Colleen McCabe, PharmD, BCOP, and Lindsay Mundy, PharmD, BCOP, led an engaging exploration of the benefits and potential pitfalls with the use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) in an oncology setting in their presentation, GLP-1s in Oncology: Miracle, Misdirection, or Missed Opportunity? This presentation was featured at the 2026 HOPA annual conference, March 25-27, in New Orleans, LA.
Early research targeting GLP-1 with oral glucose showed that it resulted in greater increases in insulin release and decreases in glucagon secretion compared with intravenous glucose. Further research was disappointing, however, because GLP-1 RAs had a half-life of only 1 to 2 minutes. Additional research centered on making it a more druggable target with a longer half-life, and in 2005, a half-life of approximately 2.4 hours was achieved, which allowed for subcutaneous (SC) dosing twice a day. In 2010, these agents became available in daily SC formulation; in 2014, a weekly SC formulation was developed, and the indication for weight loss was approved by the FDA. In 2021, the oral formulation of semaglutide became available, and this new dosing option resulted in much greater uptake.
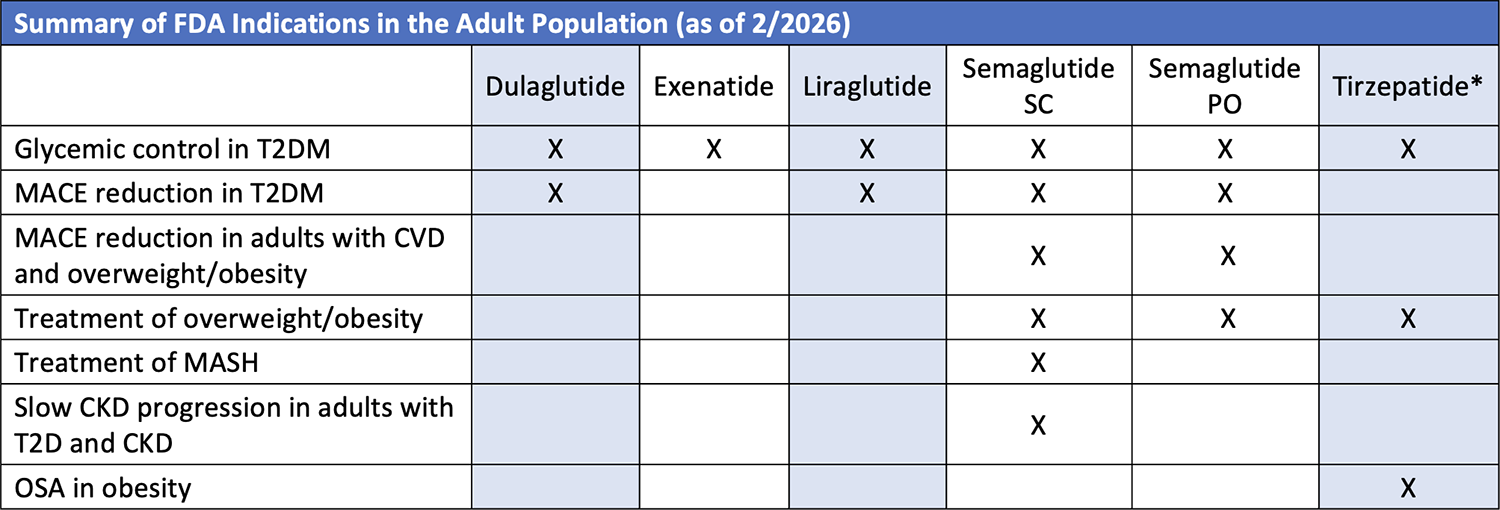
Now, what was originally developed for glucose control has demonstrated decreased gastric emptying, increased satiety, and decreased appetite; furthermore, weight loss of 5% to 19% of body weight, depending on the agent, has been documented. Currently, these medications are being assessed for cardiac, renal, and potentially even neuroprotective effects (Table 1). In this sense, they may be thought of as disease-modifying agents instead of glucose-lowering therapy.
Table 1. GLP-1 RAs: FDA Indications as of February 2026
*Combination GLP-1 RA and glucose-dependent insulinotropic polypeptide RA.
