Connecting Science to Practice
The authors evaluated the real-world occurrence of infusion-
related reactions (IRRs) associated with the programmed
cell death protein 1 inhibitors pembrolizumab and nivolumab.
A retrospective chart review was conducted at an academic
medical center over a 2-year period (January 1, 2021–
January 1, 2023) to determine the incidence of IRRs and
assess the effectiveness of subsequent mitigation strategies.
Findings from this analysis provide insight into the frequency,
management, and outcomes of IRRs in clinical practice,
which appear to differ from those reported in clinical trials.
These results highlight the importance of early recognition,
consistent documentation, and proactive mitigation to optimize
patient safety and support the continued delivery of
immunotherapy in oncology practice.
The use of programmed cell death protein 1 (PD-1) inhibitors has significantly increased as a promising therapy in cancer treatment.1 Clinical trials have shown that infusion-related reactions (IRRs) associated with PD-1 inhibitors, particularly nivolumab and pembrolizumab, occur at relatively low rates. In combined results of safety trials, IRRs range from 0.5% to 6.4% with nivolumab and approximately 3% with pembrolizumab.2,3 These reactions can occur throughout the treatment course and range in severity from mild to anaphylactic-like reactions, although severe reactions were observed in <1% of patients.2-4 The prescribing information for pembrolizumab and nivolumab recommends appropriate measures, such as interrupting or slowing down the infusion rate for grade 1 or 2 IRRs, and discontinuing the agent permanently for grade 3 or 4 reactions.3,4
It is essential to acknowledge that clinical trials may not fully capture the entire spectrum of reactions that can occur within a broader patient population. Therefore, conducting investigations in real-world settings is critical to obtaining a more comprehensive understanding of PD-1 inhibitor–related infusion reactions and developing effective mitigation strategies. With indications for PD-1 inhibitors expanding and the expected approval of new drugs in this class, the likelihood of encountering IRRs associated with PD-1 inhibitors will likely increase.5
To address this research gap, we conducted a retrospective analysis of reported cases of PD-1 inhibitor–related IRRs at The University of Kansas Health System, a large academic medical center with 5 infusion centers caring for approximately 13,000 patients annually. Our aim was to provide valuable insights into the incidence, characteristics, and factors associated with PD-1 inhibitor–related IRRs in a real-world setting.
Methods
This study was a retrospective analysis to identify patients who had IRRs related to the PD-1 inhibitors pembrolizumab or nivolumab between January 1, 2021, and January 1, 2023. Patients who received ≥1 dose of either medication were included in the study; those enrolled in clinical trials were excluded.
The primary end point was the incidence of IRRs. Secondary end points included the severity and type of IRR, the time from the first dose of the PD-1 inhibitor to the first IRR, the percentage of patients with recurrent IRRs, the proportion who discontinued infusion after an IRR, the percentage who received medications after an IRR, and the interventions applied for subsequent doses, such as extended administration time and added premedications.
Patients who had an IRR to either nivolumab or pembrolizumab were identified through a review of adverse drug reactions reported via a safety event-reporting software (Datix). Although anyone within the health system could submit a report, nurses followed established protocols to ensure that IRRs were documented in the incident reporting system.
Demographic and clinical information were gathered through a retrospective chart review. This included data on cancer type, line of therapy, the specific PD-1 inhibitor used, the dose administered, cycle length, and cycle number. Progress notes and medication administration records were examined to document the administration time of the PD-1 inhibitor, the types of IRRs (which were graded for severity using the National Cancer Institute’s Common Terminology Criteria for Adverse Events, Version 5), and the onset and resolution times of these reactions. In addition, information on medications administered during the IRRs was collected.
Data were also collected regarding IRRs that occurred during cycles after the first reported reaction. Information collected included the use of premedications, any extensions to administration time, and whether the patient had another reaction after the initial event.
