Connecting Science to Practice
For patients with relapsed/refractory multiple myeloma,
B-cell maturation antigen (BMCA)–directed bispecific antibodies
(BsAbs) offer deep responses and improved disease
control. However, certain adverse events (AEs), such as cytokine
release syndrome, immune effector cell–associated
neurotoxicity syndrome, and infection, require special patient
management considerations. This review article compiled
published data, AE risk mitigation concepts, and real-world
experiences to provide practical guidance for managing
patients receiving BCMA-directed BsAbs. Given the differences
among clinical centers, practitioners should design
patient management approaches based on their existing infrastructure,
resource accessibility, and individual patient
needs.
Before the 2020s, patients with triple class–exposed (immunomodulatory drugs, proteasome inhibitors, and CD38-targeting antibodies), relapsed or refractory multiple myeloma (RRMM) had limited treatment options and outcomes were extremely poor.1-3 The advent of T-cell–redirecting therapies, including CAR T and bispecific antibodies, has changed the outlook in RRMM, offering high response rates and prolonged disease control for patients.2,4,5 However, these novel immunotherapies are also associated with a risk for adverse events (AEs), specifically cytokine release syndrome (CRS), immune effector cell–associated neurotoxicity syndrome (ICANS), and infections.2,6,7
Two B-cell maturation antigen (BCMA)–directed bispecific antibodies have been approved by the FDA, elranatamab-bcmm and teclistamab-cqyv.8,9 These agents are an attractive option for patients who need an off-the-shelf treatment, and they are increasingly being used in academic centers, as well as in community settings.10 The administration of BCMA-directed bispecific antibodies requires special considerations for monitoring; patient, caregiver, and care team education; and supportive care.6,8,9 This review article describes the practical considerations for patient management during treatment with BCMA-directed bispecific antibodies.
Bispecific antibodies are constructs engineered to recognize 2 distinct antigens. In multiple myeloma (MM), these antigens are a target that is present on MM cells and CD3 that is present on T cells.8,9,11 Engagement of the cognate antigen and CD3 results in T-cell activation and release of the cytolytic proteins, including perforin, which creates transmembrane pores in the target cell, and granzyme B, which migrates through those pores to induce apoptosis.12 Activated T cells and monocytic cells in the microenvironment also release cytokines, including interferon (IFN)-γ, interleukin (IL)-2, IL-6, IL-10, and tumor necrosis factor (TNF)-α, that may contribute to antitumor activity.11,13,14
BCMA is a rational target for bispecific antibodies in MM because it is strongly expressed on plasma cells and plasma blasts while being virtually absent on hematopoietic stem cells and nonhematopoietic tissues.15,16 Furthermore, BCMA plays important roles in myelomagenesis, including promoting the proliferation and survival of MM cells, maintaining an immunosuppressive bone marrow microenvironment, mediating drug resistance, and promoting disease progression.15-17 This may be one reason why antigen loss after exposure to BCMA-directed CAR T-cell or bispecific antibody therapy, while possible, is not a common event.18 Resistance mechanisms to BCMA-directed bispecific antibody and CAR T-cell therapy require further investigation.
The mechanism of antimyeloma activity of BCMA-directed bispecific antibodies also contributes to the characteristic AEs associated with these agents. Cytokine release caused by T-cell activation may trigger an inflammatory cascade resulting in CRS or, less often, ICANS.7,13,14 Patients receiving BCMA-directed bispecific antibodies have an increased risk for infections in part because of treatment-emergent cytopenias and hypogammaglobulinemia, which may be related to BCMA being essential for the survival of functional plasma cells.4,5,15,16,19,20
Indications for BCMA-Directed Bispecific Antibodies
The approval of teclistamab was based on the phase 1/2 MajesTEC-1 study (NCT03145181,21 NCT0455709822), and the approval of elranatamab was based on the phase 2 MagnetisMM-3 study (NCT0464935923). In the United States, teclistamab-cqyv and elranatamab-bcmm are indicated for the treatment of RRMM in adults who have received at least 4 lines of therapy, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.8,9 Ongoing studies are investigating earlier lines of treatment with bispecific antibodies, as well as in combinations with standard anti-myeloma regimens.24,25
Dosing and Administration of BCMA-Directed Bispecific Antibodies
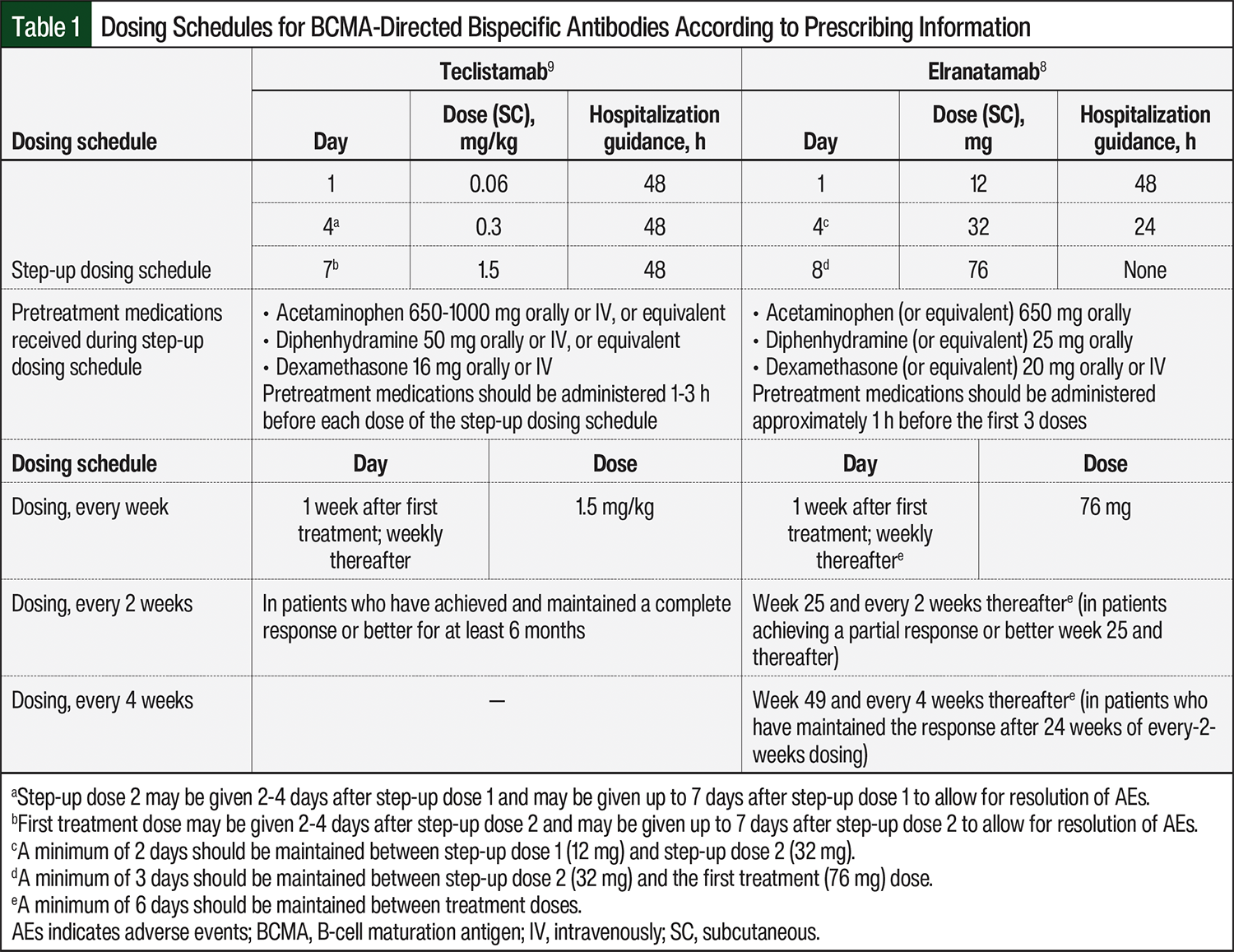
Teclistamab and elranatamab are administered as subcutaneous injections.8,9 Both drugs are available in 2 sizes of single-use, ready-to-use vials that do not need dilution before administration. Teclistamab, however, is supplied in 2 concentrations of 10 mg/mL (30 mg/3 mL) and 90 mg/mL (153 mg/1.7 mL).9 The concentration of elranatamab is 40 mg/mL in both vial sizes (76 mg/1.9 mL and 44 mg/1.1 mL).8 Because the dosing of teclistamab is based on actual body weight, the total dose, injection volume, and the number of vials must be calculated at each administration.9 By contrast, elranatamab is administered as a fixed dose.8 The dosing for both agents is initiated according to a step-up schedule (Table 1).8,9 During the step-up dosing schedule, pretreatment medications (consisting of antipyretics, a histamine 1-receptor antagonist, and corticosteroids) are administered to reduce the risk for CRS.26,27 The prescribing information for teclistamab and elranatamab include differing durations of hospitalization8,9; securing a bed is therefore a component of initiating BCMA-directed bispecific antibody treatment (Table 1).8,9 However, strategies for outpatient step-up dosing are an ongoing area of investigation.28,29
The low severity of CRS associated with bispecific antibodies4,30 favors the feasibility of initiating these agents in the community setting.10 However, some patients may require referral to an academic center for step-up dosing if the infrastructure for inpatient monitoring is not available locally or if there is concern about tolerability with bispecific antibodies.
