Connecting Science to Practice
When dosing the calcineurin inhibitor tacrolimus, adjustments
are likely because of its narrow therapeutic index and
variations in renal function, as well as the addition or removal
of concomitant agents with drug–drug interactions
that can affect tacrolimus’s metabolism. This retrospective,
single-center chart review sought to determine whether ideal
body weight, adjusted body weight, or actual body weight
was most often associated with achieving therapeutic tacrolimus
levels in allogeneic hematopoietic stem cell transplant
(HSCT) recipients. This research highlights a potential
method to optimize immunosuppression levels after HSCT
to prevent graft-versus-host disease and tacrolimus-related
adverse events.
The calcineurin inhibitor tacrolimus is frequently used for the prevention of graft-versus-host disease (GVHD) after allogeneic hematopoietic stem cell transplant (HSCT). The initial doses of tacrolimus are weight-based, but the prescribing information for tacrolimus does not specify if the calculations are based on ideal body weight (IBW), adjusted body weight, or actual body weight (ABW).1 In clinical practice, the definition of therapeutic tacrolimus levels varies by institution, but the literature has defined a target trough range between 5 and 20 ng/mL.2
Therapeutic tacrolimus dosing is important to optimize patients’ outcomes after HSCT. According to the National Comprehensive Cancer Network (NCCN)’s guidelines for HSCT, GVHD is a common complication of allogeneic HSCT that results in immune-mediated cellular injury of the skin, gastrointestinal tract, and/or liver because of donor lymphocytes mounting an alloimmune response against the recipient’s tissues.3 Despite the use of immunosuppression for GVHD prophylaxis, the guidelines note that up to 50% of patients undergoing allogeneic HSCT will develop acute GVHD.3 GVHD is the leading cause of nonrelapse mortality in allogeneic HSCT recipients; therefore, the prevention of GVHD by optimizing immunosuppression after an HSCT is a critical component of care.3 Several studies have demonstrated the benefit of optimized immunosuppressive blood levels in the reduction of GVHD rates.4-8 We hypothesize that a consequence of using IBW for initial tacrolimus dosing may delay therapeutic levels, leading to an increased risk for GVHD.
Despite the importance of achieving therapeutic immunosuppression with tacrolimus, providers must also be cautious when dosing this medication because of its narrow therapeutic index. Aside from the intended immunosuppression that is used as the indication in patients undergoing allogeneic HSCT, the adverse events (AEs) related to tacrolimus treatment include nephrotoxicity, particularly in patients with existing renal dysfunction or who are receiving concomitant cytochrome (CY) P3A inhibitors; neurotoxicity, including tremors, headache, delirium, and posterior reversible encephalopathy syndrome; electrolyte disturbances, such as hyperkalemia and hypomagnesemia; gastrointestinal disturbances; and hypertension.1 Per tacrolimus’s prescribing information, neurotoxicity as well as nephrotoxicity and its associated electrolyte disturbances have been correlated with elevated serum concentrations of tacrolimus and may be responsive to dose reductions.1
Since the introduction of tacrolimus to the market in 1994, several studies have evaluated the relationship between tacrolimus blood concentrations and the associated AEs. In a 1996 multicenter evaluation of tacrolimus whole blood levels in kidney transplant recipients, there was a trending higher incidence of AEs with increasing whole blood tacrolimus levels and a significant increase in AEs requiring dose reduction with rising tacrolimus concentrations.9 Of note, the patients in this concentration-ranging trial received tacrolimus doses to generate trough levels from 5 to 40 ng/mL.9 From those results, the researchers concluded that a tacrolimus blood level between 5 and 15 ng/mL best optimized the balance between prevention of transplant organ rejection and the minimization of AEs.9 Although the current literature has been unable to establish a precise estimate at which blood levels these AEs can be expected, we hypothesize that the use of ABW for the initial doses of tacrolimus could result in higher serum tacrolimus levels, resulting in a potential increased risk for these AEs.
Although a cohesive clinical guideline outlining the use of calcineurin inhibitors in patients who have had an HSCT is not currently available to guide practitioners on weight-based dosing of these agents, some literature is available regarding methods for intravenous (IV) tacrolimus dosing at large academic medical centers.10,11 However, IV tacrolimus poses several challenges in clinical practice, for example, patients require a dedicated line for the accuracy of tacrolimus laboratory testing collection, fluid and medication shortages can limit drug availability, and oral immunosuppression provides a more practical transition to outpatient care. In addition, the lack of a standardized dose-conversion ratio because of the variable bioavailability of oral tacrolimus makes it difficult to predict accurate doses when transitioning from IV to oral tacrolimus.1,12,13 To our knowledge, no studies to date have evaluated the impact of dosing weight on therapeutic tacrolimus level achievement for patients initiated on oral tacrolimus for immunosuppression after an initial allogeneic HSCT.
