Connecting Science to Practice
Pegaspargase is a critical chemotherapy drug for treating pediatric
acute lymphoblastic leukemia, but allergic reactions
can necessitate that patients be switched to alternative medications
that require more frequent dosing and may be in
short supply. This study evaluated whether giving premedications
and monitoring drug levels could reduce medication
switches. Although the premedication strategy did not significantly
reduce medication switches or allergic reactions,
reactions occurred later in treatment, potentially saving patients
additional doses of alternative drugs. These findings
may help institutions understand which premedication
strategies work best, but continued protocol refinement to
optimize patient care and reduce healthcare costs is needed.
Pegaspargase is a modified, pegylated version of L-asparaginase, an enzyme that causes the depletion of asparagine in leukemic cells, which ultimately leads to the inhibition of protein synthesis and apoptosis.1 Pegaspargase is an essential component of multiagent chemotherapy that is used for the first-line treatment of acute lymphoblastic leukemia (ALL) in pediatric patients and adults.1,2 Asparaginase drugs can cause antibody-mediated clinical hypersensitivity reactions (eg, urticaria, dizziness, respiratory symptoms) that may be associated with asparaginase-neutralizing antibodies.2-5 Reaction severity has not been correlated with the risk for neutralizing antibodies.2-4 In addition, patients can present with neutralizing antibodies that result in asparaginase inactivity without any clinical symptoms of a reaction, which is known as silent inactivation.2-4 Although the incidence of silent inactivation is low (approximately 0%-8% of treated patients), detection is paramount because neutralizing antibodies can render asparaginase therapy ineffective.6
Asparaginase drugs can also cause a distinct non–antibody-mediated, nonallergic infusion reaction that may result from spikes in serum ammonia levels, a byproduct of the asparaginase enzyme–catalyzed reaction, which can be difficult to distinguish from an antibody-mediated clinical hypersensitivity reaction.3-5,7 Despite this risk, premedication has historically been avoided before pegaspargase administration for fear of masking hypersensitivity reactions.2 Therapeutic drug monitoring (TDM) using serum asparaginase activity (SAA) levels has increased the ability to differentiate among clinical hypersensitivity, subclinical hypersensitivity (silent inactivation), and non–antibody-mediated infusion reactions.2-4,7 Although goal SAA levels can be debated depending on the collection time frame, in general, therapy adjustment is recommended if an SAA level collected 3 to 7 days after infusion results is <0.1 IU/mL.2-4
