Connecting Science to Practice
This review clarifies a newer approach to the practice of hematology/
oncology pharmacy by emphasizing individualized
venetoclax dosing when used concurrently with azole antifungals.
Here, we describe pharmacokinetic and clinical evidence
that specifically supports a reduced venetoclax dose of 50 mg
when coadministered with posaconazole.
Venetoclax is an oral chemotherapy agent that selectively inhibits BCL-2, a family of proteins that regulate programmed cell death.1 As a selective BCL-2 inhibitor, venetoclax induces cell death in malignant B cells making it a staple of therapy in hematologic malignancies, such as chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL). Venetoclax is also used in acute myeloid leukemia (AML) because many myeloid blasts overexpress BCL-2.1
The metabolism of venetoclax is primarily via cytochrome (CY)P3A4 mechanisms, allowing for interactions with CYP3A4 inhibitors, such as azole antifungals, a medication class that is frequently used prophylactically in patients receiving chemotherapy.2 Prophylaxis with azole therapy has become the standard of care for patients receiving treatment for hematologic malignancies, because they reduce morbidity and mortality by decreasing invasive fungal infection.3,4 Posaconazole and voriconazole are strong inhibitors of CYP3A4, whereas fluconazole and isavuconazole are considered moderate inhibitors.5 The prescribing information for venetoclax recommends a dose of 400 mg daily in patients who are not receiving concomitant azole therapy, whereas a reduction to 200 mg daily is recommended in patients receiving moderate CYP3A4 inhibitors, to 100 mg in the presence of strong CYP3A4 inhibitors, and to 70 mg with posaconazole.6
Although the NCCN’s guideline for AML acknowledges that receiving concomitant interacting medications may require dose adjustments and refer clinicians to the prescribing information for guidance, they do not provide explicit dose recommendations.7 Similar dose reductions are endorsed in the Infectious Diseases Working Party of the German Society for Haematology and Medical Oncology’s guideline, which recommends reducing the dose of venetoclax by ≥75% when coadministered with posaconazole or voriconazole and by 50% in combination with fluconazole or isavuconazole.8
The objective of this review was to address the lack of clarity, consistency, and quality of previous studies evaluating the combination of venetoclax and azole therapy. We also sought to highlight the concerns raised by clinicians and to challenge the current practices based on the available evidence.
Discussion
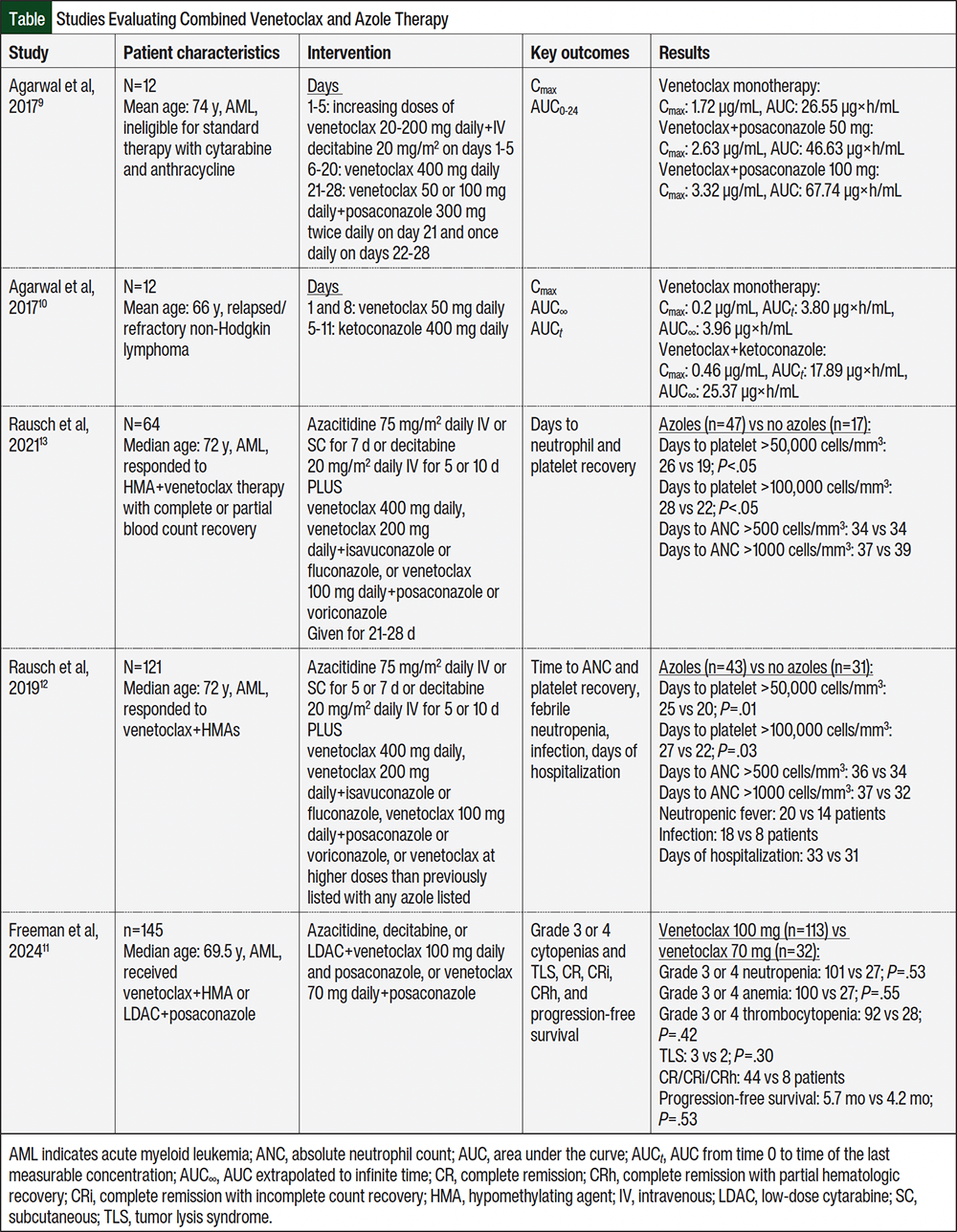
A summary of the studies that were included in our review is shown in the Table.9-13 The coadministration of venetoclax with posaconazole was evaluated in a phase 1b, open-label, drug–drug interaction study where 12 patients with AML received escalating doses of 20 to 200 mg of venetoclax along with 20 mg/m2 of decitabine on days 1 through 5, followed by 400 mg of venetoclax as monotherapy on days 6 through 20.9 Reduced doses of venetoclax 50 or 100 mg were given on days 21 through 28 to account for increased plasma concentrations because of the drug–drug interaction with the addition of posaconazole (300 mg twice on day 21 and once daily on days 22-28).9