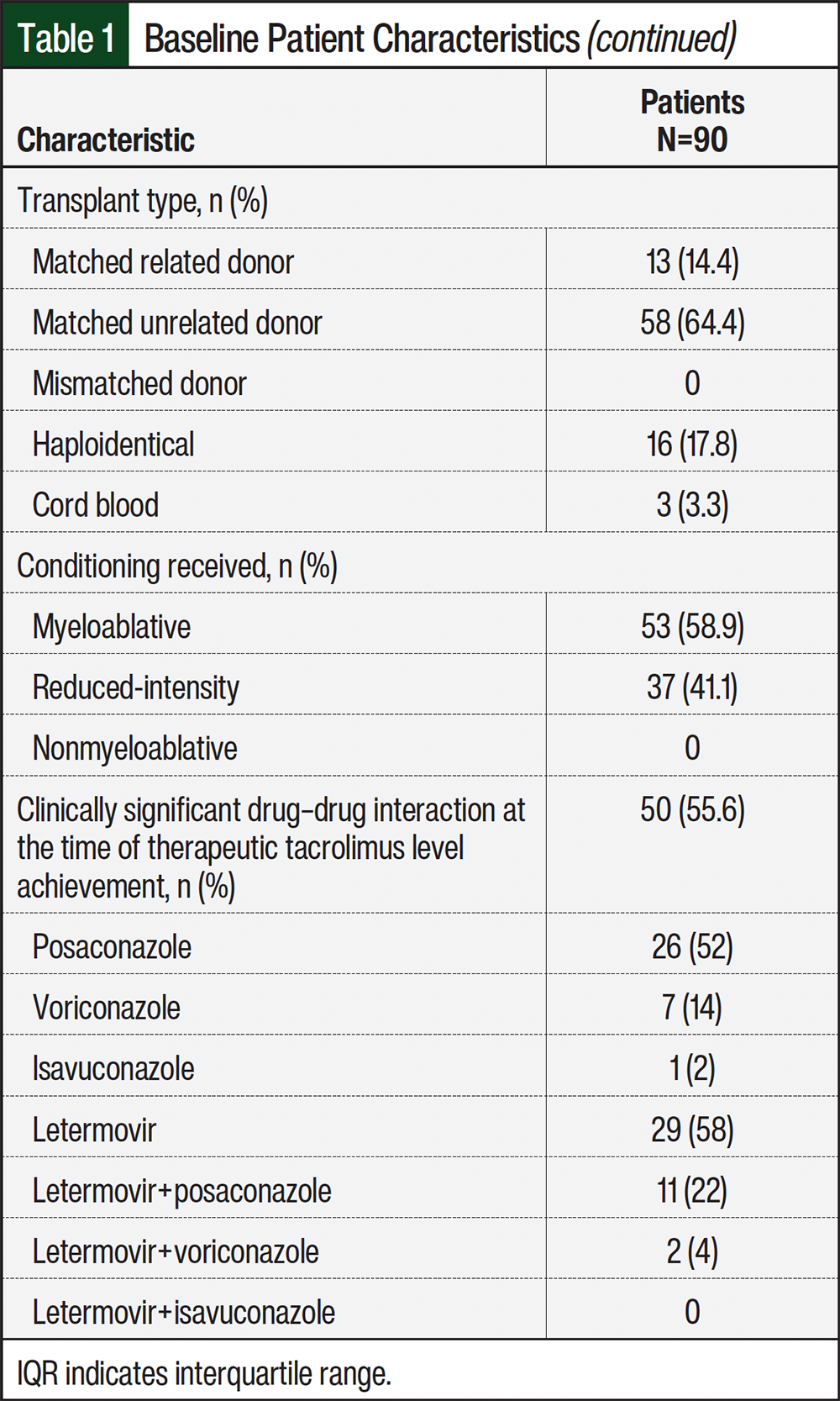
A limited review of immunosuppression guidance highlights the need for data to direct weight-based tacrolimus dosing strategies. The Blood and Marrow Transplant Clinical Trials Network often recommends using institution-specific immunosuppression protocols and practice for the dosing of tacrolimus.14-16 The current practice for the study institution is to use IBW to calculate the initial tacrolimus dose of 0.09 mg/kg daily administered orally in divided doses twice daily. The purpose of this study is to determine whether to use IBW, adjusted body weight, or ABW when calculating initial oral tacrolimus doses for patients undergoing an allogeneic HSCT to minimize the time to achieve therapeutic tacrolimus levels and facilitate optimal immunosuppression for the prevention of GVHD and the minimization of tacrolimus-related AEs.
Methods
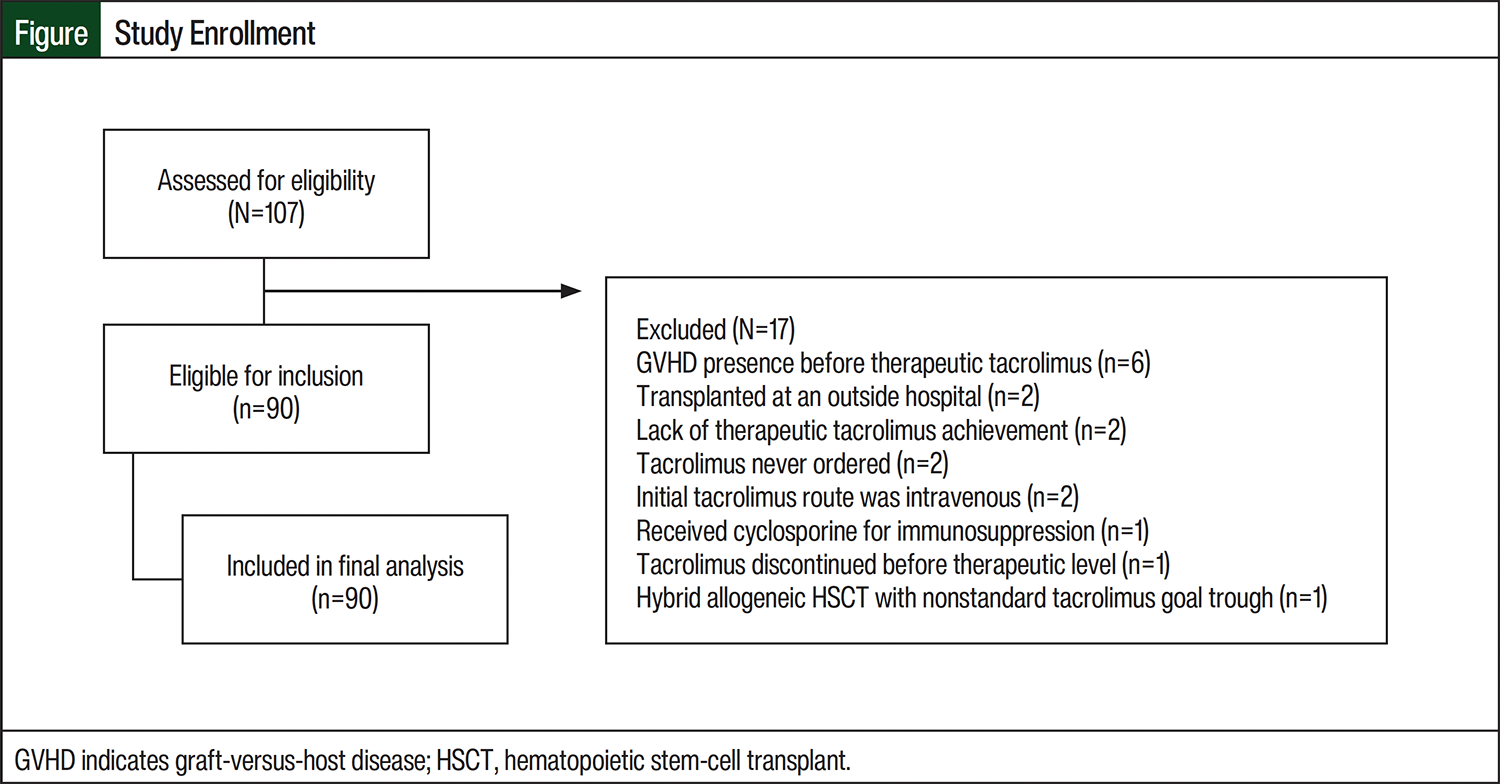
This retrospective chart review was conducted at SSM Health Saint Louis University Hospital in St Louis, MO, an academic medical center that offers inpatient and outpatient care for patients undergoing HSCT. Adults who received oral tacrolimus for immunosuppression after their initial allogeneic HSCT between July 1, 2018, and March 31, 2023, at SSM Health Saint Louis University Hospital were eligible for inclusion in this study. Patients were excluded from the study if they discontinued tacrolimus therapy before achieving a therapeutic trough level, presented with acute GVHD before achieving a therapeutic tacrolimus trough level, had an initial tacrolimus dosing route other than oral, or underwent a transplant at an outside hospital for which transplant data were not accessible.