Because of TDM with SAA, we no longer rely on reactions being the only sign of antibody development and premedications can be considered without hesitation in all patients before intravenous (IV) pegaspargase administration.2,3 Premedications can reduce the incidence and severity of adverse events (AEs), as well as the need to switch to asparaginase Erwinia chrysanthemi or asparaginase E. chrysanthemi (recombinant)-rywn.2 This is important because asparaginase E. chrysanthemi was historically on prolonged shortage, and both asparaginase E. chrysanthemi and asparaginase E. chrysanthemi (recombinant)-rywn require 6 intramuscular doses for every 1 dose of pegaspargase.2
The implementation of TDM with SAA and premedication strategies have varied across institutions. The University of Michigan Medicine implemented a universal premedication policy requiring the administration of acetaminophen, diphenhydramine, and hydrocortisone before pegaspargase, which significantly reduced the need to switch from pegaspargase to asparaginase E. chrysanthemi, from 21% to 7%, with corresponding cost-savings.6 A similar premedication strategy (acetaminophen, diphenhydramine, and methylprednisolone) implemented at Children’s Minnesota showed a numerical reduction in grade 3 or 4 allergic reactions.8 Both institutions also implemented routine SAA monitoring.8 The Hospital for Sick Children in Toronto, Canada, chose a different strategy and used a clinical decision tree (continue, modify, premedicate, or discontinue therapy) to guide future pegaspargase administration based on SAA results.9 Utilizing this strategy, treatment with pegaspargase was continued in 98.9% of cases without requiring premedications.9 Of 129 patients in this study, only 1 presented with a grade 2 AE that subsequently required premedication before pegaspargase doses.9 This patient was able to continue pegaspargase treatment with the addition of diphenhydramine and famotidine, while also maintaining adequate asparaginase activity.9
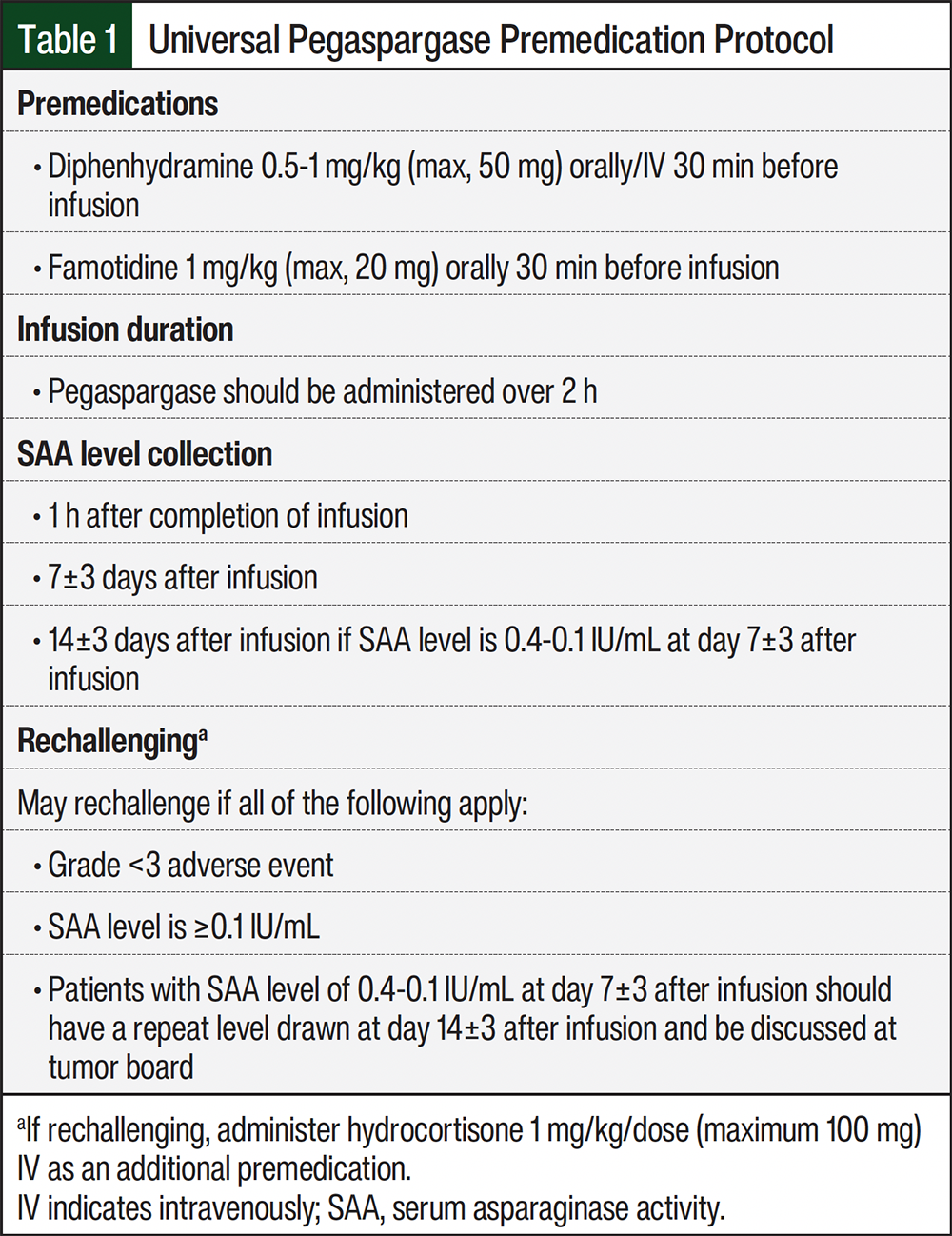
The pediatric oncology department at University of North Carolina Medical Center (UNCMC) implemented routine SAA TDM and pegaspargase premedication with diphenhydramine and famotidine in July 2019 (Table 1). This strategy was based on a study that showed that similar universal premedication reduced asparaginase E. chrysanthemi substitutions and AEs while maintaining pegaspargase SAA and increasing cost-savings.10 We did not elect to add steroids up front in hopes of avoiding unnecessary steroid use; instead, we recommended steroid treatment with rechallenges. In addition, at our institution the infusion duration of pegaspargase was prolonged from 1 to 2 hours. To confirm the efficacy of this TDM, premedication, and infusion duration strategy, we evaluated the incidence of AEs and asparaginase E. chrysanthemi switch rates in patients before and after the implementation of a premedication protocol.
