Connecting Science to Practice
This study investigates the drug interaction between vincristine
and aprepitant in pediatric patients with rhabdomyosarcoma
receiving vincristine, dactinomycin, and cyclophosphamide
therapy. We evaluated whether rates of vincristineinduced
adverse events (AEs) were higher among children
receiving aprepitant as antiemetic prophylaxis compared with
those who did not receive aprepitant. This single-center, retrospective
chart review showed that aprepitant use was not
associated with an increased rate of clinically significant AEs,
as defined by vincristine dose modification in subsequent
cycles. We recommend additional studies to explore the clinical
significance of the interaction between vincristine and
aprepitant in other pediatric populations and across other
cancer therapies.
Vincristine is a vinca alkaloid that is frequently used for the treatment of pediatric malignancies.1 Vincristine exhibits its cytotoxic effects by binding to tubulin and stopping mitotic spindle formation, resulting in tumor cell death.2 Peripheral neuropathy, constipation, and other neurologic adverse events (AEs) are well-known and debilitating complications in pediatric patients with cancer.3 Vincristine-induced peripheral neuropathy may be sensory, motor, or autonomic, and can be severe and dose-limiting.4 As a cytochrome (CY)P3A4 substrate, the coadministration of vincristine with CYP3A4 inhibitors has the potential to increase such AEs.4
The combination of vincristine and strong CYP3A4 inhibitors, such as posaconazole and voriconazole, has increased vincristine-induced AEs with the potential to lead to significant dose modifications for vincristine.5,6 However, there is conflicting evidence about the clinical significance of weak-to-moderate CYP3A4 inhibitors received in combination with vincristine.7,8 Vincristine is minimally emetogenic,9 but it may be received in combination with other moderately to highly emetic chemotherapy agents, necessitating the use of guideline-concordant multiagent antiemetic regimens.10 Moderately and highly emetogenic chemotherapy should be received with a 5-HT3 receptor antagonist, dexamethasone, and aprepitant, which is a neurokinin-1 receptor antagonist. Aprepitant is a known dose- and duration-dependent weak-to-moderate CYP3A4 inhibitor.11 Although the interaction is known, there is limited literature addressing its significance in pediatric patients with cancer.
Despite this potential drug interaction, patients with newly diagnosed rhabdomyosarcoma receiving the standard-of-care chemotherapy regimen of vincristine, dactinomycin, and cyclophosphamide may receive aprepitant during each cycle as a component of antiemetic prophylaxis. The objective of this study is to determine whether the rates of vincristine-induced AEs are higher among children receiving aprepitant as antiemetic prophylaxis during rhabdomyosarcoma therapy than in children who did not receive aprepitant.
Methods
This was a single-center, retrospective cohort study conducted at Texas Children’s Hospital in Houston. Patients were included if they received treatment for rhabdomyosarcoma with vincristine, dactinomycin, and cyclophosphamide from January 2011 through December 2022. Patients were identified by an electronic medical record (EMR)-generated report for patients with rhabdomyosarcoma with treatment plans that included vincristine, dactinomycin, and cyclophosphamide therapy, standard of care in pediatric patients with rhabdomyosarcoma.12-15 Patients were included in the study if they received at least 2 cycles of vincristine, dactinomycin, and cyclophosphamide at the facility. The vincristine doses in the study were 1.5 mg/m2 per dose, with a maximum of 2 mg, for up to 30 doses total over 40 weeks. Dose banding was used for patients with a body surface area of <0.6 m2, depending on the year and reference treatment protocol. Patients were excluded from the study if they received concurrent, strong CYP3A4/CYP3A5 inhibitors, had preexisting neuropathies before therapy, or required a dose adjustment of vincristine for hepatic impairment, which was defined as a total bilirubin of ≥2.1 mg/dL per the institutional guidelines. This study was approved by the institutional review board.
