Connecting Science to Practice
The increasing use of biomarker testing across the oncology
care continuum has led to more frequent identification
of HER2-positive cancers, driving greater utilization
of trastuzumab and other HER2-targeted
therapies. Although these agents offer significant survival
benefits, their potential to induce or exacerbate
heart failure presents a critical clinical challenge. Understanding
left ventricular ejection fraction (LVEF)
dynamics is essential for balancing therapeutic efficacy
with cardiac safety and guiding decisions regarding
when to hold, resume, or discontinue therapy. Clinicians
must also evaluate the role of adjunctive cardioprotective
strategies, such as angiotensin-converting
enzyme inhibitors or beta-blockers, and their effectiveness
in mitigating risk. Ultimately, integrating evidence-
based monitoring protocols with individualized
risk assessment ensures that the benefits of targeted
therapy are maximized without compromising cardiovascular
health. This overview of decreased LVEF as a
complication from use of trastuzumab synthesizes current
guidelines and clinical practice.
HER2-directed agents, such as trastuzumab, have become a cornerstone of treatment for patients with HER2-overexpressing breast cancer.1 The American Cancer Society reports that 15% to 20% of breast tumors have a higher level of HER2 proteins.2 HER2 gene amplification is assessed using in situ hybridization and protein overexpression by immunohistochemistry. Guidance for HER2 testing in patients with breast cancer is provided by ASCO and the College of American Pathologists in their 2018 clinical guideline.3,4 In addition, the NCCN has formally endorsed this clinical guideline within its invasive breast cancer guidelines.1 HER2-positive breast cancers are often considered to be more favorable because they can be treated with drugs that target HER2, unlike those with negative HER2 expression.2 Trastuzumab works by binding the extracellular domain of HER2-overexpressing cells, which inhibits the proliferation of cancer cells.5
Indications for the use of trastuzumab have expanded beyond breast cancer into colorectal, endometrial, and gastric cancers.5-8 These subsequent indications offer opportunities for additional research on managing adverse events (AEs) with trastuzumab; this article, however, will focus on the management of this agent’s cardiotoxicity in breast cancer only. In addition, we will focus only on the HER2 overexpression in breast cancer as defined in the 2018 ASCO guideline.3 Research is ongoing on the relevance of HER2 point mutations and overexpression and mutations in the ERBB2 gene in breast cancer. ERBB2 and HER2 are often referred to as the same, but it should be noted that the HER2 protein is a receptor, whereas ERBB2 is a gene that encodes the protein.
The oncology landscape is seeing rising healthcare costs and reduced reimbursements, and numerous biosimilars of trastuzumab have been developed to better control costs. To date, 6 biosimilars are available: trastuzumab-strf (Hercessi), trastuzumab-pkrb (Herzuma), trastuzumab-anns (Kanjinti), trastuzumab-dkst (Ogivri), trastuzumab-dttb (Ontruzant), and trastuzumab-qyyp (Trazimera). These biosimilars are highly similar with no clinically significant differences in safety or efficacy. None, however, are interchangeable without physician order, collaborative practice agreement, or institution-approved therapeutic interchange based on their FDA approval statuses. Many institutions have developed biosimilar interchanges that allow the most cost-effective agent to be given to inpatients and allow outpatient clinics to interchange based on insurance requirements. As additional biosimilars continue to be approved, it is imperative to continue to evaluate practices and processes related to selecting, purchasing, and stocking these agents.