Emerging Relevance of GLP-1 RAs in Oncology
Evidence is emerging that GLP-1 RAs have potential benefits in patients with cancer. GLP-1 RAs have shown antitumor effects via metabolic and immune modulation, reduced incidence of obesity-related cancers, and improved outcomes in some patients. However, these agents are associated with increased thyroid cancer risk, and the packaging for these medications in the United States has a boxed warning stating that they are contraindicated in people with a personal or family history of medullary thyroid cancer and in patients with a diagnosis of multiple endocrine neoplasia syndrome type 2.
It is important to note that the use of GLP-1 RAs for weight loss or metabolic control in people who are subsequently diagnosed with cancer pose potential risks. Adverse events such as diarrhea, constipation, nausea, and vomiting associated with the GLP-1 RAs overlap with those seen with cancer treatments, so the patient who is undergoing treatment with an emetogenic regimen for cancer will likely experience additional weight loss, which can lead to nutritional deficiencies, loss of lean muscle mass, and loss of bone mass. In these situations, the pharmacist can monitor the patient who is rapidly losing weight with an eye to closely watching weight-based chemotherapy dosing. In addition, bioavailability issues with oral chemotherapy can stem from the patient’s loss of appetite, and delayed gastric emptying can alter drug absorption. Finally, serious nausea and vomiting may result in lack of adherence to oral chemotherapy.
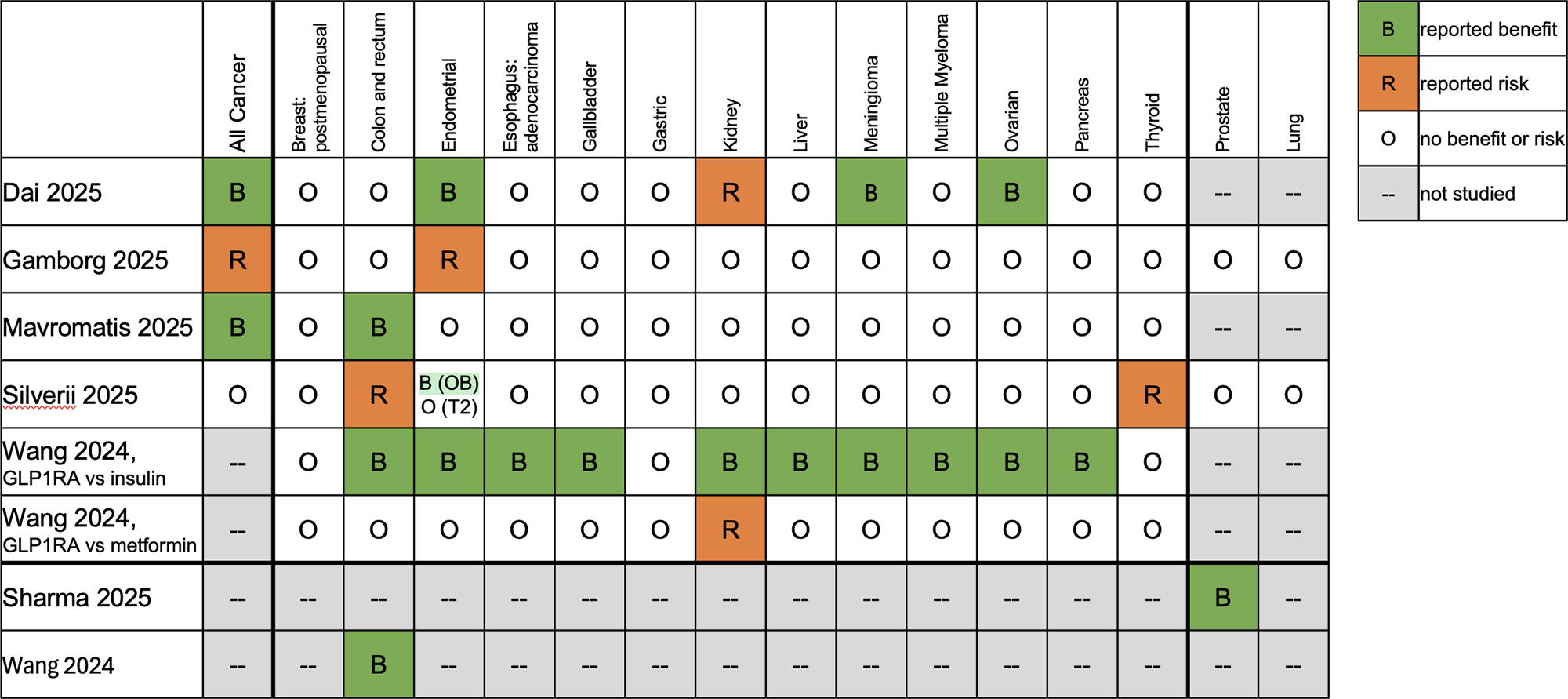
