This section provides a summary of symptomatic conditions in oncology and their management. Readers are invited to submit summaries following the guidelines.
Connecting Science to Practice
The use of antibody–drug conjugates (ADCs) as part of the treatment of patients with cancer has increased in recent years. The ADCs tisotumab vedotin and mirvetuximab soravtansine are often used to treat gynecologic cancers, but these agents can result in ocular adverse events (AEs). The symptoms and pathophysiology of ocular AEs occurring with tisotumab vedotin and mirvetuximab soravtansine differ, however, and approaches to the prevention and treatment of these AEs also vary between these 2 agents. This symptom management overview provides essential information needed for pharmacists to manage ocular AEs in patients receiving ADCs for the treatment of gynecologic malignancies.
Symptom Overview
The use of antibody–drug conjugates (ADCs) within the treatment paradigms of patients with cancer has increased in recent years. Some ADCs cause a range of ocular adverse events (AEs), affecting the structure and/or function of the ophthalmic system. Not all ocular AEs of ADCs are the same; they can differ in their pathophysiology and symptomatology. In particular, ADCs with ocular AE risks gained a major role in the treatment of patients with gynecologic malignancies after the FDA’s approvals of tisotumab vedotin for the treatment of recurrent or metastatic cervical cancer after disease progression on or after receiving chemotherapy and mirvetuximab soravtansine for the treatment of folate receptor alpha (FRα)-expressing, platinum-resistant epithelial ovarian, fallopian tube, and primary peritoneal cancers after receiving 1 to 3 systemic treatment regimens.1,2
ADCs for the treatment of gynecologic malignancies will continue to expand considering the ADC drug development pipeline and the most recent data supporting the use of fam-trastuzumab deruxtecan across uterine, ovarian, and cervical tumors with HER2 amplification, among other solid tumors. Practitioners who manage patients with gynecologic cancers should be aware of the differences in ocular AE profiles, the mechanisms of ocular AEs, and the prevention, monitoring, and management strategies for these medications.
Etiology
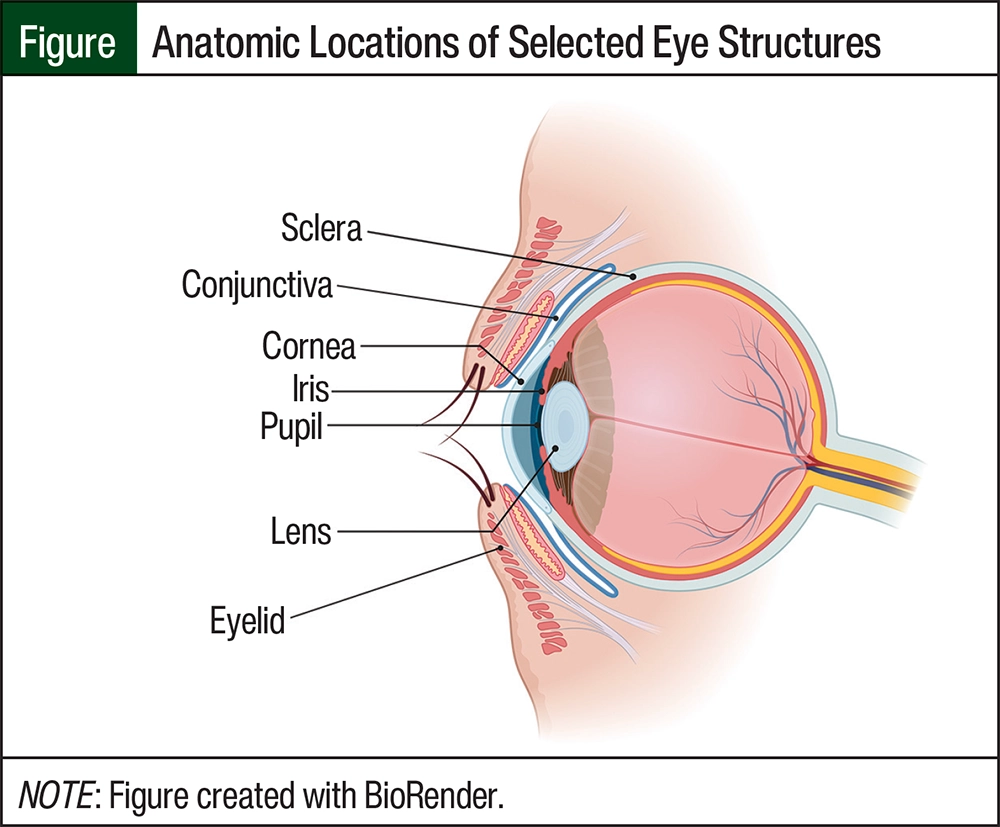