Data were summarized using descriptive statistics, and nominal data were analyzed using chi-square tests, conducted with R (R Core Team, 2023), with an alpha level of 0.05 for statistical significance.
Results
During the 2-year study period, a total of 1968 patients received ≥1 dose of either pembrolizumab (n=1297) or nivolumab (n=671). Among these patients, 17 had a PD-1 inhibitor IRR: 15 were associated with nivolumab and 2 were associated with pembrolizumab, resulting in an overall incidence of an IRR of 2.2% for nivolumab and 0.15% for pembrolizumab.
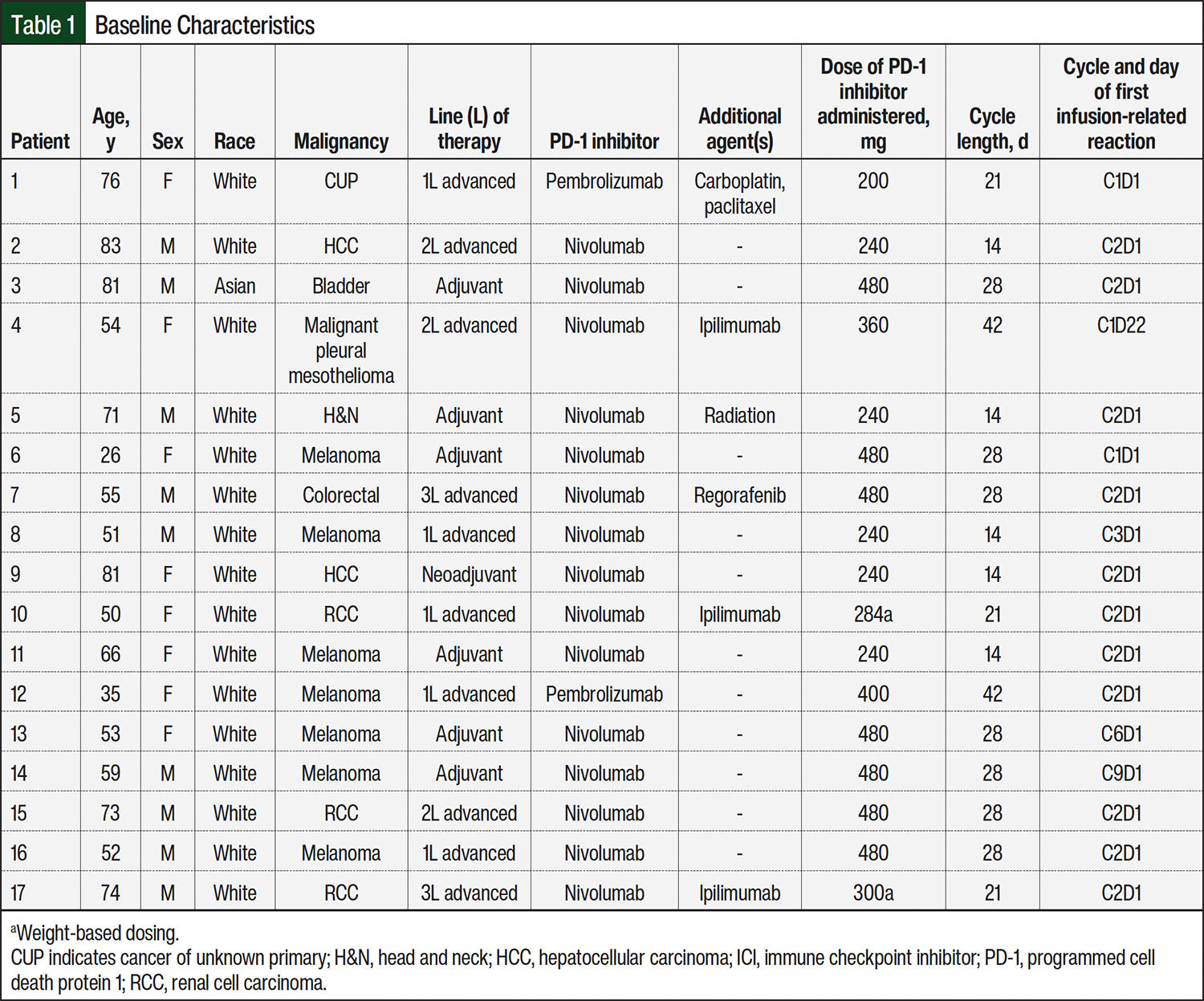
The median age of patients who had an IRR was 59 years (range, 26-83), with 9 men and 8 women. The underlying malignancies comprised melanoma (n=7), renal cell carcinoma (n=3), hepatocellular carcinoma (n=2), head and neck cancer (n=1), bladder cancer (n=1), colorectal cancer (n=1), mesothelioma (n=1), and cancer of unknown primary (n=1). The median dose of nivolumab was 360 mg (range, 240-480 mg), and the median dose of pembrolizumab was 300 mg (range, 200-400 mg). Fifteen patients received flat dosing, whereas 2 patients received weight-based dosing. Additional details regarding baseline characteristics are provided in Table 1.