After the step-up dosing schedule is completed, BCMA-directed bispecific antibodies are administered weekly. According to the prescribing information, the dose interval for teclistamab may be extended to every 2 weeks for patients who maintained a complete response or better for at least 6 months.9 The dose frequency of elranatamab should be reduced to every 2 weeks after 24 weeks in patients who have achieved a partial response or better for at least 2 months; then the dose frequency should be further reduced to every 4 weeks in patients who maintained the response.8 Responses are maintained after the switch to every-2-weeks dosing, whereas the incidence of some grade 3 or 4 AEs may decrease.4,31
Efficacy Outcomes with BCMA-Directed Bispecific Antibodies in RRMM
The MajesTEC-1 and MagnetisMM-3 trials enrolled patients with triple class–exposed RRMM and evaluated the overall response rate (ORR) as the primary end point.4,5 The primary analyses for both clinical trials reported the outcomes of patients who did not previously receive BCMA-directed therapy. The responses were rapid; the median time to response was 1.2 months for both agents (range, 0.2-5.5 months with teclistamab and 0.9-7.4 months with elranatamab). The ORRs for teclistamab and elranatamab were 63% (95% confidence interval [CI], 55.2-70.4) and 61% (95% CI, 51.8-69.6), respectively, with 39.4% and 35%, respectively, of patients achieving a complete response or better.4,5 The median duration of response (mDOR) was not mature for teclistamab or elranatamab at the time of the primary analysis.4,5,32
Extended follow-up data for MajesTEC-1 and MagnetisMM-3 have demonstrated continued clinical benefit with each agent, ongoing responses after a switch to every-2-weeks dosing, and updated time-to-event analyses.31,33-35 With longer follow-up (median follow-up of 30.4 months with teclistamab and 28.4 months by reverse Kaplan–Meier curve for elranatamab), the complete response rates continued to deepen for teclistamab and elranatamab (rates of complete response or better, 46.1% and 37.4%, respectively).32,36 The mDOR was 24 months for teclistamab and was not reached for elranatamab, with an estimated 30-month DOR rate of 61% (95% CI, 47.8-71.8).32,35-37 The median progression-free survival was 11.4 months for teclistamab and 17.2 months for elranatamab (95% CI, 9.8-not estimable).36-38 The median overall survival was 22.2 months for teclistamab and 24.6 months for elranatamab (95% CI, 13.4-not estimable).35,36
The outcomes in patients who previously received BCMA-directed therapies have also been reported. Among a separate cohort of 40 patients who previously received BCMA antibody–drug conjugate or CAR T-cell therapy and were enrolled in MajesTEC-1, the ORR was 52.5% (95% CI, 36.1-68.5) and the mDOR was not reached at a median follow-up of 12.5 months (range, 0.7-14.4 months).39 In a pooled analysis of 87 patients who previously received BCMA CAR T-cell therapy or antibody–drug conjugate and therapy with elranatamab across 4 studies at a median follow-up of 11.3 months (range, 0.3-32.3 months), the ORR was 46% (95% CI, 35.2-57) and the mDOR was 17.1 months (95% CI, 9.8-not estimable).40
The real-world efficacy of BCMA-directed bispecific antibodies has been consistent with the results of registrational clinical trials. ORRs with teclistamab of between 50% and 65% have been reported in real-world, BCMA treatment–naïve patients, including those who would have been ineligible for inclusion in the MajesTEC-1 study.41-43 In patients with previous BCMA-directed therapy, the ORRs to BCMA-directed bispecific antibody treatment range from 50% to 63%.40,43-45 Overall, the available evidence suggests that BCMA-directed bispecific antibodies offer clinical benefit for patients who previously received BCMA-directed therapy.
Safety of BCMA-Directed Bispecific Antibodies in RRMM
AEs of any grade occurred in 100% of the patients in the MajesTEC-1 and MagnetisMM-3 studies, and were grade ≥3 in 94.5% and 70.7% of patients in these trials, respectively.4,5 AEs resulted in dose interruption in 63% of patients who received teclistamab and in 77.2% of patients who received elranatamab. Hematologic AEs were the most common grade ≥3 AEs, including neutropenia (64.2% and 48.8%), anemia (37% and 37.4%), and lymphopenia (32.7% and 25.2%), in MajesTEC-1 and MagnetisMM-3, respectively. CRS was the most frequent nonhematologic AE, with an incidence for grade 3/4 CRS of 0.6% for teclistamab and 0% for elranatamab.4,5
Based on the results of the registrational clinical trials, the prescribing information for teclistamab and elranatamab include boxed warnings for CRS and neurologic AEs, including ICANS.8,9 Both agents are available only under a Risk Evaluation and Mitigation Strategy (REMS) program. In addition to the boxed warnings for CRS and neurologic AEs, including ICANS, warnings and precautions for infections, neutropenia, hepatotoxicity, and embryo-fetal AEs are included in both drugs’ prescribing information.8,9 The prescribing information for teclistamab has an additional warning and precaution about hypersensitivity and other administration reactions.9 More important, the safety profiles of BCMA-directed bispecific antibodies in real-world data to date have been comparable with clinical trial data, with no increased frequency or severity of AEs when these agents are received outside of clinical trials.41-45
Managing and Mitigating Key AEs With BCMA-Directed Bispecific Antibodies
As with any aspect of MM therapy, there is no one-size-fits-all approach to patient management during BCMA-directed bispecific antibody treatment. However, there are general principles that the care team may apply to mitigate the risk for AEs of special interest during BCMA-directed bispecific antibody therapy.