With tisotumab vedotin and mirvetuximab soravtansine, ocular AEs are primarily confined to the eye’s surface, which includes the conjunctiva, eyelids, and cornea (Figure).3,4 The conjunctiva is a thin, translucent, continuous tissue composed of squamous epithelium covering 2 parts of the eye: the palpebral or tarsal conjunctiva covers the inner surface of the eyelid and the bulbar conjunctiva covers the sclera.5 Goblet cells of the conjunctiva help provide the mucin layer for the tear film; therefore, the conjunctiva plays a large role in protecting the eye through lubrication. The cornea is a clear layer of avascular tissue covering the iris, pupil, and anterior chamber of the eye that ultimately provides a structural barrier. It is important to note that the cornea also refracts or focuses the entry of light into the eye.5
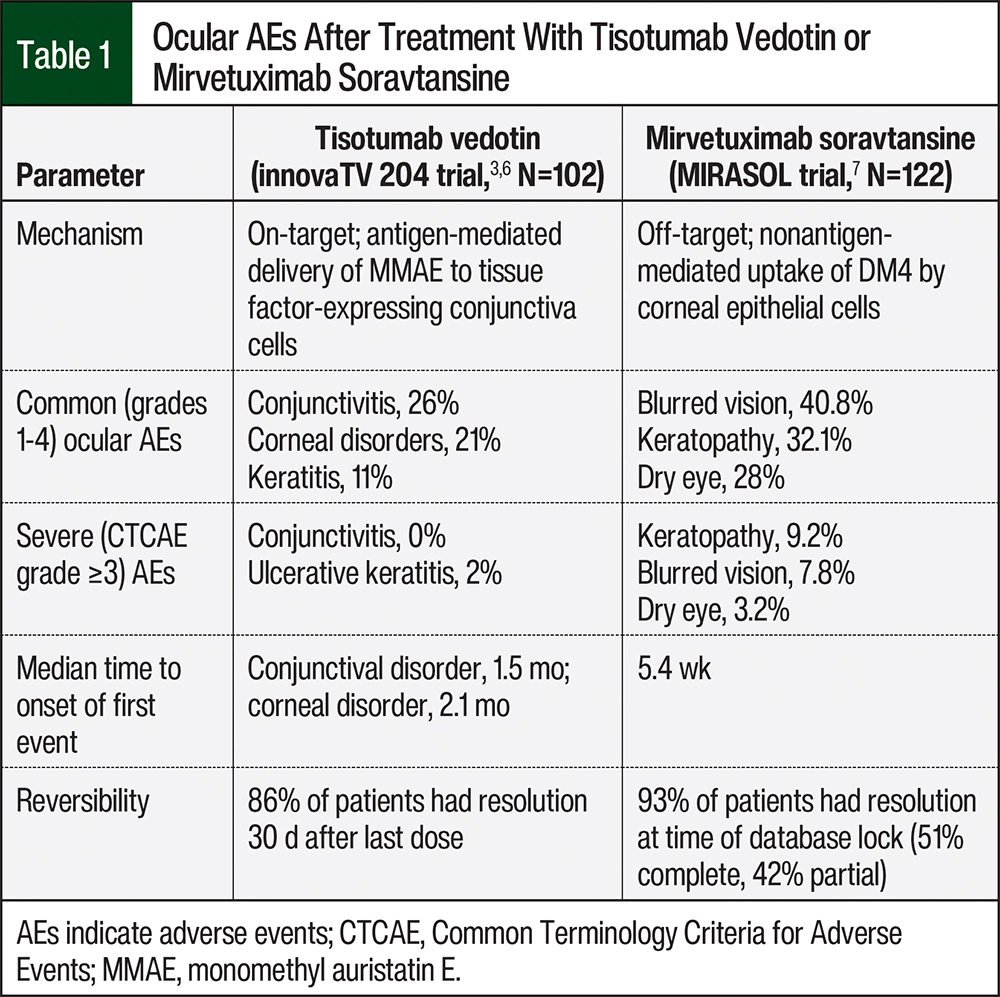
The mechanisms of ocular AEs that result from treatment with tisotumab vedotin or mirvetuximab soravtansine differ (Table 1).6,7 The mechanism of tisotumab vedotin–induced ocular AEs is antigen-mediated delivery of monomethyl auristatin E to the conjunctiva cells, which express tissue factor.8 Unlike tisotumab vedotin, the mechanism of ocular AEs from mirvetuximab soravtansine is linked to off-target uptake of the microtubule-disrupting payload, maytansinoid DM4.4 It is proposed that mirvetuximab soravtansine reaches the cornea via the vascularized limbal region, where DM4 accumulates and damages progenitor cells, resulting in the formation of microcysts.4
The ocular AEs that are frequently associated with tisotumab vedotin are conjunctivitis, dry eye, keratitis, and other periorbital and eyelid disorders.6,8 Conjunctival disorders often cause symptoms of dry eye, redness around the eyes, and irritation or pain, which may affect visual acuity.6,8 The most frequent ocular AEs with mirvetuximab soravtansine treatment include keratopathy, including, but not limited to, corneal microcysts or cysts, keratitis, and limbal stem-cell deficiency.4,6 With keratopathies, patients are likely to have blurred vision, dry eye, photophobia, and reduced visual acuity.9
Tisotumab Vedotin
Tisotumab vedotin is a tissue factor–directed monoclonal antibody that is linked to a monomethyl auristatin E antimicrotubule conjugate in a 4:1 drug-to-antibody ratio.10 A multicenter, open-label phase 2 study, innovaTV 204/GOG-3023/ENGOT-cx6, evaluated the efficacy and safety of tisotumab vedotin in the treatment of patients with recurrent or metastatic cervical cancer.6 The objective response rate (ORR) was 24%, with 7% of patients achieving a complete response, 17% a partial response, and 49% with stable disease. Results from the phase 3 innovaTV301/GOG-3057/ENGOT-cx12 trial showed significant improvements in overall survival (OS), progression-free survival (PFS), and ORR with tisotumab vedotin when compared with the investigator’s choice of single-agent chemotherapy, and a consistent safety profile.11 As of September 2024, the FDA has approved tisotumab vedotin for the treatment of recurrent or metastatic cervical cancer in patients with disease progression while or after receiving chemotherapy.12