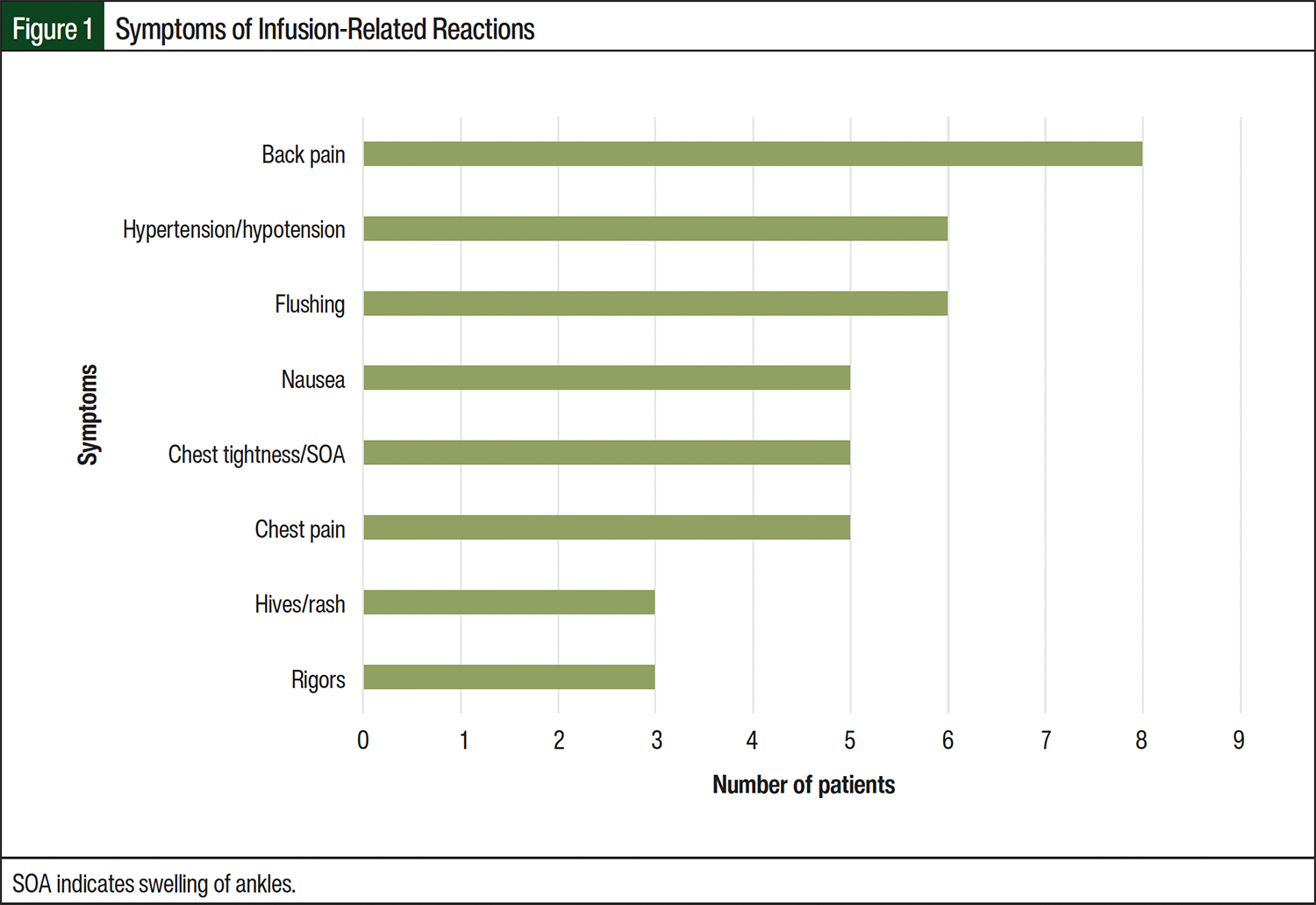
All infusions were ordered to be given over a 30-minute period, and 70.6% of patients had their first IRR during the second dose. All IRRs were observed exclusively during the infusion itself, with no occurrences of delayed reactions. All patients had multiple symptoms during the reactions (Figure 1), with back pain, flushing, and hypertension/hypotension being the most frequently reported. The severity of reactions was categorized as grade 1 (n=1), grade 2 (n=15), and grade 3 (n=1).
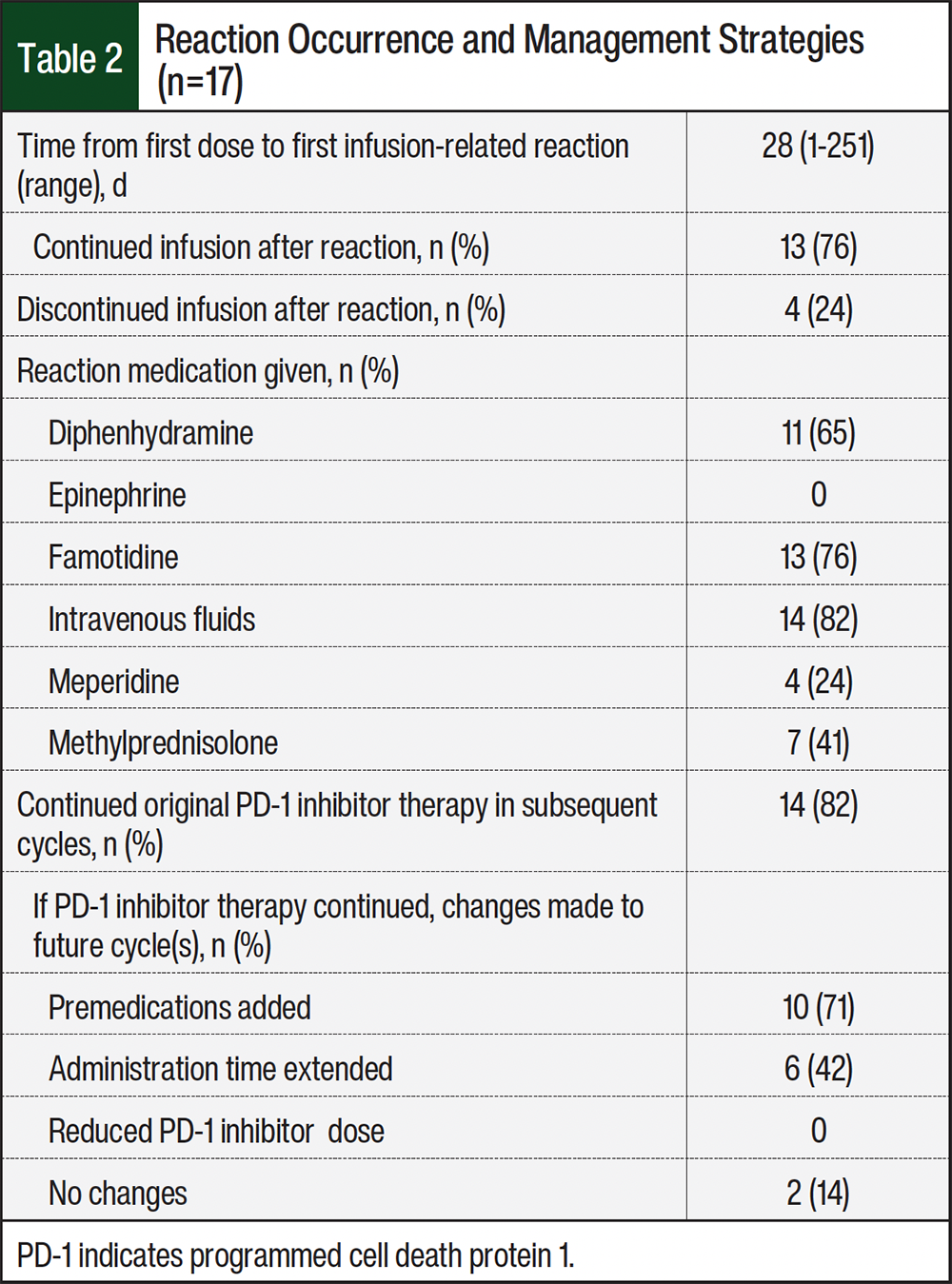
Infusions were temporarily paused for all patients after the IRR (Table 2). Each patient received intravenous fluids and ≥1 hypersensitivity medication, such as