Cytokine Release Syndrome
T-cell–redirecting therapies are universally associated with a risk for CRS.7,13,14,30 Although the pathophysiology of CRS is not completely understood, the release of IFN-γ by activated T cells is believed to be the precipitating event to a systemic inflammatory cascade.13,14,30 Secreted IFN-γ causes fever and the constitutional symptoms that are the hallmarks of grade 1 CRS, and also sets a positive feedback loop in motion by inducing monocytic cells to produce additional cytokines, specifically IL-6, IL-10, and TNF-α.7,13,14,30 TNF-α is responsible for endothelial activation and further amplification of the inflammatory response, which results in capillary leakage, hypotension, coagulopathy, hypoxia, and—in severe and uncontrolled cases—multiorgan failure.7,13,14
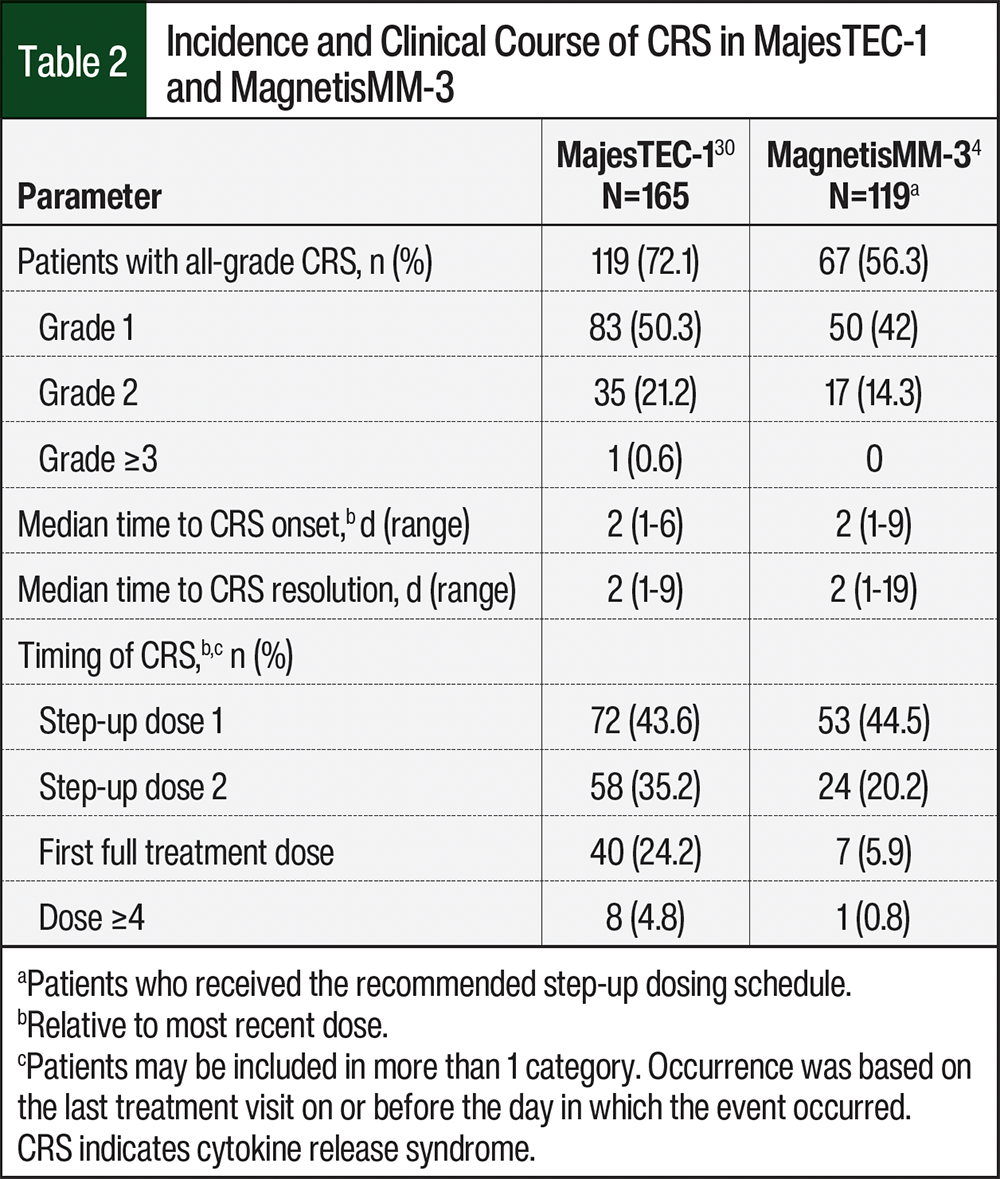
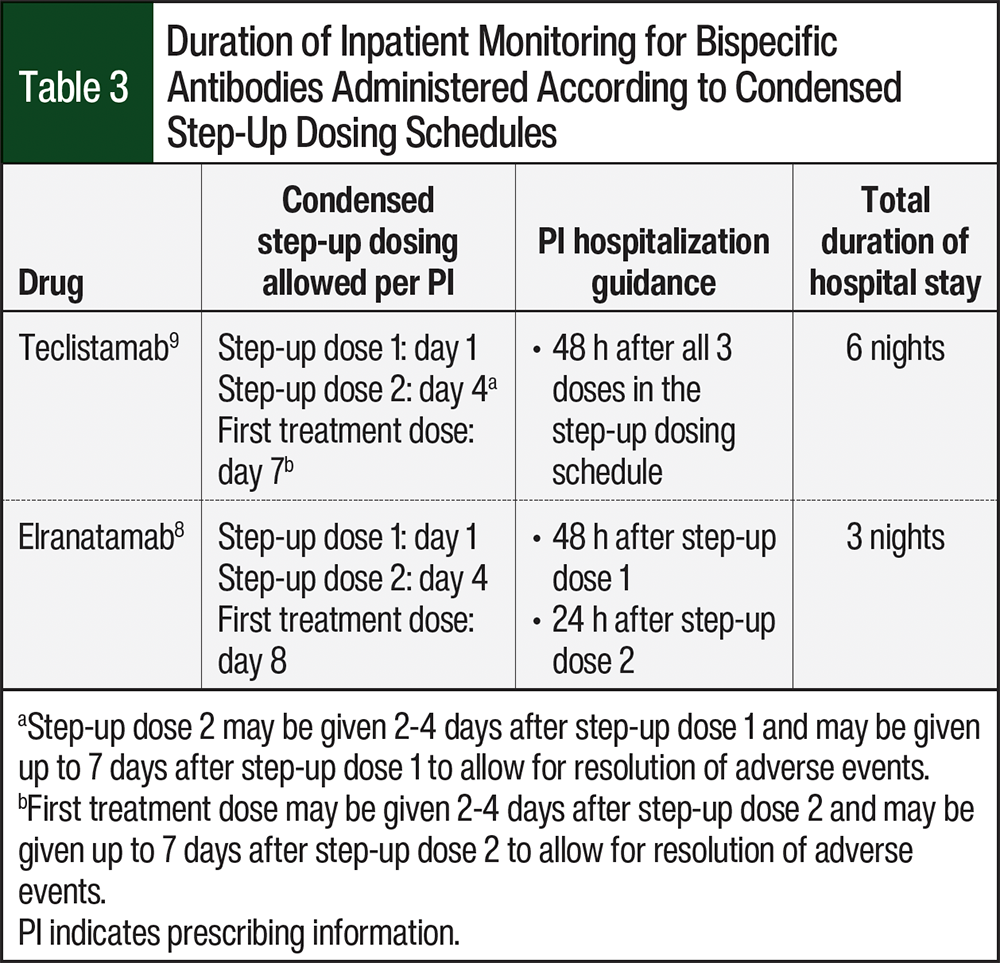
In patients who received the step-up dosing schedule in the MajesTEC-1 and MagnetisMM-3 studies, the reported incidences of CRS were 72.1% and 56.3%, respectively (Table 2).4,30 Although CRS is common with BCMA-directed bispecific antibodies, the events are typically low grade. Differing approaches to step-up dosing result in the predictability of the timing of CRS varying by agent; however, most events occur with step-up doses of BCMA-directed bispecific antibodies (Table 3).8,9 No grade 4 or 5 CRS events occurred in the MajesTEC-1 or MagnetisMM-3 clinical trials.4,30
In the real-world use of BCMA-directed bispecific antibodies, CRS has been similarly low grade with predictable timing. The results of the real-world use of BCMA-directed bispecific antibodies show incidences of CRS of 52% to 73%.41-45 The vast majority of CRS in real-world experience with BCMA-directed bispecific antibodies was grade 1 or 2 and occurred during step-up dosing; the incidences of grade ≥3 CRS have ranged from 0% to 3%.41-45 These real-world accounts are consistent with our experience; we have rarely encountered CRS outside of the step-up dosing schedule, and we have seen very few grade ≥2 CRS events.
High disease burden is a well-established predictor of CRS risk during CAR T-cell therapy.14 However, there are currently no patient- or disease-related factors that have been validated to predict CRS risk during BCMA-directed bispecific antibody treatment.
Step-up dosing and the administration of pretreatment medications reduces the risk for CRS.27 At our centers, we have largely followed the recommendations in the prescribing information,8,9 and all of the cases of CRS we have encountered have been low grade. Alternative strategies to step-up dosing, which are discussed in subsequent sections of this review article, may minimize the need for hospitalization during step up.
IL-6 receptor antagonists are part of the standard management of bispecific antibody–associated CRS.6 At the time of this writing, multiple reports described the feasibility of prophylactic tocilizumab administration during step-up dosing, with encouraging results.46-49 The rates of CRS in patients receiving prophylactic tocilizumab were <40%, with most cases being grade 1, and the incidence of neutropenia was in line with what was observed during the registrational trials. More important, efficacy outcomes were not compromised with prophylactic tocilizumab.47-49 Prophylaxis may be provided as a single intravenous infusion of tocilizumab before step-up dose 147,49 or between step-up dose 1 and step-up dose 2.48 There have been no head-to-head comparisons of these approaches.