This study compared vincristine-induced AE rates, as indicated by the need for vincristine dose reduction. The data collection period spanned the period of the standardization of aprepitant use as a component of antiemetic prophylaxis for this regimen. An institutional antiemetic guideline update took place in 2018. The recommended aprepitant dosing per the guideline at that time was 3 mg/kg on day 1, followed by 2 mg/kg on days 2 and 3 of aprepitant, with a maximum dose of 125 mg on day 1 and 80 mg on days 2 and 3. This dosing schedule was used until 2021, when dose banding was implemented.16
The patients were divided into 3 groups depending on the extent of aprepitant use: group A consisted of patients who received aprepitant during all vincristine, dactinomycin, and cyclophosphamide cycles; group B consisted of patients who received aprepitant during at least 1 cycle, but not all cycles, of vincristine, dactinomycin, and cyclophosphamide treatment; and group C consisted of patients who did not receive aprepitant during any cycles of vincristine, dactinomycin, and cyclophosphamide. The study’s primary end point was the rate of dose reduction secondary to vincristine-induced AEs in children who received aprepitant compared with children who did not have these AEs. The trial’s secondary end points were the incidence and severity of vincristine-induced AEs, as well as the use of new neuropathic medications.
The baseline demographic information that was collected included age, sex, race, and ethnicity. The clinical data collected included the primary disease site; risk stratification; dose intensities of vincristine, dactinomycin, and cyclophosphamide; and aprepitant use during vincristine, dactinomycin, and