diphenhydramine, famotidine, methylprednisolone. Four patients had rigors and were given meperidine; however, no patients received epinephrine. After the resolution of the IRR, 13 (76.5%) patients resumed their infusions the same day. Of the 13 patients who resumed the infusion, 1 had chest pain on restarting, leading to the termination of the infusion. All others completed the infusion without further symptoms. After the initial IRR, 2 patients were hospitalized. One of these hospitalizations was potentially related to the IRR, whereas the other was unrelated. IRRs resulted in no patient deaths.
Of the 17 patients who had an IRR, 14 continued therapy with the initial PD-1 inhibitor (Table 2). In the subsequent cycle after the first IRR, treatment plans were modified for 13 patients, with premedications added for 10 patients and infusion times extended for 6 patients. No dose adjustments were made. In addition, 2 patients received subsequent treatments without any modifications and did not experience further reactions.
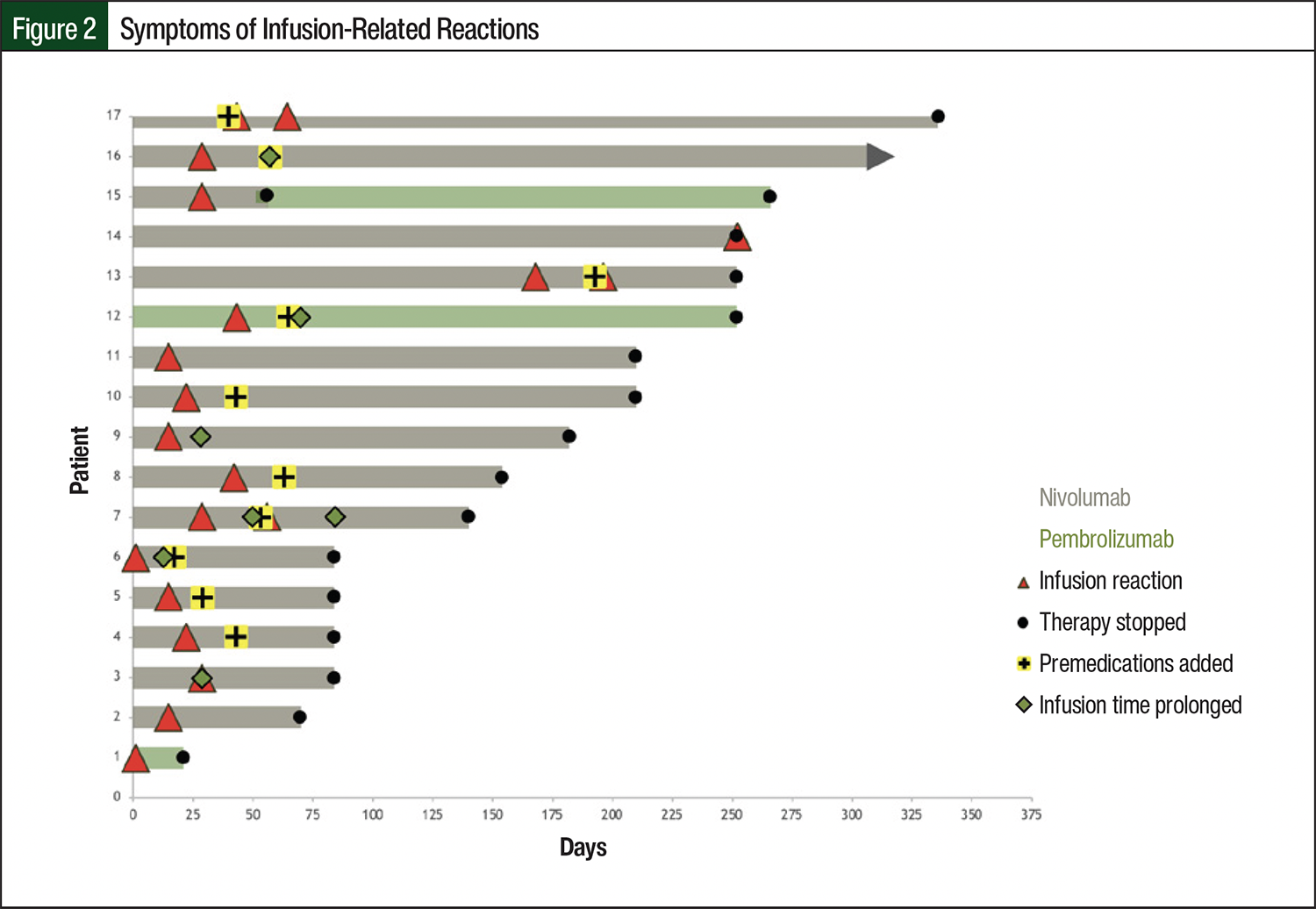
Of the 14 patients who continued PD-1 inhibitor therapy, 5 had recurrent reactions despite modifications to their subsequent treatment plans. Of these patients, 4 had reactions with the next dose but tolerated all subsequent doses. However, 1 patient did not receive future doses of nivolumab after having 2 reactions; 1 patient switched from nivolumab to pembrolizumab and tolerated the infusions without further incidents; 1 patient discontinued nivolumab due to disease progression; and 1 patient discontinued treatment as their cancer diagnosis was changed from cancer of unknown primary to bladder cancer.
Three patients received combination therapy with nivolumab and ipilimumab, with nivolumab infused first. Any IRR that occurred was during the nivolumab infusion, thus excluding ipilimumab as the cause. In a separate case, a patient with cancer of unknown primary received pembrolizumab before concomitant carboplatin and paclitaxel, indicating that the reaction was caused by pembrolizumab rather than the chemotherapy agents. Figure 2 provides a timeline of each patient and shows the time of infusion reaction until the treatment was stopped, along with the mitigation strategies used to prevent future reactions.