Trastuzumab was the first FDA-approved HER2-directed therapy that demonstrated improvement in overall survival (OS), disease-free survival (DFS), and time to distant recurrence for those with early-stage HER2-positive breast cancer.9-11 Romond and colleagues reported that DFS for the alive patients at 4 years was 67.1% in the control group and 85.3% in the trastuzumab group (absolute difference, 18.2 percentage points; 95% confidence interval (CI), 12.7-23.7).9 The researchers also reported that OS at 4 years was 86.6% in the control group and 91.4% in the trastuzumab group (absolute difference, 4.8 percentage points; 95% CI, 0.6-9.0).9 Piccart-Gebhart and colleagues reported a benefit in DFS that was 8.4 percentage points higher in the trastuzumab group at 2 years compared with the observation group (95% CI, 2.1-14.8); the 2-year DFS rate was 85.8% in the trastuzumab group and 77.4% in the observation group.10 This study also showed that time to distant recurrence was statistically significant, with an absolute value of 7.8 percentage points higher in the trastuzumab group compared with the control group (90.6% vs 82.8%; 95% CI, 0.38–0.63).10 OS was also higher, although not statistically significant, with 96% in the trastuzumab group at 2 years compared with 95.1% in the observation group (95% CI, 0.47–1.23; P<.26).10 Trastuzumab also proved to be advantageous in time to disease progression, rates of response, duration of response, and time to treatment failure in those who had disease progression on chemotherapy alone and later received trastuzumab in a crossover study by Slamon and colleagues.11
Targeted drug therapies continue to be developed with goals to maintain efficacy and minimize AEs. Despite these efforts, AEs are associated with nearly every oncologic treatment plan. Patients receiving trastuzumab are at increased risk for tolerable AEs, such as rash and gastrointestinal upset.11 Significant cardiac risks for trastuzumab were first reported in the phase 3 trial by Slamon and colleagues, which assessed trastuzumab in combination with chemotherapy in patients with metastatic breast cancer.11 This serious event warranted discontinuation in 8% of patients, although the clinical picture was complicated by the cardiotoxicity of concurrent chemotherapy agents.11
Symptom Overview
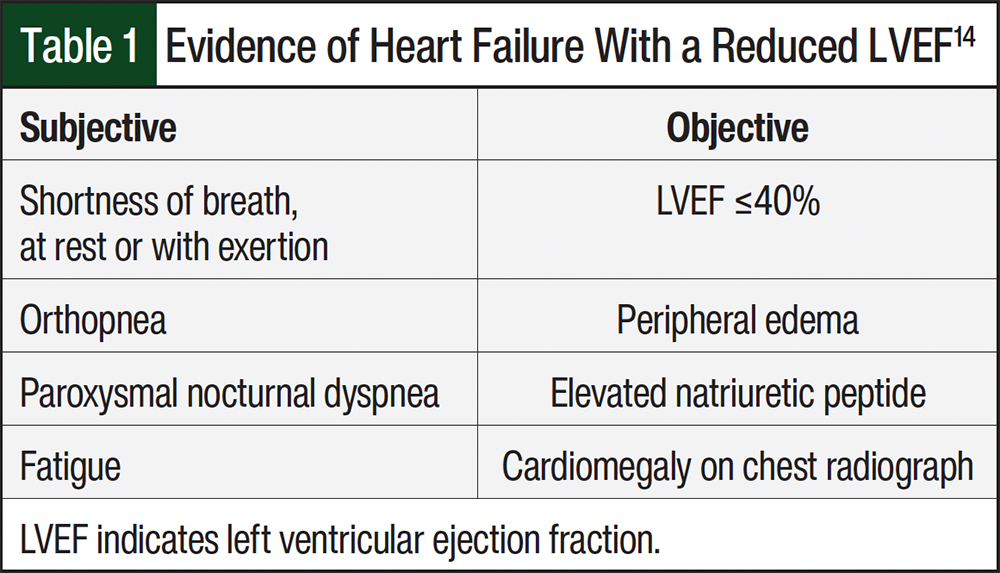
Trastuzumab can cause a range of cardiac dysfunctions, such as hypertension, arrhythmia, decreased left ventricular ejection fraction (LVEF), cardiac failure, and cardiac death.5,9,11 The FDA has required that the trastuzumab prescribing information include a boxed warning for cardiomyopathy because asymptomatic and symptomatic cardiac dysfunctions were seen in the phase 3 trials and postmarketing surveillance.5,9,11 Decreased LVEF is reported in up to 22% of patients on trastuzumab, with the highest incidence seen in those also receiving an anthracycline-containing chemotherapy regimen.12 A normal LVEF range is 50% to 70%, meaning that with each heartbeat the left ventricle is expected to pump out 50% to 70% of the blood to the rest of the body.13 In symptomatic cardiac dysfunction, patients may experience cool, moist hands; shortness of breath; pedal edema; and orthopnea. Findings on physical examination may include bilateral crackles, S3 gallop, extension of the jugular vein, sinus tachycardia, and tachypnea.14 Decreased LVEF can lead to detrimental effects on health, including death. Dunlay and colleagues reported that the median time from advanced heart failure diagnosis to death was 12.2 months.14 Advanced heart failure in this study was an operationalized definition characterized by episodes of congestion, low output or malignant arrhythmias, forms of cardiac dysfunction, exercise impairment, and severe symptoms of heart failure despite current guideline-directed medical therapies.14 Because heart failure can be fatal, monitoring LVEF in asymptomatic patients is imperative. It is important to recognize the clinical signs of cardiac dysfunction and monitor LVEF in patients receiving trastuzumab to prevent irreversible cardiac damage.