Methods
This was a single-center, retrospective analysis approved by the institutional review board that included pediatric patients who received pegaspargase as part of their therapy for ALL at UNCMC between April 2014 and September 2021. The included patients received at least 1 dose of IV pegaspargase during the study period and had leukemia care provided by a pediatric oncology provider at UNCMC with documented follow-up. Patients were excluded from the analysis if they had a nonleukemia malignancy, they had natural killer cell leukemia, they were aged <1 year, or if pegaspargase was discontinued as a result of an AE that was not related to allergies or infusion reactions. The patients were divided based on whether they received pegaspargase before or after the implementation of the premedication protocol. A third group of patients received some pegaspargase doses before and some doses after the protocol implementation; these patients were not included in the primary outcome but were included in the secondary outcomes.
The primary outcome was the incidence of asparaginase E. chrysanthemi substitution before and after the protocol implementation. Of note, this study took place before asparaginase E. chrysanthemi (recombinant)-rywn became available. The secondary outcomes included the incidence of clinically apparent allergic reactions; the incidence of clinically apparent grade ≥3 allergic reactions, which was retroactively graded by the study team based on what was documented in the electronic medical record; and the incidence of pegaspargase rechallenge after a previous allergic reaction before and after the protocol implementation. The criteria to rechallenge, as depicted in Table 1, was if a patient had a grade <3 AE and the SAA level was ≥0.1 IU/mL. In addition, the rates of protocol adherence and of silent inactivation after the protocol was implemented were also assessed.