Patients were identified for screening via the organ transplant tracking record (OTTR) database utilized by this transplant center. The presence or absence of acute and chronic GVHD were also obtained from the OTTR database. All other data points were collected from the electronic health record (EHR). The IBW, adjusted body weight, and ABW were identified for each patient using the standardized weight input sheet that the transplant center uploads to the EHR at the initial transplant visit. Our center uses a standard time before starting conditioning chemotherapy to obtain each patient’s weight to control for weight fluctuations that occur between the initial transplant visit and the time of the transplant.
The primary study outcome was the proportion of patients with a therapeutic tacrolimus dose that corresponded to IBW, adjusted body weight, or ABW. At this institution, the standard initial dose of tacrolimus is 0.09 mg/kg daily administered orally in divided doses twice daily, and IBW is used to calculate this dose. If strong CYP450-inhibiting antifungal agents, such as posaconazole or voriconazole, were initiated before the patient achieved therapeutic tacrolimus levels, a 1:3 conversion factor was used to calculate the tacrolimus dose to allow for an appropriate comparison of doses in the cohort.1 For patients who received IV tacrolimus at the time they achieved the therapeutic trough level, a 1:4 conversion factor was used to calculate the equivalent oral dose.
Therapeutic tacrolimus was defined as the first 2 consecutive therapeutic trough levels separated by at least approximately 24 hours, with no dose adjustments in the 24 hours before the first therapeutic level and no significant dose adjustments, defined as within 1 mg daily, leading up to the second therapeutic level. At the study institution, the goal tacrolimus troughs are 5 to 10 ng/mL for a matched-related HSCT, 8 to 12 ng/mL for a haploidentical or cord blood HSCT, and 10 to 15 ng/mL for a mismatched or matched-unrelated HSCT. After the first 2 consecutive therapeutic tacrolimus levels were obtained, the therapeutic total daily dose in mg was compared with the