cyclophosphamide therapy. The AE information collected included the dose of vincristine for each cycle and the vincristine dose-modification data, including the percentage of dose reduction and the reason for dose modification (eg, constipation, neuropathy, neurotoxicity). The documentation of neuropathies included the provider’s grading of peripheral motor or sensory neuropathy per the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 grading scale. In addition, chart documentation of neuropathies was identified using phrases such as neuropathy and tingling, or noting the initiation of gabapentin or duloxetine. Every cycle of vincristine, dactinomycin, and cyclophosphamide therapy received at Texas Children’s Hospital was analyzed in all included patients. The time to vincristine dose modification was defined as the time between the patient’s first dose of vincristine and a dose modification of vincristine, if applicable.
Statistical Analysis
Descriptive statistics were used to characterize the baseline patient characteristics. Statistical differences in these characteristics across the groups of interest (groups A, B, and C) were evaluated using chi-square and Fisher’s exact tests for the categorical variables and ANOVA for the continuous variables. Next, to evaluate the differences in time to dose modification across the groups, we used the Kaplan–Meier and log-rank tests. Differences where P<.05 were considered statistically significant. All tests were conducted using Stata Version 15.0 (StataCorp; College Station, TX).
Results
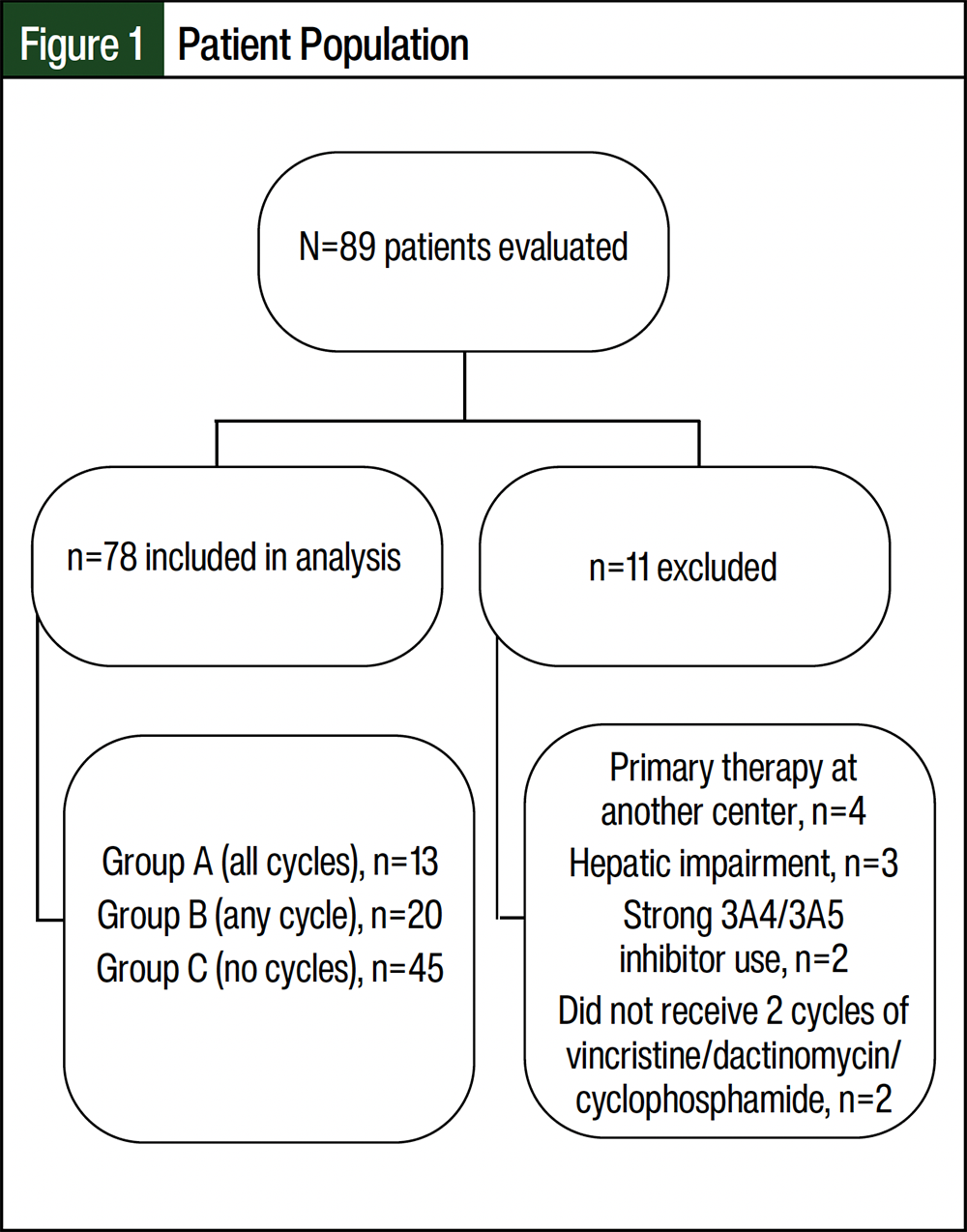
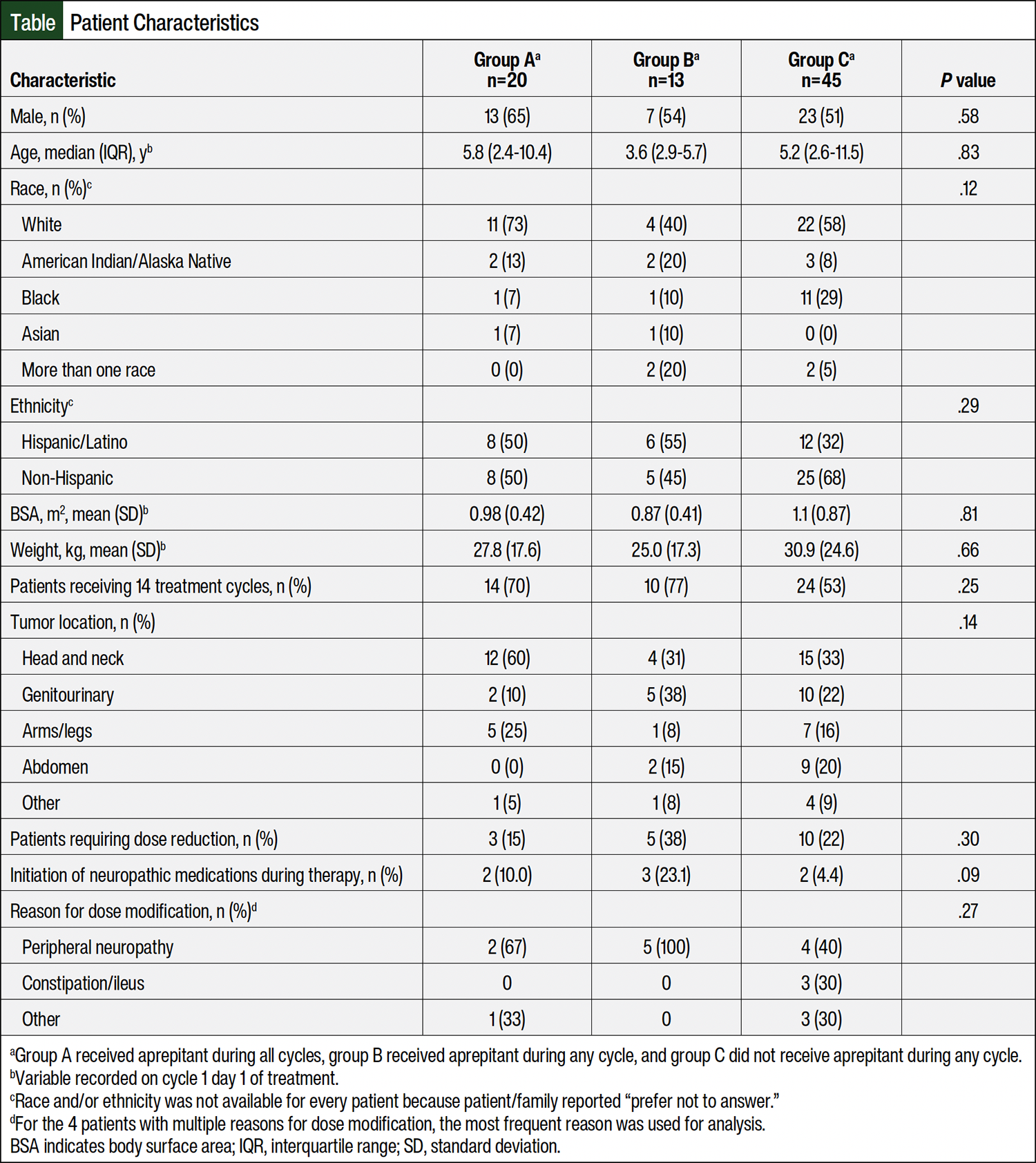