SAA levels were collected 1 hour after the completion of infusion and 7±3 days after infusion. They were considered therapeutic if they were ≥0.1 IU/mL. Because some data at the time suggested levels at even >0.1 IU/mL, we added a caveat for patients whose 7±3 days after infusion level was between 0.4 IU/mL and 0.1 IU/mL; they should have a 14±3 days level drawn and be discussed at our tumor board. In general, if the patient’s 14-day level decreased to <0.1 IU/mL, those patients would also be switched to asparaginase E. chrysanthemi. Clinically apparent allergic reactions were graded based on the Common Terminology Criteria for Adverse Events, Version 4.11
The categorical data were analyzed to assess for differences between the groups with the chi-square test or Fisher’s exact test. The continuous data were analyzed using the Analysis of Variance test.
Results
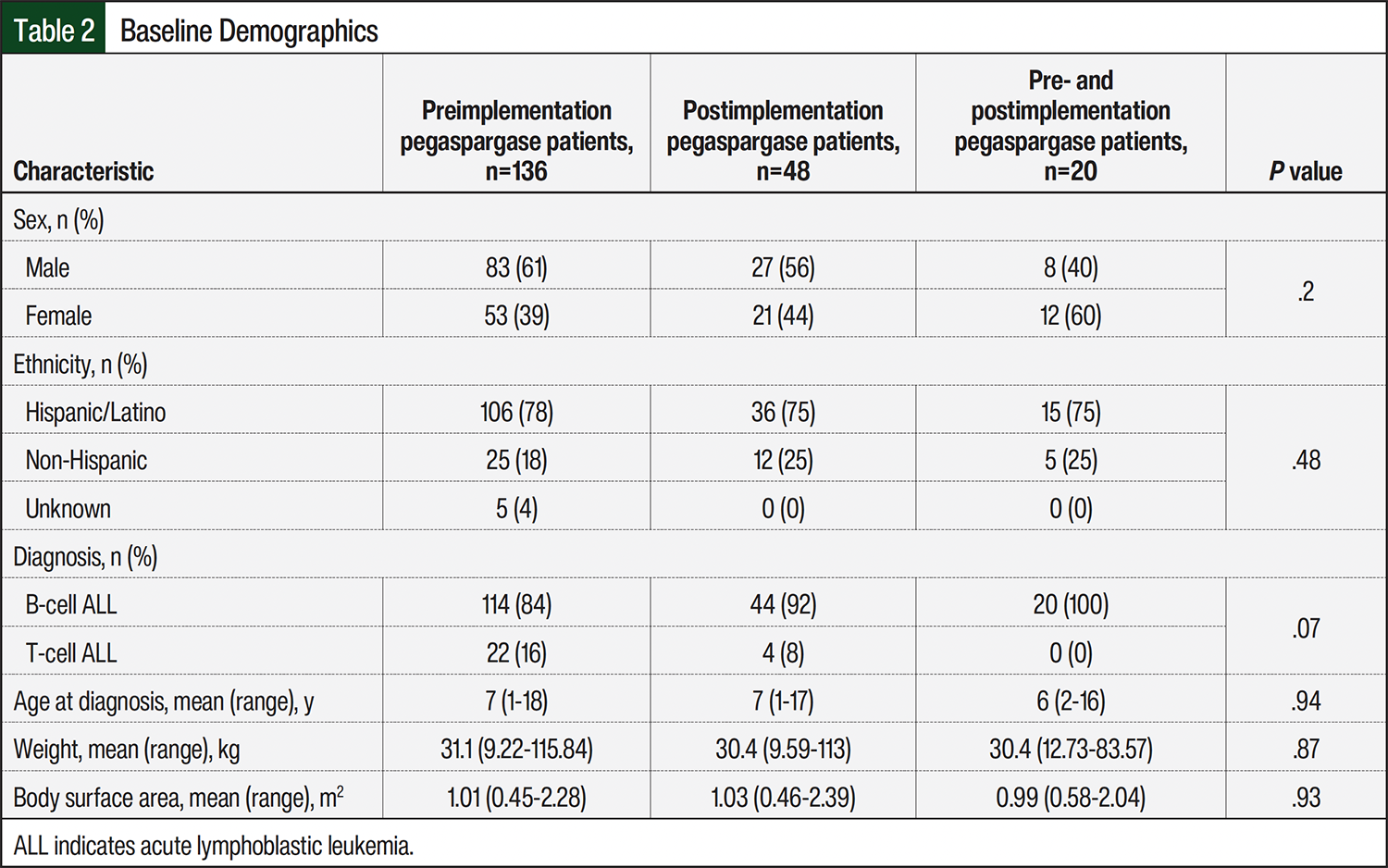
A total of 205 patients met the inclusion criteria. One patient was excluded because of being aged <1 year; therefore, a total of 204 patients were included in the analysis. Of the included patients, 136 (66%) received pegaspargase before the premedication protocol implementation, 48 (24%) received pegaspargase after the protocol implementation, and 20 (10%) received a mix of pegaspargase doses before and after the protocol implementation. The patients’ baseline demographics are shown in Table 2. The average age of the patients was 6.9 years. Most patients had B-cell ALL and identified as Hispanic or Latino. There were no statistical differences in the baseline characteristics between the groups.
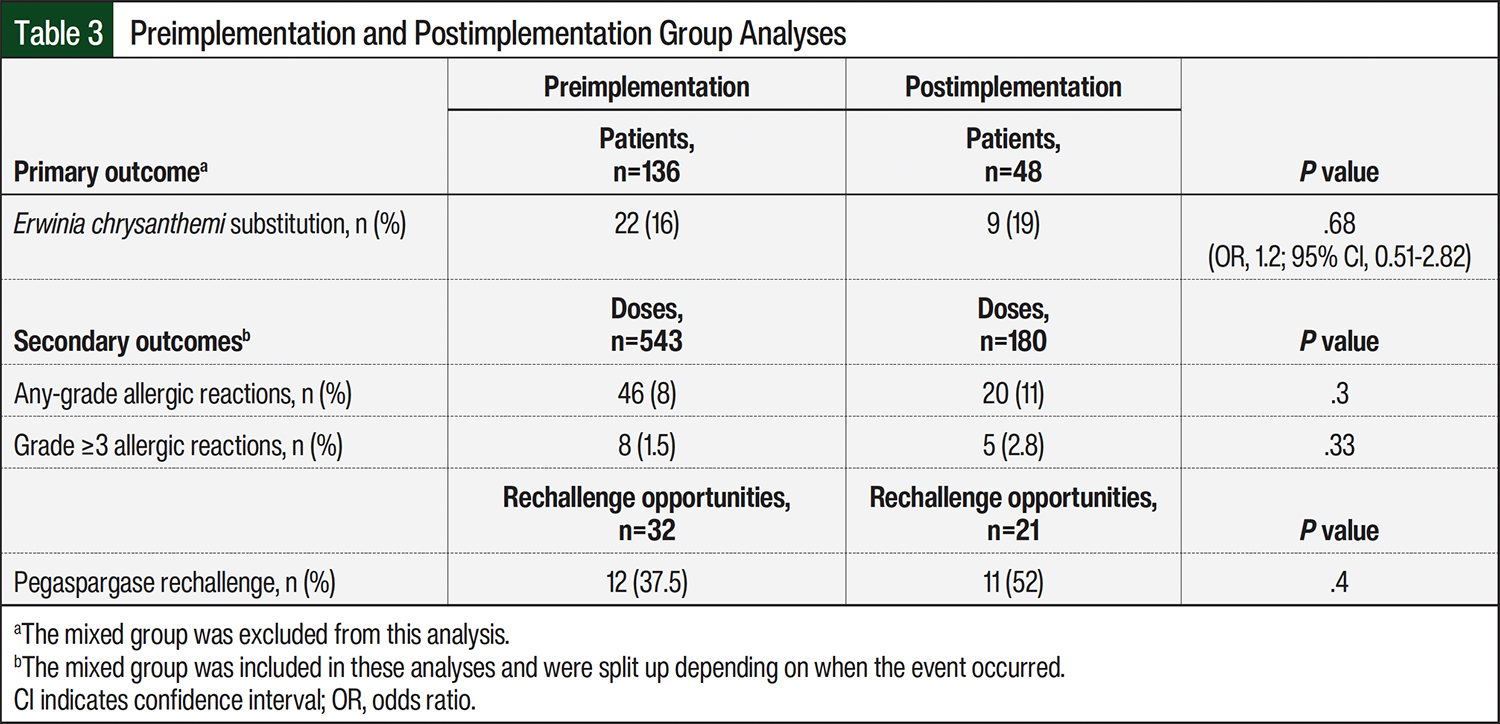
Table 3 shows the difference in outcomes between the groups. For the primary outcome, 22 (16%) patients were switched to asparaginase E. chrysanthemi after a pegaspargase hypersensitivity reaction before the protocol implementation versus 9 (19%) patients after the protocol implementation (odds ratio, 1.2; 95% confidence interval, 0.51-2.82; P=.68). For the secondary outcomes, 543 doses of pegaspargase were received before the protocol implementation versus 180 doses after the protocol implementation. Of those doses, 46 (8%) resulted in an allergic reaction of any grade before the protocol versus 20 (11%) doses after the