calculated dose at this institution’s standard initial dose of 0.09 mg/kg for IBW, adjusted body weight, and ABW. IBW was calculated using the following equations: for male patients, 50 kg+2.3 kg for each inch over 5 feet tall, and, for female patients, 45.5 kg+2.3 kg for each inch over 5 feet tall. Adjusted body weight was calculated using a 40% correction factor for each kg over the IBW. The calculated dose that was closest to the therapeutic dose was used to identify which dosing weight was most closely associated with a dose that achieves a therapeutic trough level.
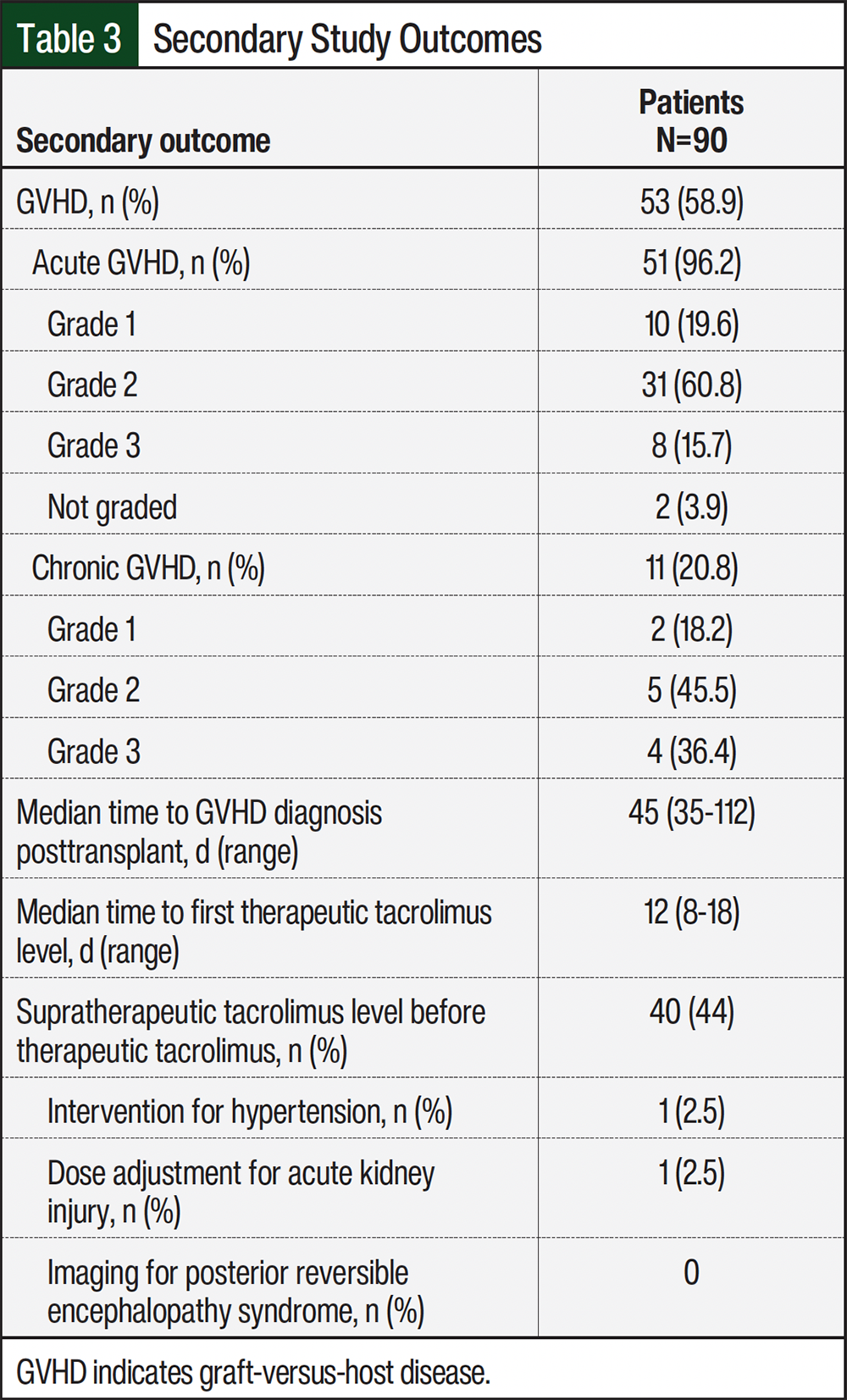
The secondary outcomes included the time to therapeutic tacrolimus achievement, number of patients with supratherapeutic tacrolimus levels before therapeutic level achievement, proportion of patients who had acute and/or chronic GVHD, severity of acute and/or chronic GVHD, and the incidence of AEs requiring intervention in the setting of supratherapeutic tacrolimus levels, which included dose adjustment for acute kidney injury, intervention for hypertension, or imaging for posterior reversible encephalopathy syndrome.
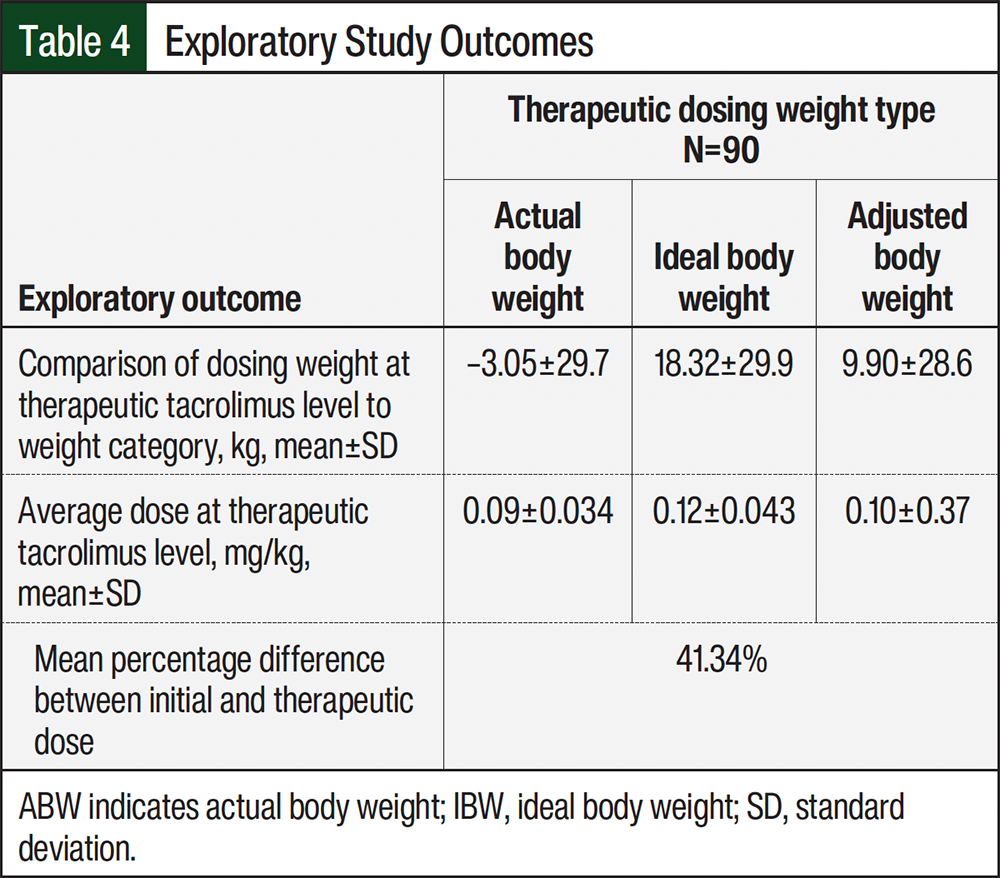
The exploratory outcomes included the observed dosing weight, which was defined as the dosing weight correlating to the therapeutic dose using 0.09 mg/kg daily; the observed mg/kg therapeutic dose; and the percentage difference between the initial therapeutic dose and the therapeutic tacrolimus dose. Multiple subgroup analyses were performed to assess the relationship between the therapeutic tacrolimus dosing weight and the baseline body mass index (BMI), the variance of baseline weight from IBW, and the transplant match status.
Statistical Analysis
All data points were analyzed using descriptive statistics. Further statistical analyses were not pursued because of the lack of comparison of 2 groups in this study. The data are presented as medians with interquartile ranges, numbers (percentages), and means with standard deviations. The analyses were performed with Microsoft Excel for Office 365 (Microsoft; Redmond, WA). This study received institutional review board approval.