In the innovaTV 204 study, 31% of patients had conjunctivitis, none of whom had grade 3 AEs, with a median time to onset of the first event at 1.4 months.6 The second most frequent (35%) ocular AE in the innovaTV204 study was dry eye, which typically includes foreign body sensation, ocular light sensitivity, and irritation.3 In all, 21% of patients had corneal disorders (keratitis in 11%), with a median time to onset of 2.1 months.3 A total of 16% of patients had periorbital and eyelid disorders, with a median time to onset of 1.7 months, which included blepharitis (inflammation of eyelids or eyelashes), entropion (lower eyelid and eyelashes turning inwards toward the eye), and meibomitis (inflammation of the meibomian glands in the eyelid).3 Blepharitis, entropion, and meibomitis were rare, occurring in 7%, 3%, and 3% of patients, respectively.3 Most (86%) of the ocular AEs had resolved 30 days after the last dose of tisotumab vedotin, with a median time to resolution of 0.7 months (interquartile range, 0.3-1.6 months).6
Mirvetuximab Soravtansine
Mirvetuximab soravtansine is an FRα-directed monoclonal antibody linked to the maytansinoid DM4 antimicrotubule conjugate in a 3.5:1 drug-to-antibody ratio.13 The phase 3 global, open-label, randomized, controlled MIRASOL trial evaluated the efficacy and safety of mirvetuximab soravtansine for the treatment of patients with platinum-resistant epithelial ovarian cancer with high FRα tumor expression (eg, ≥75% cells with ≥2+ staining intensity) by the VENTANA FOLR1 RxDx assay.7 The patients who received mirvetuximab soravtansine had a median PFS of 5.62 months (95% confidence interval [CI], 4.34-5.95) compared with 3.98 months (95% CI, 2.86-4.47) for patients who received the investigator’s choice single-agent chemotherapy (P<.001). The secondary end points of OS and ORR were also significantly improved with mirvetuximab soravtansine. The median OS was 16.46 months with mirvetuximab soravtansine versus 12.75 months with conventional chemotherapy (hazard ratio, 0.67; 95% CI, 0.50-0.89; P=.005). The ORR for mirvetuximab soravtansine was 42.3% versus 15.9% in the chemotherapy group, with 5.3% of patients achieving a complete response, 37% a partial response, and 37.9% stable disease with mirvetuximab soravtansine.7