Compared with 400 mg of venetoclax without a concomitant azole, 50 mg of venetoclax plus 300 mg of posaconazole increased venetoclax’s maximum serum concentration (Cmax) and area under the curve from time 0 to 24 hours/total daily exposure (AUC0-24) by 53% and 76%, respectively.9 The administration of 100 mg of venetoclax with the same dose of posaconazole increased Cmax by 93% and AUC0-24 by 155%. Despite the increases in Cmax and AUC, the 50-mg and 100-mg doses were well tolerated by patients. During and 2 weeks after the trial (through day 42), the most common (>30%) adverse drug reactions (ADRs) included nausea, diarrhea, anemia, thrombocytopenia, hypokalemia, and hyperphosphatemia, and venetoclax doses of 50 mg or 100 mg with posaconazole did not increase the number or severity of ADRs. Most of the ADRs occurred before the administration of an azole. Thus, reducing the venetoclax dose by at least 75% in patients undergoing treatment for AML and needing fungal prophylaxis was deemed safe and effective.9 However, although this study supports the safety of the prescribing information’s recommendation to reduce venetoclax doses to 70 mg when coadministered with posaconazole, the efficacy claim is assumed because of the venetoclax Cmax and AUC with concurrent posaconazole treatment remaining above that with venetoclax monotherapy, which previously demonstrated efficacy.6,9
Although posaconazole and ketoconazole are strong inhibitors of CYP3A and P-glycoprotein (an efflux protein), ketoconazole also inhibits BCRP (another efflux protein). In a similar phase 1b, open-label study that included 11 patients with relapsed or refractory non-Hodgkin lymphoma, the combination of venetoclax 50 mg (days 1 and 8) and ketoconazole 400 mg (days 5-11) increased venetoclax Cmax and AUC∞ 2.3- and 6.4-fold higher than venetoclax monotherapy, respectively.10 Mild-to-moderate ADRs (eg, thrombocytopenia, electrolyte abnormalities, increased serum creatinine) were more frequent with the combination, and sustained grade 3 thrombocytopenia was observed.10 Although ketoconazole is no longer used for antifungal prophylaxis in this setting, these data illustrate the magnitude of venetoclax exposure increases possible with strong CYP3A and efflux transporter inhibition, supporting the need for venetoclax dose reductions when these drug interactions are present to minimize the risk for increased ADRs.
