with Normosol (1:10 dilution). A second luer-lock syringe with Normosol-R was prepared and attached to the prepared cell syringe using a fluid dispensing connector. Then, the cells were gently mixed with Normosol-R and the calculated volume of cell suspension was withdrawn. Last, the final dose for delivery was placed in a labeled syringe (mean volume, 2.7 mL).
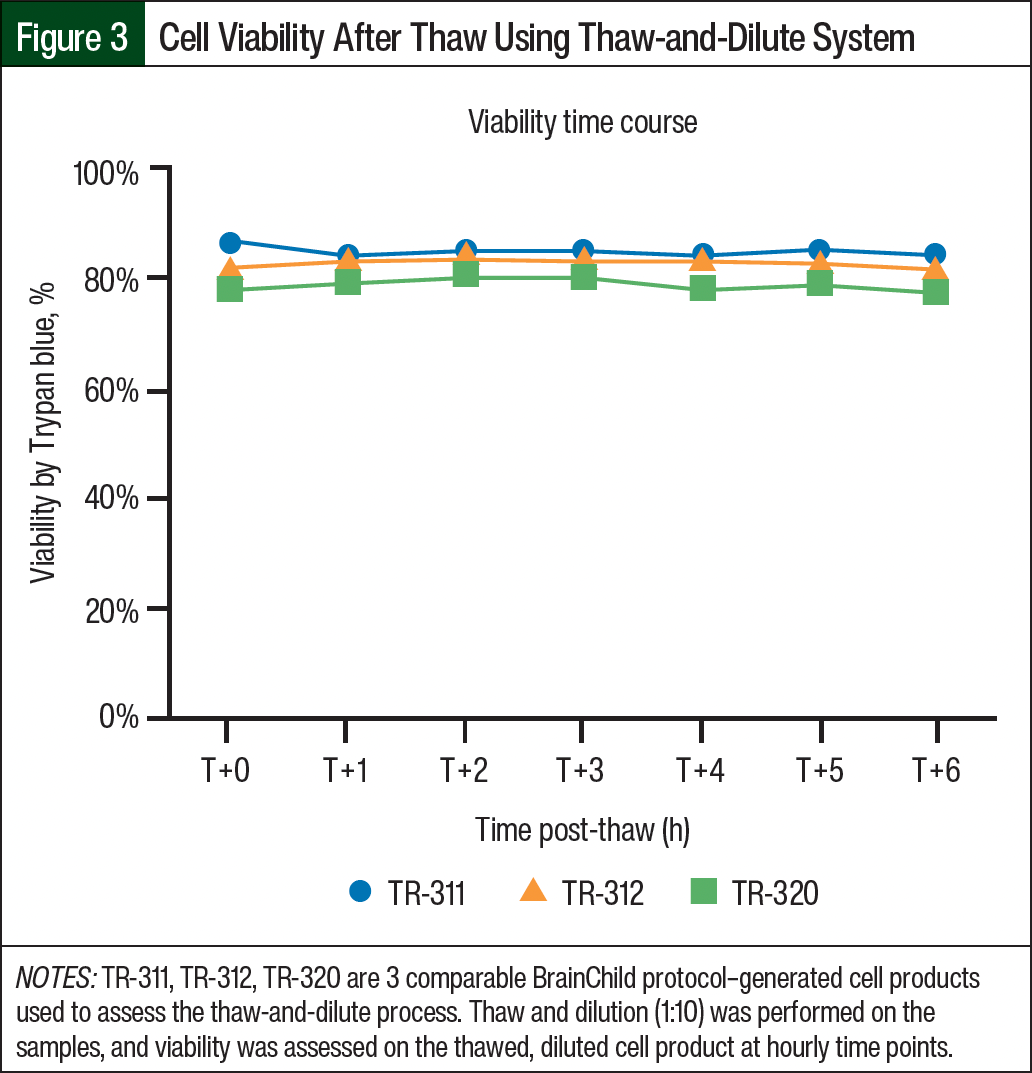
Viability assessments during the process qualification were performed by TCPC for 3 different samples from the same cryopreserved material. The cell viability was uniformly >70% (range, 74.3%-75.8%) and was always within the <10% range of the cell viability assessed at the time of cryopreservation. In comparison, the samples tested during the process qualification by thaw and wash demonstrated cell viability of 72.4% to 75.8%. This viability was preserved through 6 hours post thaw (Figure 3), which is much longer than the time from thaw to patient infusion (approximately 1 hour).