In the MIRASOL trial, blurred vision was the most frequent (40.8%) ocular AE, and keratopathy was the second most common (32.1%) ocular AE; dry eye occurred in 28% of patients.14 The median time to the onset of ocular events was 5.4 weeks. Patients who received mirvetuximab soravtansine in the MIRASOL trial did not have corneal ulcers or corneal perforations, and the majority of ocular events were reversible. Of the patients who had ocular AEs, 93% had complete or partial resolution of ocular AEs at the time of the study data lock.14
The results from MIRASOL confirmed the results of the single-arm, phase 2 SORAYA trial, which led to the FDA-accelerated approval of mirvetuximab soravtansine.14 The SORAYA study followed the phase 3 randomized FORWARD I trial that evaluated the efficacy of mirvetuximab soravtansine versus single-agent chemotherapy.15 Although the FORWARD I study included and evaluated a population with medium (50%-74% expression) and high (≥75% expression) FRα expression, it did not show a significant difference in the primary end point of PFS.15 In March 2024, the FDA has granted full approval to mirvetuximab soravtansine for the treatment of patients with FRα-positive, platinum-resistant ovarian cancer.16
Fam-Trastuzumab Deruxtecan
Fam-trastuzumab deruxtecan is a HER2-directed ADC that is composed of a humanized immunoglobulin G1 monoclonal antibody, a cleavable tetrapeptide-based linker, and topoisomerase I inhibitor in an 8:1 ratio.17,18 Compared with tisotumab vedotin and mirvetuximab soravtansine, the occurrence of ocular AEs is minimal.17 DESTINY-PanTumor02 is a phase 2 study that evaluated fam-trastuzumab deruxtecan monotherapy across patients with one of multiple tumor types, including endometrial, cervical, ovarian, and vulvar cancers, with 2+ or 3+ HER2 expression by immunohistochemistry after receiving at least 1 line of therapy or without treatment alternatives. The ORR for all tumor types was 37.1%, and the ORRs were 57.5%, 50%, and 45% for endometrial, cervical, and ovarian cancers, respectively.17
In the DESTINY-PanTumor02 study, as well as in the DESTINY-Breast04, there were no ocular AEs.17,19 The prescribing information for fam-trastuzumab deruxtecan notes that the rate of ophthalmic AEs in the form of dry eye syndrome in 11% of patients.20 Another ADC for the treatment of patients with breast cancer that targets HER2 as well as DM1, ado-trastuzumab emtansine, displays similar ocular AEs to mirvetuximab soravtansine, again supporting the proposed mechanism of AEs being related to the microtubule-disrupting payload stated above.21 As the data continue to mature with the use of fam-trastuzumab deruxtecan, reports of ocular AEs are likely to be limited but should be closely monitored and reported in clinical trials and phase 4 monitoring.