Because higher doses of posaconazole may be required to achieve adequate AUC/minimum inhibitory concentration ratios for the successful treatment of some fungal infections, Bhatnagar and colleagues validated and used physiologically based pharmacokinetic models to predict venetoclax exposures with posaconazole 500 mg daily.14 Compared with posaconazole 300 mg daily, the 500-mg daily dose increased venetoclax exposures by approximately 12% with the 50-, 70-, and 100-mg doses. The predicted median exposure for the 50- and 70-mg doses remained below the maximum studied venetoclax dose of 1200 mg, but the 100-mg venetoclax dose resulted in a predicted median exposure that was 7.5% above that of the 1200-mg dose. Thus, the researchers concluded that the dose reduction to 70 mg of venetoclax when given with posaconazole 300 mg was applicable with posaconazole doses up to 500 mg daily.14
A single-center, retrospective study evaluated a cohort of 121 patients with AML receiving venetoclax plus hypomethylating agents or low-dose cytarabine with azole prophylaxis to analyze the impact of the azole interaction on myelosuppression, neutropenic fever, infection, and duration of hospitalization.12 Patients received either venetoclax 400 mg daily, venetoclax 200 mg daily with isavuconazole or fluconazole, or venetoclax 100 mg daily with posaconazole or voriconazole. This study was conducted before the recommended dose of venetoclax when coadministered with posaconazole was further reduced to 70 mg in the prescribing information. Higher doses of venetoclax in these combinations were categorized as >400-mg venetoclax equivalent. Although there was a significant delay in platelet recovery time with azoles compared with no azoles (25 days vs 20 days to platelets >50,000 cells/mm3, and 27 days vs 22 days to platelets >100,000 cells/mm3; P<.05 for both comparisons), patients who did not receive azoles had similar frequencies of febrile neutropenia and hospitalization duration. Although not significant, there were more documented infections in the azole group than in the no-azole group (42% vs 26%, respectively).12
In another single-center, retrospective study, Rausch and colleagues investigated the use of venetoclax with strong CYP3A4 inhibitors (posaconazole and voriconazole), as well as moderate CYP3A4 inhibitors (isavuconazole and fluconazole) in 64 patients with AML to determine the duration of cytopenias.13 Patients received either venetoclax 400 mg daily or equivalent dosages in combination with isavuconazole, fluconazole, posaconazole, or voriconazole. Concomitant azoles prolonged platelet recovery, with a median time to achieve platelet count >50,000 cells/mm3 of 26 days versus 19 days without an azole (P<.001). Likewise, the median recovery time to platelet count >100,000 cells/mm3 increased to 28 days for the patients who received azoles versus 22 days without azoles (P=.008). Receiving strong CYP3A4 inhibitors resulted in significantly longer thrombocytopenia than receiving moderate CYP3A4 inhibitors (27 days vs 25 days to reach platelet count >50,000 cells/mm3; P<.05). Concomitant azole use was not associated with changes in the duration of neutropenia. There was no significant difference in the duration of thrombocytopenia between individual azoles (P=.067). Ultimately, the possible extended duration of thrombocytopenia for azole users was not associated with worse outcomes,13 although further large-scale studies, including randomized controlled trials, are needed to confirm the findings.