A significant relationship was observed between the type of PD-1 inhibitor given and the likelihood of IRR (chi-square test [χ2] =22.37, degree of freedom [df] =1, n=17, P<.001), indicating a higher risk of reaction with nivolumab compared with pembrolizumab. No significant associations were observed between race, White vs non-White: χ2=0.8, df=1, n=17, P=.37, or between sex, men vs women; χ2=0.01, df=1, n=17, P=.92, indicating that these variables were independent of IRRs. Also, no significant relationships were observed between variables in the risk of subsequent IRRs with the addition of premedications diphenhydramine, added vs not added: χ2=3.11, df=1, n=17, P=.08; famotidine, added vs not added: χ2=2.8, df=1, n=17, P=.09); or extension of administration time, time extended vs time not extended: χ2=0.034, df=1, n=17, P=.85).
Discussion
The incidence of IRRs differed significantly between patients who received nivolumab and those who received pembrolizumab, with reactions occurring in 2.2% and 0.15% of patients, respectively. These reactions were generally mild and typically observed during the second or third treatment cycle, in line with published literature.6,7
Various prevention strategies, such as adding premedications or extending the infusion time, have been employed by individual healthcare providers. Most patients continued their PD-1 inhibitor therapy with the implementation of these mitigation strategies; however, based on our data, we cannot provide definitive recommendations regarding the effectiveness of these measures in preventing future reactions. With the small number of patients who had subsequent reactions in our study, it is difficult to make conclusive comparisons between these groups. Another successful strategy reported in the literature, although yet to be implemented at our institution for immune checkpoint inhibitors (ICIs), is the use of desensitization protocols. Several publications have demonstrated that patients who previously reacted to an ICI received desensitization with either a 3-bag 12-step protocol or a 1-bag 15-step protocol, resulting in no recurrent reactions.8,9,11
Our findings align with previous studies and case reports concerning IRRs.12-14 Similar to these studies, a considerable number of patients in our analysis had reactions during subsequent treatment cycles, and comparable mitigation strategies were employed. Of note, there was 1 case report where a patient who reacted to subsequent infusions of nivolumab was successfully switched to pembrolizumab without encountering another reaction.14
Recent updates to national guidelines on managing ICI-related infusion reactions have provided insights into using mitigation strategies based on the severity of the reaction.15 Many of these strategies were consistent with those used at our institution, such as adding premedications or extending the infusion time. These updates represent a step toward standardizing mitigation strategies for patients experiencing IRRs to ICIs.
Despite many valuable findings, there are several limitations to our analysis. First, our data were collected retrospectively from a single center, which may introduce biases. Second, reliance on reported safety events may not have captured all reactions that occurred. Using electronic medical records to identify patients with documented reactions was considered, but we found that this method would have missed certain cases. Third, during our study period, none of the patients who had an infusion reaction had lung cancer, most likely due to the prescribing practices of our lung oncologists, who preferred pembrolizumab to nivolumab for lung cancer indications. In addition, our evaluation focused solely on pembrolizumab and nivolumab, excluding other PD-1 inhibitors such as cemiplimab, dostarlimab, and retifanlimab, which have demonstrated efficacy and safety in various malignancies. We included only these 2 medications in our analyses because of the high utilization of these agents at our institution, and with data already available on the higher incidences of IRRs with other PD-1 inhibitors, we focused on agents that engender relatively lower concern for IRRs.16
IRRs can occur with various ICIs, and the incidence and characteristics of these reactions may vary.7 For instance, severe or life-threatening IRRs have been reported with avelumab, necessitating premedication with antihistamines and acetaminophen before the first 4 cycles of treatment.17 Therefore, it is crucial to recognize that our findings are specific to pembrolizumab and nivolumab and may not directly apply to other ICIs.
Our study provides valuable insights into the incidence and management of IRRs with pembrolizumab and nivolumab, but more research is needed to investigate the incidence, characteristics, and optimal management strategies for reactions associated with other ICIs. A comprehensive understanding of IRRs across the range of available ICIs will enable healthcare providers to make informed decisions regarding patient care and treatment optimization.