Results
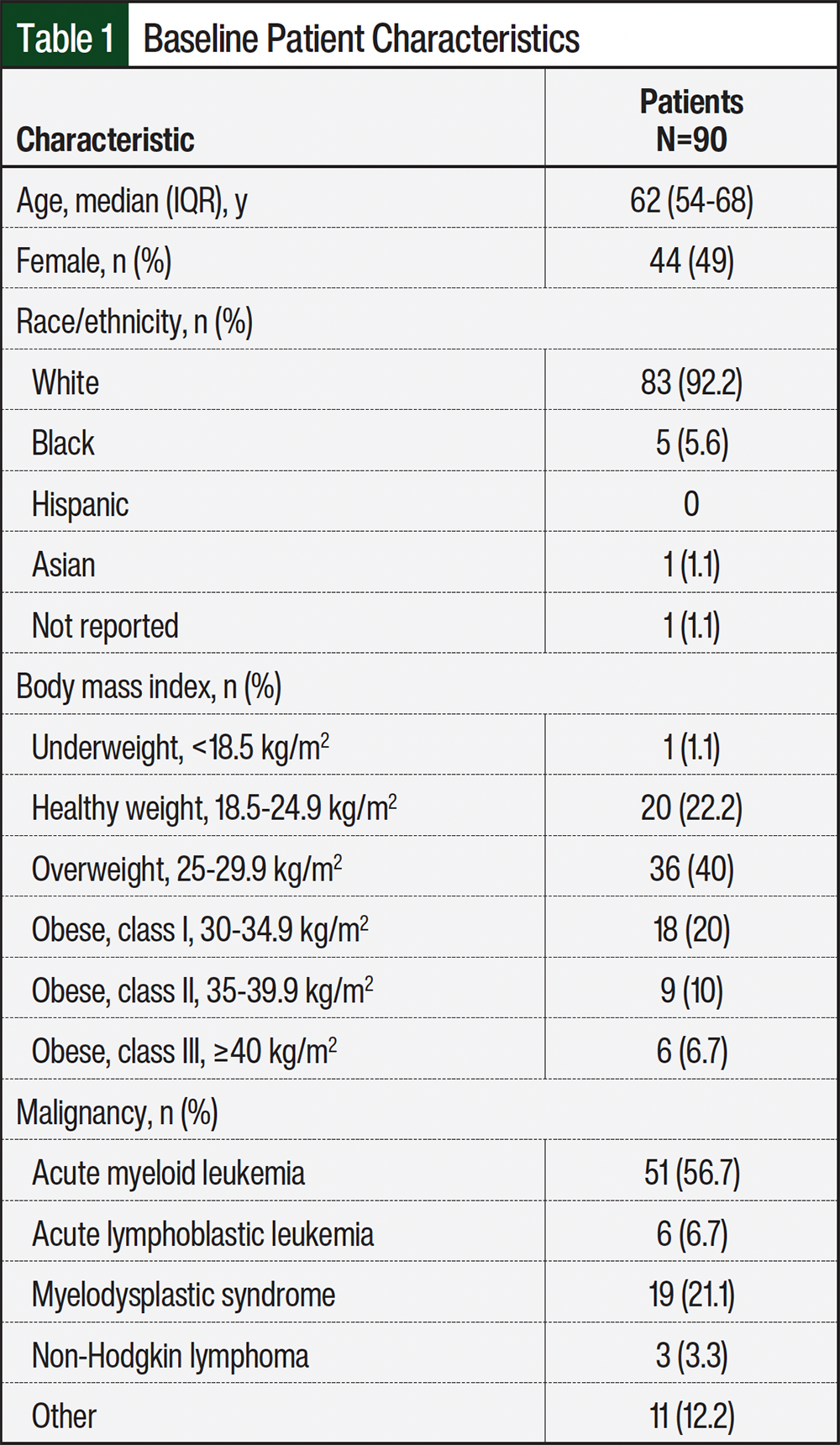
A total of 107 medical records were screened during the study period. Of those records, 90 met the study’s inclusion criteria. The most common reasons for study exclusion are shown in the Figure. The patients’ baseline characteristics are listed in Table 1. The study cohort was overall reflective of the patient population that receives treatment at our institution.
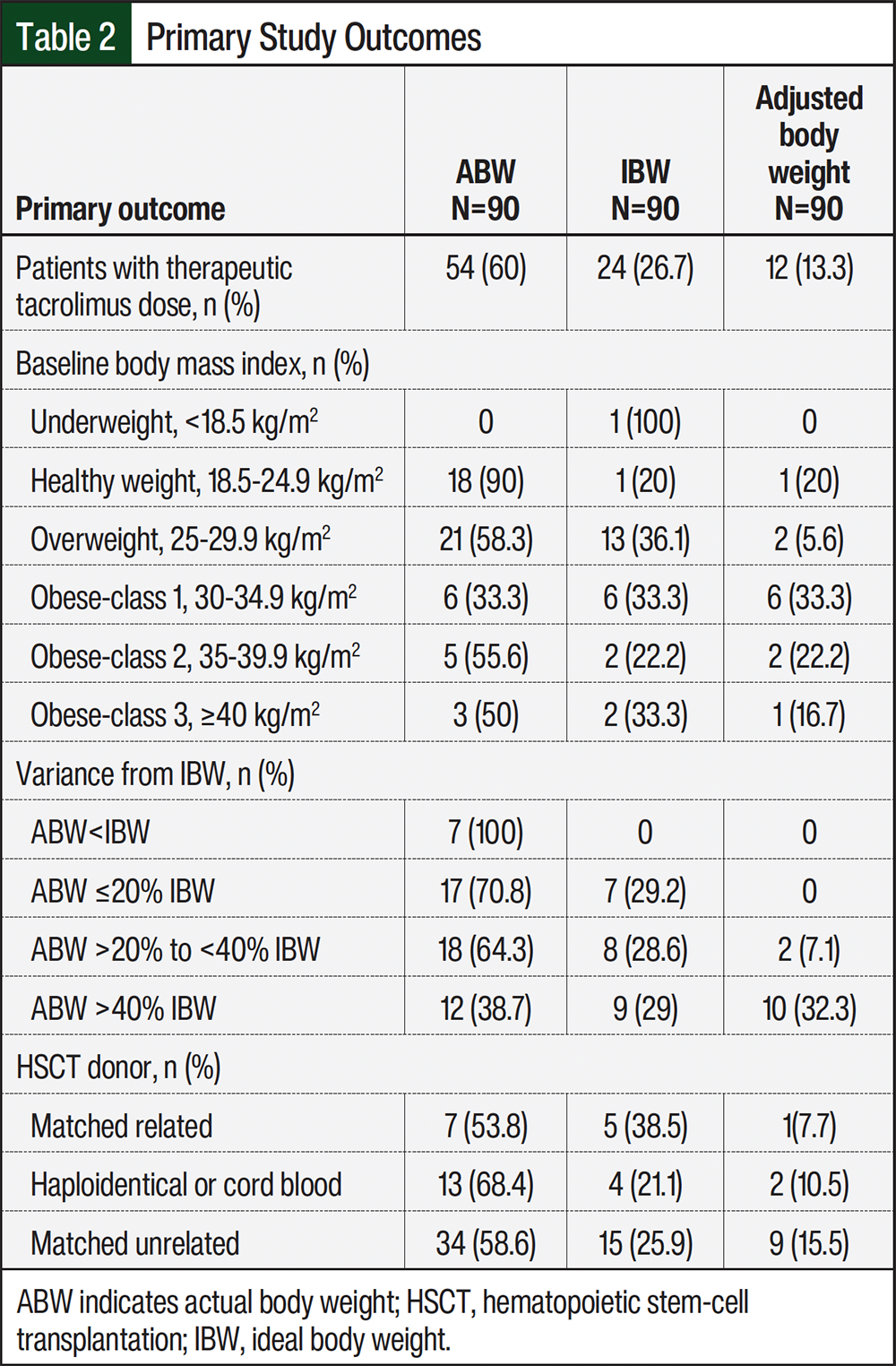
The results of the primary, secondary, and exploratory study outcomes are shown in Table 2, Table 3, and Table 4, respectively. For the primary outcome of the proportion of patients with a therapeutic dose corresponding to IBW, ABW, or adjusted body weight, ABW was identified as the dosing weight that most correlated with therapeutic tacrolimus achievement (60%), followed by IBW (27%) and adjusted body weight (13%). Dose adjustments from the initial tacrolimus dose that was calculated using IBW ranged from a reduction of 4 mg to an increase of 11 mg, for an overall dose percentage change of –75% to +160%. With our institution’s current practice of determining initial tacrolimus doses using IBW, we have observed an undetectable first tacrolimus level in some patients, which often leads to an increased dose of tacrolimus. The correlation of ABW with therapeutic tacrolimus doses is therefore expected, given our experience in clinical practice.
Table 2 shows the results of several subgroup analyses. A subgroup analysis of the impact of baseline BMI on the dosing weight associated with therapeutic tacrolimus demonstrated consistent results, with ABW being the most correlated with a therapeutic dose of tacrolimus in patients with a healthy weight BMI (ie, 18.5-24.9 kg/m2) or overweight BMI (ie, 25-29.9 kg/m2). Patients with an obese BMI had varying results, with ABW most correlated with a therapeutic dose in patients with class 2 obesity (ie, 35-39.9 kg/m2), IBW for class 3 obesity (ie, ≥40 kg/m2), and an even distribution between IBW, ABW, and adjusted body weight for class 1 obesity (ie, 30-34.9 kg/m2). Similarly, a subgroup analysis of baseline variance from IBW demonstrated that ABW is most correlated with a therapeutic dose for patients within 40% of their IBW, but the correlation for dosing weight is evenly distributed across the 3 dosing weights for patients whose weight is >40% of the IBW.
An additional subgroup analysis was performed for the primary outcome to identify whether there was an association between dosing weight and transplant match status, given the varied therapeutic tacrolimus goals per match status at the study institution. Despite the variation in therapeutic tacrolimus trough targets, the results were consistent with that of the primary outcome analysis across HSCT type, with ABW as the dosing weight most correlated with therapeutic tacrolimus levels in more than 50% of the patients in each HSCT subgroup.
For the secondary outcomes, the median time to therapeutic tacrolimus was 12 days (range, 8-18 days; Table 3). Approximately 60% of the patients had GVHD, 96% of whom had acute GVHD and 21% had chronic GVHD. Most of the cases of acute and chronic GVHD were grade 2.
