Connecting Science to Practice
This article reviews clinical evidence on the use of azacitidine
in treating patients with peripheral T-cell lymphomas,
especially nodal T-follicular helper lymphomas (nTFHL).
The phase 3 ORACLE trial showed that azacitidine has
promising single-agent activity in patients with relapsed or
refractory disease. Phase 2 studies explored combining azacitidine
with other epigenetic drugs, such as histone deacetylase
inhibitors, to improve outcomes. In addition, preclinical
research suggests azacitidine may help make tumors
more responsive to chemotherapy by altering the tumor
environment. These findings support further investigation
into azacitidine’s role in frontline therapy and combination
regimens. Oral azacitidine may offer a new treatment option
for nTFHL, especially in patients who are not candidates
for intensive chemotherapy.
Peripheral T-cell lymphomas (PTCLs) are rare and heterogenous malignancies, accounting for approximately 5% to 10% of all non-Hodgkin lymphoma cases.1 Included in the 2022 World Health Organization (WHO) classification of lymphoid malignancies, there are more than 30 types of PTCLs, which are further broken down by subtype.2 Nodal T-follicular helper cell lymphomas (nTFHLs) were first introduced in the 2016 WHO classification, and, in 2022, this entity was updated to include 3 previously recognized diseases that are now classified as nTFHL subtypes. Angioimmunoblastic T-cell lymphoma (AITL) is now nTFHL, angioimmunoblastic type; follicular T-cell lymphoma is now nTFHL, follicular type; and nodal PTCL with TFH phenotype is now nTFHL, not otherwise specified.2
This reclassification was supported by the loss-of-function genes responsible for regulating DNA and histone methylation that are frequently shared by nTFHLs. The genes, such as TET2, IDH2, and DNMT3A, ultimately result in epigenetic alterations, supporting the use of epigenetic-modifying agents in the treatment of nTFHL.2,3 The current recommendations for the frontline treatment of nTFHL include multiagent cytotoxic chemotherapy; epigenetic modifying agents, such as belinostat and romidepsin, may be considered as first-line treatment in patients pursuing palliative intent therapy.4
Epigenetic Modification in PTCL
Histone deacetylase inhibition has been a mechanism in the treatment of PTCLs since the FDA approvals of romidepsin and belinostat more than a decade ago. Romidepsin has an approximately 38% overall response rate (ORR) as a single agent in the setting of relapsed or refractory PTCL.5 In a phase 3 trial, treatment with romidepsin plus cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) chemotherapy was evaluated for the frontline treatment of PTCL.6 The combination of romidepsin plus CHOP did not show superior progression-free survival (PFS) versus CHOP alone (12 months vs 10.2 months, respectively) and there was an increase in adverse events (AEs), with 93.8% of patients in the romidepsin plus CHOP arm having grade ≥3 AEs compared with 69.7% in the group that received CHOP alone. However, in an exploratory analysis of patients with nTFHL, PFS was significantly prolonged in the romidepsin plus CHOP group at 19.5 months versus 10.6 months with CHOP alone.7 Therefore, there may be a potential benefit to the use of epigenetic modification in the frontline setting for patients with nTFHL.
The efficacy of the hypomethylating agents (HMAs) azacitidine and decitabine in patients with myeloid malignancies portends their use in other malignancies characterized by TET2, DNMT3A, and IDH2 mutations, such as nTFHL. In a retrospective case series, 12 patients with AITL received subcutaneous azacitidine for 7 days for the treatment of a concomitant myeloid malignancy or as compassionate therapy for relapsed or refractory AITL.8 In all, 9 patients responded to treatment with azacitidine, with a complete response (CR) rate of 50% and a partial response rate of 25%. Of the responding patients, 5 had a sustained response and still had a CR almost 3 years from initiating treatment with azacitidine.8
Oral azacitidine has now been incorporated into clinical practice guidelines for PTCL in the relapsed or refractory setting.4 Most of the evidence supporting HMAs is in the setting of patients with PTCL who receive oral azacitidine, which will be the focus of this review. At this time, there is limited evidence regarding decitabine treatment for PTCL to support the use of HMAs as a class in this setting9; however, this may change as additional studies are conducted.