A total of 89 patients were identified, 78 of whom met the study’s inclusion criteria and were included in the analysis (Figure 1). The median patient age was 5.3 years (range, 1 month-23 years), and there were predominantly more male patients than female patients (male-to-female ratio, 1.2:1). The most common primary cancer site was head and neck, followed by genitourinary (Table). Data for all 14 cycles of vincristine, dactinomycin, and cyclophosphamide therapy administered at our institution were available for 48 (61.5%) patients.
The largest cohort of patients represented those who did not receive aprepitant during any cycles (n=45, 57.7%), whereas 33 patients received aprepitant during any (n=20, 25.6%) or all cycles (n=13, 16.7%). The baseline characteristics were similar among the groups (Table). A total of 18 (23.1%) patients required vincristine dose modification across all groups, with an average of 3.7 cycles modified. The median percentage dose reduction (including dose omissions) was 50%. Of these patients, 11 tolerated re-escalation to the full dose after the resolution of AEs. The most common reasons for dose modification were peripheral neuropathy (n=11, 14.1%) and constipation/ileus (n=3, 3.8%).
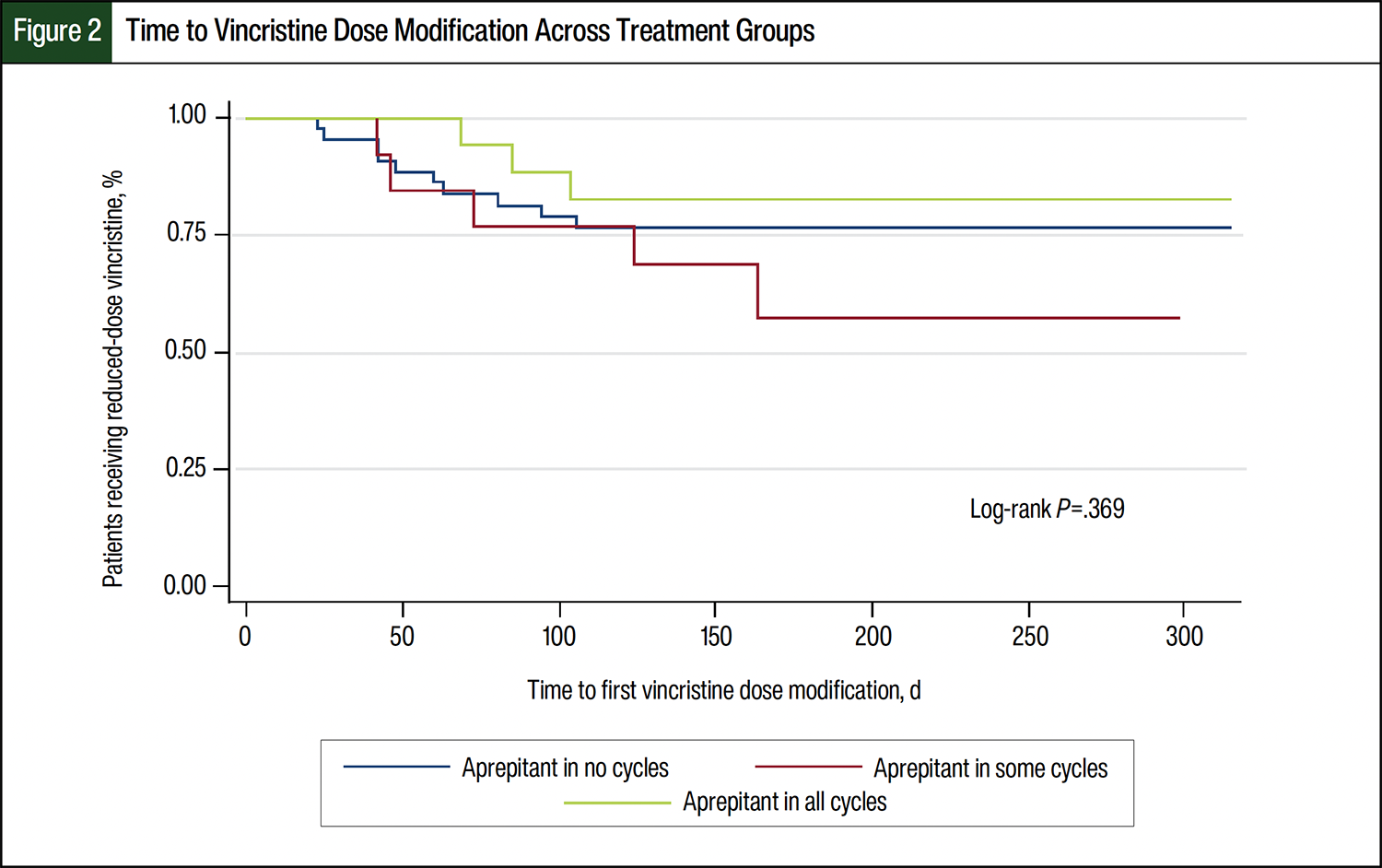
There were no significant differences in the patients’ characteristics or time to vincristine dose modification across the groups (Table and Figure 2). The incidence of dose modification and total number of modified doses were highest in group B, although these did not reach statistical significance (Figure 2). Similarly, the initiation of medications for neuropathy, most often gabapentin, was highest in group B (n=3, 23.1%, P=.09; Table).
Discussion