Etiology
The exact mechanism for cardiotoxicity from HER2-directed therapies is poorly understood; however, a few hypotheses may explain the cause. Most data in this area are gathered from murine studies, and not all findings have been reproducible in human studies, leading to uncertainty regarding the exact mechanism that cardiotoxicity occurs from trastuzumab.15 Several animal studies suggest that blockade of HER2 signaling leads to reduced protection against cardiotoxins.16-18 Evidence from murine studies suggests involvement from the ERBB2 proto-oncogene, leading to dilated cardiomyopathy.16 One hypothesis is that trastuzumab produces significantly elevated oxidative and nitrative damage in cardiomyocytes.17 This is likely due to a reduction in reactive oxygen species scavengers caused by increased glutathione expression that leads to an ultimate reactive oxygen species increase.17 Another hypothesis generated from a study involving a murine a model is that increased reactive oxygen species production may contribute to mitochondrial impairment from loss of membrane integrity and increased cytochrome C release.18 Finally, given the pro-inflammatory effects of trastuzumab on the body, another hypothesis holds that cardiotoxicity is caused from structural remodeling of the heart. Namely, that the drug may trigger a signaling cascade, including overexpression of tumor necrosis factor-alpha, interleukin-6, and transforming growth factor-beta.15
Other agents used to treat breast cancer with and without trastuzumab have increased risk for cardiotoxicity as well, such as the anthracycline class. The cardiotoxicity of trastuzumab differs from that of anthracyclines. Because it is not dose-dependent, it does not occur in all patients and is usually reversible.19 Patients who receive a treatment regimen containing an anthracycline and a HER2-directed drug are at the highest risk for developing cardiomyopathy, and patients with certain preexisting conditions are also at an increased risk. These conditions include heart failure, decreased baseline LVEF, coronary artery disease, atrial fibrillation, hypertension, diabetes, increased body mass index, dyslipidemia, renal failure, and increased age. In 2001, Slamon and colleagues discussed cardiotoxicity seen in their phase 3 trial, as symptomatic and asymptomatic events were seen in 63 of 464 patients.11 These patients were also receiving chemotherapy, and a higher incidence of cardiotoxicity was seen in those with an anthracycline agent in the regimen.11 New York Heart Association class III or IV cardiac dysfunction was reported in 16% of patients receiving an anthracycline/cyclophosphamide/trastuzumab regimen, 3% of patients receiving an anthracycline/cyclophosphamide combination, 2% of patients receiving paclitaxel and trastuzumab, and 1% of patients receiving paclitaxel alone.11