The administration of prophylactic tocilizumab is often institution-specific and may be influenced by reimbursement. To our knowledge, data for accurate CRS risk prediction are lacking, but ongoing analyses may help generate a prediction model in patients with MM similar to one developed for glofitamab in patients with large B-cell lymphoma.50 In a community setting with less resources, it may make more sense to prescribe tocilizumab proactively given the potential impact on CRS rates and to avoid more costly management of CRS events. Of note, the NCCN guidelines on the prophylactic use of tocilizumab for the management of immunotherapy-related AEs may help with reimbursement concerns moving forward.51
Some institutions are implementing outpatient step-up dosing administration for BCMA-directed bispecific antibodies.28,29 Although the particulars vary between centers, the overall approach usually includes components of patient and caregiver education, encouraging patients to stay in proximity to the healthcare facility during step-up dosing, providing a dose of dexamethasone for patients to self-administer at home at the first onset of fever, daily evaluation in an outpatient clinic, and prompt admission if patients have a fever or any other signs and symptoms of AEs. Some centers are adopting remote monitoring systems.28,29 For patients with MM who received teclistamab, step-up dosing with prescribing information–recommended pretreatment medications under the hospital-based outpatient program at the Mayo Clinic (n=57), the incidence of any-grade CRS was 31.6% (grade 1, 22.8%; grade 2, 7%; and grade 4, 1.8%).52 Of the 9 patients who completed outpatient step-up dosing at Fox Chase Cancer Center, 1 was admitted for inpatient monitoring.29 Even in centers that offer outpatient step-up dosing, some patients may not be candidates based on comorbidities, frailty, and/or a lack of caregiver assistance. Some institutions that provide outpatient dosing may have financial assistance or safety net programs that could provide accommodations for patients whose residence is not within close proximity to the treatment center.
In general, healthcare providers should carefully monitor every patient who is receiving treatment with bispecific antibodies for CRS during the step-up dosing period. If step-up dosing is administered inpatient, the duration of hospital stay and monitoring will differ between the agents, even if given according to a condensed dosing schedule as allowed in the prescribing information (Table 3). The oncology pharmacist should also educate patients on the signs and symptoms of CRS, as well as the need for promptly contacting their care team in the event of fever. Published guidelines recommend assessing patients for CRS at least twice daily while hospitalized and at any change in a patient’s condition.6 At our institutions, vital signs are assessed every 4 to 6 hours while the patients are hospitalized, and the frequency increases if there are any clinical concerns. However, as outpatient step-up dosing becomes more common, the collection of vital signs by a reliable caregiver may be feasible. Of note, some institutions such as the Mayo Clinic, the Levine Cancer Institute, the Fox Chase Cancer Center, and the Institut Universitaire du Cancer de Toulouse have leveraged technology for patient self-monitoring, but this may not be feasible for all centers.29,52-54 To appropriately tailor the monitoring approach, validated predictive factors are needed to identify patients who may have a higher risk for CRS.
The administration of step-up dosing and monitoring should be tailored based on the resources available and on patients’ needs. Reasonable approaches include admitting patients for step-up dose administration and monitoring; administering step-up dosing on an outpatient basis and admitting the patient for monitoring immediately thereafter; and administering step-up dosing and monitoring entirely in the outpatient setting if resources allow for rapid assessment, admission, and intervention in the event of CRS.
Patients and caregivers should be educated on the signs and symptoms of CRS, including instructions on when and how to notify their healthcare provider at the onset of symptoms (the prescribing information and REMS for teclistamab and elranatamab include patient education resources). The patient’s vital signs should be monitored every 4 to 6 hours during step-up dosing.
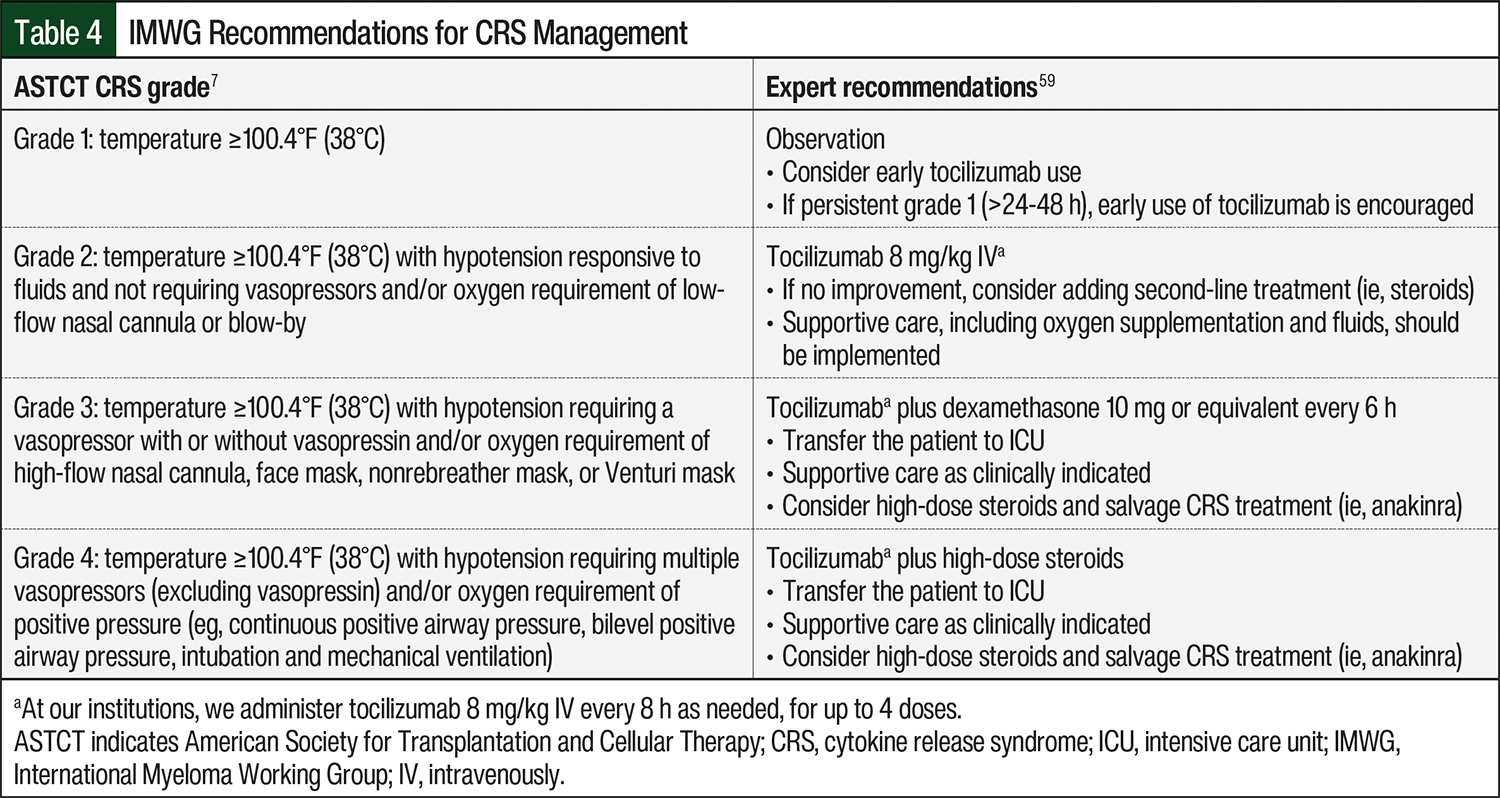
During the initial clinical development of BCMA-directed bispecific antibodies, the protocol-recommended approach to CRS management largely followed early guidelines established for CAR T-cell therapy, with tocilizumab deployed only for events that were grade ≥2.55 However, early intervention by some clinicians to prevent progression to more severe CRS, with tocilizumab at grade 1 or prophylactically, is increasingly becoming the preferred strategy for patient management during bispecific antibody treatment56-58; these are the approaches used at our institutions. Some centers also provide patients with a take-home dose of dexamethasone to be administered at the onset of fever, after discussion with a healthcare provider.53 More important, the symptoms of grade 1 CRS may overlap with those of infection,6,7 and, for this reason, we perform infectious workup concomitant with CRS management. Guidance on dose modification and patient management in the event of CRS may be included in the prescribing information for elranatamab and teclistamab.8,9 The International Myeloma Working Group has published consensus guidelines on the use of bispecific antibodies in patients with MM, including recommendations on the management of CRS, which are summarized in Table 4.7,59
Tocilizumab was administered to 36.4% and 22.7% of patients in MajesTEC-1 and MagnetisMM-3, respectively.4,5 Patients who received tocilizumab at the first onset of CRS had lower rates of recurrent CRS than those who did not receive tocilizumab.30,60 Mechanism-based pharmacokinetic and pharmacodynamic model simulations predict that a single dose of tocilizumab blocks IL-6 receptor occupancy for 10 days.61 This long duration of receptor occupancy contributes to slightly differing management approaches for grade 1 CRS in patients who received prophylactic tocilizumab compared with those who did not, because IL-6 antagonism is expected to persist if tocilizumab was administered during step-up dosing. In our experience, CRS with bispecific antibodies is mitigated with early or prophylactic tocilizumab and supportive care; however, in patients with refractory disease, we would consider repeat dosing and the addition of corticosteroids.