Treatment Options
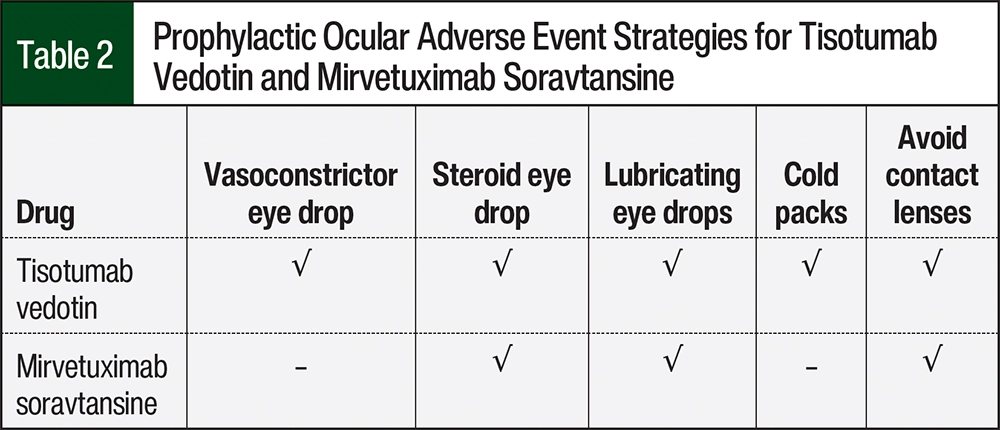
Given the rates of ocular AEs and potential effect on quality of life in patients with recurrent cancer, the clinical care of patients receiving tisotumab vedotin or mirvetuximab soravtansine requires specific and diligent pharmacologic and nonpharmacologic prevention and mitigation strategies (Table 2).
Patients receiving tisotumab vedotin or mirvetuximab soravtansine require preemptive monitoring by an eyecare provider (eg, ophthalmologist, optometrist, other eyecare specialist) at routine intervals, as determined within the prescribing information, with slit lamp and visual acuity examinations.1,2 If AEs should occur, access to additional eyecare provider visits is imperative. Patients who are receiving tisotumab vedotin should have a baseline examination from an eyecare provider before the first 9 treatment cycles with tisotumab vedotin, after which all eye examinations can be performed as clinically indicated.1 Patients receiving mirvetuximab soravtansine should receive a baseline eye examination, examinations before every other cycle for the first 8 cycles, and then examinations as needed.2 The manufacturers of tisotumab vedotin and mirvetuximab soravtansine provide eyecare forms that clinicians can use to coordinate monitoring and perform clinical decision-making.
Prophylactic strategies for the prevention of ocular AEs with tisotumab vedotin and mirvetuximab soravtansine treatment have been shown to successfully reduce the rates of eye AEs, as shown in phase 1 clinical trials.22,23 These strategies, as outlined below, were subsequently implemented in the aforementioned phase 2 clinical trials.
Tisotumab vedotin currently requires the administration of a steroid eye drop (eg, dexamethasone) followed by a vasoconstrictor eye drop (eg, brimonidine, tetryzoline) approximately 10 minutes before the tisotumab vedotin infusion.1,24 The coordination of infusion pharmacy supply or a prescription pick-up before the infusion is necessary to achieve this. Prescriptions for the necessary eye drops can be managed by the oncology team or by the eyecare provider. Patients should continue using the steroid eye drop 3 times daily, or as prescribed, for 72 hours after the infusion.1,24 In addition, patients should wear a cold pack over both eyes and the bridge of their nose starting 10 minutes before the infusion, for the duration of the 30-minute infusion, and for 20 minutes after the tisotumab vedotin infusion.1,24 Applying cold packs to these areas may reduce blood flow, and therefore drug delivery, and aid in the prevention of ocular AEs and epistaxis. Data from the phase 1/2 innovaTV 201 study show that the implementation of these measures reduced ocular event rates from 80% (n=15) to 60% (n=40), and the rates of conjunctivitis from 80% (n=12 of 15 patients) to 28% (n=11 of 40 patients).22
Mirvetuximab soravtansine currently requires the administration of a steroid eye drop (eg, prednisolone) starting the day before the infusion and continuing through day 8 of each 21-day cycle.2 The dosing of the steroid eye drop is 6 times daily starting the day before the infusion and continuing through day 4, then 4 times daily through day 8. Patients should wait at least 10 minutes after instilling steroid eye drops before the use of lubricating eye drops to avoid drug clearance. It is recommended that patients use lubricating eye drops at least 4 times daily and as needed. No vasoconstrictor eye drops or cold packs are recommended at this time with mirvetuximab.2
In addition, mirvetuximab soravtansine should be dosed based on the formula of adjusted ideal body weight=ideal body weight in kg+0.4×(actual weight in kg–ideal body weight).2 In the dose-escalation phase of a phase 1 trial of treatment with mirvetuximab soravtansine in patients with solid tumors, mirvetuximab soravtansine was initially based on actual body weight.25 This dosing strategy yielded initial plasma levels associated with a high rate of ocular AEs. Therefore, the dosing strategy was changed to reduce the variability in peak exposure levels and, more important, to reduce ocular AEs to acceptable rates.25 In the expansion-cohort phase of this trial, the daily use of lubricating eye drops with the nonpharmacologic measures outlined below reduced the rate and severity of ocular AEs.23 Prophylactic steroid eye drops reduced the number of dose reductions for mirvetuximab, despite no significant differences in blurred vision or keratopathy.23