protocol (P=.3). Of the 543 doses of pegaspargase given before protocol implementation, a grade ≥3 allergic reaction occurred after 8 pegaspargase doses (1.5%), whereas of the 180 doses of pegaspargase given after protocol implementation, a grade ≥3 allergic reaction occurred after 5 doses (2.8%; P=.33). These grade ≥3 reactions typically included hypotension, tachycardia, difficulty breathing, flushing, rash, nausea/vomiting, and less frequently, rigors.
A hypersensitivity reaction of any grade occurred most often during the second pegaspargase dose before the protocol (range, dose 1-5) versus the third dose after the protocol (range, dose 2-8). The average time to hypersensitivity reaction from the start of the infusion was 28 minutes before the protocol (range, 1-480 minutes; 1 patient had a reaction several hours after the infusion) versus 21 minutes after the protocol (range, 3-66 minutes). Of the 32 opportunities to rechallenge pegaspargase after a reaction before the protocol, 12 (37.5%) rechallenges occurred, whereas after the protocol there were 21 opportunities to rechallenge and 11 (52%) rechallenges (P=.4).
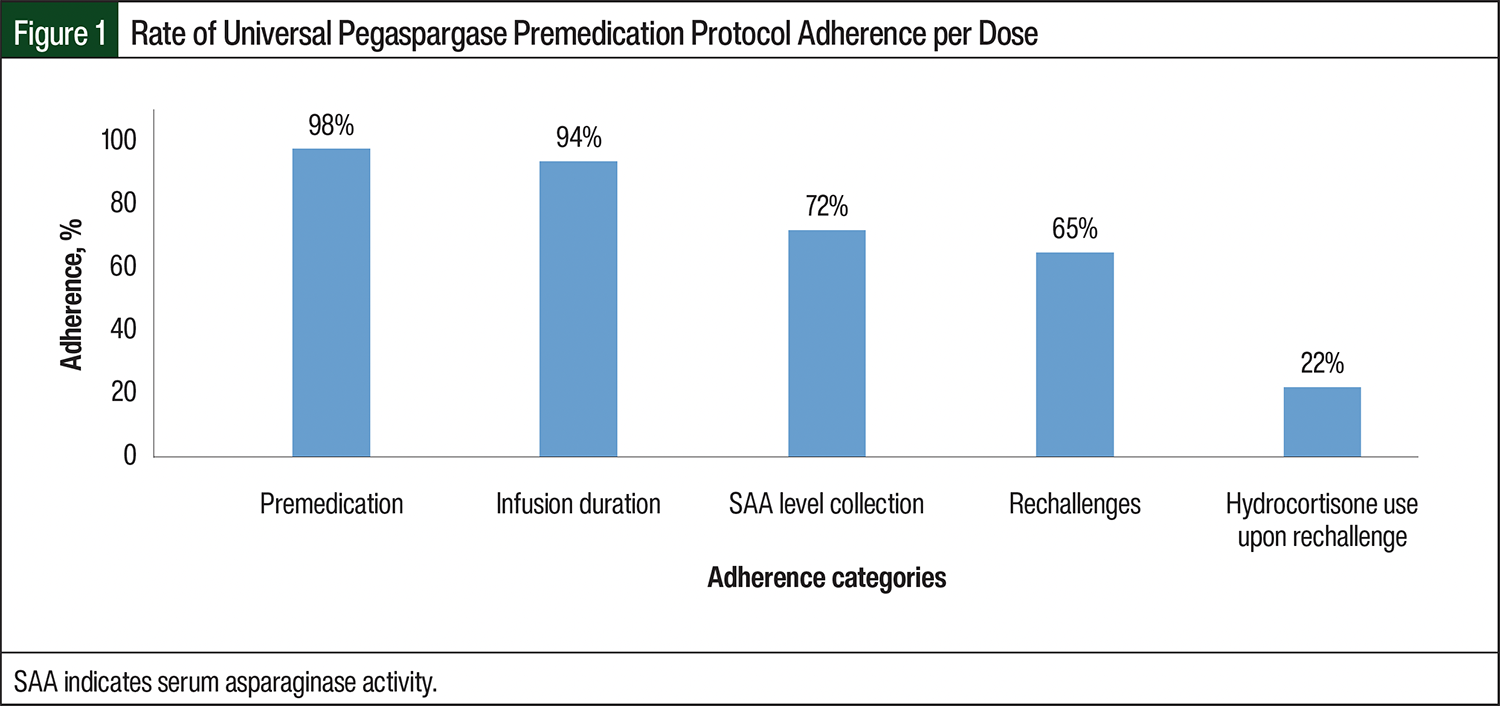
Figure 1 shows the adherence to our universal pegaspargase premedication protocol. The overall adherence rates were 98% for administering the correct premedications, 94% for the correct infusion duration, 72% for SAA level collection and/or timing, 65% to the appropriate rechallenging, and 22% to administering hydrocortisone on rechallenge.
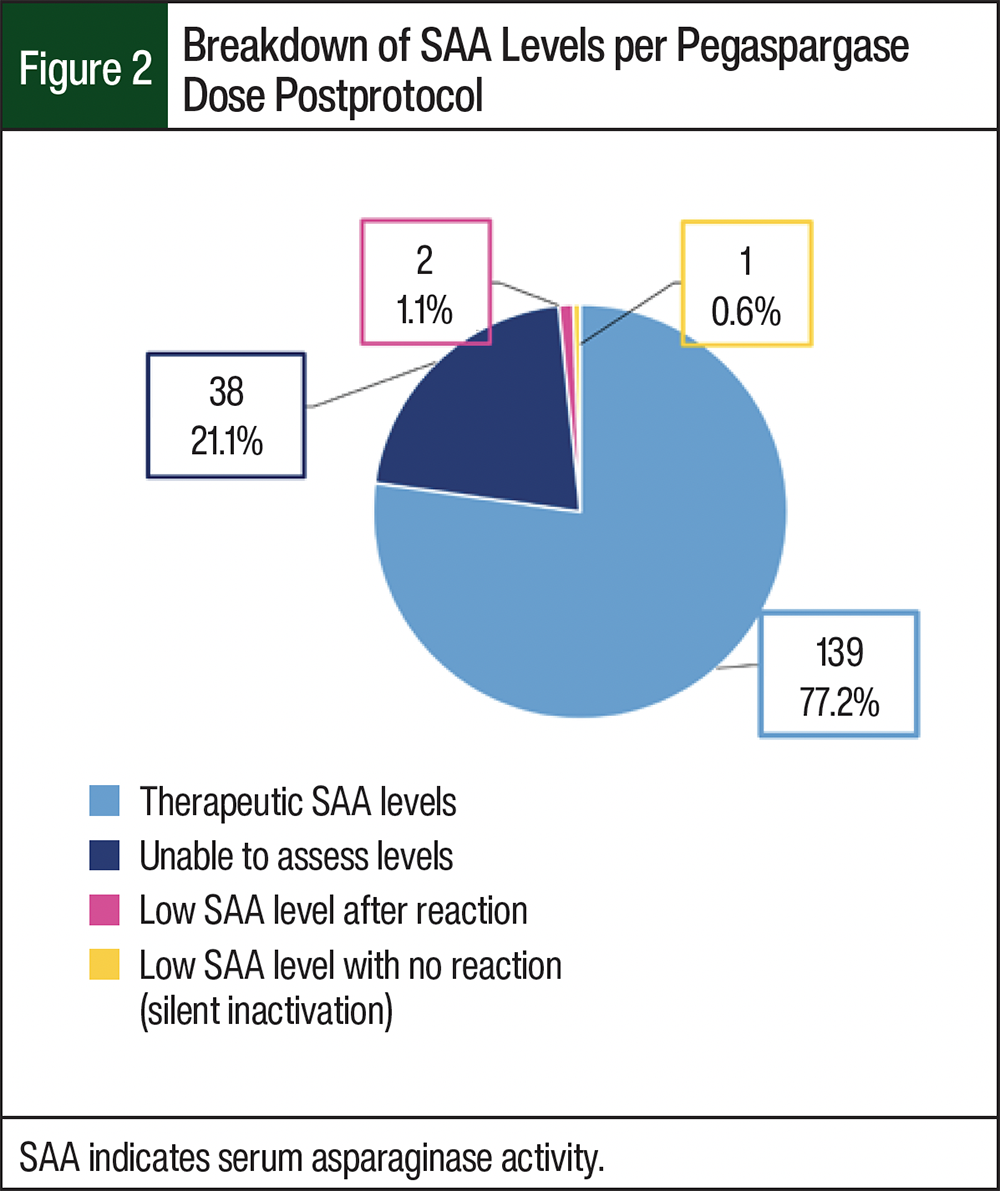
Figure 2 shows the breakdown of SAA levels after the protocol. A total of 139 (77.2%) levels resulted as therapeutic, 38 (21.1%) opportunities for levels were unable to be assessed because the levels were not collected at the appropriate times or because they were not collected at all, 2 (1.1%) levels were low after a documented hypersensitivity reaction, and 1 (0.6%) level resulted in silent inactivation (subtherapeutic level in the absence of signs or symptoms of hypersensitivity reaction).
Discussion