Combination therapies are a cause for concern due to increased cumulative cardiotoxicity in patients. Extended infusion of anthracyclines and trastuzumab should be avoided.1 Trastuzumab in combination with a taxane is generally considered safe due to low rates of severe cardiotoxicity.20 The use of lapatinib, a tyrosine kinase inhibitor, has been reported to decrease LVEF; thus, cardiotoxicity as an additive effect may be a concern when used with trastuzumab.21 Evidence suggests that these agents are not significantly more cardiotoxic together, compared with when used individually.22 Pertuzumab, another HER2-directed monoclonal antibody, also did not show significantly worse cardiotoxicity when used with trastuzumab in the CLEOPATRA trial.23
Treatment Options
Several small trials have looked at the role of beta-blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), and statin therapies for the prevention of cardiomyopathy in patients receiving trastuzumab.24,25 Gujral and colleagues published a meta-analysis that concluded that ARB prophylaxis did not prevent a decrease in LVEF or the development of new heart failure in patients receiving anthracycline chemotherapy with or without trastuzumab.24 They did find, however, that beta-blocker use was associated with lower LVEF dysfunction and lower rates of new heart failure; however, limitations in the studies make the results less useful in clinical practice.24 The PRADA trial showed modest benefit with the use of candesartan in preventing LVEF reduction as assessed by magnetic resonance imaging at 3 years compared with patients not receiving candesartan (P=.026).25
The use of ACE inhibitors, ARBs, and beta-blockers as prophylaxis is an ongoing area of investigation. ASCO does not give recommendations on the use of these agents to prevent cardiac dysfunction because of the lack of compelling data currently available.12 According to the 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure, it may be reasonable in asymptomatic patients with cancer therapy–related cardiomyopathy (ejection fraction <50%) to use an ARB, ACE inhibitor, or beta-blocker to prevent progression of heart failure and improve cardiac function.13 These guidelines also state that the use of beta-blockers, ACE inhibitors, or ARB therapy for the primary prevention of drug-induced cardiomyopathy is of uncertain benefit.13
Pooled data from 6 randomized trials (N=1070) that examined the use of dexrazoxane for cardiotoxicity prevention suggested that dexrazoxane reduced the relative risk of cardiotoxicity by 77% (odds ratio , 0.21; 95% CI, 0.08-0.51; P=.0006).26 Seymour and colleagues evaluated dexrazoxane as a cardioprotective agent in patients receiving doxorubicin and epirubicin. The authors did not review its use with trastuzumab, but this is mentioned as it may be an opportunity for future research.26 Risk of myelosuppression and secondary malignancies induced by dexrazoxane may outweigh any potential benefits.27