Notably, the currently enrolling phase 3 GLORIOSA trial that is evaluating mirvetuximab soravtansine with or without bevacizumab as maintenance therapy for platinum-sensitive ovarian cancer no longer requires patients to receive primary prophylaxis with steroid eye drops.26 Dose reductions of mirvetuximab soravtansine in compiled analyses of the SORAYA and FORWARD I trials were similar to those of the phase 1 trial, which used lubricating eye drops alone.14,15,23 This suggestion of minimal benefit with steroid eye drops, combined with the risk for cataracts and intraocular pressure, prompted this change in eyecare plan.26
Patients who are receiving tisotumab vedotin or mirvetuximab soravtansine should use lubricating eye drops (eg, propylene glycol, carboxymethylcellulose) for the duration of therapy until approximately 1 month after therapy has been discontinued.1,2 Preservative-free formulations of lubricating eye drops may be less irritating. Patients should be counseled on good eye hygiene, including gentle cleansing of the eyelid margins (ie, the junctures of the conjunctiva and skin) while receiving therapy, which generally includes using a clean, soft cloth to gently wipe the eyelids from the inner corner of the eye toward the outer corner. All patients should avoid wearing contact lenses and the use of products that may induce inflammation in or around the eyes (eg, irritating eye makeup) for the duration of therapy.1,2
Patients should be counseled on the importance of reporting any new or worsening ocular symptoms to their oncology team and eyecare provider. When symptoms arise, swift consultation and communication with an eyecare provider are necessary for the evaluation, diagnosis, and management of ocular AEs. Strategies for the management of ocular AEs may include antibiotic eye drops in the case of conjunctivitis and adjustment of the dosage and frequency of steroid eye drops. Notably, increasing steroid eye drop use may require more extensive monitoring for cataracts and increased intraocular pressure.3,27
The oncology clinician should be particularly aware of the link between eyecare provider assessment and the grading of AEs and subsequent management of chemotherapy. The prescribing information for each drug provides detailed recommendations for holding therapy and dose reductions depending on the type and grade of ocular AEs (Table 3).1,2 In general, more severe AEs require dose interruption or permanent discontinuation of treatment. Notably, the FDA does not currently require a Risk Evaluation and Mitigation Strategies program for tisotumab vedotin or mirvetuximab soravtansine.
The ocular AE rates in this review reflect those of patients in clinical trial settings who by nature of participation in a trial may have access to resources and higher adherence. Cervical cancer, in particular, is a cancer that disproportionately affects underserved populations.28 Therefore, multiple barriers, such as lower access to time or transportation to attend additional monitoring appointments and low health literacy, among others, may affect adherence to eyecare plans and, ultimately, outcomes. Especially given that social determinants of health, including geographic proximity to eyecare providers, dictate access to eyecare in the United States, the management of ocular AEs requires healthcare team resources to coordinate, communicate, and overcome barriers.29 Certainly, given these barriers in clinical practice, oncology pharmacists serve an important role in care coordination and monitoring, and are capable of adeptly meeting the clinical and logistic needs for the safe use of tisotumab vedotin and mirvetuximab soravtansine.