Based on the results of our study, a similar proportion of patients were switched to asparaginase E. chrysanthemi after a pegaspargase reaction before and after our protocol was implemented. In addition, we saw similar rates of overall and grade ≥3 hypersensitivity reactions in both study populations. One explanation for this is that this protocol is relatively new, only being recently implemented in 2019, and we had a lower number of subjects to observe in our postimplementation group. Our overall population was also relatively small given the rarity of pediatric cancer; thus, we might have been underpowered to detect any significant differences between our study groups. It is also possible that along with the awareness of the severe asparaginase E. chrysanthemi shortage and concomitant implementation of our protocol, clinicians were more vigilant in identifying and documenting pegaspargase reactions after 2019, leading to more identifications in the postimplementation group and possibly skewing our results.
As an exploratory analysis, it seems the time of reaction to pegaspargase was prolonged from the second dose to the third dose in patients after the protocol implementation while preserving our low rate of silent inactivation at 1.5% of patients after the protocol. Although our overall sample numbers are small and statistical significance testing was not performed on this observation, this trend could be a clinically relevant finding, because this extra effective administered dose of pegaspargase saves patients and the healthcare system 6 doses of asparaginase E. chrysanthemi. In addition, some patients following standard-risk protocols only receive 3 doses of pegaspargase in total, and therefore they may potentially avoid asparaginase E. chrysanthemi altogether. This saves money in terms of extra drug cost, as well as additional clinic visits and transportation costs. Future research with appropriate statistical analyses is necessary to confirm the meaning of this finding.