The management of grade 1 CRS includes supportive care and tocilizumab for those patients who did not receive tocilizumab prophylactically. Supportive care measures for grade 1 CRS include intravenous fluids, empiric antibiotics, and infectious workup, and the symptomatic management of constitutional symptoms. Reasonable approaches to managing CRS that is refractory to tocilizumab include the addition of dexamethasone or repeat dosing of tocilizumab every 8 hours for up to 3 or 4 doses. High-grade CRS is unlikely with BCMA-directed bispecific antibodies, but it generally requires management in a critical care setting in which the approach should follow the prescribing information and the current practice guidelines (Table 4).
Immune Effector Cell–Associated Neurotoxicity Syndrome
Although ICANS is the second most common nonhematologic AE associated with CAR T-cell therapy,6 it has been relatively uncommon in patients who receive BCMA-directed bispecific antibodies.4,5 The pathophysiology of ICANS is perhaps not as well understood as that of CRS, and ICANS is thought to involve endothelial activation and blood–brain barrier dysfunction, as well as proinflammatory cytokines in the cerebrospinal fluid.6,7
Patients had ICANS infrequently in MajesTEC-1 and MagnetisMM-3, occurring in 5 and 4 patients in each clinical trial, respectively; none of the patients discontinued treatment as a result of ICANS.4,5 In real-world analyses, the incidence of ICANS after receiving teclistamab has been slightly higher than that in the registrational trials. In 89 patients across 5 academic institutions, 7% had grade 1 or 2 ICANS, 4.4% had grade 3 or 4 ICANS, and 1 patient had a grade 5 event.42 The incidence of ICANS was 7.3% in 123 real-world patients in Germany,43 and 13% in 45 patients who did not meet the eligibility criteria for MajesTEC-1.41 In clinical trials and real-world experience, ICANS has predominantly occurred during step-up dosing and presented concomitantly with CRS; ICANS was managed with corticosteroids.4,5,41-43 This aligns with our experience; ICANS has been rare, and the onset was early in the patients who had ICANS.
A high index of suspicion for ICANS among the care team, patients, and caregivers is key to early recognition and intervention. The hallmark symptom of early ICANS is expressive aphasia.62 Mild lethargy, motor findings, and dysgraphia are also frequent with ICANS. Headache, although common, is a nonspecific symptom of ICANS.6,7 Because many encephalopathy-related terms are overlapping and subjective, the Immune Effector Cell-Associated Encephalopathy (ICE) score was developed to objectively evaluate neurologic AEs associated with CAR T-cell therapy; ICE is also used in patients receiving bispecific antibodies.6,7 The ICE score includes measures of orientation, naming, following commands, writing, and attention. ICE scores range from 0 to 9, with lower ICE scores associated with higher ICANS grading. The grading of ICANS considers the ICE score, level of consciousness, presence of seizures, motor findings, and cerebral edema, with the grade being determined by the most severe event.7
Baseline and at least twice-daily neurologic and ICE examinations are performed during the initiation of bispecific antibody therapy, but once neurotoxicity is suspected, more frequent serial neurologic examinations with immune effector cell–associated neurological AEs grading are performed.59 At our institutions, ICE scores are performed by the care team while patients are hospitalized during step-up dosing, and they are collected either every 4 hours (at the time of vital signs assessment) or every 12 hours. As more centers adopt outpatient step-up dosing, neurologic monitoring may be shifted to home healthcare providers or possibly even responsible caregivers (with comprehensive patient and caregiver education), as appropriate.28
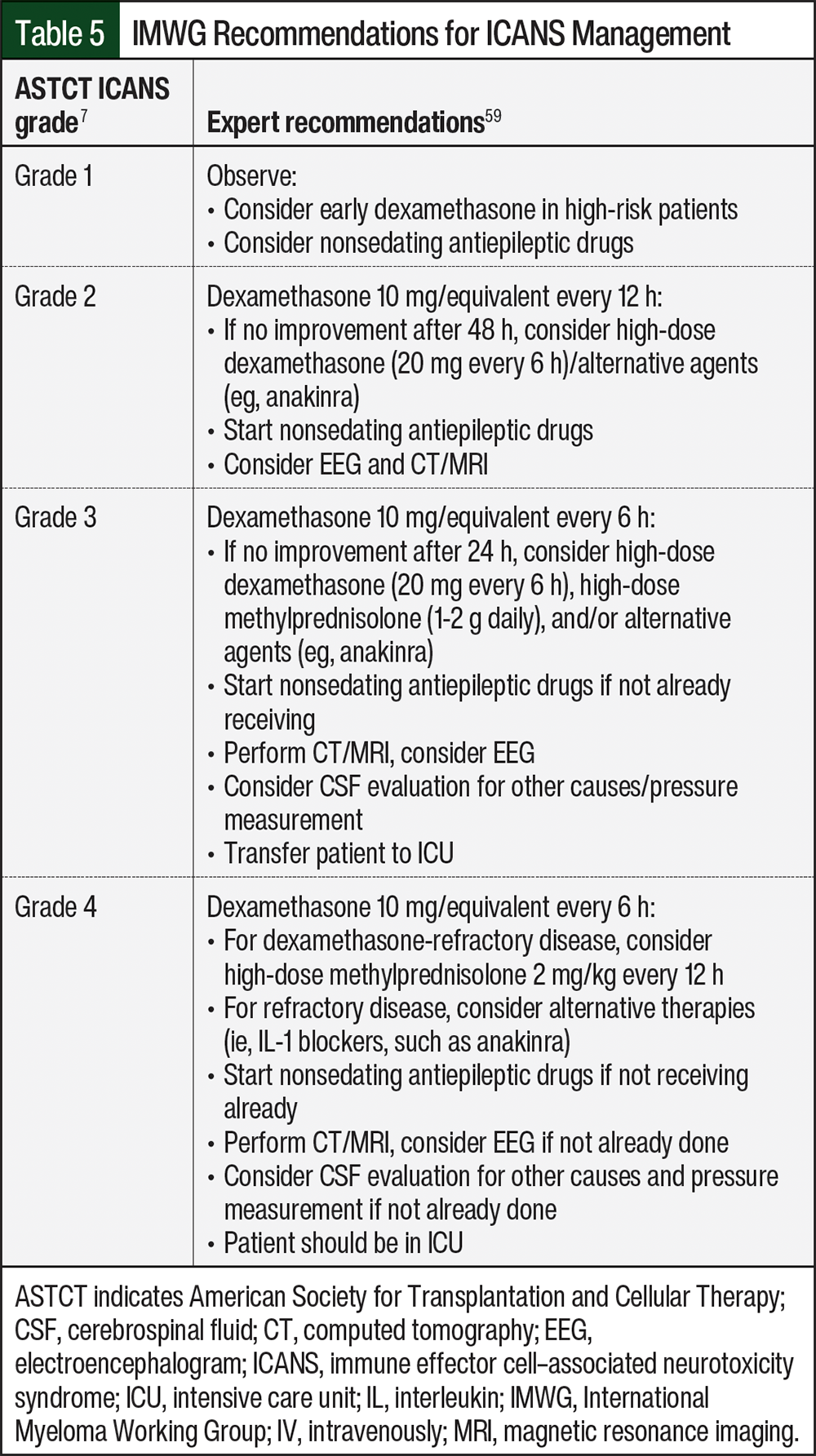
Recommendations on dose modifications and supportive care in the event of ICANS are available in the prescribing information for teclistamab9 and elranatamab,8 as well as in guidelines from the International Myeloma Working Group (Table 5).7,59
ICANS after receiving BCMA-directed bispecific antibodies has been infrequent at our centers, and we have rarely encountered any severe or refractory cases. There has been a shift toward intervention with corticosteroids in cases of grade 1 ICANS associated with BCMA-directed CAR T-cell therapy,63,64 and the algorithms we have in place for BCMA-directed bispecific antibodies include early intervention with steroids with the consideration of neurology consultation, imaging, and alternative agents for persistent events.