Conclusion
Ocular AEs are frequent AEs related to the use of currently FDA-approved ADCs for the treatment of patients with gynecologic malignancies. The majority of ocular events in clinical trials were low grade and reversible, and predominantly affected the ocular surface (eg, conjunctiva and cornea). However, some symptoms of ocular AEs in a minority of patients (eg, blurred vision, cataract, keratopathy with mirvetuximab soravtansine) were not fully reversed in clinical trials even with the use of supportive measures.
Given the importance of adherence to supportive measures and the ability of patients and their team to recognize symptoms to optimize outcomes, oncology pharmacists are pivotal to ensure proper education and the monitoring of supportive therapy. Oncology pharmacists can also play a large role in maintaining ADC dose intensity by optimizing supportive care or facilitating appropriate dose reductions. Oncology clinicians caring for patients receiving tisotumab vedotin or mirvetuximab soravtansine should be aware of evidence-based measures for the prevention of ocular AEs, the importance of patient education, communication with eyecare providers, and the management of therapy if AEs occur.
Author Disclosure Statement
Dr Hayward is on the Advisory Board at Merck, Verastem, and ImmunoGen; Dr MacDonald is on the Advisory Board at Mersana; Dr Zacholski is an Advisory Group Participant at UroGen and Speaker, Internal Medical Affairs, at Seagen; Dr Pragides and Ms Johnson have no conflicts of interest to report.
References
- Tivdak (tisotumab vedotin-tftv) injection, for intravenous use [prescribing information]. Seagen; April 2024. Accessed August 2, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/761208s007lbl.pdf
- Elahere (mirvetuximab soravtansine-gynx) injection, for intravenous use [prescribing information]. ImmunoGen; March 2024. Accessed August 2, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/761310Origs005lbl.pdf
- Kim SK, Ursell P, Coleman RL, et al. Mitigation and management strategies for ocular events associated with tisotumab vedotin. Gynecol Oncol. 2022;165:385-392.
- Matulonis UA, Birrer MJ, O’Malley DM, et al. Evaluation of prophylactic corticosteroid eye drop use in the management of corneal abnormalities induced by the antibody–drug conjugate mirvetuximab soravtansine. Clin Cancer Res. 2019;25:1727-1736.
- Sridhar MS. Anatomy of cornea and ocular surface. Indian J Ophthalmol. 2018;66:190-194.
- Coleman RL, Lorusso D, Gennigens C, et al. Efficacy and safety of tisotumab vedotin in previously treated recurrent or metastatic cervical cancer (innovaTV 204/GOG-3023/ENGOT-cx6): a multicentre, open-label, single-arm, phase 2 study. Lancet Oncol. 2021;22:609-619.
- Moore KN, Angelergues A, Konecny GE, et al. Mirvetuximab soravtansine in FRα-positive, platinum-resistant ovarian cancer. N Engl J Med. 2023;389:2162-2174.
- Vergote I, Kim S, Ursell P, et al. EP064/#192 Potential mechanism for ocular adverse events observed with tisotumab vedotin. Int J Gynecol Cancer. 2022;32(suppl 3):A74-A75.
- Lakhundi S, Siddiqui R, Khan NA. Pathogenesis of microbial keratitis. Microb Pathog. 2017;104:97-109.
- Martin J, Zacholski E, O’Cearbhaill R, Matulonis U, Chen L. Society of Gynecologic Oncology Journal Club: controversial conversations in gynecologic cancer – The ABCs of ADCs (Antibody drug Conjugates). Gynecol Oncol Rep. 2023;45:101141.
- Vergote I, González-Martín A, Fujiwara K, et al. Tisotumab vedotin as second- or third-line therapy for recurrent cervical cancer. N Engl J Med. 2024;391:44-55.