Our findings indicate that aprepitant use was not associated with an increased rate of clinically significant AEs, as defined by dose modification, in pediatric patients with rhabdomyosarcoma receiving vincristine, dactinomycin, and cyclophosphamide therapy; however, this is in the context of a limited sample size. A homogeneous population was selected to minimize confounding factors that could contribute to vincristine-induced peripheral neuropathy, but this limited our ability to meet power. Overall, our study had a similar rate of vincristine-induced peripheral neuropathy to other studies.17,18
In this cohort, aprepitant was received on days 1 through 3 of an every-3-week cycle before cyclophosphamide, and vincristine was administered on day 1 of weeks 1 to 13, week 16, weeks 19 to 25, week 28, weeks 31 to 37, and week 40. Particularly in this regimen, the overlapping time between the administration of aprepitant and vincristine may not allow for CYP enzyme saturation and inhibition, limiting the effect on vincristine exposure and the associated AEs. In addition, aprepitant was not administered on the days that vincristine was received alone. These results may not apply to multiday-interacting chemotherapy, such as ifosfamide. Multiple studies have examined the association between aprepitant use and neurotoxicity in ifosfamide-containing regimens, because ifosfamide is also a CYP3A4 substrate.19,20 The studies hypothesize that the addition of aprepitant likely causes increased serum concentrations of ifosfamide and subsequently increased production of ifosfamide’s neurotoxic metabolite chloroacetaldehyde.19,20 Further studies are needed to evaluate the clinical significance of other CYP3A4 substrate chemotherapies received on consecutive days with CYP3A4 inhibitors.