A retrospective cohort study by Freeman and colleagues that included 145 adults with AML receiving 70 mg or 100 mg of venetoclax daily with hypomethylating agents or low-dose cytarabine plus concomitant posaconazole showed no significant differences in grade 3 or 4 cytopenias, with grade 3 neutropenia occurring in 89.4% versus 84.4% of the patients, respectively (P=.53); grade 3 or 4 anemia in 88.5% versus 84.4%, respectively (P=.55); and grade 3 or 4 thrombocytopenia in 81.4% versus 87.5%, respectively (P=.42).11 The treatment response rates were also similar (50% vs 40%, respectively; P=.42). The researchers concluded that a 100-mg dose of venetoclax may be a safe alternative to 70 mg in the setting of posaconazole coadministration.11 Notably, the 70-mg dose groups had more adverse-risk patients than the 100-mg dose group per the European LeukemiaNet 2024 (18.6% vs 31.3%, respectively), so similar outcomes between the 70-mg and 100-mg dose groups support that 70 mg may be adequate.
Azoles have been established as first-line antimicrobial agents for invasive fungal prophylaxis, with guidelines recommending posaconazole as the drug of choice for intermediate- to high-risk neutropenic AML in patients with myelodysplastic syndrome.15 In this population, posaconazole has resulted in fewer invasive fungal infections than fluconazole.16 However, posaconazole coadministration with venetoclax is contraindicated during therapy initiation and ramp-up in patients with CLL or SLL, and significantly reduced venetoclax doses are required in patients with AML.6 Isavuconazole has a similar spectrum of activity, more reliable absorption, fewer drug–drug interactions, superior tolerability, and absence of QTc prolonging properties than voriconazole.17 Because isavuconazole only moderately inhibits CYP3A4, coadministration of isavuconazole with venetoclax is permitted in the treatment of CLL or SLL with a 50% dose reduction.6 A study by Bose and colleagues showed that the breakthrough invasive fungal infection rate for isavuconazole is similar to that with posaconazole, so it is reasonable to consider isavuconazole as an alternative treatment for posaconazole.18
Several limitations exist with the currently available literature, including small sample sizes, lack of a comparator group, incomplete detailing of dosing information, and the use of nonstandard dosing. In addition, the studies by Rausch and colleagues prescribed doses of 100 mg of venetoclax instead of the recommended 70 mg, which may have contributed to the longer duration of thrombocytopenia.12,13 Another possible explanation to the delay in platelet recovery may be the inconsistency in the azole exposure, because the duration to platelet recovery was longer in the patients with sustained myelosuppression than in those without prolonged myelosuppression. Other potential confounding factors include the concurrent use of hypomethylating agents, such as azacitidine or decitabine, as well as the hematologic malignancy itself or insufficient response to treatment. Overall, the limited evaluation of clinical efficacy end points makes it difficult to conclude optimal dosing beyond pharmacokinetic measures.12,13 In other words, concurrent azole therapy leads to increased venetoclax exposure, even with reduced venetoclax doses, but the association between remission rates and the increased exposure effect needs to be further assessed. In patients aged ≥65 years who have previously untreated or de novo AML, venetoclax has achieved a complete remission rate of ≥60%19,20; however, it is unclear if increased exposure is responsible for higher remission rates. To our knowledge, from the standpoint of clinical safety end points, no studies have specifically evaluated bleeding events, so future research should determine whether prolonged thrombocytopenia is associated with increased bleeding.