Although the evidence is as yet uncertain, weight loss associated with GLP-1 RAs in people without cancer may prevent obesity-related cancers. The International Agency for Research on Cancer (IARC) published a paper in 2016 that examined 1000 epidemiologic studies on the effect of body habitus on cancer risk. The researchers found that 13 obesity-related cancers showed sufficient evidence that weight loss can have a preventive effect. In addition, studies of bariatric surgery in overweight or obese individuals versus matched controls also showed decreased incidence of obesity-related cancers and decreased cancer-related mortality. Obesity-related cancers identified in the IARC study include endometrial, esophageal, renal cell, gastric, pancreas, colon/rectal, gallbladder, breast, ovarian, and thyroid cancers as well as meningioma, multiple myeloma.
The literature on oncology patients is primarily focused on those with type 2 diabetes (Table 2). These studies were mostly retrospective and have limitations, including small sample size and detection bias. In addition, variable comparators such as metformin or insulin have important differences, because insulin is known to cause weight gain. The findings, however, seem to indicate that weight loss can have a beneficial effect on the incidence of obesity-related cancers, but it is too early to assert causality.
Table 2. Current Data in Oncology Population
GLP-1 RAs and Risk for Thyroid Cancer
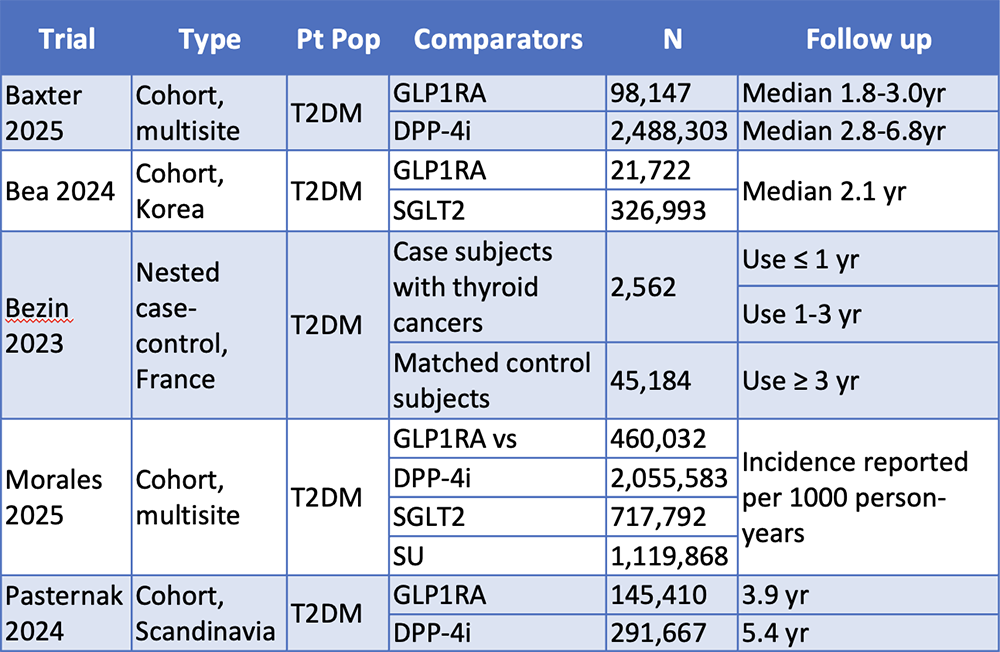
As previously stated, the packaging of GLP-1 RAs includes a boxed warning for thyroid cancers, but what can the studies in human participants tell us about this risk (Table 3)? Although Bezin and colleagues concluded that GLP-1 RAs increase the risk for all thyroid cancers and medullary thyroid cancer, this study also has limitations, including detection bias, confounding factors (obesity, family history, thyroid nodules), disproportionate number of patients with medullary thyroid cancer (15.5% vs <5% globally), lack of cumulative exposure risk, and selective reporting. Subsequently, 4 cohort studies showed no significant increase in thyroid cancers.