Study results from Smitherman and colleagues also support a limited clinical significance of vincristine with other moderate CYP3A4 inhibitors.7 In their study, pediatric patients received standard therapy for acute lymphoblastic leukemia (ALL), which included weekly vincristine and daily fluconazole, a moderate CYP3A4 inhibitor, for fungal prophylaxis. Their study’s results showed that concurrent administration of fluconazole and induction therapy for ALL did not significantly increase the risk for vincristine-induced AEs.7
Conversely, a study by Edwards and colleagues showed a greater overall incidence of vincristine-induced peripheral neuropathy in adults with non-Hodgkin lymphoma receiving the CYP3A4 inhibitor aprepitant with vincristine-containing therapy.8 Some notable differences between their research and ours include that our study evaluated pediatric patients compared with adults, and the dose, schedule, and frequency of vincristine administration. Our pediatric patient cohort received more frequent administrations of vincristine, as well as received aprepitant and vincristine on the same day when given on the weeks that were previously specified. Additional differences include the definitions of clinically significant vincristine-induced peripheral neuropathy. In the study by Edwards and colleagues, vincristine-induced peripheral neuropathy was defined as any documentation of tingling or neuropathic pain or the initiation of neuropathic medications.8 In addition, the researchers noted that all cases of vincristine-induced peripheral neuropathy were grade 1 per CTCAE version 5.0. Instead, we opted to define clinically significant neuropathies as those requiring dose modifications of vincristine to capture patients potentially receiving reduced therapy because of the interaction of vincristine and aprepitant.
Limitations
Our study has a few notable limitations. First, there was inconsistent use of aprepitant across cycles. Aprepitant was incorporated into our institutional standards for antiemetic prophylaxis in 2018; however, standardized risk-adapted antiemetic regimens were not incorporated into the EMR at that time. Rather, the use of aprepitant was based on clinician preference or pharmacist recommendations during much of the study period. To account for this, we applied simple definitions for the extent of aprepitant exposure (none, any cycle, or all cycles), which may carry limitations.
Further, a unique relationship between the use of aprepitant and vincristine-induced AEs was observed in our results, which we attributed to real-life clinical decision-making at our institution. Although not significant, we observed a higher rate of dose modification in patients receiving aprepitant in any treatment cycle (Figure 2). This likely highlights our real-world practice of holding medications with the potential to interact in patients who have AEs and not a causative relationship between receiving aprepitant and having vincristine-induced AEs.
There are additional patient-specific risk factors for vincristine-induced AEs to consider in the context of this study, including age, sex, race, administration method, and genetic polymorphisms.21 Specifically, some literature has identified the CEP72 genotype as a specific risk marker for vincristine-induced peripheral neuropathy in adults and pediatric patients.22,23 Therefore, an additional limitation of this study is that pharmacogenetic variances in CYP3A4/3A5 enzymes were not collected, but may also have influenced vincristine-induced AEs. An emerging better understanding of vincristine metabolism involving CYP3A5 as a major contributor may also explain why interactions with moderate CYP3A4 inhibitors have not consistently yielded clinically significant events.6 Further research is needed in these areas because we were not able to explore pharmacogenetic variances in this study.
Conclusion
This single-center, retrospective cohort study is the first to our knowledge that evaluated the clinical impact of the potential drug interaction between vincristine and aprepitant in pediatric patients. Our study’s results showed no increased risk for vincristine-induced AEs that required dose reduction in pediatric patients with rhabdomyosarcoma receiving concomitant aprepitant and therapy for vincristine-induced peripheral neuropathy. The interaction between aprepitant and CYP3A4 substrate chemotherapy should be further explored in other pediatric populations with cancer, in groups with a larger sample size, as well as in patients receiving multiday interacting chemotherapy.
Author Disclosure Statement
Dr Varughese, Dr Bernhardt, Ms Chambers, Dr Foster, Dr Lindsay, Dr Lupo, and Dr Sauer have no conflicts of interest to report.
References
- van de Velde ME, Kaspers GL, Abbink FCH, et al. Vincristine-induced peripheral neuropathy in children with cancer: a systematic review. Crit Rev Oncol Hematol. 2017;114:114-130.
- Stryckmans PA, Lurie PM, Manaster J, Vamecq G. Mode of action of chemotherapy in vivo on human acute leukemia—II. Vincristine. Eur J Cancer. 1973;9:613-620.
- Courtemanche H, Magot A, Ollivier Y, et al. Vincristine-induced neuropathy: atypical electrophysiological patterns in children. Muscle Nerve. 2015;52:981-985.
- Triarico S, Romano A, Attinà G, et al. Vincristine-induced peripheral neuropathy (VIPN) in pediatric tumors: mechanisms, risk factors, strategies of prevention and treatment. Int J Mol Sci. 2021;22:4112.