Conclusion
Based on the results of our single-institution study, nivolumab and pembrolizumab are associated with a low risk of IRRs. Most patients who had a reaction tolerated future doses. With the increasing use of PD-1 inhibitors and other ICIs, further investigation is needed to identify risk factors and optimize mitigation strategies.
Author Disclosure Statement
Dr Martin is on the APP Advisory Board for GlaxoSmithKline; Dr Tran and Dr Brunk have no conflicts of interest to report.
References
- Liu K, Sun Q, Liu Q, et al. Focus on immune checkpoint PD-1/PD-L1 pathway: new advances of polyphenol phytochemicals in tumor immunotherapy. Biomed Pharmacother. 2022;154:113618.
- Roselló S, Blasco I, García Fabregat L, et al. Management of infusion reactions to systemic anticancer therapy: ESMO Clinical Practice Guidelines. Ann Oncol. 2017;28(suppl 4):iv100-iv118. Erratum in: Ann Oncol. 2018;29(suppl 4):iv260.
- Opdivo (nivolumab) injection, for intravenous use [prescribing information]. Bristol-Myers Squibb Company; May 2025. Accessed June 5,2025. https://packageinserts.bms.com/pi/pi_opdivo.pdf
- Jeytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck & Co., Inc; June 2025. Accessed July 5, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2025/125514s173lbl.pdf
- Gong J, Chehrazi-Raffle A, Reddi S, Salgia R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive review of registration trials and future considerations. J Immunother Cancer. 2018;6:8.
- Chung CH. Managing premedications and the risk for reactions to infusional monoclonal antibody therapy. Oncologist. 2008;13:725-732.
- Rombouts MD, Swart EL, Van Den Eertwegh AJM, Crul M. Systematic review on infusion reactions to and infusion rate of monoclonal antibodies used in cancer treatment. Anticancer Res. 2020;40:1201-1218.
- Labella M, Castells M. Hypersensitivity reactions and anaphylaxis to checkpoint inhibitor-monoclonal antibodies and desensitization. Ann Allergy Asthma Immunol. 2021;126:623-629.
- Hong DI, Madrigal-Burgaleta R, Banerji A, et al. Controversies in allergy: chemotherapy reactions, desensitize, or delabel? J Allergy Clin Immunol Pract. 2020;8:2907-2915.E1.
- Agrawal S, Statkevich P, Bajaj G, et al. Evaluation of immunogenicity of nivolumab monotherapy and its clinical relevance in patients with metastatic solid tumors. J Clin Pharmacol. 2017;57:394-400.
- Isabwe GAC, de Las Vecillas Sanchez L, Castells M. Management of adverse reactions to biologic agents. Allergy Asthma Proc. 2017;38:409-418.
- Thomas M, Wazir A, Poudel A. Pembrolizumab-induced acute skin reaction: a case report and review of literature. Cureus. 2022;14:e26143.
- White C, Borlagdan J, Wing J, Lopez CD. Localized muscle spasm as a manifestation of pembrolizumab-induced infusion reaction. J Oncol Pharm Pract. 2021;27:1270-1274.
- Choi B, McBride A, Scott AJ. Treatment with pembrolizumab after hypersensitivity reaction to nivolumab in a patient with hepatocellular carcinoma. Am J Health Syst Pharm. 2019;76:1749-1752.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: management of immunotherapy-related toxicities. Version 1.2025. December 20, 2024. Accessed July 28, 2023. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf.
- Fujiwara Y, Tsuchiya K, Mako Koseki, et al. Infusion-related reactions from immune checkpoint inhibitors: a proportional and network meta-analysis. J Clin Oncol. 2024;42(suppl 23):Abstract 38.
- Bavencio (avelumab) injection, for intravenous use [prescribing information]. EMD Serono, Inc; June 2025. Accessed July 5, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2025/761049s021lbl.pdf