Table 3. Thyroid Cancer Risk: Human Studies
Shared Decision-Making: The Patient With Cancer Who Is Also Receiving a GLP-1 RA
It is important to discuss these potential adverse events (such as excess nausea and vomiting, nutritional deficiencies, changes in bioavailability of oral chemotherapies) with the patient newly diagnosed with cancer who is receiving a GLP-1 RA before initiating chemotherapy to determine the optimal treatment plan. The most common plan of action has been to hold the GLP-1 RA for at least the first cycle to monitor for unwanted effects. If appropriate, the weight-loss drug can be added back. Some patients, however, elect to continue the GLP-1 RA because the thought of regaining weight is unacceptable.
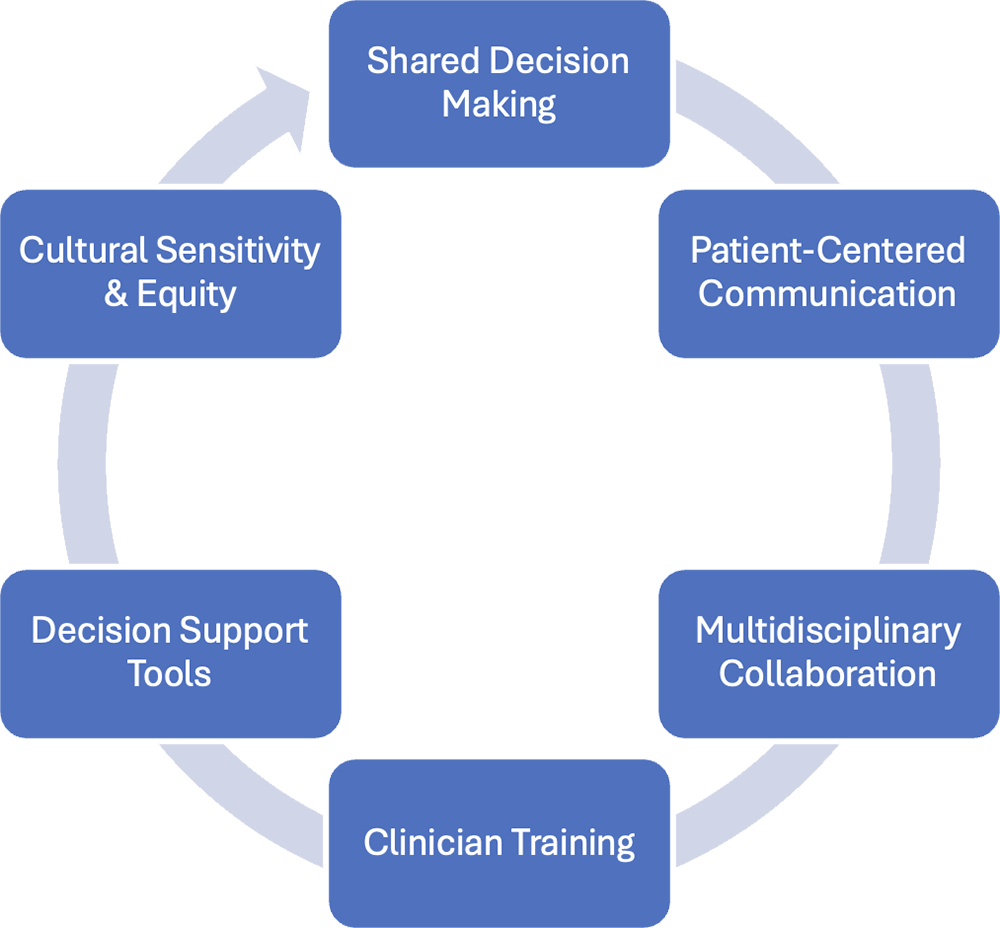
Key points in a shared decision-making strategy (Figure) include patient-centered communication to ensure that the patient who may be reluctant to discontinue the GLP-1 RA feels heard and like an equal contributor to the decision. Multidisciplinary collaboration is also important. For example, a nutritionist is well positioned to educate the patient on the potential for malnutrition, and in the event the patient desires to continue the GLP-1 RA, how that decision may unfold. Clinician training is also important to ensure that pharmacists can navigate these challenging discussions. Possible discussion points may include, “We want to make sure that your treatment plan aligns with your goals, which includes managing your weight safely while undergoing chemotherapy. When you’re receiving highly emetogenic chemotherapy, we expect weight loss.” The use of decision-support tools, such as visual aids or summary sheets, during these discussions is also beneficial. Cultural sensitivity and equity in this scenario entail being respectful of the patient’s attitude toward the use of the GLP-1 RA, which can be an emotional topic in some.