The studies ensured that the final product quality included inter- and intraoperator assessment of processed samples for stability. We determined that the thaw-and-dilute protocol yielded final concentrations of DMSO to <1% (range, 0.6%-0.7%) and improved cell recovery compared with the previous thaw-and-wash protocol. Part of the process qualification to thaw and dilute was to demonstrate that viability assessments were no longer required after release to the IDS pharmacy and that viability testing for cell products prepared by thaw and dilute were no longer required by protocol, thereby further reducing labor and resources.
The initial thaw-and-wash and thaw-and-dilute processes were performed within the hospital inpatient pharmacy under a sterile biological safety cabinet. All equipment, materials, and calculations are recorded on the standard operating procedure document along with the operator or verifier check. Although the thaw-and-wash process took approximately 2 to 2.5 hours, the thaw-and-dilute process requires only 30 to 40 minutes of preparation time. Once the cell dose has been prepared, the syringe is labeled and delivered to the bedside nurse by the IDS pharmacy staff. Cell product administration occurs over approximately 1 minute. As of 2024, the IDS pharmacy has successfully prepared and dispensed more than 500 doses of CAR T-cell product.
Conclusion
We present the preliminary clinical experience of manufacturing and preparing outpatient repeated intracranial CAR T-cell doses for children in Seattle Children’s Hospital’s phase 1 BrainChild clinical trials. By leveraging the experience of our cellular therapeutic team, including clinical neuro-oncology, outpatient oncology, TCPC, and IDS pharmacy, our team devised an efficient roadmap for intracranial CAR T-cell doses. The T-cell journey begins at Seattle Children’s Hospital, before undergoing manufacturing at TCPC and storage and preparation at the IDS pharmacy, and then begins its therapeutic mission through intracranial outpatient CAR T-cell infusions. We have demonstrated a feasible role for pharmacists in the collaborative delivery of CAR T-cell therapy and potentially other cellular-based therapies. Ultimately, as cellular therapies reach FDA approval, they will come to market as medications or drugs. The currently approved CAR T-cell products (eg, tisagenlecleucel, axicabtagene ciloleucel, betibeglogene autotemcel) are assigned National Drug Codes that identify these products as drugs. These drugs are regulated by the Federal Food, Drug, and Cosmetic Act and use common drug codes for billing and reimbursement. Pharmacists have skill sets and expertise with the medication use process (ie, safe delivery of drugs and products to a patient) to manage the delivery of cellular products.
Although the preparation of this specific CAR T-cell product required additional training and equipment that are not traditionally available in pharmacies, the overarching medication processes remain and use the same skill sets as traditional pharmacy practices, including dose calculation, order verification, complex sterile compounding, and dispensing of patient doses. Through collaboration with the pharmacy, cell therapy laboratories, nursing, and provider teams can leverage their existing infrastructure, medication safety policies, and institutional guidelines. There are unique requirements for the procurement, storage, and handling of cell therapies that can be incorporated into existing pharmacy practice models to allow for efficiencies that create opportunities to optimize the delivery of cutting-edge therapies to patients. We hope this article will aid in the shared goal of providing more immunotherapeutic clinical trial options to children and young adults with CNS tumors.
Acknowledgments
We thank the children and families who bravely shoulder the burden of their disease and place their trust in Seattle Children’s Hospital. We are indebted to A. Thomsen for her expert immunotherapy clinical coordination. We would like to thank our clinical research team, including J. Stevens, H. Ullom, S. Bakotich, S. Bagchi, J. Chau, D. Chen, M. Guthrie, K. Hoon, C. Krein, R. Persona, Z. Maino, M. MacQuivey, and A. Westby.
Funding Sources
Funding has been provided by Cookies for Kid’s Cancer Young Investigator Grant (NAV), DIPG All-In (NAV), DIPG/DMG Research Funding Alliance (NAV), Matthew Larson Research Grant (NAV), the We Love You Connie Foundation (NAV), and St Baldrick’s Stand Up to Cancer Dream Team Translational Cancer Research Grants (SU2C-AACR-DT-27-17; NAV, RAG, RR, MCJ, JRP; Stand Up to Cancer is a division of the Entertainment Industry Foundation, and research grants are administered by the American Association for Cancer Research); and U01 TR002487 (JRP).