For grade 1 ICANS, treatment with corticosteroids is appropriate. High-grade or refractory ICANS should be managed according to the prescribing information and expert guidelines (Table 5).
Other Neurologic AEs
Peripheral neuropathy, inclusive of motor dysfunction and sensory neuropathies, may occur in patients with MM, as an AE associated with immunomodulatory drugs and proteasome inhibitors, or as a direct complication of the disease itself.65-67 Motor dysfunction and sensory neuropathy of any grade were reported in 16% and 15%, respectively, of patients in MajesTEC-1,9 and in 17.1% and 13.8%, respectively, of patients in MagnetisMM-3.4 Based on the available data of real-world experiences with bispecific antibodies, treatment-emergent peripheral neuropathy is not common.41,43-45 These real-world reports are consistent with the experience at our centers; we have not seen any indication that BCMA-directed bispecific antibodies have a neurotoxicity signal outside of the known risk for ICANS. Although we have not seen peripheral neuropathy events with BCMA-directed bispecific antibodies out of proportion to what we observe in other patients with RRMM receiving ongoing therapy of any kind at our institutions, prescribing information includes recommendations for dose modifications for neurologic AEs other than ICANS.8,9
Infections
Patients with MM are up to 7 times more likely to have infections than the general population.68 BCMA-directed bispecific antibodies can induce immunosuppression via multiple mechanisms; therefore, patients receiving these agents warrant close monitoring for infections, as well as prophylaxis.10,20,68
The disruption of the B-cell compartment with malignant plasma cells, dysregulation of the bone marrow microenvironment, and monoclonal antibody production all contribute to impaired innate and adaptive immunity in patients with MM, manifesting as defects in monocyte activation and pathogen clearance, reduced circulating B cells, immunoparesis, and perturbations in T-cell counts and functions.69,70 This disease-related immunosuppression is further exacerbated during BCMA-directed bispecific antibody treatment, during which neutropenia frequently emerges and the elimination of healthy plasma cells results in hypogammaglobulinemia.4,71,72 Furthermore, sustained treatment with bispecific antibodies may result in T-cell exhaustion.73
These known mechanisms of impaired immunity are borne out in the clinical data, and patients receiving BCMA-directed bispecific antibodies are more likely to have grade ≥3 infections compared with patients who receive bispecific antibodies directed against other targets.72 Infections of any grade occurred in 80% (grade 3 or 4, 55.2%) of patients in MajesTEC-1 and in 69.9% (grade 3 or 4, 39.8%) of patients in MagnetisMM-3.4,71 The rates of fatal infections in patients who received BCMA-directed bispecific antibodies ranged from 4.2% to 12.7%.4,9,71 The potential spectrum of infectious etiologies includes bacterial, viral, and fungal pathogens, as well as opportunistic infections associated with compromised T-cell immunity, such as Pneumocystis jirovecii pneumonia, cytomegalovirus, and aspergillosis.4,71,72
Expert consensus statements recommend risk adaptation of infection monitoring and prophylaxis based on patients’ characteristics, previous treatment, disease-related factors, and infection history.6,20,59 The patient characteristics that are associated with higher infection risk include advanced age, impaired performance status, comorbidities, immunoparesis, and prolonged cytopenias.20 High cumulative glucocorticoid exposure, triple class–exposed status, and recent treatment with intensive therapy, another bispecific antibody, or CAR T-cell therapy are all treatment-related risk factors.20 Disease-related factors include high tumor burden, triple-class refractory status, disease type, and renal dysfunction.20 Finally, a history of numerous infections, severe infections, hospitalization caused by infections, or baseline exposure to DNA viruses are predictors of future infections.20,68,71
Unlike acute-phase AEs (ie, CRS and ICANS), the risk for infections persists long after step-up dosing and is likely present for as long as a patient is receiving therapy as well as after the discontinuation of treatment.10,69,72 Careful monitoring is warranted for the duration of therapy. However, the incidence of some grade 3 or 4 infections may decrease after a transition to every-2-weeks dosing.72 Approximately half of all viral infections in MajesTEC-1 occurred within the first 7 months, and most fungal infections occurred within the first 3 months.72 Most cases of P. jirovecii pneumonia occurred in patients who were not receiving prophylaxis.4,71 In MagnetisMM-3, higher exposure–adjusted infection rates were observed in patients with hypogammaglobulinemia and in those who did not receive immunoglobulin (Ig)G replacement therapy.74 If a hematologic AE is present, it can be managed with dose delays and supportive care strategies.59 Our institutions are generally in alignment with the principles of infection monitoring and prophylaxis available in published guidelines,6,20,59,68 which would also be reasonable models for implementation in community cancer centers.
The monitoring for and prevention of infection in patients receiving BCMA-directed bispecific antibodies include screening and watching for the signs and symptoms of infection. At baseline, serologic or molecular testing for Epstein-Barr virus, cytomegalovirus, hepatitis B virus, hepatitis C virus, and HIV should be considered and a CD4 count should be obtained. With every dose of BCMA-directed bispecific antibodies, a complete blood count with differential and complete metabolic panel should be obtained. At regular intervals, the patient’s IgG level (ie, monthly with MM response assessment) should be tested. If viral infection or reactivation is suspected, polymerase chain reaction–based viral panels should be obtained.
In patients receiving antimicrobial or antiviral prophylaxis, for those with bacterial infection, levofloxacin or an equivalent drug should be prescribed during periods of neutropenia; universal prophylaxis may be considered during step-up dosing, with early discontinuation in patients who do not have neutropenia. For patients with fungal infections, antiyeast prophylaxis can be used for the treatment of neutropenia, and antimold prophylaxis can be used if the duration of neutropenia is >2 weeks or if the patient received >1 dose of tocilizumab.
In patients with P. jirovecii pneumonia, treatment with trimethoprim-sulfamethoxazole or dapsone can be considered, and is used universally in patients receiving BCMA-directed bispecific antibodies. Pentamidine treatment should be considered for patients with cytopenias. In those with COVID-19 or influenza, seasonal vaccination of patients and their close contacts is recommended, and in patients with herpes simplex virus or varicella zoster virus, treatment with acyclovir or valacyclovir is recommended. In terms of supportive measures, for patients with neutropenia, growth factor support may be used for the treatment of neutropenia after the risk for CRS has passed, and in patients with hypogammaglobulinemia (IgG <400 mg/dL), patients should receive immunoglobulin replacement monthly (even off treatment) until the resolution of hypogammaglobulinemia.
Conclusion
In this article, we offer practical guidance for the management of patients receiving BCMA-directed bispecific antibodies, based on published data, expert recommendations, and our experience at our own academic institutions. We recognize, however, that infrastructure and resources available to an oncology care team vary by practice setting, and patients’ needs are as diverse as the patients themselves. Other considerations for disease management may include drug cost, reimbursement, hospitalization, and drug waste. Therefore, centers should tailor their approach with the considerations listed here, based on their existing resources and individual patient needs. As we gain more familiarity with these novel agents, we must continue to share our experiences, report real-world outcomes, and collaborate on management guidelines to increase comfort level across the oncology community and to ensure that effective therapies are accessible to all patients who can benefit from them.
Acknowledgment
Medical writing support was provided by Sam Million-Weaver, PhD, and Amy Pazzalia, MPAS, PA-C, on behalf of BOLDSCIENCE, and was funded by Pfizer. Editorial support was provided in accordance with Good Publication Practice guidelines.
Funding Source
This research was funded by Pfizer.
Author Disclosure
Dr Wagner is a consultant to Pfizer and has received research funding from Jazz; Dr Steinbach is on the Speakers Bureau for Bristol Myers Squibb, Janssen, and Pfizer, and is on the Advisory Board for Janssen and Pfizer; Mr Verducci is a consultant to Pfizer; and Dr Kowalski is a consultant to Pfizer.
References
- Dhanasiri S, Hollier-Hann G, Stothard C, et al. Treatment patterns and outcomes in triple-class exposed patients with relapsed and refractory multiple myeloma: findings from the multinational ITEMISE study. Clin Ther. 2021;43:1983-1996.