Figure. Shared Decision-Making in Oncology
The ultimate goals in shared decision-making are a comprehensive, transparent discussion of risks and benefits of each treatment option, including an individualized approach that balances the metabolic, weight loss, and oncologic priorities; a decision-making approach that is centered around the patient; clinician training to meet the challenges inherent in difficult discussions; and cultural sensitivity and equity.
Sources
Zhao X, et al. Front Endocrinol. 2021;12.
Alicic RZ, et al. Curr Opin Nephrol Hypertens. 2023;32:377-385.
J.P. Morgan. The increase in appetite for obesity drugs. 2023.
Trulicity (dulaglutide) [prescribing information]. Eli Lilly and Company. 2025.
Byetta (exenatide) [prescribing information]. Amylin Pharmaceuticals, Inc. 2009.
Victoza (liraglutide) [prescribing information]. Novo Nordisk Inc. 2017.
Ozempic (semaglutide) [prescribing information]. Novo Nordisk Inc. 2025.
Rybelsus (semaglutide) [prescribing information]. Novo Nordisk Inc. 2025.
Wegovy (semaglutide) [prescribing information]. Novo Nordisk Inc. 2025.
Mounjaro (tirzepatide) [prescribing information]. Eli Lilly and Company. 2025.
Zepbound (tirzepatide) [prescribing information]. Eli Lilly and Company. 2025.
Saxenda (liraglutide) [prescribing information]. Novo Nordisk Inc. 2026.
Morales DR, et al. Diabetes Care. 2025;48:1386-1394.
Aminian A, et al. JAMA. 2022;327:2423-2433.
Lauby-Secretan B, et al. N Engl J Med. 2016;375:794-798.
Dai H, et al. JAMA Oncol. 2025;11:1186-1193.
Gamborg M, et al. Lancet Reg Health Eur. 2025;55:101346.
Mavromatis LA, et al. Diabetes Obes Metab. 2025;27:4571-4575.
Silverii GA, et al. Diabetes Obes Metab. 2025;27:4454-4468.
Wang L, et al. JAMA Netw Open. 2024;7:e2412345.
Sharma N, et al. Int Urol Nephrol. 2025;57:1039-1049.
Wang L, et al. JAMA Oncol. 2024;10:256-258.
Bjerre Knudsen L, et al. Endocrinology. 2010;151:1473-1486.
Bulchandani D, et al. Eur J Pharmacol. 2012;691:292-296.
Byrd RA, et al. Endocrinology. 2015;156:2417-2428.
Vahle JL, et al. Endocrinology. 2015;156:2409-2416.
Baxter SM, et al. Thyroid. 2025;35:69-78.
Bea S, et al. Diabetes Obes Metab. 2024;26:108-117.
Bezin J, et al. Diabetes Care. 2023;46:384-390.
Morales DR, et al. Diabetes Care. 2025;48:1386-1394.
Pasternak B, et al. BMJ. 2024;385:e078225.
Thompson CA, et al. Diabetes Care. 2023;46:249-251.
Mozaffarian D, et al. Obesity. 2025;33:1475-1503.
Heersche N, et al. Cancer Commun. 2025;45:9114-9118.
Gilligan T, et al. J Clin Oncol. 2017;35:3618-3632.