Symptom Overview
Rasburicase is a recombinant urate oxidase that catalyzes the oxidation of uric acid into allantoin, a more soluble metabolite. Within hours of intravenous administration, rasburicase directly reduces serum uric acid concentrations, making it an essential therapy for the prevention and treatment of hyperuricemia in patients with tumor lysis syndrome (TLS).1 Prompt administration of rasburicase in high-risk malignancies is therefore key for preventing uric acid–induced acute kidney injury, which can exacerbate concomitant electrolyte abnormalities of TLS and potentially delay chemotherapy elimination.
It is important to be cognizant, however, of the potential severe side effects of rasburicase administration. Specifically, rasburicase is contraindicated in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency, because of the drug’s potential to cause hemolytic anemia.2
Hemolytic anemia can be normocytic or macrocytic, can occur extravascularly or intravascularly, and is characterized by a sudden drop in hemoglobin and oxygen saturation, with concurrent reticulocytosis not explained by active bleeding or by nutrient deficiency.3 After rasburicase administration, in patients with G6PD deficiency, hemolysis may occur intravascularly, in which hemoglobin is being released into the blood circulation and eliminated by the kidneys, often giving urine a dark, brownish appearance.4
In addition, the free hemoglobin in the serum binds to haptoglobin and is then converted to bilirubin in the liver. This results in low-to-undetectable levels of haptoglobin and elevated bilirubin levels. Lactate dehydrogenase is also released after red blood cell (RBC) destruction, resulting in elevated levels of lactate dehydrogenase.3
Rasburicase has also been known to induce methemoglobinemia in patients with G6PD deficiency. Methemoglobinemia is a disorder characterized by an increased production (>1%) of methemoglobin—an oxidized form of hemoglobin that contains Fe3+ in place of Fe2+—which results in a greater affinity for oxygen binding and decreased release of oxygen in tissues.5 Early symptoms of methemoglobinemia include altered mental status, dyspnea, cyanosis, and an asymptomatic drop in O2 saturation.6,7
Etiology
Hemolytic anemia can be caused by autoimmune or nonimmune processes. Autoimmune hemolytic anemia involves RBC destruction caused by immunoglobulin G antibodies.4 Autoimmune hemolytic anemia is often secondary to diseases involving the immune system, such as HIV, rheumatoid arthritis, and chronic lymphocytic leukemia. To test for autoimmune hemolytic anemia, a direct antiglobulin test, also known as the direct Coombs test, would yield a positive result and would necessitate further testing. Negative direct antiglobulin test results suggest that the hemolytic process is not likely to be immune-related, and that other causes should be evaluated.
G6PD is an enzyme involved in the pentose phosphate pathway that maintains levels of nicotinamide adenine dinucleotide phosphate (NADPH), a common reducing agent that is a co-factor in several biochemical reactions.8 Deficiency in G6PD is an inherited, X-linked disorder that affects more than 400 million people worldwide, with its highest frequencies in Africa, Southeast Asia, and the Mediterranean to the Middle East regions. As with most X-linked diseases, G6PD deficiency is more common in men than in women. Heterozygous women can have cells deficient in G6PD similar to cells of hemizygous men, but the clinical manifestations in such women are typically less severe than in men.8
Individuals with G6PD deficiency are uniquely sensitive to exposure to oxidative stress, which can result in intravascular hemolytic anemia. Hemolytic anemia caused by oxidative stress is a nonimmune-related process, and would result in negative direct antiglobulin test results.4 Moreover, the oxidative stress mediates an intracorpuscular mechanism for RBC destruction and is often caused by a hereditary condition, such as G6PD deficiency. Mature RBCs lack mitochondria, relying instead on continuous enzymatic restoration of redox environments to protect them from significant oxidation involved in their oxygen transport role.8
The only source of NADPH in an RBC is the pentose phosphate pathway. Therefore, increased exposure to oxidation in the setting of diminished G6PD expression leads to denatured hemoglobin, characterized as “Heinz bodies,” which are then destroyed by macrophages in the spleen.9 After rasburicase administration, hydrogen peroxide is created as the byproduct of the enzymatic oxidation of uric acid into allantoin. With low levels of G6PD expression, the normal mechanisms to convert hydrogen peroxide to water are overloaded, resulting in extensive hemolysis.