Aside from the general approach to management of heart failure, the 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure provides specific recommendations for cardio-oncology patients.13 Management of patients at risk for cardiac dysfunction should include multidisciplinary discussions around healthy lifestyle habits, a recommendation that is supported by the strongest level of evidence.13 The general approach is to use guideline-directed medical therapies for all patients with a reduced ejection fraction.13
Course of Therapy
Cardiotoxicity from trastuzumab is most likely to occur within 6 months of therapy initiation and is characterized by a decrease in LVEF.28 Trastuzumab-induced reduction of LVEF after completion of trastuzumab therapy has also been reported.12 For this reason, LVEF should be evaluated routinely before, during, and after treatment. Clinicians are recommended to complete a thorough history and screening regarding the patient’s cardiac status and previous cumulative chemotherapies, specifically anthracyclines because of the increased risk for cardiotoxicity.12 Table 1 provides objective and subjective signs of reduced LVEF that a patient with heart failure may display.14
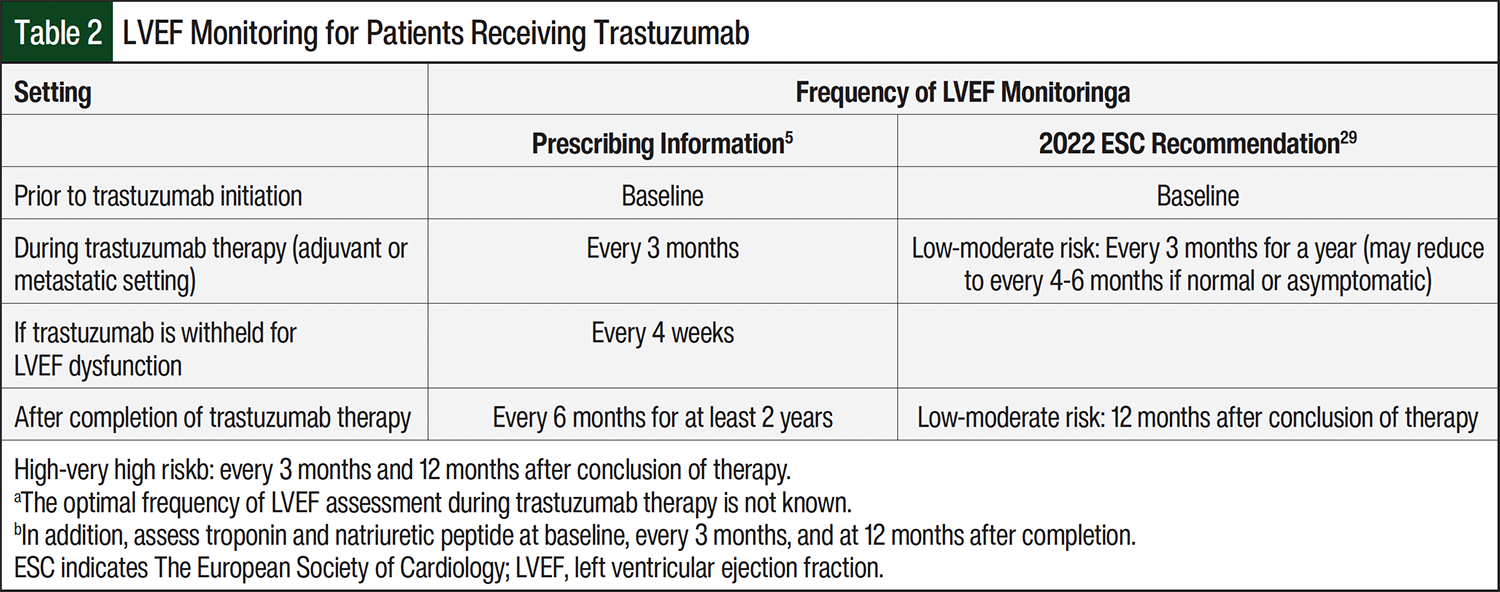
A baseline transthoracic echocardiogram is preferred because it is noninvasive while providing appropriate cardiac structure and functional views. A multigated acquisition scan may also be used if an echocardiogram is not feasible.29 Although optimal frequency of assessment has not been determined, the FDA recommends LVEF measurements before beginning treatment and every 3 months while on therapy.5 Table 2 provides the guideline-based recommended monitoring frequency for LVEF in patients undergoing treatment with trastuzumab in adjuvant and metastatic settings.5,29
A pause to assess current medication therapies is warranted when a decrease in LVEF has been determined. The FDA prescribing information for trastuzumab recommends withholding treatment for ≥4 weeks if LVEF has decreased ≥16% from baseline or if LVEF is below institutional limits of normal and ≥10% absolute decrease in LVEF from pretreatment values.5 Therapy may be resumed if LVEF returns to normal limits and the absolute decrease from baseline is ≤15% within 4 to 8 weeks.5 Trastuzumab should be permanently discontinued if LVEF decline persists for >8 weeks or if trastuzumab treatment was held on >3 occasions for cardiomyopathy.5 Anthracyclines should be avoided, if possible, to reduce any further cardiac dysfunction.5 Ultimately, the decision to stop other cardiotoxic agents is a clinical decision that may vary based on provider experience.