For the exploratory outcomes, therapeutic levels were achieved using the observed dosing weights closest to ABW, followed by adjusted body weight and then IBW for the standard dose of 0.09 mg/kg daily. The average therapeutic doses were 0.09 mg/kg for ABW, 0.12 mg/kg for IBW, and 0.10 mg/kg for adjusted body weight (Table 4). Patients required an overall mean dose adjustment of 41% of their original tacrolimus dose that was calculated using IBW compared with the therapeutic dose of tacrolimus. Table 3 shows the incidence of AEs. Although tacrolimus treatment has many potential AEs, we elected to evaluate only those that required interventions in the form of dose adjustment or imaging.
Discussion
The results of this retrospective chart review indicate that ABW was more frequently correlated with a therapeutic tacrolimus dose than IBW. Dose adjustments from the initial tacrolimus dose that was calculated using IBW were required in 83 of the 90 (92%) patients analyzed and ranged from a reduction in 4 mg to an increase of 11 mg. Comparatively, the true therapeutic dose for the cohort would have required an adjustment between –5 mg and +6 mg or –4 and +9 mg if the initial doses were calculated using ABW or adjusted body weight, respectively. Based on these results, the researchers hypothesize that calculating the initial tacrolimus doses with ABW may reduce the time required to identify the dose at which the drug becomes therapeutic. ABW was most associated with therapeutic tacrolimus doses across the different transplant types and most patients across the weight spectrum in the cohort. For patients with high extremes of body weight, such as a BMI of ≥40 kg/m2 or an ABW differing from IBW by ≥40%, the optimal dosing weight for tacrolimus remains unknown based on the findings of this study.
Although the literature on the relationship between tacrolimus dosing and weight is limited in the HSCT population, some data are available in solid-organ transplant recipients that external factors may play a role in tacrolimus dosing for overweight or obese patients.17,18 In 2012, a small Korean study of kidney transplant recipients that examined the effect of body composition on serum tacrolimus concentrations was unable to identify an association between BMI and serum tacrolimus levels, but the researchers observed higher tacrolimus levels at 0 hours and 4 hours after treatment in patients with a high fat mass compared with those with a low fat mass.17 This study suggests that body composition, especially lean body mass, may have a substantial effect on tacrolimus’s pharmacokinetics and should therefore be a consideration in the dosing of tacrolimus.17 Similarly, the results of a 2017 study also conducted in kidney transplant recipients showed supratherapeutic steady-state tacrolimus levels in more than 50% of patients with overweight or obese BMIs who were dosed based on ABW.18 This 2017 study used their results to develop dosing guidance for patients with a BMI of >25 kg/m2 and recommended dosing at 70% to 85% of the standard dose for patients with a healthy weight BMI.18 Like the results of these 2 studies, our study supports the need for continued research on the factors that affect weight-based tacrolimus dosing, given the observed variation in the optimal dosing weight for patients with an obese BMI.