Author Disclosure Statement
Dr Vitanza serves as the scientific advisory board chair for BrainChild Bio, Inc. Dr Foster is a consultant for Teladoc and has received honoraria/royalties from Tmunity; Dr Gust is a consultant at Johnson & Johnson; Dr Gardner has received royalties from Juno Therapeutics and has received research support from 2seventy bio; Dr Jensen is the Chief Scientific Officer of BrainChild Bio and has received research and clinical trial support from Umoja Biopharma and 2seventy bio. Drs Vitanza, Foster, Gardner, and Jensen are inventors on issued and pending patents related to CAR T-cell therapies. Dr Choe, Mr Brown, Mr Beebe, Dr Kong, Dr Rogers, Dr Jacob, Ms Mano, Ms Abuan, Ms Mgebroff, Ms Lindgren, Dr Gustafson, Dr Wilson, Ms Noll, Dr Ronsley, Dr Crotty, Dr Leary, and Dr Park have no conflicts of interest to report.
References
- Gardner RA, Finney O, Annesley C, et al. Intent-to-treat leukemia remission by CD19 CAR T cells of defined formulation and dose in children and young adults. Blood. 2017;129:3322-3331.
- Vitanza NA, Monje M. Diffuse intrinsic pontine glioma: from diagnosis to next-generation clinical trials. Curr Treat Options Neurol. 2019;21:37.
- Louis DN, Perry A, Reifenberger G, et al. The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 2016;131:803-820.
- Donovan LK, Delaidelli A, Joseph SK, et al. Locoregional delivery of CAR T cells to the cerebrospinal fluid for treatment of metastatic medulloblastoma and ependymoma. Nat Med. 2020;26:720-731.
- Brown CE, Badie B, Barish ME, et al. Bioactivity and safety of IL13Rα2-redirected chimeric antigen receptor CD8+ T cells in patients with recurrent glioblastoma. Clin Cancer Res. 2015;21:4062-4072.
- Brown CE, Alizadeh D, Starr R, et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N Engl J Med. 2016;375:2561-2569.
- HER2-specific CAR T cell locoregional immunotherapy for HER2-positive recurrent/refractory pediatric CNS tumors. NLM identifier: NCT03500991. Updated December 12, 2023. Accessed June 27, 2024. https://clinicaltrials.gov/study/NCT03500991
- EGFR806-specific CAR T cell locoregional immunotherapy for EGFR-positive recurrent or refractory pediatric CNS tumors. NLM identifier: NCT03638167. Updated October 27, 2023. Accessed June 27, 2024. https://clinicaltrials.gov/study/NCT03638167
- Study of B7-H3-specific CAR T cell locoregional immunotherapy for diffuse intrinsic pontine glioma/diffuse midline glioma and recurrent or refractory pediatric central nervous system tumors. NLM identifier: NCT04185038. Updated December 20, 2023. Accessed June 27, 2024. https://clinicaltrials.gov/study/NCT04185038
- Vitanza NA, Johnson AJ, Wilson AL, et al. Locoregional infusion of HER2-specific CAR T cells in children and young adults with recurrent or refractory CNS tumors: an interim analysis. Nat Med. 2021;27:1544-1552.
- Vitanza NA, Wilson AL, Huang W, et al. Intraventricular B7-H3 CAR T cells for diffuse intrinsic pontine glioma: preliminary first-in-human bioactivity and safety. Cancer Discov. 2023;13:114-131.
- Ravanpay AC, Gust J, Johnson AJ, et al. EGFR806-CAR T cells selectively target a tumor-restricted EGFR epitope in glioblastoma. Oncotarget. 2019;10:7080-7095.
- Ercikan-Abali EA, Mineishi S, Tong Y, et al. Active site-directed double mutants of dihydrofolate reductase. Cancer Res. 1996;56:4142-4145.
- Zhang C, Deng Y, Dai H, et al. Effects of dimethyl sulfoxide on the morphology and viability of primary cultured neurons and astrocytes. Brain Res Bull. 2017;128:34-39.