Pharmacokinetics and Pharmacodynamics of Oral Azacitidine
Oral azacitidine is nonbioequivalent to the subcutaneous and intravenous formulations of azacitidine, as is evidenced by a mean bioavailability of 6.3% to 20% and decreased DNA methylation when received on the same 7-day schedule.10 Because the mechanism of azacitidine is dependent on DNA incorporation during S phase, it was theorized that efficacy would be improved by administering oral azacitidine over a longer period; this is further supported by the fact that methylation levels returned to baseline at the end of a 28-day cycle with 7-day dosing.10 Extended dosing of azacitidine was further evaluated with 14- and 21-day cohorts at a lower dose than administered in the 7-day study (300 mg vs 480 mg).10,11 DNA methylation was significantly reduced at all points of the cycle for the 14- and 21-day cohorts, with methylation being more substantially reduced at cycle end with 21-day dosing compared with 14-day dosing.11 Safety outcomes were not reported with the extended-dosing evaluation11; however, in the 7-day dosing study, the most frequently reported AEs were gastrointestinal disorders, headache, fatigue, and peripheral edema.10 In clinical practice, antiemetic premedication is routinely given with the first 2 cycles of oral azacitidine.
Treatment with HMAs results in the re-expression of tumor suppressor genes that have been aberrantly repressed through methylation.12 HMAs have improved tumor infiltration and killing ability of T cells via increased expression of effector cytokines TNF and IFN-γ.13 The result of HMAs on the tumor microenvironment beyond their direct anticancer effect suggests that combination treatment or priming with HMA therapy could result in chemosensitization and improved response. Oral azacitidine priming before romidepsin plus CHOP in patients with diffuse large B-cell lymphoma has clinical efficacy, with an ORR rate of 94.9% and an acceptable safety profile according to the results of a phase 1 study.14 Methylation studies in this population showed increased expression of immune-related genes, providing further evidence of the impact of HMAs on the tumor microenvironment.14
Clinical Efficacy of Oral Azacitidine in PTCL