- O’Neill C, van de Donk NWCJ. T-cell redirecting bispecific antibodies in multiple myeloma: current landscape and future directions. eJHaem. 2023;4:811-822.
- Madduri D, Hagiwara M, Parikh K, et al. Real-world treatment patterns, healthcare use and costs in triple-class exposed relapsed and refractory multiple myeloma patients in the USA. Future Oncol. 2021;17:503-515.
- Lesokhin AM, Tomasson MH, Arnulf B, et al. Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results. Nat Med. 2023;29:2259-2267.
- Moreau P, Garfall AL, van de Donk NWCJ, et al. Teclistamab in relapsed or refractory multiple myeloma. N Engl J Med. 2022;387:495-505.
- Ludwig H, Terpos E, van de Donk NWCJ, et al. Prevention and management of adverse events during treatment with bispecific antibodies and CAR T cells in multiple myeloma: a consensus report of the European Myeloma Network. Lancet Oncol. 2023;24:e255-e269.
- Lee DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25:625-638.
- Elrexfio (elranatamab-bcmm) injection, for subcutaneous use [prescribing information]. Pfizer Inc; July 2025. Accessed July 17, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2023/761345Orig1s000lbl.pdf
- Tecvayli (teclistamab-cqyv) injection, for subcutaneous use [prescribing information]. Janssen Biotech, Inc; November 2024. Accessed June 3, 2025. www.janssenlabels.com/package-insert/product-monograph/prescribing-information/TECVAYLI-pi.pdf
- Berdeja JG. Multiple myeloma: a paradigm for blending community and academic care. Hematol Am Soc Hematol Educ Program. 2023;2023:318-323.
- Kazandjian D, Kowalski A, Landgren O. T cell redirecting bispecific antibodies for multiple myeloma: emerging therapeutic strategies in a changing treatment landscape. Leuk Lymphoma. 2022;63:3032-3043.
- Abramson HN. B-cell maturation antigen (BCMA) as a target for new drug development in relapsed and/or refractory multiple myeloma. Int J Mol Sci. 2020;21:5192.
- Leclercq-Cohen G, Steinhoff N, Albertí Servera L, et al. Dissecting the mechanisms underlying the cytokine release syndrome (CRS) mediated by T-cell bispecific antibodies. Clin Cancer Res. 2023;29:4449-4463.
- Shimabukuro-Vornhagen A, Gödel P, Subklewe M, et al. Cytokine release syndrome. J Immunother Cancer. 2018;6:56.
- Shah N, Chari A, Scott E, et al. B-cell maturation antigen (BCMA) in multiple myeloma: rationale for targeting and current therapeutic approaches. Leukemia. 2020;34:985-1005.
- Yu B, Jiang T, Liu D. BCMA-targeted immunotherapy for multiple myeloma. J Hematol Oncol. 2020;13:125.
- Cho SF, Anderson KC, Tai YT. Targeting B cell maturation antigen (BCMA) in multiple myeloma: potential uses of BCMA-based immunotherapy. Front Immunol. 2018;9:1821.
- Zhou X, Rasche L, Kortüm KM, et al. BCMA loss in the epoch of novel immunotherapy for multiple myeloma: from biology to clinical practice. Haematologica. 2023;108:958-968.
- Mohan M, Nagavally S, Dhakal B, et al. Risk of infections with B-cell maturation antigen-directed immunotherapy in multiple myeloma. Blood Adv. 2022;6:2466-2470.
- Raje N, Anderson K, Einsele H, et al. Monitoring, prophylaxis, and treatment of infections in patients with MM receiving bispecific antibody therapy: consensus recommendations from an expert panel. Blood Cancer J. 2023;13:116.
- ClinicalTrials.gov. Dose escalation study of teclistamab, a humanized BCMA*CD3 bispecific antibody, in participants with relapsed or refractory multiple myeloma (MajesTEC-1). NCT03145181. Accessed June 23, 2025. https://clinicaltrials.gov/study/NCT03145181
- ClinicalTrials.gov. A study of teclistamab in participants with relapsed or refractory multiple myeloma (MajesTEC-1). NCT04557098. Accessed June 23, 2025. https://clinicaltrials.gov/study/NCT04557098
- ClinicalTrials.gov. MagnetisMM-3: study of elranatamab (PF-06863135) monotherapy in participants with multiple myeloma who are refractory to at least one PI, one IMiD and one anti-CD38 mAb. NCT04649359. Accessed June 23, 2025. https://clinicaltrials.gov/study/NCT04649359
- Searle E, Quach H, Wong SW, et al. Teclistamab in combination with subcutaneous daratumumab and lenalidomide in patients with multiple myeloma: results from one cohort of MajesTEC-2, a phase 1b, multicohort study. Blood. 2022;140(suppl 1):394-396.
- Grosicki S, Crafoord J, Koh Y, et al. MagnetisMM-5: an open-label, multicenter, randomized phase 3 study of elranatamab as monotherapy and in combination with daratumumab in patients with relapsed/refractory multiple myeloma. J Clin Oncol. 2022;40(16 suppl):Abstract TPS8074.
- Chen X, Kamperschroer C, Wong G, Xuan D. A modeling framework to characterize cytokine release upon T-cell-engaging bispecific antibody treatment: methodology and opportunities. Clin Transl Sci. 2019;12:600-608.
- Elmeliegy M, Viqueira A, Vandendries E, et al. Dose optimization to mitigate the risk of CRS with elranatamab in multiple myeloma. Blood. 2022;140(suppl 1):7174-7175.
- Bansal R, Paludo J, Corraes A, et al. Outpatient management of CAR-T and teclistamab for patients with lymphoma and multiple myeloma. Blood. 2023;142(suppl 1):253-255.
- Varshavsky-Yanovsky AN, Styler M, Khanal R, et al. P940: an outpatient model for teclistamab step-up dosing administration–initial experiences at Fox Chase Cancer Center BMT program. HemaSphere. 2023;7(suppl 3):1777-1778.
- Martin TG, Mateos MV, Nooka A, et al. Detailed overview of incidence and management of cytokine release syndrome observed with teclistamab in the MajesTEC-1 study of patients with relapsed/refractory multiple myeloma. Cancer. 2023;129:2035-2046.
- Usmani SZ, Karlin L, Benboubker L, et al. Durability of responses with biweekly dosing of teclistamab in patients with relapsed/refractory multiple myeloma achieving a clinical response in the majesTEC-1 study. J Clin Oncol. 2023;41(16 suppl):Abstract 8034.
- Tomasson MH, Iida S, Niesvizky R, et al. Long-term survival and safety of elranatamab in patients with relapsed or refractory multiple myeloma: update from the MagnetisMM-3 study. HemaSphere. 2024;8:e136.
- van de Donk NWCJ, Moreau P, Garfall AL, et al. Long-term follow-up from MajesTEC-1 of teclistamab, a B-cell maturation antigen (BCMA) x CD3 bispecific antibody, in patients with relapsed/refractory multiple myeloma (RRMM). J Clin Oncol. 2023;41(16 suppl):Abstract 8011.
- Tomasson MH, Iida S, Niesvizky R, et al. Long-term efficacy and safety of elranatamab monotherapy in the phase 2 MagnetisMM-3 trial in relapsed or refractory multiple myeloma. Poster presented at American Society of Hematology 2023 Annual Meeting and Exposition; December 9-12, 2023. Poster 3385.
- Mohty M, Iida S, Bahlis NJ, et al. Long-term survival after elranatamab monotherapy in patients with relapsed or refractory multiple myeloma (RRMM): MagnetisMM-3. Presented at the European Hematology Association 2024 Meeting. Abstract P932.
- Garfall AL, Nooka AK, van de Donk NWCJ, et al. Long-term follow-up from the phase 1/2 MajesTEC-1 trial of teclistamab in patients with relapsed/refractory multiple myeloma. J Clin Oncol. 2024;42(16 suppl):Abstract 7540.
- Prince HM, Bahlis NJ, Rodriguez-Otero P, et al. MagnetisMM-3: long-term update and efficacy and safety of less frequent dosing of elranatamab in patients with relapsed or refractory multiple myeloma. Blood. 2024;144:4738-4739.
- Tomasson M, Iida S, Niesvizky R, et al. Long-term efficacy and safety of elranatamab monotherapy in the phase 2 Magnetismm-3 trial in relapsed or refractory multiple myeloma (RRMM). Blood. 2023;142(suppl 1):3385-3386.