Although one potential solution proposed for the interaction between azoles and venetoclax has been to consider treatment alternatives, such as isavuconazole,21 the wholesale acquisition cost of isavuconazole is $2366.28 for the initiation phase and $394.38 daily versus a cost of $404.58 for the initiation phase and $202.29 daily for posaconazole.22 In clinical practice, some providers still prescribe posaconazole concurrently with venetoclax and select the 100-mg dose of venetoclax. The recommended dose of venetoclax when received with posaconazole is 70 mg6; however, because venetoclax is only supplied in 10-, 50-, and 100-mg tablets,6 a single 100-mg tablet is more practical than 3 tablets (a 50-mg tablet plus two 10-mg tablets) to reach the 70-mg dose.
The literature has suggested that 100-mg venetoclax with posaconazole does not significantly increase ADRs in comparison to other azoles.9,10,13 However, with very small sample sizes, a short time frame for monitoring ADRs, and a lack of specific safety data regarding bleeding, true safety concerns cannot be ruled out. Venetoclax 100 mg dosed concomitantly with posaconazole 300 mg has resulted in greater exposure than venetoclax 1200 mg (the maximum safe dose studied) without azole therapy.14 Exceeding this safety margin suggests the possibility for safety concerns with this combination. Because a 50-mg dose of venetoclax with posaconazole 300 mg has also resulted in higher exposure than the FDA-approved dose of venetoclax 400-mg monotherapy without exceeding concentrations achieved with the maximum safe dose of venetoclax 1200-mg monotherapy, at least equal efficacy would be expected from the combination of venetoclax 50 mg and posaconazole 300 mg.14 In addition, 50-mg venetoclax would be more affordable than 100-mg venetoclax at a cost of $65.08 versus $130.15, respectively.22 Although further investigation of venetoclax 50-mg dosing is supported by the European Hematology Association expert consensus,23 higher doses are still often used in clinical practice.
Conclusion
The 50-mg dose of venetoclax may be just as practical as the 100-mg dose without exceeding the maximum safe dose. Although consideration should be given for other practical implications of prescribing lower doses of venetoclax, including insurance coverage for specific medication strengths and the potential impact on insurance coverage if dose titrations are needed, it is necessary to evaluate whether a 50-mg dose of venetoclax would be optimal when combined with posaconazole. Until clearer clinical evidence emerges, the existing data suggest that the use of venetoclax 50 mg with posaconazole may be reasonable, cost-effective, safe, and practical.
References
- Anderson MA, Deng J, Seymour JF, et al. The BCL2 selective inhibitor venetoclax induces rapid onset apoptosis of CLL cells in patients via a TP53-independent mechanism. Blood. 2016;127:3215-3224.
- Liu H, Michmerhuizen MJ, Lao Y, et al. Metabolism and disposition of a novel B-cell lymphoma-2 inhibitor venetoclax in humans and characterization of its unusual metabolites. Drug Metab Dispos. 2017;45:294-305.
- Cornely OA, Ullmann AJ, Karthaus M. Evidence-based assessment of primary antifungal prophylaxis in patients with hematologic malignancies. Blood. 2003;101:3365-3372.
- Lee R, Cho SY, Lee DG, et al. Infections of venetoclax-based chemotherapy in acute myeloid leukemia: rationale for proper antimicrobial prophylaxis. Cancers (Basel). 2021;13:6285.