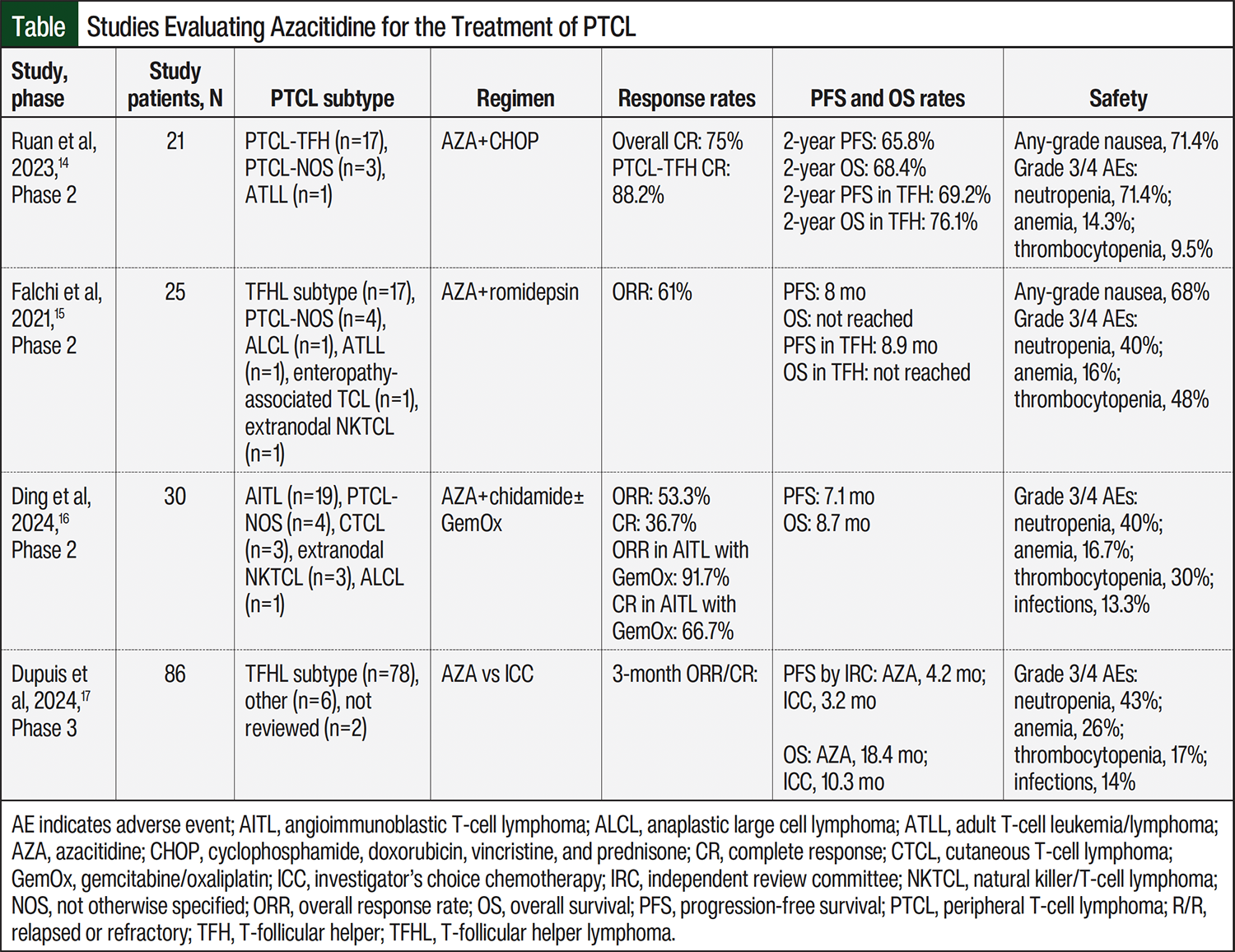
The Table lists several studies that evaluated the use of azacitidine for the treatment of PTCL.14-17 A multicenter, open-label, single-arm, phase 2 study evaluated the efficacy and safety of oral azacitidine plus standard CHOP in patients with treatment-naïve nTFHL, which included PTCL-TFH, PTCL-not otherwise specified (NOS), and T-cell leukemia or lymphoma in adults.15 Azacitidine was initiated as a priming dose of 300 mg orally per day for 7 days before cycle 1, day 1 of CHOP. Oral azacitidine 300 mg daily was then administered on days 8 to 21 during CHOP cycles 1 to 5. The primary efficacy end point was CR at the end of treatment. The secondary end points included PFS and overall survival (OS).15
A total of 21 patients were enrolled in the study, of whom 1 patient withdrew consent before the completion of cycle 1 as a result of compliance issues.15 Therefore, only 20 patients were evaluable for the study. Most patients had PTCL-TFH subtype (n=17; 81%). The CR rate was 75%, and all responses in the study were CRs at the end of the treatment. The estimated 2-year PFS and OS rates were 65.8% and 68.4%, respectively. Among the patients with PTCL-TFH, the CR rate was 88.2%; the estimated 2-year PFS and OS rates were 69.2% and 76.1%, respectively. A total of 10 patients received a consolidative stem-cell transplant after the completion of therapy; however, this did not impact PFS or OS (P=.56 and P=.75, respectively). The researchers concluded that azacitidine added to CHOP is an active regimen that leads to a high CR rate, and warrants evaluation in a larger, randomized trial.15
A multicenter, open-label, single-arm, phase 2 trial evaluated the combination of oral azacitidine and romidepsin in patients with treatment-naive or relapsed or refractory PTCL.16 Treatment was delivered in 35-day cycles and consisted of once-daily oral azacitidine 300 mg on days 1 to 14 and intravenous romidepsin 14 mg/m2 on days 8, 15, and 22. The treatment was continued until disease progression or unacceptable AEs. The primary end point was investigator-assessed ORR. The secondary end points included PFS, OS, and duration of response (DOR).