Trastuzumab-related cardiomyopathy is generally reversible over a period of 1 to 3 months after discontinuation.19 For example, a phase 3 trial looked at 33 patients receiving trastuzumab for a median of 26 weeks despite developing a cardiac event (most often an mild decline in LVEF).30 The cardiac status of 28 (85%) of these patients improved or remained the same.30 Another trial examined 235 patients receiving trastuzumab in combination with chemotherapy. Of these patients, 63 developed cardiac dysfunction, and of these 63, 44 patients were treated with standard medical care. Symptoms were reversible for 75% of patients receiving standard medical care.11
Consensus is lacking as to the ideal management of patients with a history of cardiac conditions or persistent cardiac dysfunction despite discontinuation of therapy. NCCN does not have specific monitoring recommendations; however, NCCN recommends follow-up based on patient cardiovascular risk assessments and manufacturer’s prescribing information.1 The prescribing information for trastuzumab recommends cardiac monitoring with echocardiography or multigated acquisition scan at baseline, every 3 months while on treatment and every 6 months following treatment for at least 2 years.5 The European Society of Cardiology (ESC) also published guidelines in 2022 that discuss monitoring recommendations for those on HER2-targeted therapies, such as trastuzumab.29 For low- to moderate-risk patients, there is a Class 1 recommendation for echocardiography at baseline, every 3 months during treatment for 12 months, and 12 months post treatment.29 For patients at high or very high risk, ESC recommends an additional echocardiogram at 3 months after completion of therapy in addition to the 12-month posttreatment echocardiography.29 Clinicians may choose to use echocardiography surveillance in patients who continue to receive trastuzumab long-term at an interval determined by their clinical judgment.12
Conclusion
Trastuzumab has contributed to an improvement in outcomes in patients with breast cancer and is proving to have growing benefit in other cancer diagnoses as well. Because of the increased risk for cardiac dysfunction, especially in those who have received anthracycline therapy, close monitoring and management is often required. Clinicians could benefit from increased research and group consensus on management of risks and modifications to therapy in this setting.
Author Disclosure Statement
Dr Muche and Dr Mattila have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): breast cancer. Version 4.2025. April 17, 2025. Accessed May 29, 2025. www.nccn.org/professionals/physician_gls/pdf/breast.pdf
- Breast Cancer HER2 Status. American Cancer Society. August 25, 2022. Accessed December, 20, 2025. www.cancer.org/cancer/types/breast-cancer/understanding-a-breast-cancer-diagnosis/breast-cancer-her2-status.html
- Wolf AC, Somerfield MR, Dowsett M, et al. Human epidermal growth factor receptor 2 testing in breast cancer: ASCO–College of American Pathologists guideline update. J Clin Oncol. 2023;41:3867-3872.
- College of American Pathologists. HER2 Testing in Breast Cancer - 2023 Guideline Update. Accessed November 15, 2025. www.cap.org/protocols-and-guidelines/cap-guidelines/current-cap-guidelines/recommendations-for-human-epidermal-growth-factor-2-testing-in-breast-cancer
- Herceptin (trastuzumab) injection, for intravenous use [prescribing information]. Genentech, Inc; November 2018. Accessed May 29, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2024/103792s5354lbl.pdf
- Sartore-Bianchi A, Trusolino L, Martino C, et al. Dual-targeted therapy with trastuzumab and lapatinib in treatment-refractory, KRAS codon 12/13 wild-type, HER2-positive metastatic colorectal cancer (HERACLES): a proof-of-concept, multicentre, open-label, phase 2 trial. Lancet Oncol. 2016;17:738-746. Erratum in: Lancet Oncol. 2016;17:e420.
- Santin AD, Fader AN. Randomized phase II trial of carboplatin-paclitaxel compared to carboplatin-paclitaxel-trastuzumab in advanced or recurrent uterine serous carcinomas that overexpress Her2/neu (NCT01367002). Gynecol Oncol. 2018;149:12.
- Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687-697. Erratum in: Lancet. 2010;376:1302.
- Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353:1673-1684.
- Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659-1672.
- Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344:783-792.
- Armenian SH, Lacchetti C, Barac A, et al. Prevention and monitoring of cardiac dysfunction in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2017;35:893-911.
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Joint Committee on clinical practice guidelines. Circulation. 2022;145:e895-e1032. Errata in: Circulation. 2022;145:e1033; Circulation. 2022;146:e185; Circulation. 2023;147:e674.
- Dunlay SM, Roger VL, Killian JM, et al. Advanced heart failure epidemiology and outcomes. JACC Heart Fail. 2021;9:722-732.