- US Food and Drug Administration. For healthcare professionals: FDA’s examples of drugs that interact with CYP enzymes and transporter systems. Updated June 25, 2025. Accessed January 8, 2024. www.fda.gov/drugs/drug-interactions-labeling/healthcare-professionals-fdas-examples-drugs-interact-cyp-enzymes-and-transporter-systems
- Venclexta (venetoclax) tablets, for oral use [prescribing information]. AbbVie Inc; July 2024. Accessed December 12, 2025. www.rxabbvie.com/pdf/venclexta.pdf
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: acute myeloid leukemia. Version 3.2026. November 24, 2025. Accessed January 7, 2026. www.nccn.org/professionals/physician_gls/pdf/aml.pdf
- Stemler J, Mellinghoff SC, Khodamoradi Y, et al. Primary prophylaxis of invasive fungal diseases in patients with haematological malignancies: 2022 update of the recommendations of the Infectious Diseases Working Party (AGIHO) of the German Society for Haematology and Medical Oncology (DGHO). J Antimicrob Chemother. 2023;78:1813-1826.
- Agarwal SK, DiNardo CD, Potluri J, et al. Management of venetoclax-posaconazole interaction in acute myeloid leukemia patients: evaluation of dose adjustments. Clin Ther. 2017;39:359-367.
- Agarwal SK, Salem AH, Danilov AV, et al. Effect of ketoconazole, a strong CYP3A inhibitor, on the pharmacokinetics of venetoclax, a BCL-2 inhibitor, in patients with non-Hodgkin lymphoma. Br J Clin Pharmacol. 2017;83:846-854.
- Freeman T, Habib A, Huang Y, et al. Real-world experience of venetoclax target dosing with concomitant posaconazole in adult patients with acute myeloid leukemia. Blood. 2024;144:2432-2433.
- Rausch CR, DiNardo CD, Maiti A, et al. Venetoclax dosing in combination with antifungal agents: real world experience in patients with acute myeloid leukemia. Blood. 2019;134(suppl_1):2640.
- Rausch CR, DiNardo CD, Maiti A, et al. Duration of cytopenias with concomitant venetoclax and azole antifungals in acute myeloid leukemia. Cancer. 2021;127:2489-2499.
- Bhatnagar S, Mukherjee D, Salem AH, et al. Dose adjustment of venetoclax when co-administered with posaconazole: clinical drug-drug interaction predictions using a PBPK approach. Cancer Chemother Pharmacol. 2021;87:465-474.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: prevention and treatment of cancer-related infections. Version 1.2025. June 20, 2025. Accessed January 7, 2026. www.nccn.org/professionals/physician_gls/pdf/infections.pdf
- Cornely OA, Maertens J, Winston DJ, et al. Posaconazole vs. fluconazole or itraconazole prophylaxis in patients with neutropenia. N Engl J Med. 2007;356:348-359.
- Ananda-Rajah MR, Kontoyiannis D. Isavuconazole: a new extended spectrum triazole for invasive mold diseases. Future Microbiol. 2015;10:693-708.
- Bose P, McCue D, Wurster S, et al. Isavuconazole as primary antifungal prophylaxis in patients with acute myeloid leukemia or myelodysplastic syndrome: an open-label, prospective, phase 2 study. Clin Infect Dis. 2021;72:1755-1763.
- DiNardo CD, Pratz KW, Letai A, et al. Safety and preliminary efficacy of venetoclax with decitabine or azacitidine in elderly patients with previously untreated acute myeloid leukaemia: a non-randomised, open-label, phase 1b study. Lancet Oncol. 2018;19:216-228.
- DiNardo CD, Pratz K, Pullarkat V, et al. Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood. 2019;133:7-17.
- Falci DR, Pasqualotto AC. Profile of isavuconazole and its potential in the treatment of severe invasive fungal infections. Infect Drug Resist. 2013;6:163-174.
- Micromedex RED BOOK. Merative. August 2022. Accessed November 6, 2025. www.merative.com/documents/micromedex-red-book
- Stemler J, de Jonge N, Skoetz N, et al. Antifungal prophylaxis in adult patients with acute myeloid leukaemia treated with novel targeted therapies: a systematic review and expert consensus recommendation from the European Hematology Association. Lancet Haematol. 2022;9:e361-e373. Erratum in: Lancet Haematol. 2022;9:e398.