A total of 25 patients were enrolled in the trial; 11 (44%) patients were treatment-naïve and 14 (56%) had relapsed or refractory disease. The patients with relapsed or refractory PTCL received a median of 2 previous lines of therapy (range, 1-6 lines). The most common PTCL subtypes included AITL (n=14; 56%), PTCL-NOS (n=4; 16%), and PTCL-TFH (n=3; 12%). The ORR and CR rates were 61% and 43%, respectively. Among the patients with PTCL of TFH origin (AITL and PTCL-TFH), higher ORR and CR rates of 80% and 60%, respectively, were observed. The median PFS was numerically higher among patients with AITL or PTCL-TFH compared with other PTCL subtypes (8.9 months vs 2.3 months, respectively; hazard ratio (HR), 0.3; 95% confidence interval (CI), 0.09-1.06; P=.05).
After a median follow-up of 13.5 months, the median OS was not reached for patients with PTCL of TFH origin compared with 9.4 months in patients with other PTCL subtypes (HR, 0.2; 95% CI, 0.03-1; P=.03). The responses were durable, with a median DOR of 20.3 months. The researchers concluded that the combination of azacitidine and romidepsin is a highly active treatment in PTCL, particularly in those patients with a TFH phenotype, such as AITL or PTCL-TFH.
A multicenter, open-label, single-arm, phase 2 trial conducted in China evaluated the combination of azacitidine and chidamide with or without gemcitabine and oxaliplatin (GemOx) for the treatment of relapsed or refractory PTCL in patients who received at least 1 previous line of therapy.17 Chidamide is an oral histone deacetylase inhibitor approved for the treatment of PTCL in China and is prescribed as a dose of 20 mg orally twice weekly. Azacitidine was administered subcutaneously at a dose of 100 mg daily on days 1 through 7 of a 21-day cycle. Gemcitabine 1000 mg/m2 and oxaliplatin 100 mg/m2 were received on day 7. The patients received up to 8 cycles of induction with azacitidine plus chidamide with or without GemOx, followed by azacitidine and chidamide maintenance at the same dosing and schedule as above until disease progression or unacceptable AEs.17
A total of 30 patients were enrolled in the trial.17 The most common PTCL subtypes included AITL (n=19; 63.3%) and PTCL-NOS (n=4; 13.3%). In all, 12 (40%) patients had received ≥2 previous lines of therapy. Induction with concomitant GemOx was administered to 16 patients. Among the entire cohort, the ORR was 53.3% and the CR rate was 36.7%. The response rates were higher among patients with AITL than in those with non-AITL subtypes (with GemOx, 91.7% vs 50%, respectively; without GemOx, 42.9% vs 0%, respectively). The median PFS and OS for the entire cohort were 7.1 months and 8.7 months, respectively. In the patients with AITL who received GemOx-containing induction therapy, their median PFS and OS were markedly higher than the general cohort, at 17.2 months and 38.8 months, respectively. The researchers concluded that dual epigenetic therapy with azacitidine and a histone deacetylase inhibitor with or without chemotherapy induced high response rates in patients with relapsed or refractory AITL.17
The international, open-label, randomized phase 3 ORACLE trial compared azacitidine with the investigator’s choice chemotherapy in patients with relapsed or refractory TFHL.18 Azacitidine was administered once daily at a dose of 300 mg orally on days 1 through 14 of a 28-day cycle and was continued until disease progression or unacceptable AEs. The investigator’s choice chemotherapy included gemcitabine or bendamustine for up to 6 cycles, or romidepsin until disease progression or AEs. The patients had relapsed or refractory disease after receiving at least 1 previous line of therapy. The primary efficacy end point was PFS, and the key secondary end points included OS, ORRs, and DOR.