Some studies in the HSCT population have attempted to establish the relationship between tacrolimus dosing and body weight.10,11 A 2016 study at The Johns Hopkins Hospital evaluated initial tacrolimus levels after receiving a standard daily IV dose of tacrolimus of 1 mg over 4 hours starting just after transplant and showed no association between the various dosing weights and achieving therapeutic tacrolimus trough levels.10 However, the primary objective of that study was to determine the impact of a flat (ie, identical dose for all patients) 1-mg dose of IV tacrolimus on initial serum tacrolimus concentrations, with patient-specific factors, including weight, as a secondary objective. Across the spectrum of dosing weights in the cohort, the John Hopkins Hospital study concluded that regardless of the dosing weight used, all patients would have required a starting dose of IV tacrolimus that was more than the 1-mg standard dose in their protocol. The researchers suggested that a flat initial tacrolimus dose may be preferred in the HSCT population because they did not identify a correlation between weight and therapeutic dose in their cohort.10
Similarly, the 2021 study at NYU Langone Health that evaluated IV tacrolimus did not find an association between dosing weight and achieving therapeutic tacrolimus.11 Patients were dosed using ABW (70% of the cohort) or adjusted body weight (30% of the cohort) for those who weighed >125% of their IBW. At an initial dose of 0.02 mg/kg daily for IV tacrolimus, approximately 50% of the patients were able to achieve therapeutic levels at the first measured tacrolimus level. More than 50% of the cohort had supratherapeutic tacrolimus levels, yet there were limited AEs, including nephrotoxicity, hepatotoxicity, or neurotoxicity, associated with these levels. The researchers noted minimal changes in serum creatinine, despite the initial supratherapeutic tacrolimus levels, and <33% of patients required the new initiation of antihypertensive agents for tacrolimus-related AEs. This finding is consistent with the results of our study in which 44% of patients had supratherapeutic tacrolimus levels, but only 5% of the patients had associated AEs that required intervention.11
Of note, the NYU Langone Health study used a goal tacrolimus trough of 5 to 12 ng/mL, and approximately 75% of the patients’ tacrolimus levels would have been considered therapeutic at their initial level if this range was extended to 5 to 15 ng/mL.11 Overall, the rates of acute GVHD by day 100 were low in this study at 22%; however, the sample size was limited to 64 patients. The NYU Langone Health trial supports further investigation of the appropriate dosing weight for oral tacrolimus, given the possible association between ABW dosing and a faster achievement of a therapeutic tacrolimus level and limited AEs.11
Published data have demonstrated the benefit of timely therapeutic immunosuppression levels in lowering the rates of GVHD in post–allogeneic HSCT recipients.4-6 A 2016 retrospective-cohort study of adults undergoing allogeneic HSCT who received GVHD prophylaxis with tacrolimus and methotrexate identified a lower risk for grade 1 to 4 acute GVHD with tacrolimus levels >12 ng/mL in the first week after transplant.4 However, this study was unable to attain a significant correlation between GVHD rates and tacrolimus concentrations for weeks 2 through 4 after transplant.4
In contrast, a 2012 single-center study of 60 allogeneic HSCT recipients who received tacrolimus and methotrexate for GVHD prophylaxis revealed that lower serum tacrolimus concentrations during the second and third weeks after HSCT was a significant risk factor for grade 2 to 4 acute GVHD.5 Most recently, a large retrospective analysis of allogeneic HSCT recipients conducted in 2021 identified an association between acute GVHD in patients with tacrolimus levels during the first week after transplant, with the benefit of lower incidence of grade 2 to 4 acute GVHD in patients with first-week tacrolimus levels ≥10.15 ng/mL.6 This study also evaluated the association between tacrolimus levels immediately after transplant with chronic GVHD and observed a non–statistically significant but marginally lower risk for chronic GVHD in patients with tacrolimus levels between 7.4 and 10.1 ng/mL in the first week after transplant.6
To our knowledge, our study is the first to evaluate dosing weight for patients initiated on oral tacrolimus treatment for immunosuppression after undergoing an initial allogeneic HSCT. When a therapeutic dose was reached, most patients corresponded to an initial dose calculated using ABW. Dose adjustments are likely to occur for patients receiving tacrolimus, regardless of the dosing weight used to calculate their initial dose, because of the narrow therapeutic index of tacrolimus in addition to fluctuations in renal function and the addition or removal of concomitant medications with drug–drug interactions that impact tacrolimus’s metabolism. Further research is needed to determine whether calculating the initial therapeutic doses of tacrolimus with ABW instead of IBW could reduce the time to therapeutic tacrolimus doses and thereby optimize immunosuppression earlier to promote a reduction in the incidence of GHVD.
This study has several strengths. Standardized input of dosing weight at the study’s institution ensures accurate initial tacrolimus dosing for all patients. This process provides consistency and accuracy in the dosing of medications at our transplant center. As previously mentioned, our study population primarily included patients with healthy weight and overweight BMIs, both of which had ABW as the most correlated with a therapeutic tacrolimus dose. This cohort is reflective of the study institution’s typical patients undergoing allogeneic HSCT based on demographics, including race, transplant type, and the malignancy associated with the transplant indication. The subgroup stratification of results by BMI or variance from IBW subgroups further highlights the best individualized dosing strategy by patient.
Limitations
This study has multiple limitations. The retrospective study design limits the interpretation of the data and entrusts its accuracy to the documentation in the EHR and the OTTR database. This limitation is highlighted in the assessment of tacrolimus levels for patients whose tacrolimus levels became therapeutic in the outpatient setting; in this population, appropriate tacrolimus administration cannot be guaranteed, and dose changes were not always documented with clarity in the EHR.
The protocol design did not account for variance in tacrolimus levels from the standard trough goals when considering whether a level should be therapeutic. For example, if a patient’s tacrolimus trough goal was 8 to 12 mcg/mL and their level was 7.9 mcg/mL, this level would have been considered therapeutic in clinical practice but did not count as therapeutic for data collection purposes.