- Eaton H, Timm KN. Mechanisms of trastuzumab induced cardiotoxicity – is exercise a potential treatment?. Cardiooncology. 2023;9:22.
- Özcelik C, Erdmann B, Pilz B, et al. Conditional mutation of the ErbB2 (HER2) receptor in cardiomyocytes leads to dilated cardiomyopathy. Proc Natl Acad Sci U S A. 2002;99:8880-8885.
- ElZarrad MK, Mukhopadhyay P, Mohan N, et al. Trastuzumab alters the expression of genes essential for cardiac function and induces ultrastructural changes of cardiomyocytes in mice. PLoS. 2013:8:e79543. Erratum in: PLos One. 2014;9:10.
- Gordon LI, Burke MA, Singh AT, et al. Blockade of the erbB2 receptor induces cardiomyocyte death through mitochondrial and reactive oxygen species-dependent pathways. J Biol Chem. 2009;284:2080-2087.
- Florido R, Smith KL, Cuomo KK, Russell SD. Cardiotoxicity from human epidermal growth factor receptor-2 (HER2) targeted therapies. J Am Heart Assoc. 2017;6:e006915.
- Telli ML, Witteles RM. Trastuzumab related cardiac dysfunction. J Natl Compr Canc Netw. 2011;9:243 249.
- Tykerb (lapatinib) tablets, for oral use [prescribing information]. Novartis Pharmaceuticals Corporation; December 2018. Accessed July 1, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2022/022059s031lbl.pdf
- Blackwell KL, Burstein HJ, Storniolo AM, et al. Randomized study of lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer. J Clin Oncol. 2010;28:1124-1130.
- Swain SM, Im YH, Im SA, et al. Safety profile of pertuzumab with trastuzumab and docetaxel in patients from Asia with human epidermal growth factor receptor 2-positive metastatic breast cancer: Results from the phase III trial CLEOPATRA. Oncologist. 2014;19:693-701.
- Gujral DM, Lloyd G, Bhattacharyya S. Effect of prophylactic betablocker or ACE inhibitor on cardiac dysfunction & heart failure during anthracycline chemotherapy ± trastuzumab. Breast. 2018;37:64-71.
- Gulati G, Heck SL, Ree AH, et al. Prevention of cardiac dysfunction during adjuvant breast cancer therapy (PRADA): a 2 × 2 factorial, randomized, placebo-controlled, double-blind clinical trial of candesartan and metoprolol. Eur Heart J. 2016;37:1671-1680.
- Seymour L, Bramwell V, Moran LA. Use of dexrazoxane as a cardioprotectant in patients receiving doxorubicin or epirubicin chemotherapy for the treatment of cancer. The Provincial Systemic Treatment Disease Site Group. Cancer Prev Control. 1999;3:145-159.
- Zinecard (dexrazoxane for injection) [prescribing information]. FDA Center for Drug Evaluation and Research. Updated May 2025. Accessed January 2, 2026. www.accessdata.fda.gov/drugsatfda_docs/label/2025/020212s021lbl.pdf
- Seferina SC, de Boer M, Derksen MW, et al.; Southeast Netherlands Breast Cancer Consortium. Cardiotoxicity and cardiac monitoring during adjuvant trastuzumab in daily Dutch practice: a study of the Southeast Netherlands Breast Cancer Consortium. Oncologist. 2016;21:555–562.
- Lyon AR, López-Fernández T, Couch LS, et al; ESC Scientific Document Group. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J. 2022;43:4229-4361. Erratum in: Eur Heart J. 2023;44:1621.30.
- Tripathy D, Seidman A, Keefe D, et al. Effect of cardiac dysfunction on treatment outcomes in women receiving trastuzumab for HER2-overexpressing metastatic breast cancer. Clin Breast Cancer. 2004;5:293-298.
- Telli ML, Witteles RM. Trastuzumab related cardiac dysfunction. J Natl Compr Canc Netw. 2011;9:243 249.