A total of 86 patients were randomized to receive either azacitidine (n=42) or investigator’s choice (n=44). Most patients had received at least 1 or 2 previous lines of therapy (n=71; 82.5%). The primary end point of median PFS was 5.6 months with azacitidine compared with 2.8 months in the control arm (HR, 0.63; 95% CI, 0.38-1.07; 2-sided P value=.084). Although there was not a statistically significant PFS advantage, there was an improvement in median OS with azacitidine compared with the investigator’s choice of therapy (18.4 months vs 10.3 months, respectively; HR, 0.56; 95% CI, 0.32-0.96). The ORR at 6 months was 31% in the azacitidine arm compared with 23% in the control group. Among the responders, the DOR was longer in the azacitidine arm than in the investigator’s choice of treatment arm (10.4 months vs 3.4 months, respectively; HR, 0.73; 95% CI, 0.3-1.7). The researchers concluded that although the study’s primary end point was not met, azacitidine had a favorable safety profile and promising overall survival and could add to the available treatment options for relapsed or refractory TFHL.18
In the phase 2 trial evaluating azacitidine plus CHOP in PTCL, the most common grade 3/4 AEs were hematologic, including neutropenia (71%), anemia (14%), and thrombocytopenia (10%).15 More important, the addition of azacitidine in this study did not seem to impact CHOP dose intensity, because all patients received standard CHOP without any dose modifications. During the trial, 3 patients’ azacitidine dose was lowered; however, none of the patients discontinued any study treatment as a result of an AE.15
In the phase 3 ORACLE trial, there was a lower rate of grade 3 or 4 AEs with azacitidine than the investigator’s choice of therapy (76% vs 98%, respectively).18 The most common grade 3 or 4 AEs with azacitidine were neutropenia (43%), anemia (26%), thrombocytopenia (17%), and infections (14%). Treatment discontinuation as a result of an AE was also lower in the azacitidine arm than in the control arm (7% vs 21%, respectively).18
Similar to the aforementioned trials, the safety events with azacitidine-containing combinations in studies by Ding and colleagues and Falchi and colleagues in patients with PTCL consisted primarily of hematologic AEs, including neutropenia, anemia, and thrombocytopenia.16,17 One patient receiving azacitidine plus romidepsin discontinued the study treatment as a result of reactivation of the Epstein–Barr virus.16 In general, the incorporation of azacitidine into various treatment paradigms in PTCL has a safety profile expected of azacitidine and does not significantly impact the dose intensity of concomitantly administered multiagent chemotherapy.
Discussion
With the 2022 reclassification of nTFHL and subsequent subtypes, there are new opportunities for the development of treatment regimens that align pharmacologically with disease biology. Drug development in PTCL has been challenging, particularly because of the heterogeneity of PTCL, which is in large part because of the inherent heterogeneity of T-lymphocytes. It has long been purported that targeting the underlying epigenetic dysregulation in PTCLs is a path forward in treatment and improving outcomes.19 This has led to the development of histone deacetylase inhibitors, such as belinostat and romidepsin, and investigations into targeting other molecular driver pathways, such as JAK/STAT and PI3K.
Improvements in the understanding of the biology and reclassifications of PTCL will hopefully lead to improvements in drug development and clinical trial design to select the patient populations that are likely to benefit. An example of this is the randomized phase 3 Ro-CHOP trial that evaluated romidepsin plus CHOP versus CHOP in patients with previously untreated PTCL.6,7 Although the primary survival end point was not improved with the addition of romidepsin in the general patient population, the results of a subgroup analysis of patients with TFH lymphomas showed that there was an improvement in median PFS with romidepsin plus CHOP.6,7 Unfortunately, the results of the trial of romidepsin plus CHOP trial in not meeting its primary end point have led to the withdrawal of the PTCL indication for romidepsin.20 By having a better understanding of PTCL disease biology, clinical trial designs that enrich specific subtypes may lead to improvements in drug development.