Regarding adherence to the protocol, excellent adherence rates were observed with the administration of premedications and infusion duration. The adherence rates were lower for SAA monitoring. In response to these lower rates, we have now made SAA levels part of our chemotherapy treatment plan builds in our electronic health record, which should decrease reliance on memory alone for obtaining these levels. Despite higher rates of rechallenge after a potential reaction in our postprotocol implementation group, our overall rate of adherence to the rechallenge criteria set forth in the protocol was low. It is possible that this nonadherence also inflated our overall switch rates in the postprotocol implementation group. In the future, we plan to amend our protocol to be more specific regarding rechallenge criteria in the hope of decreasing intraprovider variability in rechallenge patterns.
The results presented in our study may in part result from the premedications and infusion duration used. Many studies have added a corticosteroid and/or acetaminophen to their premedication protocols.6,8,12 In regard to the pegaspargase infusion duration, the University of Michigan administers 10% of the pegaspargase dose over the first hour of infusion and 90% over the second hour.6 Because our institution implemented and reviewed this protocol, another institution has also reviewed its premedication protocol, which consisted of a corticosteroid, histamine 2 receptor antagonist, and an antihistamine before pegaspargase.12 The other institution had a numerically lower—although not statistically significant—decrease in hypersensitivity reactions after the protocol implementation as well as a significant increase in cost-savings.12 The prescribing information for pegaspargase was also updated since the initial implementation of our premedication protocol, suggesting the use of acetaminophen, a histamine 1 receptor antagonist, and a histamine 2 receptor antagonist before doses of pegaspargase.1
The strengths of our study include the high adherence to our premedication and infusion duration guidelines within the protocol, as well as the use of SAA levels to monitor efficacy in patients receiving pegaspargase.