There have also been case reports of rasburicase-induced hemolytic anemia in individuals with normal G6PD function. One case involved a pediatric patient with acute lymphoblastic leukemia who experienced methemoglobinemia and hemolytic anemia after the second dose of rasburicase.10 It was determined that the rasburicase was the probable cause, based on the Naranjo probability scale.10 An additional case of rasburicase-induced hemolytic anemia was described in an adult with mycosis fungoides who was undergoing salvage chemotherapy and received a 15-mg (0.2-mg/kg) dose of rasburicase.7
Treatment Options
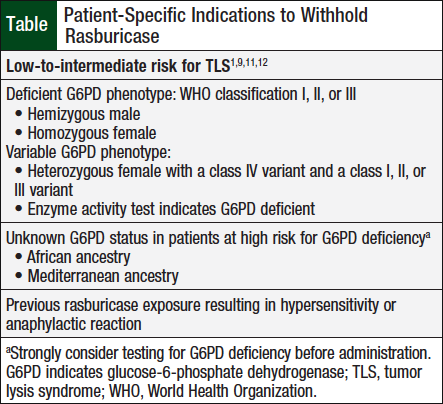
Ideally, the best course of action is prevention. Although rasburicase-induced hemolytic anemia is relatively rare, it is recommended that patients at increased risk for G6PD deficiency be tested before rasburicase administration when G6PD status is unknown.11 However, the time frame required for G6PD testing may exceed that needed for prompt rasburicase administration. This creates a clinical conundrum, because hemolytic anemia and TLS are potentially fatal. If G6PD status is unknown, it should not preclude the use of rasburicase for most patients. The Table lists the patients who should not receive rasburicase.1,9,11,12
Studies involving dapsone and primaquine have noted that the dose and frequency of the offending drug is a major determinant of the severity of acute hemolytic anemia.9 This dose-dependent oxidative attack on RBC destruction typically affects the oldest, most G6PD-deficient RBCs first. Therefore, lowering the dose or extending the dosing interval of the offending drug can lessen RBC destruction. However, this approach has yet to be demonstrated with rasburicase, because even single doses of 0.03 mg/kg and flat doses of 6 mg in patients with G6PD deficiency have resulted in hemolysis.13
It is unclear whether doses of rasburicase ≤3 mg may mitigate hemolysis in patients with G6PD deficiency. In the event that urgent rasburicase administration is required in a high-risk patient with unknown G6PD status, it would be reasonable to use single doses of 3 mg or 1.5 mg to mitigate potential hemolysis. Further studies are needed to validate this approach.
Although rasburicase-induced hemolytic anemia is potentially fatal, the majority of known cases have been mild, with treatment options mainly involving supportive care with oxygen supplementation and packed RBC transfusions.13 Any other potentially oxidative agents should be discontinued, at least until normal RBC function is restored.
It is important to note that although corticosteroids are vital in the treatment of autoimmune hemolytic anemia, they are ineffective for nonimmune-related hemolytic anemia.4 This underscores the importance of obtaining a direct antiglobulin test to rule out an autoimmune process. This is particularly important in patients with leukemia, for whom autoimmune hemolytic anemia can be secondary to the disease, or those with an underlying infectious process.
When present, methemoglobinemia often precedes hemolytic anemia; therefore, methylene blue may be contemplated as a treatment option. However, methylene blue is reduced by NADPH and will likely exacerbate hemolysis in patients with G6PD deficiency, and should be avoided in such patients.5
Alternatively, intravenous ascorbic acid exhibits reducing and chelating properties that augment the absorption of nonheme iron and has been used in successful treatment of methemoglobinemia when methylene blue cannot be used.14
Conclusion
Rasburicase is an essential agent for the prevention and treatment of hyperuricemia in patients with TLS, but it should be avoided, if possible, in those with a suspected or known G6PD deficiency, because of the risk for hemolytic anemia. Hemolytic anemia is uncommon, but it can be a serious concern, and although it is usually mild, it could lead to death in rare cases. Hemolytic anemia in patients with G6PD deficiency usually resolves with discontinuation of rasburicase and with supportive care measures, including packed RBC transfusions and/or oxygen supplementation. Corticosteroids are not likely to be effective, and methylene blue should be avoided if methemoglobinemia is present.
Author Disclosure StatementDr Kaland and Dr Borg have no conflicts of interest to report.
References
- Coiffier B, Altman A, Pui CH, et al. Guidelines for the management of pediatric and adult tumor lysis syndrome: an evidence-based review. J Clin Oncol. 2008;26:2767-2778. Erratum in: J Clin Oncol. 2010;28:708.
- Nguyen AP, Ness GL. Hemolytic anemia following rasburicase administration: a review of published reports. J Pediatr Pharmacol Ther. 2014;19:310-316.
- Lechner K, Jäger U. How I treat autoimmune hemolytic anemias in adults. Blood. 2010;116:1831-1838.
- Dhaliwal G, Cornett PA, Tierney LM Jr. Hemolytic anemia. Am Fam Physician. 2004;69:2599-2606.
- Sikka P, Bindra VK, Kapoor S, et al. Blue cures blue but be cautious. J Pharm Bioallied Sci. 2011;3:543-545.
- Sonbol MB, Yadav H, Vaidya R, et al. Methemoglobinemia and hemolysis in a patient with G6PD deficiency treated with rasburicase. Am J Hematol. 2013;88:152-154.
- Kizer N, Martinez E, Powell M. Report of two cases of rasburicase-induced methemoglobinemia. Leuk Lymphoma. 2006;47:2648-2650.
- Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet. 2008;371:64-74.
- Luzzatto L, Seneca E. G6PD deficiency: a classic example of pharmacogenetics with on-going clinical implications. Br J Haematol. 2014;164:469-480.
- Bauters T, Mondelaers V, Robays H, et al. Methemoglobinemia and hemolytic anemia after rasburicase administration in a child with leukemia. Int J Clin Pharm. 2013;35:303-305.
- Relling MV, McDonagh EM, Chang T, et al; for the Clinical Pharmacogenetics Implementation Consortium. Clinical Pharmacogenetics Implementation Consortium (CPIC) guidelines for rasburicase therapy in the context of G6PD deficiency genotype. Clin Pharmacol Ther. 2014;96:169-174.
- WHO Working Group. Glucose-6-phosphate dehydrogenase deficiency. Bull World Health Organ. 1989;67:601-611.
- Sherwood GB, Paschal RD, Adamski J. Rasburicase-induced methemoglobinemia: case report, literature review, and proposed treatment algorithm. Clin Case Rep. 2016;4:315-319.
- Park SY, Lee KW, Kang TS. High-dose vitamin C management in dapsone-induced methemoglobinemia. Am J Emerg Med. 2014;32:684.e1-684.e3.