ORACLE was the first phase 3 trial to focus on investigating the treatment of nTFHL phenotypes.18 Although this trial did not meet its primary end point of improving PFS compared with other available standard-of-care therapies, oral azacitidine was not worse than other established therapies, demonstrated antilymphoma activity, and had a lower rate of grade 3 or 4 AEs compared with the control treatment. There were also some interesting potential biologic insights realized in this study. First, the usual means of response evaluation criteria may not be adequate for azacitidine treatment in patients with PTCL. This was suggested by the fact that patients with stable disease after receiving 3 cycles of azacitidine had a median PFS of almost 9 months, which was significantly longer than expected among patients with relapsed or refractory PTCL.18 This is similar in nature to that seen with azacitidine in the setting of myeloid malignancies when the drug was first being developed, by which the response criteria needed to be adjusted based on its clinical activity.18 Second, although PFS was not significantly improved, the median OS was improved, which may be indicative of azacitidine sensitizing lymphoma cells through epigenetic modification to the next line of chemotherapy.18
The ALLIANCE 051902 randomized trial is currently evaluating azacitidine plus CHO(E)P (CHOP plus etoposide), duvelisib plus CHO(E)P, and CHO(E)P in patients with CD30-negative PTCL.21 This study includes several PTCL subtypes, including TFH-PTCL, follicular T-cell lymphoma, PTCL-NOS, AITL, enteropathy-associated T-cell lymphoma, and monomorphic epitheliotropic intestinal T-cell lymphoma.21 More important, this trial will stratify patients by their TFHL phenotype.21 This study will hopefully add to the promising results observed in the study by Ruan and colleagues of azacitidine added to CHOP for treatment-naïve patients with PTCL.15
If oral azacitidine becomes a therapy for treatment-naïve PTCL and is delivered as a priming cycle before multiagent chemotherapy in the future, this may lead to challenges in practice. PTCL is often an aggressive form of lymphoma that may necessitate emergency initiation of treatment. This could be a possible barrier in the real-world setting, where it may be difficult to wait to complete azacitidine priming before starting multiagent chemotherapy, as well as barriers posed by payers and drug access to starting treatment with azacitidine in a prompt and timely manner. The results of a real-world study that examined treatment with subcutaneous azacitidine and romidepsin showed that the ORR and CR rates were comparable with patients in a phase 2 trial of patients who received oral azacitidine plus romidepsin.16,22,23
Conclusion
Drug development for T-cell lymphomas continues to be a challenging therapeutic space because of the rarity of the disease and the biologic heterogeneity of PTCLs. With the 2022 reclassification of nTFHL, there may be potential for therapeutic approaches that focus on this PTCL phenotype and leverage epigenetic modification. Oral azacitidine has now been incorporated into clinical practice guidelines for PTCL in the relapsed or refractory setting. Although there are some promising small phase 2 trials that are evaluating azacitidine in unique treatment combinations, further investigation is warranted to evaluate azacitidine’s efficacy, safety, and ideal place in therapy in patients with PTCL.
Author Disclosure Statement
Dr Moore is on the Advisory Boards of Genentech, GSK, Incyte, and Sanofi; Dr White and Dr Elmes have no conflicts of interest to report.
References
- Vose J, Armitage J, Weisenburger D, International TCLP. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26:4124-4130.
- Alaggio R, Amador C, Anagnostopoulos I, et al. The 5th edition of the World Health Organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia. 2022;36:1720-1748. Erratum in: Leukemia. 2023;37:1944-1951.
- de Leval L, Rickman DS, Thielen C, et al. The gene expression profile of nodal peripheral T-cell lymphoma demonstrates a molecular link between angioimmunoblastic T-cell lymphoma (AITL) and follicular helper T (TFH) cells. Blood. 2007;109:4952-4963.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: T-Cell Lymphomas. Version 2.2025. May 28, 2025. Accessed January 6, 2025. www.nccn.org/professionals/physician_gls/pdf/t-cell.pdf
- Piekarz RL, Frye R, Prince HM, et al. Phase 2 trial of romidepsin in patients with peripheral T-cell lymphoma. Blood. 2011;117:5827-5834.
- Bachy E, Camus V, Thieblemont C, et al. Romidepsin plus CHOP versus CHOP in patients with previously untreated peripheral T-cell lymphoma: results of the Ro-CHOP phase III study (conducted by LYSA). J Clin Oncol. 2021;40:242-251.
- Camus V, Thieblemont C, Gaulard P, et al. Romidepsin Plus cyclophosphamide, doxorubicin, vincristine, and prednisone versus cyclophosphamide, doxorubicin, vincristine, and prednisone in patients with previously untreated peripheral T-cell lymphoma: final analysis of the Ro-CHOP trial. J Clin Oncol. 2024;42:1612-1618.