Limitations
This study has limitations. The limitations of this study include its single-center, retrospective nature; reliance on accurate clinician chart documentation; and its disproportionate numbers in our preprotocol and postprotocol implementation groups. In addition, we did not analyze whether patients were receiving steroids concomitantly with their chemotherapy at the time of the reaction, which could have impacted the risk for an AE.
Conclusion
We found similar rates of switching from pegaspargase to asparaginase E. chrysanthemi and similar rates of hypersensitivity reactions before and after our institutional pegaspargase protocol was implemented. However, the dose of pegaspargase at which these reactions occurred trended toward later administrations in the postimplementation group, potentially saving the patient and the healthcare system additional doses of asparaginase E. chrysanthemi. It is clear that additional measures may need to be taken and investigated to further prevent the need for use of alternative asparaginase drugs. These measures include the use of additional premedications (eg, acetaminophen and/or corticosteroids), the use of alternative infusion strategies (eg, titration of the infusion from slower to more rapid rates), and increased rechallenge attempts in eligible patients.
It may be prudent to involve a single interdisciplinary group in each decision to rechallenge and switch a patient from pegaspargase to an alternative drug to minimize intraprovider variability. Because strategies to prevent asparaginase hypersensitivity reactions differ vastly among institutions, as is reflected in the literature reviewed in this article, the results of this study can help direct institutions how to optimize their own protocols.
Acknowledgments
Emily Vanerian, PharmD Candidate, for their assistance with data collection, and Matthew Loop, PhD, FAHA, for their advisement regarding statistical analyses.
Author Disclosure Statement
Dr Wallace, Dr Dennison, Dr Auten, Dr Echols, Dr Phillips, and Dr Bognaski Kaplan have no conflicts of interest to report.
References
- Oncaspar (pegaspargase) injection, for intramuscular or intravenous use [prescribing information]. Servier Pharmaceuticals; March 2024. Accessed October 10, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2024/103411s5207lbl.pdf
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): pediatric acute lymphoblastic leukemia. Version 1.2026. August 11, 2025. Accessed October 10, 2025. www.nccn.org/professionals/physician_gls/pdf/ped_all.pdf
- van der Sluis IM, Vrooman LM, Pieters R, et al. Consensus expert recommendations for identification and management of asparaginase hypersensitivity and silent inactivation. Haematologica. 2016;101:279-285.
- Salzer W, Bostrom B, Messinger Y, et al. Asparaginase activity levels and monitoring in patients with acute lymphoblastic leukemia. Leuk Lymphoma. 2018;59:1797-1806.
- Burke MJ, Rheingold SR. Differentiating hypersensitivity versus infusion-related reactions in pediatric patients receiving intravenous asparaginase therapy for acute lymphoblastic leukemia. Leuk Lymphoma. 2017;58:540-551.
- Marini BL, Brown J, Benitez L, et al. A single-center multidisciplinary approach to managing the global Erwinia asparaginase shortage. Leuk Lymphoma. 2019;60:2854-2868.
- Würthwein G, Lanvers-Kaminsky C, Gerss J, et al. Therapeutic drug monitoring of asparaginase: intra-individual variability and predictivity in children with acute lymphoblastic leukemia treated with PEG-asparaginase in the AIEOP-BFM acute lymphoblastic leukemia 2009 study. Ther Drug Monit. 2020;42:435-444.
- Losasso M, Bostrom B, Messinger Y. Retrospective cohort study monitoring PEG-asparaginase activity in acute lymphoblastic leukemia patients with and without premedication. F1000Res. 2019;8:1007.
- Nadeem K, Colantonio D, Kircanski I, et al. Clinical decisions following implementation of asparaginase activity monitoring in pediatric patients with acute lymphoblastic leukemia: experience from a single-center study. Pediatr Blood Cancer. 2020;67:e28044.
- Cooper SL, Young DJ, Bowen CJ, et al. Universal premedication and therapeutic drug monitoring for asparaginase-based therapy prevents infusion-associated acute adverse events and drug substitutions. Pediatr Blood Cancer. 2019;66:e27797.
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). Version 4.03. June 14, 2010. Accessed January 14, 2021. www.eortc.be/services/doc/ctc/ctcae_4.03_2010-06-14_quickreference_5x7.pdf
- Babcock KJ, Kinnunen A, Egelund T, et al. Efficacy of a standardized premedication and therapeutic drug monitoring protocol for pegaspargase to prevent hypersensitivity reactions. J Pediatr Pharmacol Ther. 2022;27:232-236.