- US Food and Drug Administration. FDA approves tisotumab vedotin-tftv for recurrent or metastatic cervical cancer. April 29, 2024. Accessed August 7, 2024. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-tisotumab-vedotin-tftv-recurrent-or-metastatic-cervical-cancer#:~:text=On%20April%2029%2C%202024%2C%20the,progression%20on%20or%20after%20chemotherapy
- Bogani G, Coleman RL, Vergote I, et al. Mirvetuximab soravtansine-gynx: first antibody/antigen-drug conjugate (ADC) in advanced or recurrent ovarian cancer. Int J Gynecol Cancer. 2024;34:469-477.
- Matulonis UA, Lorusso D, Oaknin A, et al. Efficacy and safety of mirvetuximab soravtansine in patients with platinum-resistant ovarian cancer with high folate receptor alpha expression: results from the SORAYA study. J Clin Oncol. 2023;41:2436-2445.
- Moore KN, Oza AM, Colombo N, et al. Phase III, randomized trial of mirvetuximab soravtansine versus chemotherapy in patients with platinum-resistant ovarian cancer: primary analysis of FORWARD I. Ann Oncol. 2021;32:757-765.
- US Food and Drug Administration. FDA approves mirvetuximab soravtansine-gynx for FRα positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer. Accessed August 7, 2024. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-mirvetuximab-soravtansine-gynx-fra-positive-platinum-resistant-epithelial-ovarian
- Meric-Bernstam F, Makker V, Oaknin A, et al. Efficacy and safety of trastuzumab deruxtecan in patients with HER2-expressing solid tumors: primary results from the DESTINY-PanTumor02 phase II trial. J Clin Oncol. 2024;42:47-58.
- Linehan AS, Fitzpatrick OM, Morris PG. Profile of trastuzumab deruxtecan in the management of patients with HER2-positive unresectable or metastatic breast cancer: an evidence-based review. Breast Cancer (Dove Med Press). 2021;13:151-159.
- Modi S, Jacot W, Yamashita T, et al. Trastuzumab deruxtecan in previously treated HER2-low advanced breast cancer. N Engl J Med. 2022;387:9-20.
- Enhertu (fam-trastuzumab deruxtecan-nxki) injection, for intravenous use [prescribing information]. Daiichi Sankyo; April 2024. Accessed August 7, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/761139s028lbl.pdf
- Eaton JS, Miller PE, Mannis MJ, Murphy CJ. Ocular adverse events associated with antibody–drug conjugates in human clinical trials. J Ocul Pharmacol Ther. 2015;31:589-604.
- Hong DS, Concin N, Vergote I, et al. Tisotumab vedotin in previously treated recurrent or metastatic cervical cancer. Clin Cancer Res. 2020;26:1220-1228.
- Moore KN, Martin LP, O’Malley DM, et al. Safety and activity of mirvetuximab soravtansine (IMGN853), a folate receptor alpha-targeting antibody-drug conjugate, in platinum-resistant ovarian, fallopian tube, or primary peritoneal cancer: a phase I expansion study. J Clin Oncol. 2017;35:1112-1118.
- Seagen. Prepping for Tivdak premedication and required eye care. 2022. Accessed August 3, 2023. www.tivdakhcp.com/eye-care/
- Moore KN, Borghaei H, O’Malley DM, et al. Phase 1 dose-escalation study of mirvetuximab soravtansine (IMGN853), a folate receptor α-targeting antibody-drug conjugate, in patients with solid tumors. Cancer. 2017;123:3080-3087.
- O’Malley DM, Myers TKN, Zamagni C, et al. GLORIOSA: a randomized, open-label, phase 3 study of mirvetuximab soravtansine with bevacizumab vs. bevacizumab as maintenance in platinum-sensitive ovarian, fallopian tube, or primary peritoneal cancer. J Clin Oncol. 2023;41(16 suppl):TPS5622.
- Hendershot A, Slabaugh M, Riaz KM, et al. Strategies for prevention and management of ocular events occurring with mirvetuximab soravtansine. Gynecol Oncol Rep. 2023;47:101155.
- Perkins R, Mitchell E. Cervical cancer disparities. J Natl Med Assoc. 2023;115:S19-S25.
- Solomon SD, Shoge RY, Ervin AM, et al. Improving access to eye care: a systematic review of the literature. Ophthalmology. 2022;129:e114-e126.