Our cohort was primarily comprised of White patients, and although this is within the typical population for our transplant center, it does not address the potential variability in tacrolimus metabolism among racial groups. A study of kidney transplant recipients identified clinically significant genetic polymorphisms in the CYP3A4 enzyme in Black patients that resulted in higher tacrolimus clearance compared with White patients in the cohort.19
Nearly 50% of our population had supratherapeutic tacrolimus levels before achieving therapeutic tacrolimus. This finding can be explained by the standard practice for tacrolimus level monitoring and subsequent dose adjustments often observed at our site. At this institution, the first tacrolimus trough is collected after the third dose of tacrolimus. Although this practice gives our clinicians an estimate of how the patient will respond to tacrolimus, it is important to recognize that the first level is not a steady-state trough. Some physicians prefer to rapidly increase the tacrolimus dose based on the first trough level, which can lead to subsequent supratherapeutic levels. Finally, our study evaluated a small sample size; therefore, the participants and results of this trial may not be adequately reflective of the true population.
Conclusion
The results of this study demonstrated that therapeutic tacrolimus doses were the closest to the initial doses calculated using ABW for most patients. In our study population, 60% of therapeutic tacrolimus doses corresponded to ABW, whereas only 27% of the doses aligned with IBW. The optimal dosing weight for patients with body weight in the obese categories remains unclear based on the results of this study.
Author Disclosure Statement
Dr Thurman, Dr Laquet, Dr Knapp, and Dr Schmidt have no conflicts of interest to report.
References
- Prograf (tacrolimus) capsules, for oral use and injection, for intravenous use [prescribing information]. Astellas Pharma US, Inc; August 2023. Accessed November 4, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2023/050708s055,010115s007lbl.pdf
- Przepiorka D, Nash RA, Wingard JR, et al. Relationship of tacrolimus whole blood levels to efficacy and safety outcomes after unrelated donor marrow transplantation. Biol Blood Marrow Transplant. 1999;5:94-97.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: hematopoietic cell transplantation. Version 3.2025. September 24, 2025. Accessed November 4, 2025. www.nccn.org/professionals/physician_gls/pdf/hct.pdf
- Ganetsky A, Shah A, Miano TA, et al. Higher tacrolimus concentrations early after transplant reduce the risk of acute graft-versus-host disease in reduced-intensity allogeneic stem cell transplantation. Bone Marrow Transplant. 2016;51:568-572.
- Mori T, Kato J, Shimizu T, et al. Effect of early posttransplantation tacrolimus concentration on the development of acute graft-versus-host disease after allogeneic hematopoietic stem cell transplantation from unrelated donors. Biol Blood Marrow Transplant. 2012;18:229-234.
- Sharma N, Zhao Q, Ni B, et al. Effect of early post-transplantation tacrolimus concentration on the risk of acute graft-versus-host disease in allogenic stem cell transplantation. Cancers (Basel). 2021;13:613.
- Hagen PA, Adams W, Smith S, et al. Low mean post-transplantation tacrolimus levels in weeks 2-3 correlate with acute graft-versus-host disease in allogeneic hematopoietic stem cell transplantation from related and unrelated donors. Bone Marrow Transplant. 2019;54:155-158.
- Ram R, Storer B, Mielcarek M, et al. Association between calcineurin inhibitor blood concentrations and outcomes after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2012;18:414-422.
- Laskow DA, Vincenti F, Neylan JF, et al. An open-label, concentration-ranging trial of FK506 in primary kidney transplantation: a report of the United States Multicenter FK506 Kidney Transplant Group. Transplantation. 1996;62:900-905.
- Butts AR, Brown VT, McBride LD, et al. Factors associated with optimized tacrolimus dosing in hematopoietic stem cell transplantation. J Oncol Pharm Pract. 2016;22:275-283.
- Soskind R, Xiang E, Lewis T, et al. Initial tacrolimus weight-based dosing strategy in allogeneic hematopoietic stem-cell transplantation. J Oncol Pharm Pract. 2021;27:1447-1453.
- Kuan WYJ, Châteauvert N, Leclerc V, Drolet B. Tacrolimus dose-conversion ratios based on switching of formulations for patients with solid organ transplants. Can J Hosp Pharm. 2021;74:317-326.
- Kanamitsu K, Yorifuji T, Ishida H, et al. Clinical factors affecting the dose conversion ratio from intravenous to oral tacrolimus formulation among pediatric hematopoietic stem cell transplantation recipients. Ther Drug Monit. 2020;42:803-810.
- Cutler C, Logan B, Nakamura R, et al. Tacrolimus/sirolimus vs tacrolimus/methotrexate as GVHD prophylaxis after matched, related donor allogeneic HCT. Blood. 2014;124:1372-1377.
- Bolaños-Meade J, Reshef R, Fraser R, et al. Three prophylaxis regimens (tacrolimus, mycophenolate mofetil, and cyclophosphamide; tacrolimus, methotrexate, and bortezomib; or tacrolimus, methotrexate, and maraviroc) versus tacrolimus and methotrexate for prevention of graft-versus-host disease with haemopoietic cell transplantation with reduced-intensity conditioning: a randomised phase 2 trial with a non-randomised contemporaneous control group (BMT CTN 1203). Lancet Haematol. 2019;6:e132-e143.
- Bolaños-Meade J, Hamadani M, Wu J, et al. Post-transplantation cyclophosphamide-based graft-versus-host disease prophylaxis. N Engl J Med. 2023;388:2338-2348.
- Han SS, Kim DH, Lee SM, et al. Pharmacokinetics of tacrolimus according to body composition in recipients of kidney transplants. Kidney Res Clin Pract. 2012;31:157-162.
- Andrews LM, de Winter BCM, Tang JT, et al. Overweight kidney transplant recipients are at risk of being overdosed following standard bodyweight-based tacrolimus starting dose. Transplant Direct. 2017;3:e129.
- Tornatore KM, Meaney CJ, Attwood K, et al. Race and sex associations with tacrolimus pharmacokinetics in stable kidney transplant recipients. Pharmacotherapy. 2022;42:94-105.