- Lemonnier F, Dupuis J, Sujobert P, et al. Treatment with 5-azacytidine induces a sustained response in patients with angioimmunoblastic T-cell lymphoma. Blood. 2018;132:2305-2309.
- Han X, Liu X, Zhang C, et al. PD-1 blockade combined with decitabine to treat refractory peripheral T-cell lymphoma not otherwise specified: a case report and review of literature. J Cancer Res Ther. 2023;19:1680-1684.
- Garcia-Manero G, Gore SD, Cogle C, et al. Phase I study of oral azacitidine in myelodysplastic syndromes, chronic myelomonocytic leukemia, and acute myeloid leukemia. J Clin Oncol. 2011;29:2521-2527.
- Laille E, Shi T, Garcia-Manero G, et al. Pharmacokinetics and pharmacodynamics with extended dosing of CC-486 in patients with hematologic malignancies. PLoS One. 2015;10:e0135520.
- Noonepalle SKR, Karabon L, Chiappinelli KB, Villagra A. Editorial: genetic and epigenetic control of immune responses. Front Immunol. 2021;12:775101.
- Loo Yau H, Bell E, Ettayebi I, et al. DNA hypomethylating agents increase activation and cytolytic activity of CD8(+) T cells. Mol Cell. 2021;81:1469-1483.
- Martin P, Bartlett NL, Chavez JC, et al. Phase 1 study of oral azacitidine (CC-486) plus R-CHOP in previously untreated intermediate- to high-risk DLBCL. Blood. 2022;139:1147-1159.
- Ruan J, Moskowitz A, Mehta-Shah N, et al. Multicenter phase 2 study of oral azacitidine (CC-486) plus CHOP as initial treatment for PTCL. Blood. 2023;141:2194-2205.
- Falchi L, Ma H, Klein S, et al. Combined oral 5-azacytidine and romidepsin are highly effective in patients with PTCL: a multicenter phase 2 study. Blood. 2021;137:2161-2170. Erratum in: Blood. 2022;139:1600.
- Ding K, Liu H, Yang H, et al. A prospective phase 2 study of combination epigenetic therapy against relapsed/refractory peripheral T cell lymphoma. Med. 2024;5:1393-1401.
- Dupuis J, Bachy E, Morschhauser F, et al. Oral azacitidine compared with standard therapy in patients with relapsed or refractory follicular helper T-cell lymphoma (ORACLE): an open-label randomised, phase 3 study. Lancet Haematol. 2024;11:e406-e414.
- O'Connor OA, Bhagat G, Ganapathi K, et al. Changing the paradigms of treatment in peripheral T-cell lymphoma: from biology to clinical practice. Clin Cancer Res. 2014;20:5240-5254.
- Bristol Myers Squibb. Bristol Myers Squibb statement on Istodax (romidepsin) relapsed/refractory peripheral t-cell lymphoma U.S. indication. August 2, 2021. Accessed January 5, 2025. https://news.bms.com/news/details/2021/Bristol-Myers-Squibb-Statement-on-Istodax-romidepsin-Relapsed-Refractory-Peripheral-T-cell-Lymphoma-U.S.-Indication/default.aspx
- ClinicalTrials.gov. Testing the addition of duvelisib or CC-486 to the usual treatment for peripheral T-cell lymphoma. NCT04803201. Updated June 26, 2025. Accessed January 5, 2025. https://clinicaltrials.gov/study/NCT04803201?term=A051902&rank=1#participation-criteria
- O’Connor OA, Falchi L, Lue JK, et al. Oral 5-azacytidine and romidepsin exhibit marked activity in patients with PTCL: a multicenter phase 1 study. Blood. 2019;134:1395-1405.
- Kalac M, Jain S, Tam CS, et al. Real-world experience of combined treatment with azacitidine and romidepsin in patients with peripheral T-cell lymphoma. Blood Adv. 2023;7:3760-3763.