- Touzeau C, Krishnan AY, Moreau P, et al. Efficacy and safety of teclistamab in patients with relapsed/refractory multiple myeloma after BCMA-targeting therapies. Blood. 2024;144:2375-2388.
- Nooka AK, Lesokhin AM, Mohty M, et al. Efficacy and safety of elranatamab in patients with relapsed/refractory multiple myeloma (RRMM) and prior B-cell maturation antigen (BCMA)-directed therapies: a pooled analysis from MagnetisMM studies. Oral presentation given at the American Society for Hematology 2023 Annual Meeting & Exposition; December 9-12, 2023:Abstract 8008.
- Gordon B, Fogel L, Varma G, et al. Teclistamab demonstrates clinical activity in real-world patients ineligible for the pivotal Majestec-1 trial. Blood. 2023;142(suppl 1):4741-4743.
- Mohan M, Shah N, Luan D, et al. Teclistamab in relapsed refractory multiple myeloma: multi-institutional real-world study. Blood. 2023;142(suppl 1):545-546.
- Riedhammer C, Bassermann F, Besemer B, et al. Real-world analysis of teclistamab in 123 RRMM patients from Germany. Leukemia. 2024;38:365-371.
- Dima D, Davis JA, Ahmed N, et al. Real-world safety and efficacy of teclistamab for patients with heavily pretreated relapsed-refractory multiple myeloma. Blood. 2023;142(suppl 1):91-93.
- Grajales-Cruz AF, Castaneda O, Hansen DK, et al. Teclistamab induces favorable responses in patients with relapsed and refractory multiple myeloma after prior BCMA-directed therapy. Blood. 2023;142(suppl 1):3351-3353.
- Kowalski A, Lykon J, Diamond B, et al. Tocilizumab prophylaxis for patients with relapsed or refractory multiple myeloma treated with teclistamab, elranatamab or talquetamab. Blood. 2024;144(suppl 1):932-933.
- Kowalski A, Lykon JL, Diamond B, et al. Tocilizumab prophylaxis for patients treated with teclistamab: a single-center experience. Blood. 2023;142(suppl 1):4709-4710.
- Scott SA, Marin EM, Maples KT, et al. Prophylactic tocilizumab to prevent cytokine release syndrome (CRS) with teclistamab: a single-center experience. Blood Cancer J. 2023;13:191.
- van de Donk NWCJ, Garfall AL, Benboubker L, et al. Evaluation of prophylactic tocilizumab (toci) for the reduction of cytokine release syndrome (CRS) to inform the management of patients (pts) treated with teclistamab in MajesTEC-1. J Clin Oncol. 2023;41(16 suppl):Abstract 8033.
- Gritti G, Belousov A, Relf J, et al. Predictive model for the risk of cytokine release syndrome with glofitamab treatment for diffuse large B-cell lymphoma. Blood Adv. 2024;8:3615-3618.
- Thompson JA, Schneider BJ, Brahmer J, et al. NCCN Guidelines® Insights: management of immunotherapy-related toxicities, Version 2.2024. J Natl Compr Canc Netw. 2024;22:582-592.
- Sandahl TB, Soefje SA, Fonseca R, et al. Real-world safety and health care resource utilization of teclistamab under an outpatient model for step-up dosing administration. JCO Oncol Pract. 2025;21:702-709.
- Varga C, Ahmed F, Knight M, et al. Prophylactic dexamethasone and remote monitoring for patients with relapsed refractory multiple myeloma (RRMM) receiving bispecific antibodies (BsAb): experience at a single institution. Blood. 2024;144(suppl 1):4720.
- Hebraud B, Granell M, Lapierre L, et al. French monocentric experience of outpatient step-up dosing of teclistamab in relapsed refractory multiple myeloma. Blood. 2023;142(suppl 1):4736.
- Zhang Y, Zhou F, Wu Z, et al. Timing of tocilizumab administration under the guidance of IL-6 in CAR-T therapy for R/R acute lymphoblastic leukemia. Front Immunol. 2022;13:914959.
- Jadot G, LeBlanc R, Ahmad I, et al. Tocilizumab prophylaxis for outpatient administration of teclistamab in relapsed/refractory multiple myeloma. J Clin Oncol. 2025;43(16 suppl):e19504.
- Korst C LBM, Groen K, Bosman PWC, et al. Prophylactic tocilizumab reduces the incidence of cytokine release syndrome in relapsed/refractory myeloma patients treated with teclistamab: implications for outpatient step-up dosing. Hemasphere. 2024;8:e132.
- Trudel S, Bahlis NJ, Spencer A, et al. Pretreatment with tocilizumab prior to the CD3 bispecific cervostamab in patients with relapsed/refractory multiple myeloma (RRMM) showed a marked reduction in cytokine release syndrome incidence and severity. Blood. 2022;140(suppl 1):1363-1365.
- Rodriguez-Otero P, Usmani S, Cohen AD, et al. International Myeloma Working Group immunotherapy committee consensus guidelines and recommendations for optimal use of T-cell-engaging bispecific antibodies in multiple myeloma. Lancet Oncol. 2024;25:e205-e216.
- Niesvizky R, Arnulf B, Mohty M, et al. Clinical factors associated with cytokine release syndrome and dosing recommendations for restarting elranatamab following an interruption. Blood. 2023;142(suppl 1):3384-3386.
- Zhou J, Vishwamitra D, Guo Y, et al. Model-based exploration of the impact of prophylactic tocilizumab on IL-6 dynamics in multiple myeloma patients receiving teclistamab treatment. Blood. 2023;142(suppl 1):4670-4671.
- Rees JH. Management of Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS). In the EBMT/EHA CAR-T Cell Handbook [internet]. Kröger N, Gribben J, Chabannon C, et al, editors. Springer; 2022.
- Carvykti (ciltacabtagene autoleucel) suspension for intravenous infusion [prescribing information]. Janssen Biotech; April 2024. Accessed July 7, 2025. www.janssenlabels.com/package-insert/product-monograph/prescribing-information/CARVYKTI-pi.pdf
- Abecma (idecabtagene vicleucel) suspension for intravenous infusion [prescribing information]. Celgene Corporation; March 2025. Accessed July 7, 2025. https://packageinserts.bms.com/pi/pi_abecma.pdf
- Ludwig H, Delforge M, Facon T, et al. Prevention and management of adverse events of novel agents in multiple myeloma: a consensus of the European Myeloma Network. Leukemia. 2018;32:1542-1560.
- Rosenbaum E, Marks D, Raza S. Diagnosis and management of neuropathies associated with plasma cell dyscrasias. Hematol Oncol. 2018;36:3-14.
- Blimark C, Holmberg E, Mellqvist UH, et al. Multiple myeloma and infections: a population-based study on 9253 multiple myeloma patients. Haematologica. 2015;100:107-113.
- Mohan M, Chakraborty R, Bal S, et al. Recommendations on prevention of infections during chimeric antigen receptor T-cell and bispecific antibody therapy in multiple myeloma. Br J Haematol. 2023;203:736-746.
- Leblay N, Maity R, Hasan F, Neri P. Deregulation of adaptive T cell immunity in multiple myeloma: insights into mechanisms and therapeutic opportunities. Front Oncol. 2020;10:636.
- Caro J, Braunstein M, Williams L, et al. Inflammation and infection in plasma cell disorders: how pathogens shape the fate of patients. Leukemia. 2022;36:613-624.
- Nooka AK, Rodriguez C, Mateos MV, et al. Incidence, timing, and management of infections in patients receiving teclistamab for the treatment of relapsed/refractory multiple myeloma in the MajesTEC-1 study. Cancer. 2024;130:886-900.
- Reynolds G, Cliff ERS, Mohyuddin GR, et al. Infections following bispecific antibodies in myeloma: a systematic review and meta-analysis. Blood Adv. 2023;7:5898-5903.
- Philipp N, Kazerani M, Nicholls A, et al. T-cell exhaustion induced by continuous bispecific molecule exposure is ameliorated by treatment-free intervals. Blood. 2022;140:1104-1118.
- Leleu X, Bahlis N, Rodriguez-Otero P, et al. P-286 Impact of hypogammaglobulinemia and immunoglobulin replacement therapy on infection rate in patients with RRMM treated with elranatamab: post-hoc analysis from MagnetisMM-3. Clin Lymphoma Myeloma Leuk. 2023;23:S194.