- Harnicar S, Adel N, Jurcic J. Modification of vincristine dosing during concomitant azole therapy in adult acute lymphoblastic leukemia patients. J Oncol Pharm Pract. 2009;15:175-182.
- Dennison JB, Jones DR, Renbarger JL, Hall SD. Effect of CYP3A5 expression on vincristine metabolism with human liver microsomes. J Pharmacol Exp Ther. 2007;321:553-563.
- Smitherman AB, Faircloth CB, Deal A, et al. Vincristine toxicity with co-administration of fluconazole during induction therapy for pediatric acute lymphoblastic leukemia. Pediatr Blood Cancer. 2017;64:e26525.
- Edwards JK, Bossaer JB, Lewis PO, Sant A. Peripheral neuropathy in non-Hodgkin’s lymphoma patients receiving vincristine with and without aprepitant/fosaprepitant. J Oncol Pharm Pract. 2020;26:809-813.
- Paw Cho Sing E, Robinson PD, Flank J, et al. Classification of the acute emetogenicity of chemotherapy in pediatric patients: a clinical practice guideline. Pediatr Blood Cancer. 2019;66:e27646. Erratum in: Pediatr Blood Cancer. 2021;68:e28990.
- Patel P, Robinson PD, Cohen M, et al. Prevention of acute and delayed chemotherapy-induced nausea and vomiting in pediatric cancer patients: a clinical practice guideline. Pediatr Blood Cancer. 2022;69:e30001.
- Emend (aprepitant) capsules, for oral use [prescribing information]. Merck Sharp & Dohme LLC; May 2022. Accessed June 1, 2023. www.merck.com/product/usa/pi_circulars/e/emend/emend_pi.pdf
- Maurer HM, Beltangady M, Gehan EA, et al. The intergroup rhabdomyosarcoma study-I. A final report. Cancer. 1988;61:209-220.
- Maurer HM, Gehan EA, Beltangady M, et al. The intergroup rhabdomyosarcoma study-II. Cancer. 1993;71:1904-1922.
- Crist W, Gehan EA, Ragab AH, et al. The third intergroup rhabdomyosarcoma study. J Clin Oncol. 1995;13:610-630.
- Crist WM, Anderson JR, Meza JL, et al. Intergroup rhabdomyosarcoma study-IV: results for patients with nonmetastatic disease. J Clin Oncol. 2001;19:3091-3102.
- Bodge M, Shillingburg A, Paul S, Biondo L. Safety and efficacy of aprepitant for chemotherapy-induced nausea and vomiting in pediatric patients: a prospective, observational study. Pediatr Blood Cancer. 2014;61:1111-1113.
- Langholz B, Skolnik JM, Barrett JS, et al. Dactinomycin and vincristine toxicity in the treatment of childhood cancer: a retrospective study from the Children’s Oncology Group. Pediatr Blood Cancer. 2011;57:252-257.
- Dakik L, Basbous M, Tamim H, et al. Vincristine-induced neurotoxicity in pediatric patients with rhabdomyosarcoma: a retrospective analysis of clinical features and outcome. Pediatr Hematol Oncol. 2022;39:644-649.
- Howell JE, Szabatura AH, Hatfield Seung A, Nesbit SA. Characterization of the occurrence of ifosfamide-induced neurotoxicity with concomitant aprepitant. J Oncol Pharm Pract. 2008;14:157-162.
- Vazirian F, Samadi S, Rahimi H, et al. Aprepitant, fosaprepitant and risk of ifosfamide-induced neurotoxicity: a systematic review. Cancer Chemother Pharmacol. 2022;90:1-6.
- Yang QY, Hu YH, Guo HL, et al. Vincristine-induced peripheral neuropathy in childhood acute lymphoblastic leukemia: genetic variation as a potential risk factor. Front Pharmacol. 2021;12:771487.
- Stock W, Diouf B, Crews KR, et al. An inherited genetic variant in CEP72 promoter predisposes to vincristine-induced peripheral neuropathy in adults with acute lymphoblastic leukemia. Clin Pharmacol Ther. 2017;101:391-395.
- Wright GEB, Amstutz U, Drögemöller BI, et al. Pharmacogenomics of vincristine-induced peripheral neuropathy implicates pharmacokinetic and inherited neuropathy genes. Clin Pharmacol Ther. 2019;105:402-410.